
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Wietske Luining | + 2443 word(s) | 2443 | 2021-01-20 04:55:31 | | | |
| 2 | Vivi Li | Meta information modification | 2443 | 2021-01-26 10:37:58 | | |
Video Upload Options
Accurate staging of prostate cancer (PCa) at initial diagnosis and at biochemical recurrence is important to determine prognosis and the optimal treatment strategy. To date, treatment of metastatic PCa has mostly been based on the results of conventional imaging with abdominopelvic computed tomography (CT) and bone scintigraphy. However, these investigations have limited sensitivity and specificity which impairs their ability to accurately identify and quantify the true extent of active disease. Modern imaging modalities, such as those based on the detection of radioactively labeled tracers with combined positron emission tomography/computed tomography (PET/CT) scanning have been developed specifically for the detection of PCa. Novel radiotracers include 18F-sodium fluoride (NaF), 11C-/18F-fluorocholine (FCH), 18F-fluordihydrotestosterone (FDHT), 68Gallium and 18F-radiolabeled prostate-specific membrane antigen (e.g., 68Ga-PSMA-11, 18F-DCFPyL). PET/CT with these tracers outperforms conventional imaging. As a result of this, although their impact on outcome needs to be better defined in appropriate clinical trials, techniques like prostate-specific membrane antigen (PSMA) PET/CT have been rapidly adopted into clinical practice for (re)staging PCa.
1. Introduction
Prostate cancer (PCa) is the second-most commonly diagnosed cancer in men worldwide, and has the highest incidence of all cancers among men in the Western world. In 2018, there were an estimated 1.3 million new cases and 359,000 deaths from PCa globally [1][2][3]. The behavior of PCa varies widely, from indolent to highly aggressive. In routine practice, initial clinical suspicion of PCa is usually triggered by an elevated prostate-specific antigen (PSA) and/or an abnormal digital rectal examination (DRE). For a definitive diagnosis, histopathological confirmation is required, and typically obtained by transrectal ultrasound (TRUS) guided needle biopsies [4]. PCa has been classified into five prognostically distinct Grade Groups (GGs) by the International Society of Urological Pathology (ISUP), based on the Gleason Score (GS) [5].
The European Association of Urology (EAU) risk classification [4] (based on the D’Amico classification including initial PSA-value, clinical T-stage and biopsy GG [6]) is commonly used as a prognostic parameter to predict the risk of recurrence, dividing patients into three categories (low, intermediate and high-risk). Patients with high-risk, locally-advanced PCa have an increased risk for the development of metastases, and disease recurrence [4]. The most frequent sites of distant metastases are lymph nodes outside the pelvis (M1a) and bone (M1b) with occasional metastases elsewhere (e.g., visceral organs) (M1c).
Accurate staging of PCa, both at initial diagnosis and at biochemical recurrence (BCR) after previous curative-intent therapy, is important to determine prognosis, and for selecting the optimal treatment strategy. According to the current EAU guidelines, metastatic screening by means of “at least” an abdominopelvic computed tomography (CT)-scan and bone scintigraphy (BS) (99mTc-phosphonate), is recommended in patients with intermediate or high-risk PCa to evaluate the extent of extra-prostatic disease [4]. However, these conventional imaging modalities have limited sensitivity and specificity, affecting their ability to accurately quantify the true extent of disease, especially at low PSA-levels or in the setting of limited volume, oligometastatic disease. This has led to an ongoing search for better imaging tests. As a result of this, prostate-specific membrane antigen (PSMA) positron emission tomography (PET/CT), has been recently introduced. This modern imaging technique using a novel radiotracer has shown high-levels of diagnostic accuracy in the detection of metastatic disease [7][8][9][10], and outperforms conventional imaging in primary staging of PCa [8].
2. Modern Nuclear Imaging in Prostate Cancer: New PET/CT Radiotracers
The three studies described above were all based on the use of conventional imaging (i.e., BS, CT and/or MRI), upon which the current treatment of metastatic PCa is mainly based. However, PCa imaging is evolving rapidly. Over the last few years, modern PET/CT imaging techniques, using radioactively labelled tracers have been introduced to the diagnostic armamentarium. In clinical practice it is preferable to use PET radiopharmaceuticals with high tumor-specific uptake and low background activity, capable of diagnosing bone, lymph node, and visceral metastases. Various tracers have been developed for metastatic PCa, based on osteoblastic activity (18F-sodium fluoride (NaF)), cellular phospholipid membrane proliferation (11C-/18F-fluorocholine (FCH)), androgen receptor expression (18F-fluordihydrotestosterone (FDHT)) and targeting the prostate-specific membrane antigen (68Gallium (68Ga) or 18Flourine (18F)) [11] (Table 1).
Table 1. Diagnostic performance of selected imaging methods for the detection of prostate cancer bone metastases.
| Sensitivity (%) | Specificity (%) | Reference | Type of Article | |
|---|---|---|---|---|
| Bone scintigraphy | 79 | 82 | Shen [12] | Meta-analysis |
| CT | 8.8 | 98 | Gabriele [13] | Retrospective cohort |
| 18F-NaF PET/CT | 98 | 90 | Sheikhbahaei [14] | Meta-analysis |
| 18F-FDHT PET/CT | 63 | - | Dehdashti [15] | Prospective cohort |
| 18F-FCH PET/CT | 87 | 97 | Shen [12] | Meta-analysis |
| 68Ga-PSMA PET/CT | 77 | 97 | Perera [9] | Systematic review and meta-analysis |
| 18F-DCFPyL PET/CT | - | - | - | - |
CT: computed tomography; PET: positron emission tomography; 18F: 18Flourine; NaF: sodium fluoride; FDHT: fluorodihydrotestosterone; FCH: fluorocholine; 68Ga-PSMA: 68Gallium prostate-specific membrane antigen; DCFPyL: (2-(3-{1-carboxy-5-[(6-18F-fluoro-pyridine-3-carbonyl)-amino]-pentyl}-ureido)-pentanedioic acid).
The 18F-NaF PET/CT enables accurate detection of osseous metastases, but is nonspecific for lymph node metastatic disease and is therefore not suitable for comprehensive staging of metastatic PCa. Uptake of 18F-NaF is determined by osteoblastic activity as it attaches to sites of new bone formation [11]. A recent meta-analysis showed a pooled sensitivity and specificity of 18F-NaF PET/CT for the detection of bone metastases on a per patient basis of 98% (95%CI 95–99) and 90% (95%CI 86–93), and on per a lesion basis of 97% (95%CI 95–98) and 84% (95%CI 81–87), respectively. The diagnostic performance of 18F-NaF PET/CT is superior compared to BS [14]. However, in patients with newly diagnosed PCa scheduled for radical prostatectomy, no added value of 18F-NaF PET/CT was found for the detection of bone metastases in case of a negative BS [16]. The advantages of 18F-NaF PET/CT include: superior image quality, due to a higher bone uptake and faster blood clearance, and superior spatial resolution, with better definition of bone metastases, thus contributing to a higher diagnostic accuracy. However, BS has advantages over 18F-NaF PET/CT in terms of cost-effectiveness and availability, and therefore it remains the preferred technique for generalized use.
The relatively new oncological tracer, 18F-FDHT, is a radiolabeled analogue of dihydrotestosterone, directly binding to the androgen receptor (AR). It allows in-vivo visualization and quantification of AR expression [17][18]. The AR is crucial for PCa growth, and essential for AR-directed therapies in metastatic CRPC. 18F-FDHT PET/CT was successfully used in early phase clinical trials to demonstrate AR specific drug binding [19][20]. Larson et al. studied 18F-FDHT PET uptake in seven patients with progressive clinically metastatic PCa. Conventional imaging identified 59 lesions, and 78% of the lesions (46 of 59 lesions) were 18F-FDHT positive [21]. Dehdashti et al. [15] enrolled 19 patients with advanced PCa, with biopsy and/or radiologically proven metastatic disease, and found a sensitivity for 18F-FDHT PET of 63%. This finding suggests that 18F-FDHT PET/CT seems to be a promising predictive biomarker in the evaluation of AR status, and for treatment response assessment, rather than for the primary detection of PCa metastases. Further investigation is needed and it has not yet entered routine clinical use.
PCa cells are known for their increased proliferation and upregulation of choline kinase. Choline is a precursor for the biosynthesis of phosphatidylcholine which is a key component of cell membrane proliferation. This amino acid can be targeted with 11C- or 18F, resulting in radio-labeled choline. These radiotracers are extensively used in PCa, particularly in the setting of BCR, and as potential biomarkers of response after chemotherapy [22]. Shen et al. [12] found, on a per patient analysis, a pooled sensitivity and specificity for the detection of bone metastases using choline PET/CT in patients with PCa of 87% and 97%, respectively. Radiolabeled choline PET/CT has been shown to have a pooled sensitivity and specificity in recurrent disease for all sites (prostate, lymph nodes, bone) of 85.6% and 92.6%, respectively [23]. A limitation of 18F-Choline is the low sensitivity for the detection of PCa metastases (bone and lymph node) at low PSA-values, where it is clearly outperformed by radiolabeled-PSMA [24][25].
PSMA-PET/CT is a novel imaging technique increasingly used in routine practice. PSMA is a class II cell-surface transmembrane protein overexpressed in malignant prostatic epithelial cells, making it an excellent target for imaging. The degree of PSMA-expression is correlated with higher tumor grades, and higher risk of disease progression, leading to it being described as a marker of disease aggressiveness [26][27]. In a recent study of 90 patients with biopsy proven primary PCa, PSA-value and GG correlated with the intensity of tracer expression on 68Ga-PSMA-11 PET/CT, with a significantly higher tumor-related tracer uptake seen in patients with either PSA ≥ 10 ng/mL or GG ≥ 4 [28].
68Ga-labeled PSMA tracers are the most intensively studied, demonstrating high detection rates for both bone and lymph node metastases [7][9][29]. Perera et al. [9] evaluated the diagnostic accuracy of 68Ga-PSMA-11 PET/CT for the detection of metastatic disease in both primary (high-risk and advanced prostate cancer) and secondary staging (at BCR) (Figure 1). The lesion-based analysis showed a pooled sensitivity of 75%, and specificity of 99% for primary staging, with corresponding figures of 77% and 97%, respectively, for a per patient analysis. The positivity rate of the 68Ga-PSMA PET/CT in secondary staging increased in patients with higher PSA-levels: 33% (PSA < 0.2 ng/mL), 45% (0.2–0.49 ng/mL), 59% (0.5–0.99 ng/mL), 75% (1–1.99 ng/mL), and 95% (≥2 ng/mL). These percentages are substantially higher than those for conventional imaging techniques.
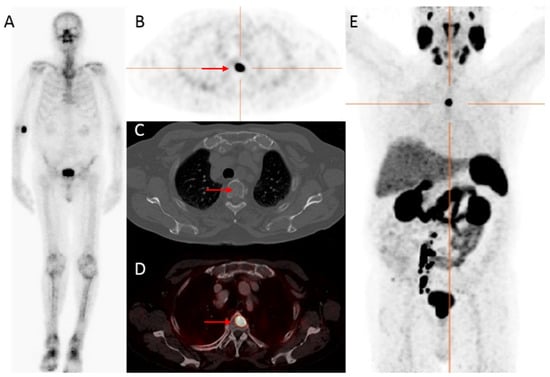
Figure 1. An 83-year-old patient, with castration-resistant prostate cancer (CRPC) after initial treatment with hormonal therapy (2009), and secondary abiraterone (2019), showed improved detection of bone metastatic prostate cancer PCa with 68Ga-PSMA PET/CT compared to bone scintigraphy. The prostate—specific antigen (PSA)—value at PET scanning was 25.9 ng/mL. On the bone scintigraphy, no suspect bone metastases were visualized (A). Transversal 68Ga-PSMA PET (B), fused PET/CT (D) and maximum intensity projection (MIP) (E) revealed a lesion located in the thoracic spine with increased PSMA expression (red arrow), with no evident substrate on CT (C). Time interval between bone scintigraphy and 68Ga-PSMA PET/CT was 5 weeks.
PSMA PET/CT has shown high detection rates (98–100%) for the primary prostate tumor [30][31][32], and provides more sensitive screening for metastatic disease at initial staging than conventional imaging modalities [8]. A recent meta-analysis confirmed the higher diagnostic accuracy of 68Ga-PSMA PET/CT compared to BS with higher sensitivity (0.97 versus 0.86) and specificity (1.00 versus 0.95) for detecting bone metastases [10]. Hofman et al. [8] prospectively compared 68Ga-PSMA PET/CT with conventional imaging in patients with high-risk PCa (n = 302). 68Ga-PSMA PET/CT showed an enhanced diagnostic accuracy for identifying either pelvic nodal or distant metastases compared to conventional imaging (p < 0.0001). In subgroup analysis, 68Ga-PSMA PET/CT was superior in detecting pelvic lymph node metastases (91% versus 59%), and distant metastases (95% versus 74%). First-line 68Ga-PSMA PET/CT (n = 148) found abdominal lymph node metastases in 13 patients (9%), bone metastases in 15 patients (10.1%), and visceral metastases in one (1%). In the primary staging of PCa, conventional imaging and 68Ga-PSMA PET/CT led to a change in treatment approach in 23 (15%) and 41 patients (28%), respectively (p = 0.008).
Next to the intensively studied 68Ga-labeled PSMA tracers, 18F-labeled tracers, such as 18F-DCFPyL [33] and 18F-PSMA-1007 [34], appear promising. 18F-labeled tracers are attractive due to a shorter positron range and higher positron yield compared to 68Ga, providing higher resolution PET-images which may improve early detection of small metastases [29], and 18F-DCFPyL has shown higher tumor to background ratios compared to 68Ga-PSMA [35]. For 18F-DCFPyL, initial experience with the detection of bone metastases in primary prostate cancer has been published (Figure 2), but the diagnostic accuracy of 18F-DCFPyL PET/CT for detecting pelvic lymph nodes metastases in initial staging is less well described. Wondergem et al. [36] enrolled 160 patients with high-risk PCa who underwent an 18F-DCFPyL PET/CT for primary staging. PSMA-positive bone metastases were detected in 49 patients (31%) and lymph nodes were found in 81 patients (51%) of which 52% (n = 42) were enlarged on CT-scan. The treatment plan was adjusted after 18F-DCFPyL in 17% (n = 27) of the patients. Jansen et al. [37] prospectively analyzed 117 patients who underwent imaging prior to robot-assisted radical prostatectomy and extended pelvic lymph node dissection, in order to determine the diagnostic accuracy of 18F-DCFPyL PET/CT for pelvic lymph node staging in intermediate- (36.8%) and high-risk (63.3%) PCa. Histological lymph node metastases were found in 17/117 (14.5%) patients, of whom seven had a suspicious PET/CT, resulting in a limited sensitivity (41.2%), but high specificity (94.0%). The low sensitivity is explained by the median tumor size of 1.5 mm for PET/CT undetected lymph node metastases and illustrates the “resolution” challenges faced by the newly developed imaging techniques.
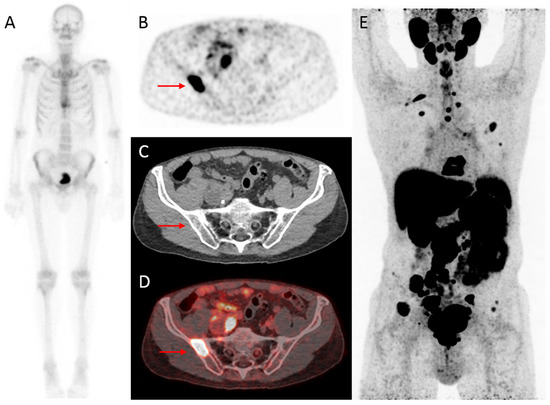
Figure 2. Initial assessment of a 75-year old patient, newly diagnosed with PCa (Grade Group (GG) 5), with an initial PSA-value of 1396 ng/mL. On bone scintigraphy, the increased uptake in the thoracic spine was attributed to an (osteoporotic) collapsed vertebra, and the faint uptake in the left third rib to a post-traumatic origin. Despite the high PSA-value, no abnormal uptake consistent with osseous metastases was visualized (A). However, extensive metastatic disease was found on 18F-DCFPyL PET/CT (B–E). For example, transversal 18F-DCFPyL PET (B) and fused PET/CT (D) showed highly increased PSMA-expression in the right iliac bone (red arrow, maximum standardized uptake value (SUVmax) 8.15), compatible with a lytic lesion on CT (C). The time interval between bone scintigraphy and 18F-DCFPyL PET/CT was 5 days.
With the better performance of novel imaging modalities, patients with BCR can be diagnosed earlier with metastatic disease. Consequently, the number of patients diagnosed with oligometastatic PCa (usually considered to be a maximum of 3–5 metastases) has increased. The true oligometastatic state is considered to be one of limited metastatic potential in which the local treatment of all visible metastases has the potential to bring about long-term survival in some patients [38]. The treatment of oligometastatic disease (in practice mostly patients with 1–2 lesions) now attracts considerable interest [39]. The most studied treatment approach in patients with oligometastatic recurrent PCa, is metastasis-directed local therapy (MDT; for example, stereotactic radiotherapy or surgery), with the potential goals of delaying the start of ADT and influencing prognosis [40]. A recent randomized phase II study of patients with oligometastatic recurrent PCa showed improved ADT-free survival in patients who underwent MDT compared to surveillance [41]. The effect of MDT on OS needs to be addressed in further clinical trials.
In summary, PSMA PET/CT has the potential for more accurate metastasis detection and (re)staging at initial diagnosis and at BCR than standard conventional imaging modalities (in practice BS and CT). This could potentially lead to changes in treatment and selection of more optimal strategies. However, the clinical benefit of (even) earlier detection of metastases has yet to be shown in well-powered randomized studies. As a result, current EAU guidelines do not recommend the routine use of PSMA (PET/CT) in their imaging algorithms [4]. Currently, at Amsterdam UMC, location VUmc, we are conducting two prospective, clinical trials looking at the diagnostic accuracy of 18F-DCFPyL PET/CT compared to conventional imaging for the detection of metastases in patients with newly diagnosed high risk PCa who have a negative BS (trial 1, VUmc IRB number: 2019.051) or a positive BS with low volume disease (trial 2, VUmc IRB number: 2019.054). The change in treatment approach will be evaluated as a secondary outcome.
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34.
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424.
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Dyba, T.; Randi, G.; Bettio, M.; Gavin, A.; Visser, O.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur. J. Cancer 2018, 103, 356–387.
- Mottet, N.; van den Bergh, R.C.N.; Briers, E.; Van den Broeck, T.; Cumberbatch, M.G.; De Santis, M.; Fanti, S.; Fossati, N.; Gandaglia, G.; Gillessen, S.; et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer-2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2020.
- Epstein, J.I.; Egevad, L.; Amin, M.B.; Delahunt, B.; Srigley, J.R.; Humphrey, P.A.; Grading, C. The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma: Definition of Grading Patterns and Proposal for a New Grading System. Am. J. Surg. Pathol. 2016, 40, 244–252.
- D’Amico, A.V.; Whittington, R.; Malkowicz, S.B.; Schultz, D.; Blank, K.; Broderick, G.A.; Tomaszewski, J.E.; Renshaw, A.A.; Kaplan, I.; Beard, C.J.; et al. Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 1998, 280, 969–974.
- Corfield, J.; Perera, M.; Bolton, D.; Lawrentschuk, N. (68)Ga-prostate specific membrane antigen (PSMA) positron emission tomography (PET) for primary staging of high-risk prostate cancer: A systematic review. World J. Urol. 2018, 36, 519–527.
- Hofman, M.S.; Lawrentschuk, N.; Francis, R.J.; Tang, C.; Vela, I.; Thomas, P.; Rutherford, N.; Martin, J.M.; Frydenberg, M.; Shakher, R.; et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): A prospective, randomised, multicentre study. Lancet 2020, 395, 1208–1216.
- Perera, M.; Papa, N.; Roberts, M.; Williams, M.; Udovicich, C.; Vela, I.; Christidis, D.; Bolton, D.; Hofman, M.S.; Lawrentschuk, N.; et al. Gallium-68 Prostate-specific Membrane Antigen Positron Emission Tomography in Advanced Prostate Cancer-Updated Diagnostic Utility, Sensitivity, Specificity, and Distribution of Prostate-specific Membrane Antigen-avid Lesions: A Systematic Review and Meta-analysis. Eur. Urol. 2020, 77, 403–417.
- Zhou, J.; Gou, Z.; Wu, R.; Yuan, Y.; Yu, G.; Zhao, Y. Comparison of PSMA-PET/CT, choline-PET/CT, NaF-PET/CT, MRI, and bone scintigraphy in the diagnosis of bone metastases in patients with prostate cancer: A systematic review and meta-analysis. Skeletal. Radiol. 2019, 48, 1915–1924.
- Beheshti, M.; Rezaee, A.; Geinitz, H.; Loidl, W.; Pirich, C.; Langsteger, W. Evaluation of Prostate Cancer Bone Metastases with 18F-NaF and 18F-Fluorocholine PET/CT. J. Nucl. Med. 2016, 57, 55S–60S.
- Shen, G.; Deng, H.; Hu, S.; Jia, Z. Comparison of choline-PET/CT, MRI, SPECT, and bone scintigraphy in the diagnosis of bone metastases in patients with prostate cancer: A meta-analysis. Skeletal. Radiol. 2014, 43, 1503–1513.
- Gabriele, D.; Collura, D.; Oderda, M.; Stura, I.; Fiorito, C.; Porpiglia, F.; Terrone, C.; Zacchero, M.; Guiot, C.; Gabriele, P. Is there still a role for computed tomography and bone scintigraphy in prostate cancer staging? An analysis from the EUREKA-1 database. World J. Urol. 2016, 34, 517–523.
- Sheikhbahaei, S.; Jones, K.M.; Werner, R.A.; Salas-Fragomeni, R.A.; Marcus, C.V.; Higuchi, T.; Rowe, S.P.; Solnes, L.B.; Javadi, M.S. (18)F-NaF-PET/CT for the detection of bone metastasis in prostate cancer: A meta-analysis of diagnostic accuracy studies. Ann. Nucl. Med. 2019, 33, 351–361.
- Dehdashti, F.; Picus, J.; Michalski, J.M.; Dence, C.S.; Siegel, B.A.; Katzenellenbogen, J.A.; Welch, M.J. Positron tomographic assessment of androgen receptors in prostatic carcinoma. Eur. J. Nucl. Med. Mol. Imaging 2005, 32, 344–350.
- Zacho, H.D.; Jochumsen, M.R.; Langkilde, N.C.; Mortensen, J.C.; Haarmark, C.; Hendel, H.W.; Jensen, J.B.; Petersen, L.J. No Added Value of (18)F-Sodium Fluoride PET/CT for the Detection of Bone Metastases in Patients with Newly Diagnosed Prostate Cancer with Normal Bone Scintigraphy. J. Nucl. Med. 2019, 60, 1713–1716.
- Cysouw, M.C.F.; Kramer, G.M.; Heijtel, D.; Schuit, R.C.; Morris, M.J.; van den Eertwegh, A.J.M.; Voortman, J.; Hoekstra, O.S.; Oprea-Lager, D.E.; Boellaard, R. Sensitivity of (18)F-fluorodihydrotestosterone PET-CT to count statistics and reconstruction protocol in metastatic castration-resistant prostate cancer. EJNMMI Res. 2019, 9, 70.
- Kramer, G.M.; Yaqub, M.; Vargas, H.A.; Schuit, R.C.; Windhorst, A.D.; van den Eertwegh, A.J.M.; van der Veldt, A.A.M.; Bergman, A.M.; Burnazi, E.M.; Lewis, J.S.; et al. Assessment of Simplified Methods for Quantification of (18)F-FDHT Uptake in Patients with Metastatic Castration-Resistant Prostate Cancer. J. Nucl. Med. 2019, 60, 1221–1227.
- Rathkopf, D.E.; Morris, M.J.; Fox, J.J.; Danila, D.C.; Slovin, S.F.; Hager, J.H.; Rix, P.J.; Chow Maneval, E.; Chen, I.; Gonen, M.; et al. Phase I study of ARN-509, a novel antiandrogen, in the treatment of castration-resistant prostate cancer. J. Clin. Oncol. 2013, 31, 3525–3530.
- Scher, H.I.; Beer, T.M.; Higano, C.S.; Anand, A.; Taplin, M.E.; Efstathiou, E.; Rathkopf, D.; Shelkey, J.; Yu, E.Y.; Alumkal, J.; et al. Antitumour activity of MDV3100 in castration-resistant prostate cancer: A phase 1–2 study. Lancet 2010, 375, 1437–1446.
- Larson, S.M.; Morris, M.; Gunther, I.; Beattie, B.; Humm, J.L.; Akhurst, T.A.; Finn, R.D.; Erdi, Y.; Pentlow, K.; Dyke, J.; et al. Tumor Localization of 16β-18F-Fluoro-5α-Dihydrotestosterone Versus 18F-FDG in Patients with Progressive, Metastatic Prostate Cancer. J. Nucl. Med. 2004, 45, 366–373.
- Oprea-Lager, D.E.; van Kanten, M.P.; van Moorselaar, R.J.; van den Eertwegh, A.J.; van de Ven, P.M.; Bijnsdorp, I.V.; Hoekstra, O.S.; Geldof, A.A. [18F]fluoromethylcholine as a chemotherapy response read-out in prostate cancer cells. Mol. Imaging Biol. 2015, 17, 319–327.
- Evangelista, L.; Zattoni, F.; Guttilla, A.; Saladini, G.; Zattoni, F.; Colletti, P.M.; Rubello, D. Choline PET or PET/CT and biochemical relapse of prostate cancer: A systematic review and meta-analysis. Clin. Nucl. Med. 2013, 38, 305–314.
- Afshar-Oromieh, A.; Zechmann, C.M.; Malcher, A.; Eder, M.; Eisenhut, M.; Linhart, H.G.; Holland-Letz, T.; Hadaschik, B.A.; Giesel, F.L.; Debus, J.; et al. Comparison of PET imaging with a (68)Ga-labelled PSMA ligand and (18)F-choline-based PET/CT for the diagnosis of recurrent prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 11–20.
- Schwenck, J.; Rempp, H.; Reischl, G.; Kruck, S.; Stenzl, A.; Nikolaou, K.; Pfannenberg, C.; la Fougere, C. Comparison of (68)Ga-labelled PSMA-11 and (11)C-choline in the detection of prostate cancer metastases by PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 92–101.
- Perner, S.; Hofer, M.D.; Kim, R.; Shah, R.B.; Li, H.; Moller, P.; Hautmann, R.E.; Gschwend, J.E.; Kuefer, R.; Rubin, M.A. Prostate-specific membrane antigen expression as a predictor of prostate cancer progression. Hum. Pathol. 2007, 38, 696–701.
- Silver, D.A.; Pellicer, I.; Fair, W.R.; Heston, W.D.; Cordon-Cardo, C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin. Cancer Res. 1997, 3, 81–85.
- Uprimny, C.; Kroiss, A.S.; Decristoforo, C.; Fritz, J.; von Guggenberg, E.; Kendler, D.; Scarpa, L.; di Santo, G.; Roig, L.G.; Maffey-Steffan, J.; et al. (68)Ga-PSMA-11 PET/CT in primary staging of prostate cancer: PSA and Gleason score predict the intensity of tracer accumulation in the primary tumour. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 941–949.
- Rowe, S.P.; Gorin, M.A.; Allaf, M.E.; Pienta, K.J.; Tran, P.T.; Pomper, M.G.; Ross, A.E.; Cho, S.Y. PET imaging of prostate-specific membrane antigen in prostate cancer: Current state of the art and future challenges. Prostate Cancer Prostatic Dis. 2016, 19, 223–230.
- Bodar, Y.J.L.; Jansen, B.H.E.; van der Voorn, J.P.; Zwezerijnen, G.J.C.; Meijer, D.; Nieuwenhuijzen, J.A.; Boellaard, R.; Hendrikse, N.H.; Hoekstra, O.S.; van Moorselaar, R.J.A.; et al. Detection of prostate cancer with 18F-DCFPyL PET/CT compared to final histopathology of radical prostatectomy specimens: Is PSMA-targeted biopsy feasible? The DeTeCT trial. World J. Urol. 2020.
- van Leeuwen, P.J.; Donswijk, M.; Nandurkar, R.; Stricker, P.; Ho, B.; Heijmink, S.; Wit, E.M.K.; Tillier, C.; van Muilenkom, E.; Nguyen, Q.; et al. Gallium-68-prostate-specific membrane antigen ((68) Ga-PSMA) positron emission tomography (PET)/computed tomography (CT) predicts complete biochemical response from radical prostatectomy and lymph node dissection in intermediate- and high-risk prostate cancer. BJU Int. 2019, 124, 62–68.
- Wondergem, M.; van der Zant, F.M.; Roeleveld, T.A.; Srbljin, S.; Kartachova, M.S.; van Dongen, A.; Franken, V.; Knol, R.J.J. 18F-DCFPyL PET/CT in primary staging of prostate cancer. Eur. J. Hybrid Imaging 2018, 2, 26.
- Szabo, Z.; Mena, E.; Rowe, S.P.; Plyku, D.; Nidal, R.; Eisenberger, M.A.; Antonarakis, E.S.; Fan, H.; Dannals, R.F.; Chen, Y.; et al. Initial Evaluation of [(18)F]DCFPyL for Prostate-Specific Membrane Antigen (PSMA)-Targeted PET Imaging of Prostate Cancer. Mol. Imaging Biol. 2015, 17, 565–574.
- Giesel, F.L.; Hadaschik, B.; Cardinale, J.; Radtke, J.; Vinsensia, M.; Lehnert, W.; Kesch, C.; Tolstov, Y.; Singer, S.; Grabe, N.; et al. F-18 labelled PSMA-1007: Biodistribution, radiation dosimetry and histopathological validation of tumor lesions in prostate cancer patients. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 678–688.
- Dietlein, M.; Kobe, C.; Kuhnert, G.; Stockter, S.; Fischer, T.; Schomacker, K.; Schmidt, M.; Dietlein, F.; Zlatopolskiy, B.D.; Krapf, P.; et al. Comparison of [(18)F]DCFPyL and [ (68)Ga]Ga-PSMA-HBED-CC for PSMA-PET Imaging in Patients with Relapsed Prostate Cancer. Mol. Imaging Biol. 2015, 17, 575–584.
- Wondergem, M.; van der Zant, F.M.; Broos, W.A.M.; Roeleveld, T.A.; Donker, R.; Ten Oever, D.; Geenen, R.W.F.; Knol, R.J.J. (18)F-DCFPyL PET/CT for primary staging in 160 high-risk prostate cancer patients; metastasis detection rate, influence on clinical management and preliminary results of treatment efficacy. Eur. J. Nucl. Med. Mol. Imaging 2020.
- Jansen, B.H.E.; Bodar, Y.J.L.; Zwezerijnen, G.J.C.; Meijer, D.; van der Voorn, J.P.; Nieuwenhuijzen, J.A.; Wondergem, M.; Roeleveld, T.A.; Boellaard, R.; Hoekstra, O.S.; et al. Pelvic lymph-node staging with (18)F-DCFPyL PET/CT prior to extended pelvic lymph-node dissection in primary prostate cancer—The SALT trial. Eur. J. Nucl. Med. Mol. Imaging 2020.
- Weichselbaum, R.R.; Hellman, S. Oligometastases revisited. Nat. Rev. Clin. Oncol. 2011, 8, 378–382.
- Ost, P.; Bossi, A.; Decaestecker, K.; De Meerleer, G.; Giannarini, G.; Karnes, R.J.; Roach, M., 3rd; Briganti, A. Metastasis-directed therapy of regional and distant recurrences after curative treatment of prostate cancer: A systematic review of the literature. Eur. Urol. 2015, 67, 852–863.
- Palma, D.A.; Olson, R.; Harrow, S.; Gaede, S.; Louie, A.V.; Haasbeek, C.; Mulroy, L.; Lock, M.; Rodrigues, G.B.; Yaremko, B.P.; et al. Stereotactic Ablative Radiotherapy for the Comprehensive Treatment of Oligometastatic Cancers: Long-Term Results of the SABR-COMET Phase II Randomized Trial. J. Clin. Oncol. 2020, 38, 2830–2838.
- Ost, P.; Reynders, D.; Decaestecker, K.; Fonteyne, V.; Lumen, N.; Bruycker, A.D.; Lambert, B.; Delrue, L.; Bultijnck, R.; Goetghebeur, E.; et al. Surveillance or metastasis-directed therapy for oligometastatic prostate cancer recurrence (STOMP): Five-year results of a randomized phase II trial. J. Clin. Oncol. 2020, 38, 10.





