
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | John Gould | + 2188 word(s) | 2188 | 2021-02-19 08:58:55 | | | |
| 2 | Vicky Zhou | Meta information modification | 2188 | 2021-03-03 05:41:20 | | |
Video Upload Options
This entry provides information regarding the proposed functions of the anuran froth nest as a mucous-based solution to the environmental challenges offspring face during development, with consideration of the functions of froth nest breakdown and communal froth nesting, as well. The adults of several anuran amphibian species deposit their eggs externally in mucus secretions that are purposely aerated to produce a froth nest. This type of clutch structure has evolved independently several times in this group and has been proposed to serve a variety and often simultaneous adaptive functions associated with protecting offspring from sub-optimal conditions during embryogenesis and later stages after hatching has occurred. These functions range from buffering offspring from sub-optimal temperatures and desiccation, to defending against predation and improving oxygenation. This versatility has likely helped facilitate the reduced reliance of egg development on water and thus the penetration of anurans into environments where permanent aquatic systems are not always available.
1. Introduction
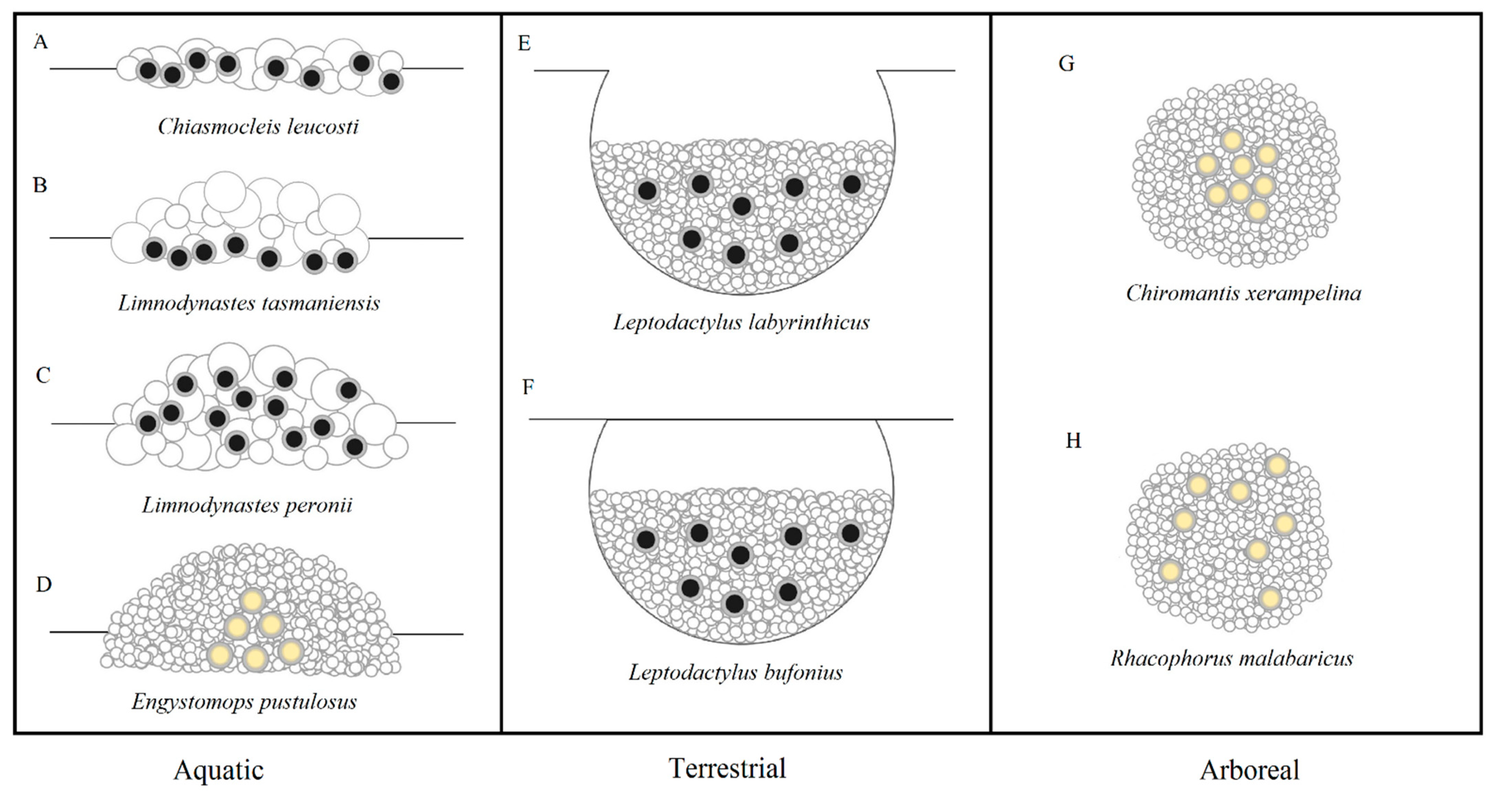
There is a great diversity of environmental settings in which froth nests are built, including aquatically on water surfaces or margins [1][2], terrestrially on moist forest floors or in burrows [3][4], and arboreally attached to plant structures such as branches and leaves [5][6]. The precursor fluid that constitutes the nest is mucus excreted from glands located on the skin of amplectant adult pairs [7], or as cloacal fluid from the female [8] or male [5], which is released prior to egg extrusions and then mixed with water obtained from the surrounding environment in aquatic species [9], or expelled from the bladder in terrestrial/arboreal species [10]. It has been suggested for several species that the froth is actually a combination of both mucus secretions and the outer layer(s) of jelly that surround each egg [11][12]. This mixture of mucus, water and possibly jelly is subsequently aerated, which involves highly synchronised movements by the adult pairs while in inguinal or axillary amplexus, though may include assistance from additional non-amplectant males in some species [13]. This results in the production of nests composed of either foam or bubbles (Figure 1) [14][15][16], bot of which are collectively defined as ‘froth nests’ [15].
2. Benefits of Froth Nesting
Currently, five main adaptive functions of froth nesting have been proposed, including increasing egg temperature, buffering eggs against sub-optimal temperatures, protection against desiccation and predation, and improved oxygenation. These are addressed in detail below.
2.1. Increasing Egg Temperature
Environmental temperature influences a variety of processes in anuran offspring, which has subsequently driven both behavioural and physiological adaptations key for reproduction in thermally stressful environments [17]. Laying eggs in froth may alter the thermal conditions they are exposed to prior to hatching (and following hatching if they remain within the nest), creating an internal environment that accelerates growth.
Empirical evidence for the heating effect of froth nesting has been shown to be contradictory among studies, even for those on the same species. For example, work conducted by Dobkin and Gettinger [18] showed that the internal temperature of Engystomops pustulosus froth nests warmed more rapidly via solar radiation than the surrounding air, exceeding ambient temperatures by as much as 4 °C during the day. However, Downie [19] found no thermal effect of froth nesting in this species across all but later stages of development, alluding to a possible flaw in Dobkin and Gettinger’s [18] study as they only measured temperatures on late stage nests that had already begun to break down and when tadpoles had already started to hatch. For eggs laid aquatically, the heating benefits of frothing are obtained as the nest floats on the surface of the water where eggs can be exposed to direct solar radiation, which likely increases internal temperatures more rapidly compared to eggs laid below the surface [20]. Here, the low albedo of anuran eggs, which are commonly black, may play a critical role in this process of heat absorption [21]. While the froth nest has been suggested to provide a thermally advantageous environment that allows for rapid development [18], experimental evidence of increased developmental rate as a result of the presence of froth is currently lacking.
Countering this proposed heating benefit of froth nesting, it has also been suggested that the high albedo of aerated secretions, which turn white as a result of the incorporation of air bubbles and become reflective as their surfaces dry (Figure 2), protect eggs from insolation, thereby keeping temperatures down [22][23]. Yet, Dobkin and Gettinger [18] showed that the frothed nests of E. pustulosus did not reflect significant amounts of solar radiation, suggesting that the high albedo of the nest does not serve to protect against overheating. Instead, the process of drying has been suggested to produce a partially water-proof barrier that may remain flexible and resistant to cracking [10], thereby preventing evaporative cooling [18][24].

2.2. Insulation against Sub-Optimal Temperatures
Evidence has also been obtained to suggest that the froth nest acts to insulate eggs from sub-optimal or variable environmental temperatures. For example, internal froth nest temperatures have been shown to be lower than that of the nest surface and surrounding air in several species [23][24][25]. Likewise, Méndez-Narváez et al. [26] found internal temperatures of both aquatic and terrestrial froth nests of Leptodactylus species were lower during hotter times of the day and higher at night when temperatures decreased, thereby stabilising internal temperatures in thermally fluctuating environments. As a buffer to the external environment, the froth nests may thus increase the chances of offspring survival by either protecting them from thermal extremes that would cause mortality or damage, or by allowing for faster developmental rates.
2.3. Environmental Control of Froth Nest Temperature
Several of these proposed thermal benefits of froth nesting have opposing effects, suggesting that they are dependent on the conditions of the oviposition site. Indeed, froth nest temperatures are likely to be influenced by their placement in the environment, as this determines the extent of their exposure to external factors such as air and substrate (water or ground) temperatures and solar radiation.
The heating benefits of froth nesting in particular is likely restricted to egg laying environments above ground and in open habitats where direct exposure to solar radiation is possible. For example, increased internal temperatures of E. pustulosus nests were the result of exposure to solar radiation during the day [18], which is in contrast to Lechriodus fletcheri nests which did not show increased daytime temperatures, possibly due to dense canopy cover overhead minimising sunlight exposure (J. Gould, unpubl. data). Nests that are deposited above ground are also more likely to be exposed to greater fluctuations in temperature that may negatively affect embryo development [24][27], particularly in temperate regions where temperatures can decline severely overnight and in temporary aquatic systems which are often poor thermal masses given their smaller size and shallowness [18]. Indeed, the eggs of some Leptodactylus species appear to be more thermally insulated in terrestrially compared to aquatically built nests, which may occur as microhabitat conditions in ground depressions or burrows provide partial or full protection from air temperature changes [26][27]. Finally, the extent of temperature variations may also be attributed to the extent of sunlight exposure. This was demonstrated in nests created by Leptodactylus knudseni and Leptodactylus labyrinthicus, which although of comparative size and placement within the environment, exhibited different temperature fluctuations as a result of differences in oviposition habitat openness [24].
2.4. Anti-Desiccation
Amphibian eggs are highly vulnerable to desiccation if moisture levels begin to drop given that they are surrounded by a highly permeable membrane and lack a protective, non-permeable shell [28]. This is a physiological challenge for species that exploit arid or seasonal environments, as well as temporary aquatic systems if rainfall is intermittent and hydroperiods are frequently below the minimum threshold time required for offspring to complete development [14][19][29]. Laying eggs within a froth nest has been suggested by several authors to offer them protection from the immediate risk of desiccating in the absence of moisture [18][32][30][31]. This may occur in three ways: (1) Drying of the spawn surface via evaporative water loss results in the formation of a reflective and/or non-permeable outer layer that reduces the rate of water loss from inner regions of the nest by deflecting solar radiation and locking in moisture [13][14][18][24][33]. (2) The incorporation of air bubbles results in a relatively larger and structured spawn body that has an insulating effect, reducing the nest’s surface area relative to its volume and placing distance between the residing eggs and external environment [12][34][35][36]. Adults of some species actively insert eggs into the interior of the nest such that few are left near to the surface, adding to this effect [37]. (3) The process of drying may also preserve this three-dimensional structure of the nest, preventing it from expanding and thinning out, which would expose embryos to the environment (Figure 3).

2.5. Predator Defence
Anuran eggs are vulnerable to predation as they are laid externally, immobile and often are not provided parental care [38]. Covering eggs in froth may act as a ‘fixed predator avoidance’ trait [39] that reduces predation risk in a variety of ways: (1) The surface of the froth nest may dry [10][13], creating a hard, shell-like protective barrier that is difficult to penetrate for predators to gain access to the eggs within. (2) If the outer layer of froth has not dried, it may remain highly adhesive (Figure 4), causing predators that make contact with it to become stuck and, therefore, incapacitated before they can reach the eggs [40]. (3) Aeration of the nest causes the eggs to become buried within a comparatively larger and more structured body [37], reducing the number of eggs near the surface. (4) For species that lay aquatically, the ability of froth nests to float causes a large portion of eggs to sit out of the water column, isolating them from aquatic predators [12][19][31]. If the spawn are free-floating and not attached to the edges of the waterbody, they may also form ‘island’ refuges that are difficult for terrestrial predators to reach. Of course, the probability of successful hatching occurring from froth nests is still highly dependent on predation pressure, which is influenced by the density of predators near the nest and the presence of other nests nearby that may relieve this pressure.

2.6. Improved Oxygenation
Similar to all other oviparous species, the anuran egg obtains oxygen directly from the surrounding environment [10]. The conundrum here is that each embryo is surrounded by a jelly capsule that acts as a partial barrier to oxygen uptake, which is exacerbated when eggs are deposited together in globular masses [41][42]. Froth nesting may improve oxygen availability in several ways related to the incorporation of air bubbles: (1) For species that oviposit aquatically, it causes the eggs to float at the surface of the water where oxygen levels are generally higher and can approach the eggs from all directions [43], and where they may become mechanically ventilated by rippling of the water’s surface [44]. (2) Each egg becomes encapsulated by a capacious oxygen store that can be exploited throughout development [10][15][19]. (3) The frothed mucus may facilitate oxygen diffusion within the nest structure, which occurs more rapidly in air than in jelly [10]. (4) The froth evenly distributes the eggs and places distance between them, thereby reducing oxygen competition in a manner similar to the function of thick egg capsules in non-froth nesting species [10][42][44].
References
- Tyler, M.J.; Davies, M. Foam Nest Construction by Australian Leptodactylid Frogs (Amphibia, Anura, Leptodactylidae). J. Herpetol. 1979, 13, 509.
- Bastos, R.P.; Haddad, C.F.; Pombal, J.P., Jr. Foam nest in Scinax rizibilis (Amphibia: Anura: Hylidae). Zoologia 2010, 27, 881–886.
- Silva, W.R.; Giaretta, A.A.; Facure, K.G. On the natural history of the South American pepper frog, Leptodactylus labyrinthicus (Spix, 1824) (Anura: Leptodactylidae). J. Nat. Hist. 2005, 39, 555–566.
- Hero, J.-M.; Galatti, U. Characteristics Distinguishing Leptodactylus pentadactylus and L. knudseni in the Central Amazon Rainforest. J. Herpetol. 1990, 24, 226–228.
- Kadadevaru, G.G.; Kanamadi, R.D. Courtship and nesting behaviour of the Malabar gliding frog, Rhacophorus malabaricus (Jerdon, 1870). Curr. Sci. 2000, 79, 377–380.
- Meegaskumbura, M.; Senevirathne, G.; Biju, S.D.; Garg, S.; Meegaskumbura, S.; Pethiyagoda, R.; Hanken, J.; Schneider, C.J. Patterns of reproductive-mode evolution in Old World tree frogs (Anura, Rhacophoridae). Zoöl. Scr. 2015, 44, 509–522.
- Shahrudin, S.; Ismail, M.N.; Kwan, S.H.; Najimudin, N. Ecology and Protein Composition of Polypedates leucomystax (Gravenhorst, 1829) (Anura: Rhacophoridae) Foam Nests from Peninsular Malaysia. Annu. Res. Rev. Biol. 2017, 14, 1–10.
- Furness, A.I.; McDiarmid, R.W.; Heyer, W.R.; Zug, G.R. Oviduct Modifications in Foam-Nesting Frogs, with Emphasis on the Genus Leptodactylus (Amphibia, Leptodactylidae). S. Am. J. Herpetol. 2010, 5, 13–29.
- Fleming, R.I.; MacKenzie, C.D.; Cooper, A.; Kennedy, M.W. Foam nest components of the túngara frog: A cocktail of proteins conferring physical and biological resilience. Proc. R. Soc. B Biol. Sci. 2009, 276, 1787–1795.
- Seymour, R.S.; Loveridge, J.P. Embryonic and larval respiration in the arboreal foam nests of the African frog Chiromantis xerampelina. J. Exp. Biol. 1994, 197, 31–46.
- Heyer, W.R.; Rand, A.S. Foam Nest Construction in the Leptodactylid Frogs Leptodactylus pentadactylus and Physalaemus pustulosus (Amphibia, Anura, Leptodactylidae). J. Herpetol. 1977, 11, 225–228.
- Ryan, M.J. The túngara Frog: A Study in Sexual Selection and Communication; University of Chicago Press: Chicago, IL, USA, 1985.
- Coe, M. Observations on the ecology and breeding biology of the genus Chiromantis (Amphibia: Rhacophoridae). J. Zoöl. 1974, 172, 13–34.
- Duellman, W.; Trueb, L. Biology of Amphibians; McGrawHill: New York, NY, USA, 1986.
- Altig, R.; McDiarmid, R.W. Morphological Diversity and Evolution of Egg and Clutch Structure in Amphibians. Herpetol. Monogr. 2007, 21, 1–32.
- Frost, D.R. Amphibian Species of the World; Allen Press and Association of Systematics Collections: Lawrence, KS, USA, 1985.
- Wells, K.D. The Ecology and Behavior of Amphibians; University of Chicago Press: Chicagi, IA, USA, 2007.
- Dobkin, D.S.; Gettinger, R.D. Thermal Aspects of Anuran Foam Nests. J. Herpetol. 1985, 19, 271.
- Downie, J. Functions of the foam in the foam-nesting leptodactylid Physalaemus pustulosus. Herpetol. J. 1988, 1, 302–307.
- Hassinger, D.D. Notes on the thermal properties of frog eggs. Herpetologica 1970, 26, 49–51.
- Salthe, S.; Mecham, J. Reproductive and courtship patterns. In Physiology of the Amphibia; Elsevier: Amsterdam, The Netherlands, 1974; Volume 2, pp. 309–521.
- Rand, A. Physalaemus pustulosus (Rana, Sapito Tingara, Foam Toad, Mud-puddle Frog). In Costa Rican Natural History; Janzen, D., Ed.; University of Chicago Press: Chicago, IA, USA, 1983; pp. 412–415.
- Gorzula, S. Foam nesting in leptodactylids: A possible function. Br. J. Herpetoloy 1977, 5, 657–659.
- Shepard, D.B.; Caldwell, J.P. From Foam to Free-living: Ecology of Larval Leptodactylus Labyrinthicus. Copeia 2005, 2005, 803–811.
- Kusano, T.; Sakai, A.; Hatanaka, S. Ecological functions of the foam nests of the Japanese treefrog, Rhacophorus arboreus (Amphibia, Rhacophoridae). Herpetol. J. 2006, 16, 163–169.
- Méndez-Narváez, J.; Flechas, S.V.; Amézquita, A. Foam Nests Provide Context-Dependent Thermal Insulation to Embryos of Three Leptodactylid Frogs. Physiol. Biochem. Zoöl. 2015, 88, 246–253.
- Philibosian, R.; Ruibal, R.; Shoemaker, V.H.; McClanahan, L.L. Nesting behavior and early larval life of the frog Leptodactylus bufonius. Herpetologica 1974, 30, 381–386.
- Touchon, J.C.; Warkentin, K.M. Reproductive mode plasticity: Aquatic and terrestrial oviposition in a treefrog. Proc. Natl. Acad. Sci. USA 2008, 105, 7495–7499.
- Spieler, M.; Linsenmair, K.E. Choice of optimal oviposition sites by Hoplobatrachus occipitalis (Anura: Ranidae) in an unpredictable and patchy environment. Oecologia 1997, 109, 184–199.
- Haddad, C.F.B., Jr.; Pombal, J.P.P.; Gordo, M. Foam Nesting in a Hylid Frog (Amphibia, Anura). J. Herpetol. 1990, 24, 225–226.
- Heyer, W.R. The adaptive ecology of the species groups of the genus Leptodactylus (Amphibia, Leptodactylidae). Evolution 1969, 23, 421–428.
- Hödl, W. Foam nest construction in South American leptodactylid frogs. In Studies in Herpetology; Roek, Z., Ed.; Charles University: Prague, Czech Republic, 1986; pp. 565–570.
- Romero-Carvajal, A.; Sáenz-Ponce, N.; Venegas-Ferrín, M.; Almeida-Reinoso, D.; Lee, C.; Bond, J.; Ryan, M.J.; Wallingford, J.B.; Del Pino, E.M. Embryogenesis and laboratory maintenance of the foam-nesting túngara frogs, genus Engystomops(= Physalaemus). Dev. Dyn. 2009, 238, 1444–1454.
- Liwanag, H.E.M.; Berta, A.; Dos Santos, D.C.; Abney, M.; Williams, T.M. Morphological and thermal properties of mammalian insulation: The evolution of fur for aquatic living. Biol. J. Linn. Soc. 2012, 106, 926–939.
- Bock, W.J. Explanatory history of the origin of feathers. Am. Zool. 2000, 40, 478–485.
- Narváez, A.; Santiago, R. Spawning behaviour of Engystomops pustulatus (Anura: Leptodactylidae). J. Nat. Hist. 2017, 5, 267–275.
- Dalgetty, L.; Kennedy, M.W. Building a home from foam-Túngara frog foam nest architecture and three-phase construction process. Biol. Lett. 2010, 6, 293–296.
- Crump, M.L. Parental Care among the Amphibia. Adv. Study Behav. 1996, 25, 109–144.
- Magnusson, W.E.; Hero, J.-M. Predation and the evolution of complex oviposition behaviour in Amazon rainforest frogs. Oecologia 1991, 86, 310–318.
- Lingnau, R.; Di-Bernardo, M. Predation on foam nests of two leptodactylid frogs by Solenopsis sp. (Hymenoptera, Formicidae) and Liophis miliaris (Serpentes, Colubridae). Biociências 2006, 14, 223–224.
- Seymour, R.S. Respiration of Aquatic and Terrestrial Amphibian Embryos. Am. Zoöl. 1999, 39, 261–270.
- Seymour, R.S. Oxygen diffusion through the jelly capsules of amphibian eggs. Isr. J. Zool. 1994, 40, 493–506.
- Haddad, F.; Hödl, W. New reproductive mode in anurans: Bubble nest in Chiasmocleis leucosticta (Microhylidae). Copeia 1997, 1997, 585–588.
- Seymour, R.S.; Roberts, J.D. Embryonic Respiration and Oxygen Distribution in Foamy and Nonfoamy Egg Masses of the Frog Limnodynastes tasmaniensis. Physiol. Zoöl. 1991, 64, 1322–1340.





