
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kinga Skoracka | + 2771 word(s) | 2771 | 2021-02-15 03:25:37 | | | |
| 2 | Dean Liu | -1059 word(s) | 1712 | 2021-02-19 03:17:08 | | |
Video Upload Options
Iodine deficiency is a global issue and affects around 2 billion people worldwide, with pregnant women as a high-risk group. Iodine-deficiency prevention began in the 20th century and started with global salt iodination programmes, which aimed to improve the iodine intake status globally.
1. Introduction
Iodine is absorbed in the stomach and the small intestine where it is transported via sodium/iodide symporter (NIS) and pendrin to the thyroid gland, and then stored in the follicular cells. Subsequently, in the presence of hydrogen peroxide (H2O2), iodine ions (I−) are oxidised by thyroid peroxidase. Then, the tyrosine residues are iodinated—first, at position 3, which forms monoiodotyrosine (MIT), and then at position 5 to form diiodotyrosine (DIT). MIT and DIT in a coupling reaction form thyroxine (T4) and triiodothyronine (T3) hormones[1][2]. As a trace element, iodine is essential in human nutrition, mostly due to its role in the thyroid hormone synthesis. Moreover, both T4 and T3 are involved in the regulation of metabolic processes in the human body and are responsible for the optimal growth of the central nervous system and brain[3][4]. Iodine also serves as an antioxidant and exhibits the protective effects of inflammatory states and cancer[5]. Iodine deficiency constitutes a global issue, and is reflected by urinary iodine concentration (UIC) < 100 µg/day[3]. It is prevalent in the eastern Mediterranean, Asia, Eastern Europe, and Africa. Additionally, mild iodine deficiency is observed in Australia, Great Britain, and New Zealand, as well as in specific groups, such as vegans or vegetarians[6]. In fact, proper iodine intake is crucial among pregnant women, as iodine deficiency is the leading cause of mental retardation in children[7]. Since 1993, World Health Organization (WHO) and United Nations Children’s Fund (UNICEF) recommend a universal salt iodisation. Currently, around 70% of households in over 120 countries have access to iodised salt—in 1990, only less than 10% had this advantage [8]. Iodine is absorbed in 90% by stomach and duodenum and is mostly delivered from the fortified salt, and other sources such as fish, seafood, dairy, water, eggs, broccoli, peas, or spinach[4][9]. However, it is vital to notice that different concentrations of iodine in food products have been noticed, which is presumably associated with different growth environments[10]. Both the deficiency and excess of iodine can lead to an impaired thyroid gland function and, subsequently, to an impaired functioning of the entire organism (Figure 1)[9]. A diet deficient in iodine can lead to mental retardation, hypothyroidism, congenital anomalies, goitre or low IQ, whereas the iodine excess can result in iodine-induced hyperthyroidism[6]. Therefore, an adequate iodine intake—150 mg/day for adults, 120 mg/day for children, and 250 mg/day for pregnant women—is essential for the proper thyroid gland function[3]. The aim of the study is to evaluate and review diets in search of the best diet to protect against iodine deficiency. Moreover, in this paper we wanted to assess which populations are at risk of iodine deficiency or excess. In order to collect the literature related to the presented topic, the PubMed database (www.pubmed.ncbi.nlm.nih.gov, accessed on 20 January 2021) was explored with reference to the terms “iodine”, “diet”, “deficiency”. Iodine Global Network materials and reports were also analyzed. We have focused on popular diets—for example; paleolithic, gluten free or vegan diets—and investigated if individuals following them may be at risk of iodine deficiency. The risk of iodine deficiency may be increased due to the type of products that are eliminated, for example dairy. However, we also discussed if there are any food substitutes, which could decrease this risk. Further, salt-restrictive and plant-based diets may be associated with iodine deficiency due to reduction or elimination of salt and animal derived foods, respectively. We also mentioned low-salicylate diet, gluten-free diet and parenteral nutrition that are associated with many nutritional deficiencies.
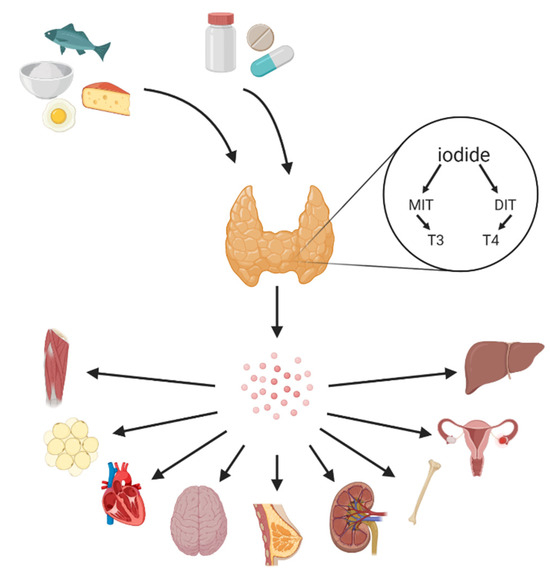
Figure 1. Iodine can be ingested through the diet and dietary supplements. The primary sources of iodine are salt, seafood, fish, algae, milk, and dairy. Iodine is taken up by the thyroid gland and used for synthesis MIT (monoiodotyrosine) and DIT (diiodotyrosine), which are used for thyroid hormone (triiodothyronine and thyroxine) biosynthesis. Receptors of thyroid hormones are on the surface of different organs; thus, thyroid hormones travel in the blood and may affect many systems among other reproductive system, skeletal or muscular system.
2. Iodine Deficiency among Various Populations
According to the WHO, iodine deficiency is defined by a median urinary iodine concentration < 100 µg/day[11]. On the basis of the Iodine Global Network report from 2019, which included school-age children, 115 countries were classified as countries with the optimal level of iodine supply (the United States of America, Canada, France, Portugal, Great Britain) and in 23 countries the iodine intake was too low—Burkina Faso, Burundi, Cambodia, Finland, Germany, Israel, Iraq, Lebanon, Madagascar, Morocco, Haiti, North Korea, Mozambique, Nicaragua, Samoa, Tajikistan, Vanuatu, Norway, Russia, South Sudan, Sudan, and Vietnam. In Angola and Italy, iodine intake was considered to be sufficient (Figure 2)[12]. As it has already been mentioned, around 2 billion are at risk of iodine deficiency, according to the WHO[13]. However, it should be noted that the report was based on the UIC results of school-age children, and thus, it cannot reflect the status of iodine, for instance, among pregnant women. Moreover, the data regarding the status of iodine in different populations remain insufficient.[14]. Most of the analyzed articles demonstrate an emerging iodine deficiency in the population of women of childbearing age, which indicates a public health issue concerning the improvement of iodine status of the abovementioned group of women[15][16]. Gizak et al. emphasised that an insufficient intake of iodine among pregnant women persists, since iodine intake among pregnant women is insufficient in 39 out of 72 countries [17].
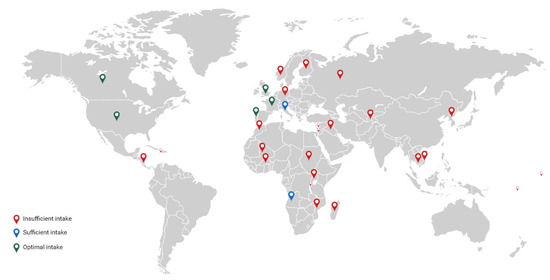
Figure 2. Insufficient iodine intake is observed in many regions, mainly African and Asian countries. Optimal intake of iodine occurs among other in the United States, Canada, and some countries in Europe. However, intake of iodine is not adequate in many European regions.
Pregnant and breastfeeding women are particularly exposed to iodine deficiency, and, therefore, iodination of salt could not be sufficient, and additional supplementation should be considered[18].
3. Health Consequences of Iodine Deficiency
Health consequences of iodine deficiency can affect all ages—adults, adolescents, children, infants and foetuses. Similarly, goitre formation and susceptibility to the nuclear radiation affect all age groups. In terms of adults, iodine deficiency has been associated with an impaired mental function, reduced work output, goitre and hypothyroidism. In children and adolescents, on the other hand, it has been linked to mental and growth retardation. Moreover, an increase in the incidence of papillary thyroid cancer (PTC) compared to follicular thyroid cancer (FTC) has been observed, amounting to 0.19 to 1.7, respectively[19]. It is now believed that the introduction of the iodine supplementation in subjects with a significant iodine deficiency may lead to an increase in the PTC/FTC ratio[20][21]. The transition from FTC to PTC may be associated with an elevated rate of the V600E BRAF (B-Raf serine/threonine kinases) mutation over time[22].
4. Iodine Deficiency and Breast Cancer
The cause–effect relationship between iodine deficiency, impaired function of the thyroid gland, and breast cancer has been observed for over 100 years[23][24][25]. Iodine is absorbed via sodium/iodide symporter in the glandular breast tissue, and its role is to promote the growth of the normal tissue[26]. In fact, research studies have demonstrated the role of iodine as an antioxidant agent in the mammary gland[27][28]. Thus, it has been suggested that iodine deficiency can increase the risk of breast cancer in two mechanisms. The first is associated with direct influence on the glandular breast tissue, which could be explained by means of an increased sensitivity to oestradiol in the case of iodine deficiency. The other is associated with an impaired thyroid gland function and, therefore, hypothyroidism as the consequence of iodine deficiency[26]. Numerous studies demonstrate the association between hypothyroidism, especially autoimmune, and breast cancer; however, this relationship remains controversial [24][29][30][31][32][33][34]. Significantly, an excessive intake of iodine, by stimulating activity of ER-α, negatively affects the risk of breast cancer[35]. Simultaneously, an impaired thyroid gland function can result in a slower tumour growth and in challenging the diagnosis with an increased tumour invasiveness[26]. In Japan, almost 3-times lower incidence of breast cancer was observed, when compared with the USA; nevertheless, the incidence of breast cancer among Japanese women living in the USA was similar to other women. It suggests that the iodine-rich diet could be preventive in the development of breast cancer. Therefore, diets comprising a low amount of iodine could be associated with an increased risk of breast cancer development, which emphasises the need to decrease iodine deficiency in all age groups.
5. Iodine Deficiency in Pregnant Women
The adequate level regarding the requirements of iodine is particularly important in the population of women in the childbearing age. It should be noted that not only severe maternal iodine deficiency, but also mild to moderate deficiencies, have been associated with the consequences appearing in the offspring.
Levie et al. conducted a meta-analysis with an individual participant data from three prospective population-based cohorts in order to investigate the relationship between the maternal iodine status during pregnancy and the infant IQ in 6180 mother-child pairs from three countries. The aim of the study was to identify the sensitive time windows of exposure to the suboptimal iodine availability. The study demonstrated that mild to moderate iodine deficiencies were associated with a reduction in verbal IQ scores of children, especially in the first trimester of pregnancy[36].
Additionally, the results obtained in the Avon Longitudinal Study of Parents and Children indicate that there is an inverse correlation between the low maternal iodine status in the first trimester and the offspring IQ scores at the age of eight, as well as reading accuracy, comprehension, and reading score at the age of nine years. The study comprised 1040 pregnant women and their children in the UK[37].
In the study by Gietka-Czernel et al.[38], which involved 100 pregnant women between the 5th and 38th week of pregnancy with normal thyroid function, only 35% of the subjects took iodine supplements, and 59% were on a diet rich in iodine carriers. Moreover, the presence of goitre was found in 28% of the pregnant women. In fact, iodine deficiency can also lead to a spontaneous abortion, stillbirth, birth cretinism, congenital disabilities, maldevelopment of the foetal brain and an increased risk of perinatal mortality[39][40].
References
- Ingbar, S.H. Autoregulation of the Thyroid. Response to Iodide Excess and Depletion. Mayo Clin. Proc. 1972, 47, 814–823.
- Pisarev, M.A. Thyroid Autoregulation. J. Endocrinol. Investig. 1985, 8, 475–484.
- O’Kane, S.M.; Mulhern, M.S.; Pourshahidi, L.K.; Strain, J.J.; Yeates, A.J. Micronutrients, Iodine Status and Concentrations of Thyroid Hormones: A Systematic Review. Nutr. Rev. 2018, 76, 418–431.
- Pironi, L.; Guidetti, M.; Agostini, F. Iodine Status in Intestinal Failure in Adults. Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 582–587.
- Zbigniew, S. Iodine Prophylaxis in the Lights of the Last Recommendation of WHO on Reduction of Daily Salt Intake. Recent Pat. Endocr. Metab. Immune Drug Discov. 2017, 11, 39–42.
- Santos, J.A.R.; Christoforou, A.; Trieu, K.; McKenzie, B.L.; Downs, S.; Billot, L.; Webster, J.; Li, M. Iodine Fortification of Foods and Condiments, Other than Salt, for Preventing Iodine Deficiency Disorders. Cochrane Database Syst. Rev. 2019, 2, CD010734.
- Angermayr, L.; Clar, C. Iodine Supplementation for Preventing Iodine Deficiency Disorders in Children. Cochrane Database Syst. Rev. 2004, CD003819.
- Pearce, E.N.; Andersson, M.; Zimmermann, M.B. Global Iodine Nutrition: Where Do We Stand in 2013? Thyroid 2013, 23, 523–528.
- Carlsen, M.H.; Andersen, L.F.; Dahl, L.; Norberg, N.; Hjartåker, A. New Iodine Food Composition Database and Updated Calculations of Iodine Intake among Norwegians. Nutrients 2018, 10, 930.
- Pyka, B.; Zieleń-Zynek, I.; Kowalska, J.; Ziółkowski, G.; Hudzik, B.; Gąsior, M.; Zubelewicz-Szkodzińska, B. Iodine Dietary Recommendations- in Search of a Consensus between Cardiologists and Endocrinologists. Folia Cardiol. 2019, 14, 156–160.
- Pearce, E.N.; Lazarus, J.H.; Moreno-Reyes, R.; Zimmermann, M.B. Consequences of Iodine Deficiency and Excess in Pregnant Women: An Overview of Current Knowns and Unknowns. Am. J. Clin. Nutr. 2016, 104 (Suppl. 3), 918S–923S.
- The Iodine Global Network. Global Scorecard of Iodine Nutrition in 2020 in the General Population Based on School-Age Children (SAC); IGN: Ottawa, ON, Canada, 2020.
- De Benoist, B.; McLean, E.; Andersson, M.; Rogers, L. Iodine Deficiency in 2007: Global Progress since 2003. Food Nutr. Bull. 2008.
- Wong, E.M.; Sullivan, K.M.; Perrine, C.G.; Rogers, L.M.; Peña-Rosas, J.P. Comparison of Median Urinary Iodine Concentration as an Indicator of Iodine Status among Pregnant Women, School-Age Children, and Nonpregnant Women. Food Nutr. Bull. 2011, 32, 206–212.
- Panth, P.; Guerin, G.; DiMarco, N.M. A Review of Iodine Status of Women of Reproductive Age in the USA. Biol. Trace Elem. Res. 2019, 188, 208–220.
- Adalsteinsdottir, S.; Tryggvadottir, E.A.; Hrolfsdottir, L.; Halldorsson, T.I.; Birgisdottir, B.E.; Hreidarsdottir, I.T.; Hardardottir, H.; Arohonka, P.; Erlund, I.; Gunnarsdottir, I. Insufficient Iodine Status in Pregnant Women as a Consequence of Dietary Changes. Food Nutr. Res. 2020, 64.
- Gizak, M.; Rogers, L.; Gorstein, J.; Zimmermann, M.; Andersson, M. Global Iodine Status in School-Age Children, Women of Reproductive Age, and Pregnant Women in 2017. In Proceedings of the Presented as a poster at Nutrition 2018, the American Society for Nutrition Annual Conference, Boston, MA, USA, 9–12 June 2018.
- Gietka-Czernel, M. Iodine Prophylaxis. Postępy Nauk Med. 2015, XXVIII, 839–845.
- Williams, E.D. Dietary iodide and thyroid cancer. In Thyroid Disorders Associated with Iodine Deficiency and Excess; Hall, R., Köbberling, J., Eds.; Raven Press: New York, NY, USA, 1985; pp. 201–207.
- Dijkstra, B.; Prichard, R.S.; Lee, A.; Kelly, L.M.; Smyth, P.P.A.; Crotty, T.; McDermott, E.W.; Hill, A.D.K.; O’Higgins, N. Changing Patterns of Thyroid Carcinoma. Ir. J. Med. Sci 2007, 176, 87–90.
- Harach, H.R.; Escalante, D.A.; Onativia, A.; Lederer Outes, J.; Saravia Day, E.; Williams, E.D. Thyroid Carcinoma and Thyroiditis in an Endemic Goitre Region before and after Iodine Prophylaxis. Acta Endocrinol. 1985, 108, 55–60.
- Mathur, A.; Moses, W.; Rahbari, R.; Khanafshar, E.; Duh, Q.-Y.; Clark, O.; Kebebew, E. Higher Rate of BRAF Mutation in Papillary Thyroid Cancer over Time: A Single-Institution Study. Cancer 2011, 117, 4390–4395.
- Eskin, B.A. Iodine Metabolism and Breast Cancer. Trans. N. Y. Acad. Sci. 1970, 32, 911–947.
- Stadel, B.V. Dietary Iodine and Risk of Breast, Endometrial, and Ovarian Cancer. Lancet 1976, 1, 890–891.
- Malya, F.U.; Kadioglu, H.; Hasbahceci, M.; Dolay, K.; Guzel, M.; Ersoy, Y.E. The Correlation between Breast Cancer and Urinary Iodine Excretion Levels. J. Int. Med. Res. 2018, 46, 687–692.
- Rappaport, J. Changes in Dietary Iodine Explains Increasing Incidence of Breast Cancer with Distant Involvement in Young Women. J. Cancer 2017, 8, 174–177.
- Venturi, S. Is There a Role for Iodine in Breast Diseases? Breast 2001, 10, 379–382.
- Smyth, P.P.A. Role of Iodine in Antioxidant Defence in Thyroid and Breast Disease. Biofactors 2003, 19, 121–130.
- Chiappa, C.; Rovera, F.; Rausei, S.; Del Ferraro, S.; Fachinetti, A.; Lavazza, M.; Marchionini, V.; Arlant, V.; Tanda, M.L.; Piantanida, E.; et al. Breast Cancer and Thyroid Diseases: Analysis of 867 Consecutive Cases. J. Endocrinol. Investig. 2017, 40, 179–184.
- Kim, E.Y.; Chang, Y.; Lee, K.H.; Yun, J.-S.; Park, Y.L.; Park, C.H.; Ahn, J.; Shin, H.; Ryu, S. Serum Concentration of Thyroid Hormones in Abnormal and Euthyroid Ranges and Breast Cancer Risk: A Cohort Study. Int. J. Cancer 2019, 145, 3257–3266.
- Adamopoulos, D.A.; Kapolla, N.; Michalakis, A.; Vassilaros, S.; Papadiamantis, J.; Georgiakodis, F. Thyroid Disease in Patients with Benign and Malignant Mastopathy. Cancer 1986, 57, 125–128.
- Turken, O.; NarIn, Y.; DemIrbas, S.; Onde, M.E.; Sayan, O.; KandemIr, E.G.; YaylacI, M.; Ozturk, A. Breast Cancer in Association with Thyroid Disorders. Breast Cancer Res. 2003, 5, R110–R113.
- Gogas, J.; Kouskos, E.; Tseleni-Balafouta, S.; Markopoulos, C.; Revenas, K.; Gogas, G.; Kostakis, A. Autoimmune Thyroid Disease in Women with Breast Carcinoma. Eur. J. Surg. Oncol. 2001, 27, 626–630.
- Smyth, P.P. The Thyroid, Iodine and Breast Cancer. Breast Cancer Res. 2003, 5, 235–238.
- He, S.; Wang, B.; Lu, X.; Miao, S.; Yang, F.; Zava, T.; Ding, Q.; Zhang, S.; Liu, J.; Zava, D.; et al. Iodine Stimulates Estrogen Receptor Singling and Its Systemic Level Is Increased in Surgical Patients Due to Topical Absorption. Oncotarget 2018, 9, 375–384.
- Levie, D.; Korevaar, T.I.M.; Bath, S.C.; Murcia, M.; Dineva, M.; Llop, S.; Espada, M.; van Herwaarden, A.E.; de Rijke, Y.B.; Ibarluzea, J.M.; et al. Association of Maternal Iodine Status With Child IQ: A Meta-Analysis of Individual Participant Data. J. Clin. Endocrinol. Metab. 2019, 104, 5957–5967.
- Bath, S.C.; Steer, C.D.; Golding, J.; Emmett, P.; Rayman, M.P. Effect of Inadequate Iodine Status in UK Pregnant Women on Cognitive Outcomes in Their Children: Results from the Avon Longitudinal Study of Parents and Children (ALSPAC). Lancet 2013, 382, 331–337.
- Gietka-Czernel, M.; Dębska, M.; Kretowicz, P.; Jastrzębska, H.; Kondracka, A.; Snochowska, H.; Ołtarzewski, M. Iodine Status of Pregnant Women from Central Poland Ten Years after Introduction of Iodine Prophylaxis Programme. Endokrynol. Pol. 2010, 61, 646–651.
- Hetzel, B.S. Iodine Deficiency Disorders (IDD) and Their Eradication. Lancet 1983, 2, 1126–1129.
- Zimmermann, M.B.; Jooste, P.L.; Pandav, C.S. Iodine-Deficiency Disorders. Lancet 2008, 372, 1251–1262.





