| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Guillermo Bordanaba | + 1467 word(s) | 1467 | 2021-02-08 06:54:05 | | | |
| 2 | Rita Xu | Meta information modification | 1467 | 2021-02-23 08:01:03 | | |
Video Upload Options
The 3D cell cultures allow cells to growth and interact between them and with the extracellular matrix in three dimensions. This conforms a culture structure closer to physiological conditions than the cell monolayers (2D) traditionally employed in cell biology, and it can be achieved by using extracellular matrix hydrogels derived from decellularized tissues, bio-printed scaffolds made of different materials, or by forcing the cells to interact between each other without physical support. 3D culture models provide a powerful tool to understand cell-to-cell interactions when used in co-cultures, and to determine the involvement of extracellular vesicles as major key interactors in cellular crosstalk.
1. Introduction
In recent years, the number of scientific groups dedicated to the study of physiological phenomena using 3D cultures has grown notably, and with it the amount of published information describing the cellular communication mechanisms in this physiological processes[1]. Released by all types of cells, extracellular vesicles (EVs) are an important tool to study cell’s biology and cell-to-cell communication. Cancer research is one of the main fields that can benefit of 3D culturing research and, specifically, in the study of vesicle-mediated cell-to-cell crosstalk in cancer progression [2]. The study of cancer biology had evolved along the last years towards culture models that reflect the biological complexity of tumoral cells and their interactions with the extracellular matrix. The reason is that the traditional bidimensional (2D) cultures differ from tridimensional (3D) cultures in their morphological characteristics, proliferation rate and degree of differentiation, the level of cell-to-cell interaction and cell-to-matrix, as well as their resistance to drugs [3][4]. However, the application of complex culture models to unravel the role of EVs in cancer research has not been yet popularized due to the difficulties that this type of cultures presents, both technically and in terms of cost. Nevertheless, several studies have highlighted the importance of 3D cultures in the study of EVs in cancer research [5][6][7].
For many years, in vitro models have been based on 2D monolayers of immortalized human cancer-derived cell lines. The popularization of 3D culturing has come with the observation that this type of cell cultures often retain heterogeneity. This feature enables the study of tumour evolution. Moreover, 3D models offer advantages over conventional monolayered cell cultures including preservation of the topology and cell-to-matrix interactions [8][9]. On the other hand, 3D cell culturing is also challenging, given the difficulties to stabilize the cultures, and the requirement of specific material to start up and maintain the culture. In Table 1, we present a comparison between 2D and 3D cultures characteristics[1]. In spite of the difficulties, 3D cultures become a great model to study the interplay between cancer and non-cancer cells in order to unveil biological mechanisms involved in cancers initiation and progression [10]. Spheroids are probably the type of 3D culture most commonly used. Spheroid formation methodologies can be divided into two categories: scaffold-based models, either incorporating materials which are components of the matrix (collagen, fibronectin, agarose, laminin, and gelatin) [11], or synthetic materials that provide cell support [12], and scaffold-free models that comprise non-adherent and in suspension cells, which are forced to aggregate and form spheroids [13].
Table 1. Main advantages and limitations of the different cellular models in cancer research [14][15].
|
Model |
Advantages |
Limitations |
|
2D Monolayers |
|
|
|
Gel based 3D Cultures |
|
|
|
Low-attachment plates |
|
|
|
Microfluidic systems |
|
|
One of the first applications of 3D cultures was the study of tumorigenesis. Typically, cells are cultured in a mouse sarcoma-derived gel (i.e., Matrigel®) but there are alternatives such as human leiomyoma discs and their matrix (Myogel). The latter has been commercialized for in vitro assays such as IncuCyte®, spheroid and sandwich assays [16].
3D culture models grown in vitro from cancer stem directly or from primary tissues are a more evolved form of organoids [17]. The usage of primary tissues has an attractive potential for personalized medicine. Organoids display a large number of features and functions of their original organs, such architecture and gene expression, reason why they have a prospective potential for the cancer research but also in other fields. The combination of organoids with the co-culture of multiple cells can mimic the tumour immune microenvironment, including key features like immune checkpoint [18]. Organoids derived from different mouse or human tumours have now been widely adopted to investigate different types of cancer. Moreover, by culturing organoids in proper media conditions, they could serve as a model of several subtypes of diseases [19]. Several other models such as prostate, brain or kidney organoids have been stablished [20].
In addition to organoids, other kinds of 3D cultures have been developed. 3D bioprinting can be defined as a layer-by-layer deposition of biomaterial, such as tissue spheroids, cell pellets, microcarriers, decellularized extracellular matrix, and cell-laden hydrogels, in a well-defined structure to generate viable 3D cultures. In the last decade, the bioprinting technologies have undergone remarkable advancements [21]. Current trends utilizing these scaffold technologies aim at capturing more of the micro-environmental cues than other model systems [22][23]. The scaffolds may act as a surrogate for the missing extracellular microenvironment (ECM), representing the available space of tumour tissue, providing the physical support for cell growth, adhesion, and proliferation, and causing the cells to form an appropriate spatial distribution and cell‑cell or cell‑ECM interaction [3].
A wide range of techniques are utilized to generate different scaffolds, including solvent casting/particulate leaching, freeze‑drying, phase inversion, electrospinning, stereolithography, selective laser sintering, shape deposition manufacturing, 3D printing, robotic microassembly, and fused deposition modelling [24]. Among these techniques, freeze-drying, phase inversion, and fibre electrospinning are utilized most of the times. Typical materials used for tumour cells 3D culture are a laminin-rich basement membrane extract gelatin (for instance Matrigel, Myogel or Cultrex BME) [25], silk fibroin proteins [26], hyaluronic acid [27], collagen [28], or decellularized material [29][30]. Scaffold-based 3D cell culture, using a biological basement membrane, captures many aspects of the spatial cues (cell-to-cell communication, cell-to-matrix adhesion, and physical characteristics) and provides a unique compromise between complexity and practicality [31]. The choice of a biological scaffold is not simply to deliver an anchorage site for cells but also to provide a complex structure enabling communication linked to cell behaviour and function. The formation of 3D structures within the culture also reproduces aspects of the nutrient and oxygen gradients found across in vivo tumours. It should be considered that those 3D scaffolds can be used not only to simulate the microenvironment but alto to assess drug research. Recently publications have showed the ability of decellularized ECM materials to encapsulate and controlled delivery of different drugs such as dexamethasone [32] or doxorubicin [33]. Therefore, 3D scaffold can have drug-carrier functions in therapeutic applications related to testing drugs and in predicting treatment efficacies.
2. 3D cultures and extracellular vesicles
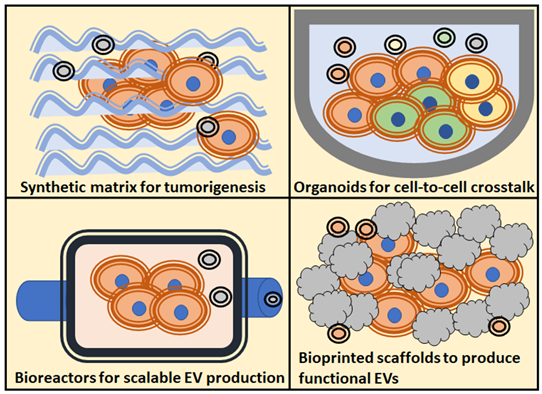
There are different 3D strategies employed to study the different roles of extracellular vesicles in cancer. The choice of a specific model depends on the research question aimed to solve; hence, there are different model options to study of the role of EVs in tumorigenicity processes (Figure 1) [1].
Figure 1. Schematic overview of the most popularized 3D culture techniques, and the main assays regarding extracellular vesicles (EVs) applications to the study of tumours biology, use of EVs as therapeutic agents, study of tumorigenesis and cell-to-cell crosstalk.
Since the first descriptions of EVs and their different types, it has been reported that tumoral cells secrete vesicles. These vesicles participate in the cellular cross-talk with the cellular matrix [34] and cancer cells are rather effective in vesicular-mediated intercellular transfer [35]. Actually, this transfer is a requirement of tumoral cells to stablish a connection with the surrounding matrix and actively regulate processes involved in cancer progression and autocrine/paracrine oncogenesis. Indeed, EVs play an important role in reprogramming stromal cells, modulating the immune system, and promoting angiogenesis (reviewed in [36]). Moreover, the dependency of tumours on vesicular communication also concerns the preparation of an extracellular niche for metastasis [37][38][39].
In either cancer research or EVs field, 3D models have already contributed to gain knowledge in pathogenesis, diagnosis and cell-to-cell communication. As mentioned in the introduction, 3D cultures reflect the in vivo biological complexity and their interactions with the extracellular matrix. They are already an appealing asset to implement in research bottom-up approaches as an intermediate step between monolayer in vitro experiments and in vivo experiments.
References
- Guillermo Bordanaba-Florit; Iratxe Madarieta; Beatriz Olalde; Juan Falcón-Pérez; Félix Royo; 3D Cell Cultures as Prospective Models to Study Extracellular Vesicles in Cancer. Cancers 2021, 13, 307, 10.3390/cancers13020307.
- Katie Meehan; Laura J Vella; The contribution of tumour-derived exosomes to the hallmarks of cancer. Critical Reviews in Clinical Laboratory Sciences 2015, 53, 121-131, 10.3109/10408363.2015.1092496.
- Donglai Lv; Zongtao Hu; Lin Lu; Husheng Lu; Xiuli Xu; Three‑dimensional cell culture: A powerful tool in tumor research and drug discovery (Review). Oncology Letters 2017, 14, 6999-7010, 10.3892/ol.2017.7134.
- Britta Weigelt; Cyrus M. Ghajar; Mina J. Bissell; The need for complex 3D culture models to unravel novel pathways and identify accurate biomarkers in breast cancer. Advanced Drug Delivery Reviews 2014, 69-70, 42-51, 10.1016/j.addr.2014.01.001.
- Takeshi Katsuda; Nobuyoshi Kosaka; Takahiro Ochiya; The roles of extracellular vesicles in cancer biology: Toward the development of novel cancer biomarkers. PROTEOMICS 2013, 14, 412-425, 10.1002/pmic.201300389.
- Carolina F. Ruivo; Bárbara Adem; Miguel Silva; Sónia A. Melo; The Biology of Cancer Exosomes: Insights and New Perspectives. Cancer Research 2017, 77, 6480-6488, 10.1158/0008-5472.can-17-0994.
- Yu-Ling Tai; Chun-Jung Lin; Tsai-Kun Li; Tang-Long Shen; Jer-Tsong Hsieh; Benjamin P C Chen; The role of extracellular vesicles in prostate cancer with clinical applications. Endocrine-Related Cancer 2020, 27, R133-R144, 10.1530/erc-20-0021.
- Anna Birgersdotter; Rickard Sandberg; Ingemar Ernberg; Gene expression perturbation in vitro—A growing case for three-dimensional (3D) culture systems. Seminars in Cancer Biology 2005, 15, 405-412, 10.1016/j.semcancer.2005.06.009.
- Marta Kapałczyńska; Tomasz Kolenda; Weronika Przybyła; Maria Zajączkowska; Anna Teresiak; Violetta Filas; Matthew Ibbs; Renata Bliźniak; Łukasz Łuczewski; Katarzyna Lamperska; et al. 2D and 3D cell cultures – a comparison of different types of cancer cell cultures. Archives of Medical Science 2016, 12, 910-919, 10.5114/aoms.2016.63743.
- Elena Fiorini; Lisa Veghini; Vincenzo Corbo; Modeling Cell Communication in Cancer With Organoids: Making the Complex Simple. Frontiers in Cell and Developmental Biology 2020, 8, 166, 10.3389/fcell.2020.00166.
- Maddaly Ravi; V. Paramesh; S.R. Kaviya; E. Anuradha; F.D. Paul Solomon; 3D Cell Culture Systems: Advantages and Applications. Journal of Cellular Physiology 2014, 230, 16-26, 10.1002/jcp.24683.
- Elsie S. Place; Julian H. George; Charlotte K. Williams; Molly M. Stevens; Synthetic polymer scaffolds for tissue engineering. Chemical Society Reviews 2009, 38, 1139-1151, 10.1039/b811392k.
- Geeta Mehta; Amy Y. Hsiao; MaryLou Ingram; Gary D. Luker; Shuichi Takayama; Opportunities and challenges for use of tumor spheroids as models to test drug delivery and efficacy. Journal of Controlled Release 2012, 164, 192-204, 10.1016/j.jconrel.2012.04.045.
- Sarah-Louise Ryan; Anne-Marie Baird; Gisela Vaz; Aaron J. Urquhart; Mathias Senge; Derek J. Richard; Kenneth J. O'byrne; Anthony M. Davies; Drug Discovery Approaches Utilizing Three-Dimensional Cell Culture. ASSAY and Drug Development Technologies 2016, 14, 19-28, 10.1089/adt.2015.670.
- Pranav Joshi; Moo-Yeal Lee; High Content Imaging (HCI) on Miniaturized Three-Dimensional (3D) Cell Cultures. Biosensors 2015, 5, 768-790, 10.3390/bios5040768.
- Tuula Salo; Mauricio Rocha Dourado; Elias Sundquist; Ehsanul Hoque Apu; Ilkka Alahuhta; Katja Tuomainen; Jenni Vasara; Ahmed Al-Samadi; Organotypic three-dimensional assays based on human leiomyoma–derived matrices. Philosophical Transactions of the Royal Society B: Biological Sciences 2017, 373, 20160482, 10.1098/rstb.2016.0482.
- Mototsugu Eiraku; Yoshiki Sasai; Self-formation of layered neural structures in three-dimensional culture of ES cells. Current Opinion in Neurobiology 2012, 22, 768-777, 10.1016/j.conb.2012.02.005.
- James T. Neal; Xingnan Li; Junjie Zhu; Valeria Giangarra; Caitlin L. Grzeskowiak; Jihang Ju; Iris H. Liu; Shin-Heng Chiou; Ameen A. Salahudeen; Amber R. Smith; et al.Brian C. DeutschLillian LiaoAllison J. ZemekFan ZhaoKasper KarlssonLiora M. SchultzThomas J. MetznerLincoln D. NadauldYuen-Yi TsengSahar AlkhairyCoyin OhPaula KeskulaDaniel Mendoza-VillanuevaFrancisco M. De La VegaPamela L. KunzJoseph C. LiaoJohn T. LeppertJohn B. SunwooChiara SabattiJesse S. BoehmWilliam C. HahnGrace X.Y. ZhengMark M. DavisCalvin J. Kuo Organoid Modeling of the Tumor Immune Microenvironment. Cell 2018, 175, 1972-1988.e16, 10.1016/j.cell.2018.11.021.
- Laura Broutier; Gianmarco Mastrogiovanni; Monique M.A. Verstegen; Hayley E. Francies; Lena Morrill Gavarró; Charles R Bradshaw; George E Allen; Robert Arnes-Benito; Olga Sidorova; Marcia P. Gaspersz; et al.Nikitas GeorgakopoulosBon-Kyoung KooSabine DietmannSusan E. DaviesRaaj K. PraseedomRuby LieshoutJan N. M. IjzermansStephen J WigmoreKourosh Saeb-ParsyMathew J. GarnettLuc J.W. Van Der LaanMeritxell Huch Human primary liver cancer–derived organoid cultures for disease modeling and drug screening. Nature Medicine 2017, 23, 1424-1435, 10.1038/nm.4438.
- Claudia Corrò; Laura Novellasdemunt; Vivian S.W. Li; A brief history of organoids. American Journal of Physiology-Cell Physiology 2020, 319, C151-C165, 10.1152/ajpcell.00120.2020.
- Pallab Datta; Madhuri Dey; Zaman Ataie; Derya Unutmaz; Ibrahim T. Ozbolat; 3D bioprinting for reconstituting the cancer microenvironment. npj Precision Oncology 2020, 4, 1-13, 10.1038/s41698-020-0121-2.
- Emily Burdett; F. Kurtis Kasper; Antonios G. Mikos; Joseph A. Ludwig; Engineering Tumors: A Tissue Engineering Perspective in Cancer Biology. Tissue Engineering Part B: Reviews 2010, 16, 351-359, 10.1089/ten.teb.2009.0676.
- John W. Haycock; 3D Cell Culture: A Review of Current Approaches and Techniques. Methods in Molecular Biology 2010, 695, 1-15, 10.1007/978-1-60761-984-0_1.
- Eleonora Carletti; Antonella Motta; Claudio Migliaresi; Scaffolds for Tissue Engineering and 3D Cell Culture. Methods in Molecular Biology 2010, 695, 17-39, 10.1007/978-1-60761-984-0_2.
- Xiaojun Yan; Lyu Zhou; Zhaozhao Wu; Xun Wang; Xiuyuan Chen; Fan Yang; Yanan Guo; Min Wu; Yuyang Chen; Wenjing Li; et al.Jun WangYanan Du High throughput scaffold-based 3D micro-tumor array for efficient drug screening and chemosensitivity testing. Biomaterials 2019, 198, 167-179, 10.1016/j.biomaterials.2018.05.020.
- Chinmoy Patra; Sarmistha Talukdar; Tatyana Novoyatleva; Siva R. Velagala; Christian Mühlfeld; Banani Kundu; Subhas C. Kundu; Felix B. Engel; Silk protein fibroin from Antheraea mylitta for cardiac tissue engineering. Biomaterials 2012, 33, 2673-2680, 10.1016/j.biomaterials.2011.12.036.
- Sara Pedron; Eftalda Becka; Brendan A.C. Harley; Regulation of glioma cell phenotype in 3D matrices by hyaluronic acid. Biomaterials 2013, 34, 7408-7417, 10.1016/j.biomaterials.2013.06.024.
- Donglai Lv; Shi-Cang Yu; Yi-Fang Ping; Haibo Wu; Xilong Zhao; Huarong Zhang; Youhong Cui; Bing Chen; Xia Zhang; Jianwu Dai; et al.Xiu-Wu BianXiao-Hong Yao A three-dimensional collagen scaffold cell culture system for screening anti-glioma therapeutics. Oncotarget 2016, 7, 56904-56914, 10.18632/oncotarget.10885.
- Lina W. Dunne; Zhao Huang; Weixu Meng; Xuejun Fan; Ningyan Zhang; Qixu Zhang; Zhiqiang An; Human decellularized adipose tissue scaffold as a model for breast cancer cell growth and drug treatments. Biomaterials 2014, 35, 4940-4949, 10.1016/j.biomaterials.2014.03.003.
- Anna T. Stratmann; David Fecher; Gaby Wangorsch; Claudia Göttlich; Thorsten Walles; Heike Walles; Thomas Dandekar; Gudrun Dandekar; Sarah L. Nietzer; Establishment of a human 3D lung cancer model based on a biological tissue matrix combined with a Boolean in silico model. Molecular Oncology 2013, 8, 351-365, 10.1016/j.molonc.2013.11.009.
- Hynda K. Kleinman; George R. Martin; Matrigel: Basement membrane matrix with biological activity. Seminars in Cancer Biology 2005, 15, 378-386, 10.1016/j.semcancer.2005.05.004.
- Xian Xu; Chandran R. Sabanayagam; Daniel A. Harrington; Mary C. Farach-Carson; Xinqiao Jia; A hydrogel-based tumor model for the evaluation of nanoparticle-based cancer therapeutics. Biomaterials 2014, 35, 3319-3330, 10.1016/j.biomaterials.2013.12.080.
- Christopher M. Mahoney; Arta Kelmindi-Doko; Malik J. Snowden; J. Peter Rubin; Kacey G. Marra; Adipose derived delivery vehicle for encapsulated adipogenic factors. Acta Biomaterialia 2017, 58, 26-33, 10.1016/j.actbio.2017.05.046.
- Mary Poupot; Jean-Jacques Fournié; Spontaneous membrane transfer through homotypic synapses between lymphoma cells.. The Journal of Immunology 2003, 171, 2517-2523, 10.4049/jimmunol.171.5.2517.
- Ariel Savina, Michel Vidal, Maria I Colombo; Faculty Opinions recommendation of The exosome pathway in K562 cells is regulated by Rab11.. J Cell Sci. 2002, 115, 2505-2515, 10.3410/f.11414964.12436063.
- Joana Maia; Sergio Caja; Maria Carolina Strano Moraes; Nuno Couto; Bruno Costa-Silva; Exosome-Based Cell-Cell Communication in the Tumor Microenvironment. Frontiers in Cell and Developmental Biology 2018, 6, 18, 10.3389/fcell.2018.00018.
- Yingkuan Shao; Ting Chen; Xi Zheng; Sheng Yang; Kailun Xu; Xuewen Chen; Fei Xu; Lantian Wang; Yanwei Shen; Tingyang Wang; et al.Mengwen ZhangWangxiong HuChenyang YeXiaofang YuJimin ShaoShu Zheng Colorectal cancer-derived small extracellular vesicles establish an inflammatory premetastatic niche in liver metastasis. Carcinogenesis 2018, 39, 1368-1379, 10.1093/carcin/bgy115.
- Miranda Y. Fong; Weiying Zhou; Liang Liu; Aileen Y. Alontaga; Manasa Chandra; Jonathan Ashby; Amy Chow; Sean Timothy Francis O’Connor; Shasha Li; Andrew R. Chin; et al.George SomloMelanie R PalomaresZhuo LiJacob R. TremblayAkihiro TsuyadaGuoqiang SunMichael A. ReidXiwei WuPiotr SwiderskiXiubao RenYanhong ShiMei KongWenwan ZhongYuan ChenShizhen Emily Wang Breast-cancer-secreted miR-122 reprograms glucose metabolism in premetastatic niche to promote metastasis. Nature 2015, 17, 183-194, 10.1038/ncb3094.
- Lucía Robado De Lope; Olwen Leaman Alcíbar; Ana Amor López; Marta Hergueta-Redondo; Héctor Peinado; Tumour–adipose tissue crosstalk: fuelling tumour metastasis by extracellular vesicles. Philosophical Transactions of the Royal Society B: Biological Sciences 2017, 373, 20160485, 10.1098/rstb.2016.0485.