
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Tatyana Savchenko | + 3423 word(s) | 3423 | 2021-04-12 06:01:58 | | | |
| 2 | Dean Liu | Meta information modification | 3423 | 2021-04-20 09:32:03 | | |
Video Upload Options
Oxidative stress is an integral component of various stress conditions in plants, and this fact largely determines the substantial overlap in physiological and molecular responses to biotic and abiotic environmental challenges. Despite of the significant variation in oxidative stress responses among different plant species and tissues, dynamic and transient character of stress-induced changes in metabolites, and the strong dependence of metabolic responses on the intensity of stress, the available literature data reveal the specific characteristic changes in sugars, sugar derivatives, tricarboxylic acid cycle metabolites, and amino acids, associated with adaptation to oxidative stress. The information about metabolic signature of oxidative stress provides the theoretical base for the selection/generation of plants with improved tolerance to oxidative stress and the development of metabolic markers applicable in research and routine agricultural practice.
1. Introduction
The most common cause of oxidative stress in plants is the saturation of the photosynthetic electron transport chain, which leads to the increased production of O2− and 1O2. This saturation is promoted by excessive illumination, and also a reduced rate of CO2 fixation [1]. CO2 deficiency can occur as a result of rapid assimilation under high light conditions or due to the stomata closure caused by drought and high temperatures. In addition to over-reduction of the photosynthetic electron transport chain, the lack of CO2 leads to an increase in photorespiration and the production of H2O2 in peroxisomes [2]. The low temperatures lead to a significant reduction in the activity of the enzymes of the Calvin cycle and antioxidant enzymes, while light absorption processes are suppressed only slightly. This disbalance causes oxidative stress [3]. Heavy metal ions have a high affinity for protein cysteine residues. Therefore, heavy metal poisoning leads to the inactivation of antioxidant enzymes and enzymes of the Calvin cycle [4]. Moreover, transition metal ions catalyze the Fenton reaction and thus increase the production of hydroxyl radicals. Oxidative stress can be induced by herbicides, such as methyl viologen, transferring electrons from the photosynthetic chain to O2, or diuron and atrazine, blocking the electron transport chain and increasing the production of O2− [3].
Thus, it is clear that in plant cells, the processes occurring within chloroplasts are the major contributors to oxidative stress. At present, the attention of researchers has largely shifted to the study of the fundamental signaling functions of ROS and their interaction with other signaling systems [5][6][7][8]. Importantly, ROS transfer signals, not only within the cell, but also between different plant organs [8][9][10]. The damaging and the signaling activities of ROS will lead to different effects on the level of gene expression profile and metabolism, including the activation of the mechanisms that are simply targeted at the removal of ROS (i.e., the induction of the antioxidant system), or the induction of mechanisms with broader adaptive consequences accompanied by the transcriptomic reprogramming and redistribution of metabolic fluxes. Interestingly enough, these effects can manifest themselves independently of one another. For example, in rice, the overexpression of glycolate oxidase confers tolerance to high light and increased temperature through the involvement of H2O2 as a signaling molecule triggering the protective responses, and does not activate the antioxidant protection [11].
2. Metabolic Signature of Oxidative Stress in Plants
2.1. Sugars and Sugar Derivatives
Major rearrangements of metabolic pathways under oxidative stress conditions observed in independent studies include the down-regulation of the glycolysis and tricarboxylic acid (TCA) cycle and activation of the oxidative pentose phosphate pathway [12][13][14][15][16]. The rerouting of glycolytic carbon flow into the oxidative pentose phosphate pathway mainly determines the changes in the sugar profile and alters the levels of sugar phosphates and soluble carbohydrates with antioxidant properties (Figure 1) [12][17]. Antioxidant properties have been confirmed for numerous soluble carbohydrates, such as sucrose, fructose, raffinose, sorbitol, mannitol, and fructans in plant tissues, in vitro, and in specific model systems, such as fish oil-in-water emulsion [18][19][20][21][22][23][24][25][26]. Being directly associated with ROS-producing processes, such as respiration and photosynthesis, and simultaneously with anti-oxidative processes, such as the oxidative pentose-phosphate pathway and carotenoid biosynthesis, soluble sugars (mainly mono- and disaccharides) are involved in the formation of the prooxidant and antioxidant intracellular environment [19][27]. Based on the strong antioxidant properties of sucrose, a primary stable carbohydrate of photosynthesis and major transported sugar in plants, the antioxidant functions of this metabolite in plant tissue have been suggested, especially in tissues with a high sucrose content [28][29][30]. Fast accumulation of sucrose and fructose, preceded by an increase in glucose level, was observed in the Arabidopsis cell culture treated with H2O2 [15]. High ozone concentrations known to induce oxidative stress in plants [31] led to a significant increase in water soluble carbohydrates in leaves of tree species Phoebe bournei and Phoebe zhennan [32]. Transgenic potato plants with increased sugar content resulted from the insertion of a yeast-derived invertase gene, coding for sucrose metabolism enzyme, and displayed a higher tolerance to hypothermia-induced oxidative stress, wherein the protective effect of sugars was caused by their ability to scavenge ROS nonspecifically under stress conditions [33]. The absence of either alkaline or neutral invertases in Arabidopsis was associated with higher expression of the oxidative stress defense gene, while transient overexpression of invertase gene in leaf mesophyll protoplasts downregulated the oxidative stress-responsive promoter of ascorbate peroxidase 2 [34]. The reprogramming of sugar metabolism under oxidative stress conditions was also confirmed by the decreased starch accumulation in transgenic Arabidopsis plants constitutively experiencing oxidative stress due to the overexpression of glycolate oxidase gene [35]. It is important to keep in mind that sugars perform not only antioxidant, but also signaling, functions. This, in particular, is confirmed by the data revealing the requirement of mitochondria-associated hexokinase for the development of oxidative stress-induced programmed cell death (PCD) in plant cells, suggesting a link between the signaling functions of glucose and apoptosis [36].
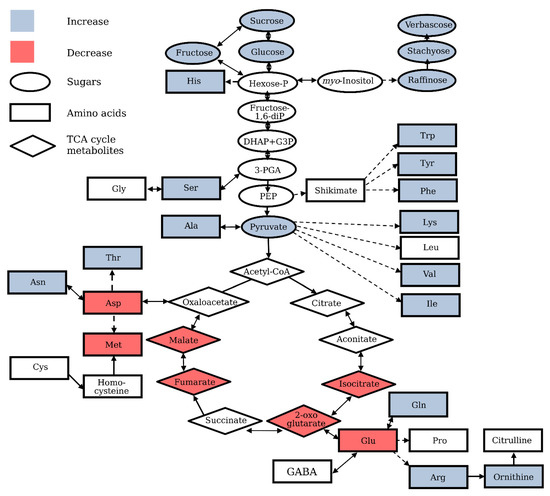
Figure 1. Oxidative stress-specific alteration of central metabolism in plants. Schematic representation of the major metabolic pathways and central metabolites, sugars, amino acids, and tricarboxylic acid (TCA) cycle metabolites. Based on the results of multiple studies, the levels of numerous metabolites were shown to be affected by oxidative stress, increased (indicated in blue) or decreased (indicated in red). Please note, that the scheme reflects the trends observed in the majority of studies, although the individual metabolites may display a different pattern in selected studies.
Stresses that give rise to excess concentrations of ROS, such as drought, freezing, heat, and excessive light irradiation, are accompanied by an accumulation of raffinose family oligosaccharides (RFO), which are α-galactosyl extensions of sucrose [37][38][39]. Among these oligosaccharides, raffinose was found in many plants, and oligosaccharides of a higher degree of polymerization, such as stachyose and verbascose, were present only in selected species [40], where they function as osmoprotectants and stabilizers of cellular membranes. Several lines of evidence suggest that these metabolites are involved in plant adaptation to conditions of oxidative stress [28][29][38] and act as antioxidants and scavengers of hydroxyl radicals, the most deleterious form of ROS [37]. The induction of genes of galactinol synthase and raffinose synthase, the enzymes of the RFO biosynthesis pathway, and heat shock transcription factors regulating the expression of these genes by hydrogen peroxide treatment, confirms the specific connection between this particular pathway and oxidative stress [38]. It has been established that raffinose accumulation in Arabidopsis is not regulated by the abscisic acid (ABA)-dependent CBF/DREB1 (the C-repeat (CRT)-binding factor/dehydration-responsive element (DRE) binding protein 1) pathway [41], but Boea hygrometrica genes of galactinol synthase and raffinose synthase contain W box cis-elements in their promoters, known targets for the ABA-inducible WRKY family of transcription factors that mediate oxidative stress responses [42][43]. The overexpression of galactinol synthase, raffinose synthase, or the heat shock transcription factor A2, leading to an increase in intracellular levels of RFO in transgenic Arabidopsis plants, resulted in the improvement in ROS scavenging capacity and oxidative stress tolerance [38][44][45]. The presence of specific raffinose transporters, enabling the transport of RFO to chloroplasts, justifies the antioxidant functions of these metabolites in plastids [46]. The level of myo-inositol, a substrate for galactinol biosynthesis [47][48], which is in turn a substrate for raffinose biosynthesis, also was reported to be altered under oxidative stress conditions [18][49][50]. In apple, myo-inositol regulates ROS-induced programmed cell death through the salicylic acid-dependent and ethylene-dependent pathways [49]. Arabidopsis mutant for a specific myo-inositol phosphate synthase (MIPS1) under long day conditions or at increased growth irradiance shows a lesion phenotype that is similar to the hypersensitive response used by plants to prevent the spreading of infection [51][52]. Since myo-inositol also serves as a substrate for the biosynthesis of other metabolites and signaling molecules, contributing to plant adaptation to adverse environmental conditions, the level of this metabolite does not necessarily increase under stress conditions [53]. For instance, in Arabidopsis cat2 mutant, which is constitutively in a state of oxidative stress due to the absence of the functional CATALASE2 gene, myo-inositol level was reduced under stress conditions [54][55].
Mannose, an epimer of glucose, is directly involved in the biosynthesis of the major antioxidant, ascorbate [56], and mannose derivative mannitol has protective functions under oxidative stress conditions. Transgenic Nicotiana tabacum plants, overexpressing mannitol-1-phosphate dehydrogenase gene coding for the chloroplasts protein, and, consequently, accumulating an increased level of mannitol in chloroplasts, display enhanced tolerance to methyl viologen-induced oxidative stress [24]. Threonate, a derivative of four-carbon monosaccharide threose, was also observed to accumulate in menadione-treated Arabidopsis cells [12], though it is important to remember that threonic acid can be formed from threose or as a result of the degradation of ascorbate [57].
Among the known di- and oligosaccharides with protective functions under oxidative stress conditions, the most studied are trehalose, a disaccharide consisting of two glucose molecules, and short-chain fructans, polymers of fructose molecules terminated by a single glucose molecule. Foliar application of trehalose led to a significant decrease in symptoms of oxidative stress in quinoa and wheat plants [58][59]. Transgenic tomato plants, accumulating higher levels of trehalose due to the overexpression of yeast trehalose-6-phosphate synthase gene, exhibited enhanced tolerance to oxidative stress [60]. The antioxidant properties of fructans have been demonstrated in plants and intestinal lumen [23][61][62]. Furthermore, in plants the metabolism of fructans is modulated by foliar application of S-nitrosoglutathione, a NO donor, wherein an increase in the fructans level helps to mitigate oxidative stress [62]. Importantly, fructans also scavenge hydroxyl radicals, and a predominant outcome of the ·OH–fructans interaction is the splitting of oligosaccharides, resulting in the formation of non-radical products, which can be used as markers for nonenzymatic sugar–radical interactions in vivo [23].
While all the above-mentioned sugars were shown to be involved in the oxidative stress response, a certain specificity of their protective functions is evident. For example, tolerant rice species accumulated galactose and raffinose under chill-stress conditions whereas these saccharides declined in sensitive species, and at the same time, the tolerance mechanism in the more salt- and water-deficit-tolerant species was associated with the accumulation of osmoprotectants, such as glucose, trehalose, and mannitol [63].
2.2. Tricarboxylic Acid Cycle Metabolites
Many tricarboxylic acid (TCA) cycle intermediates serve as substrates for several biosynthetic pathways, and therefore their levels display a very dynamic character, reflecting the processes of formation and consumption of these metabolites. Nevertheless, the levels of TCA cycle metabolites have been shown to increase when respiration is stimulated [64], and the characteristic decrease in the pool of TCA cycle metabolites has been observed in plant cells under oxidative stress conditions (Figure 1) [65][66][13]. Modulation of the activity of TCA cycle enzymes seems to contribute significantly to the alteration of the metabolic profile under oxidative stress conditions. Many enzymes of the tricarboxylic acid cycle, such as aconitase, pyruvate-dehydrogenase, and 2-oxoglutarate-dehydrogenase, are sensitive to oxidative inhibition [17][67][68][69], while other enzymes have been shown to be subject to redox modifications under stress conditions (reviewed in [66]). A decrease in TCA cycle metabolites was detected in heterotrophic Arabidopsis and rice cultured cells [12][13][15] and roots under oxidative stress conditions [17][16]. The suppression of operation of mitochondria TCA cycle activity in nonphotosynthetic cells upon menadione treatment [12][13][17][16] is rather expected, because the mitochondrion is one of the main sites of ROS production upon addition of menadione to nonphotosynthetic cells. H2O2 and methyl viologen treatments also affect the TCA cycle significantly [14][15]. Unfortunately, it is impossible to compare the effects induced by menadione, methyl viologen, and H2O2, since, to best of our knowledge, a comparative analysis of the effects of different treatments on the metabolic profile of the same biological sample has not been carried out yet.
A kinetic analysis of 13C-labeled compounds in Arabidopsis cultured cells and roots, following labeling with exogenously supplied 13C-glucose, confirmed a reduction of carbon flux through the TCA cycle, manifested in decreased labeling of almost all metabolites under menadione-induced oxidative stress conditions [12][16]. Expectedly, such a reduction in the carbon flow through the TCA cycle was accompanied by a steady accumulation of pyruvate, which upon oxidation produces acetyl-CoA, feeding the TCA cycle. Interestingly, a decrease of carbon flux through TCA cycle intermediates is not necessarily accompanied by a decreased metabolite abundance, due to the simultaneous decrease in the activity of downstream pathways; and the pattern of changes in the amount of individual TCA metabolites can be dynamic and variable [16]. For example, the citrate abundance was significantly decreased after 0.5 h of treatment with menadione, but then increased steadily under stress conditions, and even during the recovery from stress, the 2-oxoglutarate level showed a transient increase after the menadione treatment, then decreased below control level in 2 h, followed by three-fold control level increase in 6 h after the treatment. Succinate, fumarate, and malate were decreased up to two-fold [16]. A significant decline in the levels of glycolytic and TCA cycle intermediates was also observed in wild type Arabidopsis upon methyl viologen treatment, reflecting oxidative stress in plastids and inhibition of photosynthesis and respiration [14]. The pyruvate and TCA cycle metabolites displayed the most prominent differences between methyl viologen-treated wild type Arabidopsis and rcd1 (radical-induced cell death1) mutant, showing improved tolerance to methyl viologen-induced oxidative stress [14]. In rcd1 mutant, the levels of all TCA cycle intermediates, except fumarate, increased or remained high after methyl viologen treatment.
Although most studies have pointed to the suppression of TCA cycle activity, the protective role of TCA cycle metabolites under oxidative stress conditions has been also discussed. For example, the role of malate in the induction of protective antioxidant defense was demonstrated in green algae Scenedesmus obliquus and banana fruits [70][71], and citrate presence in the exudate of aluminum-tolerant Indica rice cultivars was shown to contribute to the tolerance of aluminum-induced oxidative stress [72].
2.3. Amino Acids
Amino acids are the building blocks of proteins and substrates for numerous secondary metabolites’ biosynthesis. Amino acid profile has been shown to be significantly affected by oxidative stress in plants (Figure 1) [73][15][17][16][74]. An increase in the content of individual amino acids during stress can result from the activation of biosynthetic pathways and from enhanced protein degradation [13][17][75]. The high contribution of protein degradation to the pool of free amino acids during oxidative stress has been confirmed in the analysis of labeled isotopes redistribution, revealing the suppression of the activity of pathways associated with amino acids metabolism [17][16]. Analysis of the available microarray dataset and amino acid profiles showed that under various abiotic stress conditions, abundant amino acids such as proline, arginine, asparagine, glutamine, and gamma-aminobutyric acid (GABA) are synthesized during abiotic stresses to be used by the plant as compatible osmolytes, precursors for secondary metabolites, or storage forms of organic nitrogen, while low abundance amino acids are not synthesized, but accumulate due to the increased protein degradation under stress-induced carbohydrate starvation [75].
The shikimate pathway, derived from phosphoenolpyruvate, the precursor of pyruvate, leads to the formation of aromatic amino acids and various secondary metabolites possessing antioxidant properties, such as lignin, flavonoids, alkaloids, and phytoalexins. Phenolic compounds containing one (phenolic acids) or more (polyphenols) aromatic rings, such as caffeic acid, chlorogenic acid, and ferulic acid, are effective antioxidants and free radical scavengers, and accumulation of these compounds is one of the most effective strategies used by plants to avoid oxidative damage [76][77][78]. Aromatic amino acids are used for the production of pigments, hormones, various polyphenols, and cell wall components [79]. Thus, these amino acids are directly and indirectly associated with antioxidant defense in plants. It has been demonstrated that phenylalanine and tyrosine accumulate in Arabidopsis roots under oxidative stress conditions [17], and the activation of the Shikimate pathway has been observed in Scrophularia striata cell culture under jasmonate-induced oxidative stress [80].
An increase in the branched-chain amino acids isoleucine and valine has also been observed in the roots of menadione-treated plants [17]. These amino acids are critical for normal plant growth, while also serving as substrates for protective secondary metabolites, such as cyanogenic glycosides, glucosinolates, and acyl-sugars [81].
Sulfur-containing amino acids, free and in proteins, together with other sulfur-containing metabolites (especially glutathione), represent an important part of the antioxidant system in animal and plant cells [82][83][84]. Methionine and cysteine are the major end products of sulfate assimilation comprising up to 90% of the total sulfur in most plants [85]. Based on the analysis of a 13C-redistribution in roots of plants supplied with 13C isotope labeled glucose, a significant decrease in methionine occurred under oxidative stress conditions, while the level of O-acetylserine, the precursor of cysteine, was increased significantly [15][17][16]. This suggests a shift in the balance in sulfur metabolism under stress conditions, which is most likely directed toward the production of cysteine, a substrate for glutathione biosynthesis. It was shown that the synthesis of glutathione itself, as well its key precursor cysteine, can be modulated by the ratio between reduced and oxidized glutathione [86][87]. Moreover, exogenously applied cysteine alleviates the oxidative stress induced by cobalt in Ocimum basilicum L. [88].
The level of alanine usually correlates to the intensity of starch catabolism. In the roots of menadione-treated Arabidopsis plants, alanine level was increased [17], although it did not show any significant response to 13C-redistribution in labeled glucose feeding experiments [16]. It was stated that the maintenance of high alanine levels under oxidative stress conditions correlates with plant tolerance to this stress [14].
Glutamate and aspartate derived from the TCA cycle are important substrates for the synthesis of several amino acids and donors of the amino group. The levels of aspartate derived from oxaloacetate and glutamate derived from 2-oxoglutarate decreased drastically in Arabidopsis roots under oxidative stress conditions induced by menadione treatment [17][16]. Importantly, in contrast to other metabolites, the levels of these amino acids did not return to the initial level, even during plant recovery from stress. At the same time, the downstream products asparagine and threonine showed an increase [17]. Glutamate and aspartate levels were also significantly decreased in menadione-treated rice suspension cells, despite an increase in most other amino acids, even glutamate and aspartate derivatives [13]. In Arabidopsis, oxidative stress induced by ultraviolet B (UV-B) treatment [89] moderately increased glutamate, and very strongly increased glutamine [90]. It seems that the ability to maintain the level of aspartate and glutamate under oxidative stress conditions also correlates with plant tolerance [14]. The increase in the levels of derivatives of glutamate and aspartate has been observed in several studies [13][17][74][90]. In wheat, TCA cycle-derived aspartic acid, asparagine, and GABA accumulated under oxidative stress conditions [74]. GABA was also strongly increased under oxidative stress in Arabidopsis roots [16]. The GABA often acts as a signal molecule in the regulation of stress responses, and antioxidant properties of GABA have been suggested [91].
In Scrophularia striata suspension cells the levels of asparagine and also histidine increased under oxidative stress induced by methyl jasmonate application [80]. UV-B treatment also significantly increased the level of lysine and histidine in Arabidopsis [90].
Proline, a general stress response metabolite associated with chloroplasts and photosynthesis [92], was reported to decrease in Arabidopsis cell culture treated with H2O2 [15], but increased in roots experiencing oxidative stress [17][16]. Oxidative stress resulting from aluminum toxicity was accompanied by a significant increase in proline in aluminum-tolerant rice cultivars [72]. The accumulation of proline under oxidative stress conditions and its role in the maintenance of redox balance have been demonstrated, although the ROS-scavenging ability of proline is still debated [93][94]. The level of glycine, another abundant stress response metabolite, was also decreased under oxidative stress conditions in Arabidopsis cell culture treated with H2O2 [15].
Citrulline is a nonessential amino acid that has been reported to be an efficient hydroxyl radical scavenger and a strong antioxidant [95][96]. Citrulline accumulation correlated with the tolerance to salt and drought stress [97][98]. Ornithine, the precursor of citrulline, was shown to increase transiently under oxidative stress [16].
The data on the major central metabolites involved in oxidative stress response, possessing antioxidant properties, or whose levels are affected by oxidative stress are summarized in Table 1. Thus, in this review, we have collected and analyzed the available data on oxidative stress-associated changes of the central metabolism in model plants and agricultural species. A thorough examination of the available data reveals common trends in the redistribution of metabolic fluxes during the plant adaptation to oxidative stress, targeted to the enhancement of plant performance under unfavorable environmental conditions. Adaptive metabolic changes are aimed at three major targets: (1) prevention of ROS accumulation, (2) maintenance of the biosynthesis of indispensable metabolites, and (3) production of protective compounds. The presented analysis provides the theoretical basis for the selection/generation of plants with improved tolerance to oxidative stress and the development of metabolic markers applicable in research and routine agricultural practice.
References
- Sharma, P.; Jha, A.B.; Dubey, R.S.; Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012, 2012, 217037.
- Noctor, G.; Veljovic-Jovanovic, S.; Driscoll, S.; Novitskaya, L.; Foyer, C.H. Drought and oxidative load in the leaves of C3 plants: A predominant role for photorespiration? Ann. Bot. 2002, 89, 841–850.
- Bartosz, G. Oxidative stress in plants. Acta Physiol. Plant 1997, 19, 47–64.
- Fryzova, R.; Pohanka, M.; Martinkova, P.; Cihlarova, H.; Brtnicky, M.; Hladky, J.; Kynicky, J. Oxidative Stress and Heavy Metals in Plants. In Reviews of Environmental Contamination and Toxicology; De Voogt, P., Ed.; Springer: Cham, Switzerland, 2017; Volume 245, pp. 129–156.
- Noctor, G.; Reichheld, J.-P.; Foyer, C.H. ROS-related redox regulation and signaling in plants. Semin. Cell Dev. Biol. 2018, 80, 3–12.
- Foyer, C.H. Reactive oxygen species, oxidative signaling and the regulation of photosynthesis. Environ. Exp. Bot. 2018, 154, 134–142.
- Hasanuzzaman, M.; Bhuyan, M.H.M.B.; Zulfiqar, F.; Raza, A.; Mohsin, S.M.; Mahmud, J.A.; Fujita, M.; Fotopoulos, V. Reactive Oxygen Species and Antioxidant Defense in Plants under Abiotic Stress: Revisiting the Crucial Role of a Universal Defense Regulator. Antioxidants 2020, 9, 681.
- Dietz, K.-J.; Turkan, I.; Krieger-Liszkay, A. Redox- and Reactive Oxygen Species-Dependent Signaling into and out of the Photosynthesizing Chloroplast. Plant Physiol. 2016, 171, 1541–1550.
- Gilroy, S.; Białasek, M.; Suzuki, N.; Górecka, M.; Devireddy, A.R.; Karpinski, S.; Mittler, R. ROS, Calcium, and Electric Signals: Key Mediators of Rapid Systemic Signaling in Plants. Plant Physiol. 2016, 171, 1606–1615.
- Choi, W.-G.; Miller, G.; Wallace, I.; Harper, J.; Mittler, R.; Gilroy, S. Orchestrating rapid long-distance signaling in plants with Ca2+, ROS and electrical signals. Plant J. 2017, 90, 698–707.
- Cui, L.-L.; Lu, Y.-s.; Li, Y.; Yang, C.; Peng, X.-X. Overexpression of Glycolate Oxidase Confers Improved Photosynthesis under High Light and High Temperature in Rice. Front. Plant Sci. 2016, 7, 1165.
- Baxter, C.J.; Redestig, H.; Schauer, N.; Repsilber, D.; Patil, K.R.; Nielsen, J.; Selbig, J.; Liu, J.; Fernie, A.R.; Sweetlove, L.J. The metabolic response of heterotrophic Arabidopsis cells to oxidative stress. Plant Physiol. 2007, 143, 312–325.
- Ishikawa, T.; Takahara, K.; Hirabayashi, T.; Matsumura, H.; Fujisawa, S.; Terauchi, R.; Uchimiya, H.; Kawai-Yamada, M. Metabolome Analysis of Response to Oxidative Stress in Rice Suspension Cells Overexpressing Cell Death Suppressor Bax Inhibitor-1. Plant Cell Physiol. 2009, 51, 9–20.
- Sipari, N.; Lihavainen, J.; Shapiguzov, A.; Kangasjärvi, J.; Keinänen, M. Primary Metabolite Responses to Oxidative Stress in Early-Senescing and Paraquat Resistant Arabidopsis thaliana rcd1 (Radical-Induced Cell Death1). Front. Plant Sci. 2020, 11, 194.
- Chen, Y.; Hoehenwarter, W. Changes in the Phosphoproteome and Metabolome Link Early Signaling Events to Rearrangement of Photosynthesis and Central Metabolism in Salinity and Oxidative Stress Response in Arabidopsis. Plant Physiol. 2015, 169, 3021–3033.
- Lehmann, M.; Laxa, M.; Sweetlove, L.J.; Fernie, A.R.; Obata, T. Metabolic recovery of Arabidopsis thaliana roots following cessation of oxidative stress. Metabolomics 2012, 8, 143–153.
- Lehmann, M.; Schwarzländer, M.; Obata, T.; Sirikantaramas, S.; Burow, M.; Olsen, C.E.; Tohge, T.; Fricker, M.D.; Møller, B.L.; Fernie, A.R.; et al. The metabolic response of Arabidopsis roots to oxidative stress is distinct from that of heterotrophic cells in culture and highlights a complex relationship between the levels of transcripts, metabolites, and flux. Mol. Plant 2009, 2, 390–406.
- Savchenko, T.; Rolletschek, H.; Heinzel, N.; Tikhonov, K.; Dehesh, K. Waterlogging tolerance rendered by oxylipin-mediated metabolic reprogramming in Arabidopsis. J. Exp. Bot. 2019, 70, 2919–2932.
- Couée, I.; Sulmon, C.; Gouesbet, G.; El Amrani, A. Involvement of soluble sugars in reactive oxygen species balance and responses to oxidative stress in plants. J. Exp. Bot. 2006, 57, 449–459.
- Ende, W.V.; Peshev, D. Sugars as Antioxidants in Plants; Springer: New York, NY, USA, 2013.
- Faraji, H.; Lindsay, R.C. Characterization of the antioxidant activity of sugars and polyhydric alcohols in fish oil emulsions. J. Agric. Food Chem. 2004, 52, 7164–7171.
- Keunen, E.; Peshev, D.; Vangronsveld, J.; Van den Ende, W.; Cuypers, A. Plant sugars are crucial players in the oxidative challenge during abiotic stress: Extending the traditional concept. Plant Cell Environ. 2013, 36, 1242–1255.
- Peshev, D.; Vergauwen, R.; Moglia, A.; Hideg, E.; Van den Ende, W. Towards understanding vacuolar antioxidant mechanisms: A role for fructans? J. Exp. Bot 2013, 64, 1025–1038.
- Shen, B.; Jensen, R.G.; Bohnert, H.J. Increased Resistance to Oxidative Stress in Transgenic Plants by Targeting Mannitol Biosynthesis to Chloroplasts. Plant Physiol. 1997, 113, 1177–1183.
- Stoyanova, S.; Geuns, J.; Hideg, É.; Van Den Ende, W. The food additives inulin and stevioside counteract oxidative stress. Int. J. Food Sci. Nutr. 2011, 62, 207–214.
- Van den Ende, W.; Peshev, D.; De Gara, L. Disease prevention by natural antioxidants and prebiotics acting as ROS scavengers in the gastrointestinal tract. Trends Food Sci. Technol. 2011, 22, 689–697.
- Roitsch, T. Source-sink regulation by sugar and stress. Curr. Opin. Plant Biol. 1999, 2, 198–206.
- Morelli, R.; Russo-Volpe, S.; Bruno, N.; Lo Scalzo, R. Fenton-Dependent Damage to Carbohydrates: Free Radical Scavenging Activity of Some Simple Sugars. J. Agric. Food Chem. 2003, 51, 7418–7425.
- Smirnoff, N.; Cumbes, Q.J. Hydroxyl radical scavenging activity of compatible solutes. Phytochemistry 1989, 28, 1057–1060.
- Van den Ende, W.; Valluru, R. Sucrose, sucrosyl oligosaccharides, and oxidative stress: Scavenging and salvaging? J. Exp. Bot. 2009, 60, 9–18.
- Iriti, M.; Faoro, F. Oxidative Stress, the Paradigm of Ozone Toxicity in Plants and Animals. Water Air Soil Pollut. 2008, 187, 285–301.
- Chen, Z.; Cao, J.; Yu, H.; Shang, H. Effects of Elevated Ozone Levels on Photosynthesis, Biomass and Non-structural Carbohydrates of Phoebe bournei and Phoebe zhennan in Subtropical China. Front. Plant Sci. 2018, 9, 1764.
- Sin’kevich, M.S.; Deryabin, A.N.; Trunova, T.I. Characteristics of oxidative stress in potato plants with modified carbohydrate metabolism. Russ. J. Plant Physiol. 2009, 56, 168–174.
- Xiang, L.; Le Roy, K.; Bolouri-Moghaddam, M.-R.; Vanhaecke, M.; Lammens, W.; Rolland, F.; Van den Ende, W. Exploring the neutral invertase–oxidative stress defence connection in Arabidopsis thaliana. J. Exp. Bot. 2011, 62, 3849–3862.
- Fahnenstich, H.; Scarpeci, T.E.; Valle, E.M.; Flügge, U.-I.; Maurino, V.G. Generation of Hydrogen Peroxide in Chloroplasts of Arabidopsis Overexpressing Glycolate Oxidase as an Inducible System to Study Oxidative Stress. Plant Physiol. 2008, 148, 719–729.
- Kim, M.; Lim, J.-H.; Ahn, C.S.; Park, K.; Kim, G.T.; Kim, W.T.; Pai, H.-S. Mitochondria-associated hexokinases play a role in the control of programmed cell death in Nicotiana benthamiana. Plant Cell 2006, 18, 2341–2355.
- ElSayed, A.I.; Rafudeen, M.S.; Golldack, D. Physiological aspects of raffinose family oligosaccharides in plants: Protection against abiotic stress. Plant Biol. 2014, 16, 1–8.
- Nishizawa, A.; Yabuta, Y.; Shigeoka, S. Galactinol and Raffinose Constitute a Novel Function to Protect Plants from Oxidative Damage. Plant Physiol. 2008, 147, 1251–1263.
- Urano, K.; Maruyama, K.; Ogata, Y.; Morishita, Y.; Takeda, M.; Sakurai, N.; Suzuki, H.; Saito, K.; Shibata, D.; Kobayashi, M.; et al. Characterization of the ABA-regulated global responses to dehydration in Arabidopsis by metabolomics. Plant J. 2009, 57, 1065–1078.
- Janeček, Š.; Lanta, V.; Klimešová, J.; Doležal, J. Effect of abandonment and plant classification on carbohydrate reserves of meadow plants. Plant Biol. 2011, 13, 243–251.
- Cook, D.; Fowler, S.; Fiehn, O.; Thomashow, M.F. A prominent role for the CBF cold response pathway in configuring the low-temperature metabolome of Arabidopsis. Proc. Natl. Acad. Sci. USA 2004, 101, 15243–15248.
- Wang, Z.; Zhu, Y.; Wang, L.; Liu, X.; Liu, Y.; Phillips, J.; Deng, X. A WRKY transcription factor participates in dehydration tolerance in Boea hygrometrica by binding to the W-box elements of the galactinol synthase (BhGolS1) promoter. Planta 2009, 230, 1155–1166.
- Wu, X.; Kishitani, S.; Ito, Y.; Toriyama, K. Accumulation of raffinose in rice seedlings overexpressing OsWRKY11 in relation to desiccation tolerance. Plant Biotechnol. 2009, 26, 431–434.
- Bolouri-Moghaddam, M.R.; Le Roy, K.; Xiang, L.; Rolland, F.; Van den Ende, W. Sugar signalling and antioxidant network connections in plant cells. FEBS J. 2010, 277, 2022–2037.
- Nishizawa-Yokoi, A.; Yabuta, Y.; Shigeoka, S. The contribution of carbohydrates including raffinose family oligosaccharides and sugar alcohols to protection of plant cells from oxidative damage. Plant Signal. Behav. 2008, 3, 1016–1018.
- Schneider, T.; Keller, F. Raffinose in Chloroplasts is Synthesized in the Cytosol and Transported across the Chloroplast Envelope. Plant Cell Physiol. 2009, 50, 2174–2182.
- Loewus, F.A.; Kelly, S.; Neufeld, E.F. Metabolism of Myo-Inositol in Plants: Conversion to Pectin, Hemicellulose, D-Xylose, and Sugar Acids. Proc. Natl. Acad. Sci. USA 1962, 48, 421–425.
- Loewus, F.A.; Murthy, P.P.N. Myo-Inositol metabolism in plants. Plant Sci. 2000, 150, 1–19.
- Hu, L.; Zhou, K.; Ren, G.; Yang, S.; Liu, Y.; Zhang, Z.; Li, Y.; Gong, X.; Ma, F. Myo-inositol mediates reactive oxygen species-induced programmed cell death via salicylic acid-dependent and ethylene-dependent pathways in apple. Hortic. Res. 2020, 7, 138.
- Lytvyn, D.I.; Raynaud, C.; Yemets, A.I.; Bergounioux, C.; Blume, Y.B. Involvement of Inositol Biosynthesis and Nitric Oxide in the Mediation of UV-B Induced Oxidative Stress. Front. Plant Sci. 2016, 7, 430.
- Meng, P.H.; Raynaud, C.; Tcherkez, G.; Blanchet, S.; Massoud, K.; Domenichini, S.; Henry, Y.; Soubigou-Taconnat, L.; Lelarge-Trouverie, C.; Saindrenan, P.; et al. Crosstalks between myo-inositol metabolism, programmed cell death and basal immunity in Arabidopsis. PLoS ONE 2009, 4, e7364.
- Donahue, J.L.; Alford, S.R.; Torabinejad, J.; Kerwin, R.E.; Nourbakhsh, A.; Ray, W.K.; Hernick, M.; Huang, X.; Lyons, B.M.; Hein, P.P.; et al. The Arabidopsis thaliana Myo-inositol 1-phosphate synthase1 gene is required for Myo-inositol synthesis and suppression of cell death. Plant Cell 2010, 22, 888–903.
- Valluru, R.; Van den Ende, W. Myo-inositol and beyond–Emerging networks under stress. Plant Sci. 2011, 181, 387–400.
- Mhamdi, A.; Queval, G.; Chaouch, S.; Vanderauwera, S.; Van Breusegem, F.; Noctor, G. Catalase function in plants: A focus on Arabidopsis mutants as stress-mimic models. J. Exp. Bot. 2010, 61, 4197–4220.
- Chaouch, S.; Noctor, G. Myo-inositol abolishes salicylic acid-dependent cell death and pathogen defence responses triggered by peroxisomal hydrogen peroxide. New Phytol. 2010, 188, 711–718.
- Smirnoff, N.; Conklin, P.L.; Loewus, F.A. Biosynthesis of ascorbic acid in plants: A renaissance. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2001, 52, 437–467.
- Truffault, V.; Fry, S.C.; Stevens, R.G.; Gautier, H. Ascorbate degradation in tomato leads to accumulation of oxalate, threonate and oxalyl threonate. Plant J. 2017, 89, 996–1008.
- Aldesuquy, H.; Ghanem, H.E. Exogenous Salicylic Acid and Trehalose Ameliorate Short Term Drought Stress in Wheat Cultivars by Up-regulating Membrane Characteristics and Antioxidant Defense System. J. Hortic. 2015, 2, 1–10.
- Sadak, M.S.; El-Bassiouny, H.M.S.; Dawood, M.G. Role of trehalose on antioxidant defense system and some osmolytes of quinoa plants under water deficit. Bull. Natl. Res. Cent. 2019, 43, 5.
- Cortina, C.; Culiáñez-Macià, F.A. Tomato abiotic stress enhanced tolerance by trehalose biosynthesis. Plant Sci. 2005, 169, 75–82.
- Franco-Robles, E.; López, M.G. Implication of Fructans in Health: Immunomodulatory and Antioxidant Mechanisms. Sci. World J. 2015, 2015, 289267.
- Rigui, A.P.; Carvalho, V.; Wendt dos Santos, A.L.; Morvan-Bertrand, A.; Prud’homme, M.-P.; Machado de Carvalho, M.A.; Gaspar, M. Fructan and antioxidant metabolisms in plants of Lolium perenne under drought are modulated by exogenous nitric oxide. Plant Physiol. Biochem. 2019, 145, 205–215.
- Morsy, M.R.; Jouve, L.; Hausman, J.F.; Hoffmann, L.; Stewart, J.M. Alteration of oxidative and carbohydrate metabolism under abiotic stress in two rice (Oryza sativa L.) genotypes contrasting in chilling tolerance. J. Plant Physiol. 2007, 164, 157–167.
- Dastogeer, K.M.G.; Li, H.; Sivasithamparam, K.; Jones, M.; Du, X.; Ren, Y.; Wylie, S.J. Metabolic responses of endophytic Nicotiana benthamiana plants experiencing water stress. Environ. Exp. Bot. 2017, 143, 59–71.
- Obata, T.; Matthes, A.; Koszior, S.; Lehmann, M.; Araújo, W.L.; Bock, R.; Sweetlove, L.J.; Fernie, A.R. Alteration of mitochondrial protein complexes in relation to metabolic regulation under short-term oxidative stress in Arabidopsis seedlings. Phytochemistry 2011, 72, 1081–1091.
- Dumont, S.; Rivoal, J. Consequences of Oxidative Stress on Plant Glycolytic and Respiratory Metabolism. Front. Plant Sci. 2019, 10, 166.
- Morgan, M.J.; Lehmann, M.; Schwarzländer, M.; Baxter, C.J.; Sienkiewicz-Porzucek, A.; Williams, T.C.R.; Schauer, N.; Fernie, A.R.; Fricker, M.D.; Ratcliffe, R.G.; et al. Decrease in Manganese Superoxide Dismutase Leads to Reduced Root Growth and Affects Tricarboxylic Acid Cycle Flux and Mitochondrial Redox Homeostasis. Plant Physiol. 2008, 147, 101–114.
- Sweetlove, L.J.; Heazlewood, J.L.; Herald, V.; Holtzapffel, R.; Day, D.A.; Leaver, C.J.; Millar, A.H. The impact of oxidative stress on Arabidopsis mitochondria. Plant J. 2002, 32, 891–904.
- Verniquet, F.; Gaillard, J.; Neuburger, M.; Douce, R. Rapid Inactivation of Plant Aconitase by Hydrogen-Peroxide. Biochem. J. 1991, 276, 643–648.
- Du, C.; Zhang, B.; He, Y.; Hu, C.; Ng, Q.X.; Zhang, H.; Ong, C.N.; Lin, Z. Biological effect of aqueous C(60) aggregates on Scenedesmus obliquus revealed by transcriptomics and non-targeted metabolomics. J. Hazard. Mater. 2017, 324, 221–229.
- Huang, H.; Jian, Q.; Jiang, Y.; Duan, X.; Qu, H. Enhanced chilling tolerance of banana fruit treated with malic acid prior to low-temperature storage. Postharvest. Biol. Technol. 2016, 111, 209–213.
- Awasthi, J.P.; Saha, B.; Panigrahi, J.; Yanase, E.; Koyama, H.; Panda, S.K. Redox balance, metabolic fingerprint and physiological characterization in contrasting North East Indian rice for Aluminum stress tolerance. Sci. Rep. 2019, 9, 8681.
- Li, K.; Chen, J.; Zhu, L. The phytotoxicities of decabromodiphenyl ether (BDE-209) to different rice cultivars (Oryza sativa L.). Environ. Pollut. 2018, 235, 692–699.
- Liu, Q.; Liu, M.; Wu, S.; Xiao, B.; Wang, X.; Sun, B.; Zhu, L. Metabolomics Reveals Antioxidant Stress Responses of Wheat (Triticum aestivum L.) Exposed to Chlorinated Organophosphate Esters. J. Agric. Food Chem. 2020, 68, 6520–6529.
- Hildebrandt, T.M. Synthesis versus degradation: Directions of amino acid metabolism during Arabidopsis abiotic stress response. Plant Mol. Biol. 2018, 98, 121–135.
- Alvarez, S.; Marsh, E.L.; Schroeder, S.G.; Schachtman, D.P. Metabolomic and proteomic changes in the xylem sap of maize under drought. Plant Cell Environ. 2008, 31, 325–340.
- Tzin, V.; Galili, G. The Biosynthetic Pathways for Shikimate and Aromatic Amino Acids in Arabidopsis thaliana. Arab. Book 2010, 8, e0132.
- Watkins, J.M.; Chapman, J.M.; Muday, G.K. Abscisic Acid-Induced Reactive Oxygen Species Are Modulated by Flavonols to Control Stomata Aperture. Plant Physiol. 2017, 175, 1807–1825.
- Vogt, T. Phenylpropanoid Biosynthesis. Mol. Plant 2010, 3, 2–20.
- Sadeghnezhad, E.; Sharifi, M.; Zare-Maivan, H. Profiling of acidic (amino and phenolic acids) and phenylpropanoids production in response to methyl jasmonate-induced oxidative stress in Scrophularia striata suspension cells. Planta 2016, 244, 75–85.
- Binder, S. Branched-Chain Amino Acid Metabolism in Arabidopsis thaliana. Arab. Book 2010, 8, e0137.
- Capaldi, F.R.; Gratão, P.L.; Reis, A.R.; Lima, L.W.; Azevedo, R.A. Sulfur Metabolism and Stress Defense Responses in Plants. Trop. Plant Biol. 2015, 8, 60–73.
- Colovic, M.B.; Vasic, V.M.; Djuric, D.M.; Krstic, D.Z. Sulphur-containing Amino Acids: Protective Role Against Free Radicals and Heavy Metals. Curr. Med. Chem. 2018, 25, 324–335.
- Mukwevho, E.; Ferreira, Z.; Ayeleso, A. Potential Role of Sulfur-Containing Antioxidant Systems in Highly Oxidative Environments. Molecules 2014, 19, 19376–19389.
- Giovanelli, J. Sulfur amino acids of plants: An overview. Methods Enzymol. 1987, 143, 419–426.
- Bick, J.-A.; Setterdahl, A.T.; Knaff, D.B.; Chen, Y.; Pitcher, L.H.; Zilinskas, B.A.; Leustek, T. Regulation of the Plant-type 5‘-Adenylyl Sulfate Reductase by Oxidative Stress. Biochemistry 2001, 40, 9040–9048.
- Queval, G.; Thominet, D.; Vanacker, H.; Miginiac-Maslow, M.; Gakiere, B.; Noctor, G. H2O2-activated up-regulation of glutathione in Arabidopsis involves induction of genes encoding enzymes involved in cysteine synthesis in the chloroplast. Mol. Plant 2009, 2, 344–356.
- Azarakhsh, M.R.; Asrar, Z.; Mansouri, H. Effects of seed and vegetative stage cysteine treatments on oxidative stress response molecules and enzymes in Ocimum basilicum L. under cobalt stress. J. Soil Sci. Plant Nutr. 2015, 15, 651–662.
- Gao, Q.; Zhang, L. Ultraviolet-B-induced oxidative stress and antioxidant defense system responses in ascorbate-deficient vtc1 mutants of Arabidopsis thaliana. J. Plant Physiol. 2008, 165, 138–148.
- Kusano, M.; Tohge, T.; Fukushima, A.; Kobayashi, M.; Hayashi, N.; Otsuki, H.; Kondou, Y.; Goto, H.; Kawashima, M.; Matsuda, F.; et al. Metabolomics reveals comprehensive reprogramming involving two independent metabolic responses of Arabidopsis to UV-B light. Plant J. 2011, 67, 354–369.
- Janse van Rensburg, H.C.; Van den Ende, W. Priming with γ-Aminobutyric Acid against Botrytis cinerea Reshuffles Metabolism and Reactive Oxygen Species: Dissecting Signalling and Metabolism. Antioxidants 2020, 9, 1174.
- Aswani, V.; Rajsheel, P.; Bapatla, R.B.; Sunil, B.; Raghavendra, A.S. Oxidative stress induced in chloroplasts or mitochondria promotes proline accumulation in leaves of pea (Pisum sativum): Another example of chloroplast-mitochondria interactions. Protoplasma 2019, 256, 449–457.
- Signorelli, S.; Imparatta, C.; Rodríguez-Ruiz, M.; Borsani, O.; Corpas, F.J.; Monza, J. In vivo and in vitro approaches demonstrate proline is not directly involved in the protection against superoxide, nitric oxide, nitrogen dioxide and peroxynitrite. Funct. Plant Biol. 2016, 43, 870–879.
- Szabados, L.; Savouré, A. Proline: A multifunctional amino acid. Trends Plant Sci. 2010, 15, 89–97.
- Akashi, K.; Miyake, C.; Yokota, A. Citrulline, a novel compatible solute in drought-tolerant wild watermelon leaves, is an efficient hydroxyl radical scavenger. FEBS Lett. 2001, 508, 438–442.
- Rimando, A.M.; Perkins-Veazie, P.M. Determination of citrulline in watermelon rind. J. Chromatogr. A 2005, 1078, 196–200.
- Kusvuran, S.; Dasgan, H.Y.; Abak, K. Citrulline is an important biochemical indicator in tolerance to saline and drought stresses in melon. Sci. World J. 2013, 253414.
- Yokota, A.; Kawasaki, S.; Iwano, M.; Nakamura, C.; Miyake, C.; Akashi, K. Citrulline and DRIP-1 protein (ArgE homologue) in drought tolerance of wild watermelon. Ann. Bot. 2002, 89, 825–832.





