| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Amira Azmi | + 1510 word(s) | 1510 | 2021-01-22 04:48:10 |
Video Upload Options
The synthesis and secretion of cortisol are controlled by the hypothalamic–pituitary–adrenal axis. Cortisol exhibits a proper 24-h circadian rhythm that affects the brain, the autonomic nervous system, the heart, and the vasculature that prepares the cardiovascular system for optimal function during these anticipated behavioral cycles.
1. Introduction
Cortisol has a particular circadian rhythm that is affected by sleep. Based on normal physiology, lower levels of cortisol are present at the initial part of sleep, whereas there is an increase at the end of the sleep period, which reaches its peak just minutes before the individual wakes up. This rhythm is formed in close association with the sleep–wakefulness cycle[1]. Thus, it supports the fact that cortisol contributes to a significant role in the initiation of wakefulness[2]. In other words, cortisol is the key player in the circadian system that affects nearly every tissue and organ of the body, controlling certain biological cyclical functions, including cardiovascular functions[3]. Distinct variation in the levels of cortisol during the day and night may help to explain the function of cortisol in health and disease[4]. Cortisol status appears to be an objective biological marker of the stress response that can be related to the majority of cardiovascular diseases[5]. The timing of cardiovascular events like acute myocardial infarction and ventricular fibrillation can influence the severity of disease and survival, with poor prognoses being associated with early morning occurrences[6]. In the general population, the levels of cortisol measured in blood, urine, or hair are positively associated with elevated risk factors for cardiovascular disease[7], death from cardiovascular disease, and all-cause mortality[8].
2. Synthesis and Metabolism of Cortisol
A significant component of the human neuroendocrine system is the hypothalamic–pituitary–adrenal gland (the HPA axis), which is of paramount importance to the survival of mammals, including humans[9]. The physiological function of the HPA axis is important to preserve homeostasis, adapt to environmental surroundings, and regulate human behavior, emotion, and cognitive functions. The main end product of the HPA axis is cortisol, a glucocorticoid hormone that is highly associated with circadian rhythms[10]. Cortisol is considered the major glucocorticoid in both humans and nonhuman primates, and it is produced in response to a variety of stressors[11].
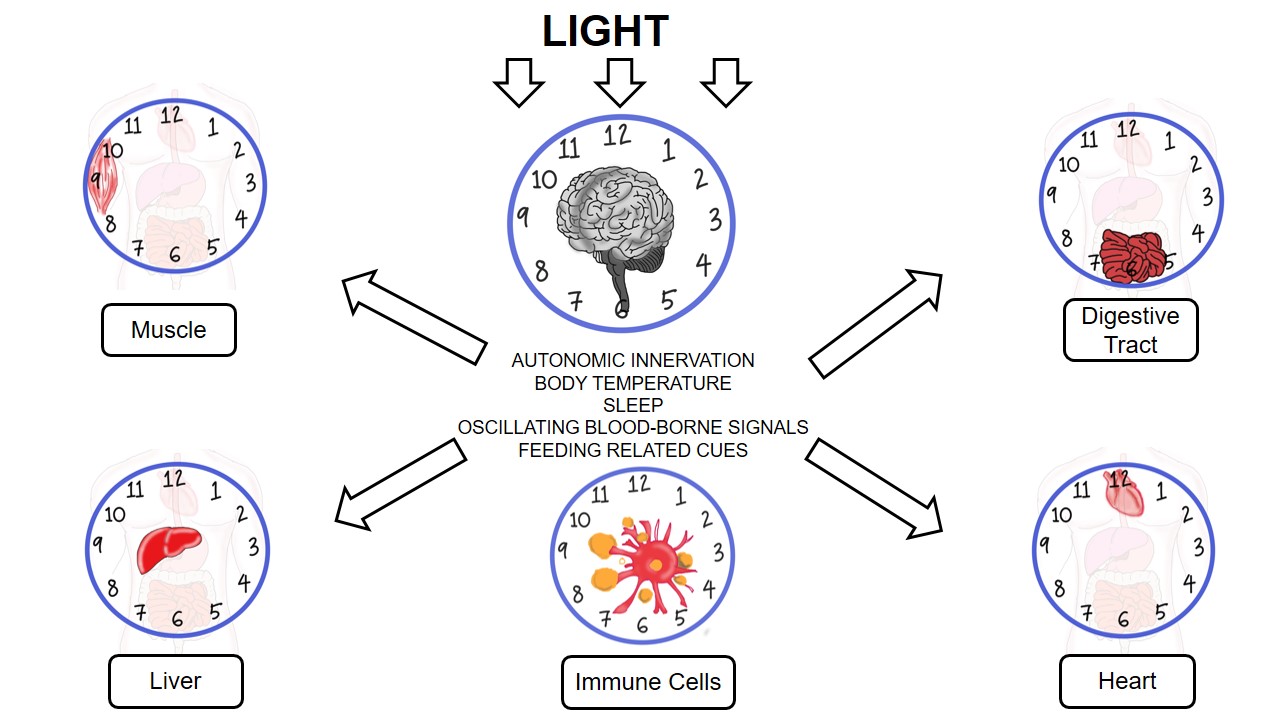
Cortisol plays important roles in various physiological functions, such as metabolism, electrolyte balance, development, and cognition, and is implicated in multiple organ systems, namely, neuroendocrine, immune, reproductive, cardiovascular, and nervous systems[12][13]. Cortisol and other glucocorticoid hormones are mainly derived from cholesterol in the mitochondria of zona fasciculata in the adrenal cortex[14]. Metabolically, cholesterol is first converted to pregnenolone by removing the side chain at C20, a process catalyzed by desmolase (cytochrome P450scc). Pregnenolone is subsequently converted to progesterone by 3β-hydroxysteroid dehydrogenase. Enzyme 17-hydroxylase (CYP17) catalyzes the conversion of progesterone to 17-hydroxyprogesterone, which is then converted to 11-deoxycortisol by the 21α-hydroxylase activity of CYP21. The final step involves the 11β-hydroxylation of 11-deoxycortisol to the end product, cortisol, by 11-hydroxylase (CYP11B1 or CYP11B2)[12][15]. The figure illustrates the biological clock in heart, digestive tract, liver, muscle, and immune cells.
3. Effect of Cortisol on the Cardiovascular System
3.1. Acute Coronary Syndromes
Acute coronary syndromes (ACSs) are characterized as ST-segment elevation of myocardial infarction (STEMI), non-ST-segment elevation of myocardial infarction (NSTEMI), and unstable angina (UA), which lead to morbidity and mortality in industrialized nations[16]. In smaller cross-sectional studies, there have been contradictory findings of the relationship between cortisol and adverse cardiovascular outcomes in AMI patients, despite the majority showing a poorer outcome with increased levels of cortisol[17]. Specifically, most of the smaller cross-sectional studies have reported increased morbidity and mortality risks with elevated levels of cortisol in AMI patients[18][19], whereas some found no correlation[20] and others found an inverse association[21].
Moreover, previous studies have also reported the association of levels of cortisol with an increase in myocardial infarct size[22], ventricular remodeling post-acute myocardial infarction (AMI)[23], and high mortality in patients with chronic heart failure [24]. Jutla et al. (2014) found the prognostic importance of cortisol in post-AMI patients at a single center with respect to major adverse cardiovascular events (MACEs) and a combination of all-cause mortality, as well as rehospitalization for heart failure immediately following myocardial necrosis[17]. Meanwhile, a case–control study by Reynolds et al. (2010) found a reverse association in which lower serum cortisol levels in post-AMI patients were associated with increased 30-day mortality[17].
3.2. Atrial and Ventricular Arrhythmias
Cardiac arrhythmias are described as the conduction of irregular electrical impulses via the myocardium, leading to altered heartbeat, altered muscle contraction, and disrupted rhythm[25]. With respect to the pathophysiology of arrhythmias and sudden cardiac death, an acute precipitating stimulus in the neurons of the brain and chronic electrical instability of the cardiomyocyte in the heart may be correlated with an increase in circulating cortisol[26].
Atrial fibrillation (AF) is the commonest type of arrhythmia in adults and a significant cause of morbidity and death. Specifically, AF is an atrial tachyarrhythmia that is categorized by irregular atrial activation and impaired atrial contractile function[27]. Cortisol is a steroid hormone that enhances in response to stress and plays an important part in the synthesis of catecholamine. Catecholamine has arrhythmic potential by influencing the cardiac conduction system[28]. Glucocorticoids are also involved in the synthesis of Na-ATP, K-ATP, and catecholamine. Furthermore, in this regard, it is responsible for a partial positive inotropic impact on the heart[29]. However, in a case–control study by Akseli et al. (2013), which aimed to determine whether serum cortisol levels were different in AF patients in comparison to a control group, it was reported that the abnormality in the levels of serum cortisol with respect to diurnal rhythm did not correlate with the etiology and permanence of AF[30].
Ventricular tachycardia and ventricular fibrillation occur as a result of dynamic and complex interactions between the arrhythmogenic substrate (i.e., myocardial scar boundary zone) and myocardial electrophysiological properties (i.e., electrical stimulation and propagation)[31]. Previous studies have reported that repeated bouts of high cortisol in view of psychological stress or neural mechanisms can predispose to low-grade inflammation, causing poor processes of repair, with low macrophages and excess neutrophils and lymphocytes. Thus, this subsequently results in myocardial dysfunction-induced arrhythmias[32][33]. Moreover, any inflammatory focus in the myocardium due either to an acute increase in cortisol or myocardial ischemia may inhibit the myocardial cell coordination, leading to ventricular fibrillation[34].
3.3. Sudden Cardiac Death
Sudden cardiac death remains a significant health concern that is responsible for a large proportion of all cardiac deaths[35]. A high incidence of sudden cardiac death may result from coronary heart disease, left ventricular hypertrophy, cardiac fibrosis, and heart failure[36]. In addition, arrhythmias also may result in sudden cardiac death due to focal or general inflammation induced by high concentrations of circulating cortisol[26]. According to the World Health Organization, sudden cardiac death is defined as sudden and unexpected death observed within an hour of symptom onset[37]. The underlying myocardial electrophysiological alteration in most patients with sudden cardiac death is ventricular fibrillation with underlying inflammation due to high cortisol concentration[5][25][26]. Long-term or extreme emotional stress may predispose patients to HPA axis dysfunction, with the resultant dysregulation of cortisol release causing ventricular fibrillation and sudden cardiac death[5].
3.4. Stroke
Stroke is the dominant cause of long-term impairment and disability among adults in modern communities. A stroke normally happens when a blood clot prevents blood from flowing to a part of the brain. Consequently, brain cells that lack blood will start to die within minutes. Patients surviving a stroke are likely to experience cognitive, visual, and motor deficits, depending on the location and severity of the brain tissue damage[38]. Acute ischemic stroke acts as a stressor and thereby activates the HPA axis, leading to high levels of glucocorticoid[39]. Based on the systematic review by Barugh et al. (2014), cortisol levels are elevated in most patients for at least seven days after stroke and are within the normal range by three months. High levels of cortisol following stroke is correlated with increased dependency, morbidity, and mortality. However, at present, there is limited evidence to presume that these connections are independent of the severity of stroke[40].
Another study by Zi and Shuai (2013) suggested that cortisol can be used as an independent short-term prognostic marker of functional outcome and death in Chinese acute ischemic stroke patients[41]. This is consistent with the findings of previous studies that have reported that hypercortisolemia was associated with older age, greater severity of neurological impairment, larger ischemic lesions on computed tomography (CT) scan, and poorer prognosis (increased disability and mortality) in patients with stroke[42][43]. Furthermore, patients with stroke and elevated levels of cortisol are more vulnerable to adverse cardiac complications, which may result in higher mortality rates[44]. A poor prognosis after stroke is the development of infectious diseases associated with immune dysregulation due to neuroendocrine disorder after stroke[45]. Increased levels of cortisol make patients more susceptible to infections[46].
4. Conclusion
Cortisol is a key player in the circadian system that significantly regulates cardiac function. Previous studies have emphasized that excessive cortisol is correlated with an increased risk of cardiovascular events, namely, acute coronary syndromes,arrhythmias, sudden cardiac death, and stroke.
References
- Scher, A.; Hall, W.A.; Zaidman-Zait, A.; Weinberg, J. Sleep quality, cortisol levels, and behavioural regulation in toddlers. Dev. Psychobiol. 2010, 52, 44–53.
- Dahlgren, A.; Kecklund, G.; Theorell, T.; Akerstedt, T. Day-to-day variation in saliva cortisol—Relation with sleep, stress and self-rated health. Biol. Psychol. 2009, 82, 149–155.
- Benarroch, E.E. Suprachiasmatic nucleus and melatonin: Reciprocal interactions and clinical correlations. Neurology 2008, 71, 594–598.
- Ice, G.H.; Katz-Stein, A.; Himes, J.; Kane, R.L. Diurnal cycles of salivary cortisol in older adults. Psychoneuroendocrinology 2004, 29, 355–370.
- Singh, R.B.; Isaza, A.; Wilczynska, A.; Kartikey, K.; Kareem, Z.; Alshihaby, W.; Almansory, A.; Hadi, N.R.; Bassm, M. View Point. Cortisol and the heart. World Heart J. 2019, 11, 73–77.
- Manfredini, R.; Boari, B.; Salmi, R.; Fabbian, F.; Pala, M.; Tiseo, R.; Portaluppi, F. Twenty-four-hour patterns in occurrence and pathophysiology of acute cardiovascular events and ischemic heart disease. Chronobiol. Int. 2013, 30, 6–16.
- Cagnacci, A.; Cannoletta, M.; Caretto, S.; Zanin, R.; Xholli, A.; Volpe, A. Increased cortisol level: A possible link between climacteric symptoms and cardiovascular risk factors. Menopause 2011, 18, 273–278.
- Jokinen,J.;Nordstrom,P.HPAaxishyperactivityandcardiovascularmortalityinmooddisorderinpatients. J.Affect. Disord. 2009, 116, 88–92.
- Nicolaides, N.C.; Charmandari, E.; Chrousos, G.P.; Kino, T. Circadian endocrine rhythms: The hypothalamic–pituitary– adrenal axis and its actions. Ann. N. Y. Acad. Sci. 2014, 1318, 71–80.
- Huang, Y.; Xu, C.; He, M.; Huang, W.; Wu, K. Saliva cortisol, melatonin levels and circadian rhythm alterations in Chinese primary school children with dyslexia. Medicine 2020, 99, e19098.
- Qin, D.D.; Rizak, J.; Feng, X.L.; Yang, S.C.; Lü, L.B.; Pan, L.; Yin, Y.; Hu, X.T. Prolonged secretion of cortisol as a possible mechanism underlying stress and depressive behaviour. Sci. Rep. 2016, 6, 30187.
- Tomas, C.; Newton, J.; Watson, S. A Review of hypothalamic-pituitary-adrenal axis function in chronic fatigue syndrome. ISRN Neurosci. 2013, 784520.
- Timmermans, S.; Souffriau, J.; Libert, C. A general introduction to glucocorticoid biology. Front. Immunol. 2019, 10, 1545.
- Miller, G.E.; Chen, E.; Fok, A.K.; Walker, H.; Lim, A.; Nicholls, E.F.; Cole, S.; Kobor, M.S. Low early-life social class leaves a biological residue manifested by decreased glucocorticoid and increased proinflammatory signaling. Proc. Natl. Acad. Sci. USA 2009, 106, 14716–14721.
- Katsu, Y.; Iguchi, T. Cortisol. In Handbook of Hormones: Comparative Endocrinology for Basic and Clinical Research; Takei, Y., Ando, H., Tsutsui, K., Eds.; Academic Press: Cambridge, MA, USA, 2015.
- Sheridan, P.J.; Crossman, D.C. Critical review of unstable angina and non-ST elevation myocardial infarction. Postgrad. Med. J. 2002, 78, 717–726.
- Jutla, S.K.; Yuyun, M.F.; Quinn, P.A.; Ng, L.L. Plasma cortisol and prognosis of patients with acute myocardial infarction. J. Cardiovasc. Med. 2014, 15, 33–41.
- Nito, I.; Waspadji, S.; Harun, S.; Markum, H.M. Correlation between cortisol levels and myocardial infarction mortality among intensive coronary care unit patients during first seven days in hospital. Acta Med. Indones. 2004, 36, 8–14.
- Adair, R.; Kasahara, M. Serum cortisol response to acute myocardial infarction in the aged. J. Am. Geriatr. Soc. 1980, 28, 472–474.
- Zouaghi,H.;Savu,L.;Guerot,C.;Gryman,R.;Coulon,A.;Nunez,E.A.Totalandunboundcortisol-,progesterone-,oestrone-,and transcortin-binding activities in sera from patients with myocardial infarction: Evidence for differential responses of good and bad prognostic cases. Eur. J. Clin. Investig. 1985, 15, 365–370.
- Reynolds, R.M.; Walker, B.R.; Haw, S.; Newby, D.E.; Mackay, D.F.; Cobbe, S.M.; Pell, A.C.; Fischbacher, C.; Pringle, S.; Murdoch, D.; et al. Low serum cortisol predicts early death after acute myocardial infarction. Crit. Care Med. 2010, 38, 973–975.
- Bain, R.J.; Fox, J.P.; Jagger, J.; Davies, M.K.; Littler, W.A.; Murray, R.G. Serum cortisol levels predict infarct size and patient mortality. Int. J. Cardiol. 1992, 37, 145–150.
- Weir, R.A.; Tsorlalis, I.K.; Steedman, T.; Dargie, H.J.; Fraser, R.; McMurray, J.J.; Connell, J.M. Aldosterone and cortisol predict medium-term left ventricular remodelling following myocardial infarction. Eur. J. Heart Fail. 2011, 13, 1305–1313.
- Gueder, G.; Bauersachs, J.; Frantz, S.; Weismann, D.; Allolio, B.; Ertl, G.; Angermann, C.E.; Stork, S. Complementary and incremental mortality risk prediction by cortisol and aldosterone in chronic heart failure. Circulation 2007, 115, 1754–1761.
- Clauss, S.; Bleyer, C.; Schuttler, D.; Tomsits, P.; Renner, S.; Klymiuk, N.; Wakili, R.; Massberg, S.; Wolf, E.; Kaab, S. Animal models of arrhythmia: Classic electrophysiology to genetically modified large animals. Nat. Rev. Cardiol. 2019, 16, 457–475.
- Elkilany, G.; Singh, R.B.; Adeghate, E.; Singh, J.; Bidasee, K.; Fedacko, J.; Hristova, K. Sudden cardiac death, mini review. World Heart J. 2017, 9, 51–62.
- Fuster, V.; Ryden, L.E.; Cannom, D.S. ACC/AHA/ESC 2006 guidelines for the management of patients with atrial fibrillation: AreportoftheAmericanCollegeofCardiology/AmericanHeartAssociationTaskForceonPracticeGuidelinesandtheEuropean Society of Cardiology Committee for Practice Guidelines (Writing Committee to Revise the 2001 Guidelines for the Management of Patients with Atrial Fibrillation). J. Am. Coll. Cardiol. 2006, 48, 149–246.
- Hollander, J.E. Acute coronary syndromes: Acute myocardial infarction and unstable angina. In Emergency Medicine a Comprehensive Study Guide, 6th ed.; Tintinalli, J.E., Kelen, G.D., Stapczynski, J.S., Eds.; McGraw-Hill: New York, NY, USA, 2004; pp. 343–351.
- Marik, P.E.; Zaloga, G.P. Adrenal insufficiency in the critically ill: A new look to an old problem. Chest 2002, 122, 1784–1796.
- Akseli, A.; Topacoglu, H.; Ucku, R. The comparison of the serum steroid levels of the patients with or without atrial fibrillation: Case control study. Int. Med. J. 2013, 20, 1–3.
- Mittnacht, A.J.; Dukkipati, S.; Mahajan, A. Ventricular tachycardia ablation: A comprehensive review for anesthesiologists. Anesth. Analg. 2015, 120, 737–748.
- Singh, R.B.; Cornelissen, G.; Chibisov, S.; Fedacko, J. Atherosclerosis? A disease of the brain. World Heart J. 2017, 9, 99–106.
- Horckmans, M.; Ring, L.; Duchene, J.; Santovito, D.; Schloss, M.J.; Drechsler, M.; Weber, C.; Soehnlein, O.; Steffens, S. Neutrophils orchestrate post-myocardial infarction healing by polarizing macrophages towards a reparative phenotype. Eur. HeartJ.2017, 38, 187–197.
- Vargova,V.;Singh,R.B.; Chibisov,S.;Bawareed,A.O.;Isaza,A.Molecularmechanismsinrelationtocortisolandleucocytesinthe pathogenesis of ventricular arrhythmias. World Heart J. 2019, 11, 131–140.
- Mahmoud, K.D.; de Smet, B.J.; Zijlstra, F.; Rihal, C.S.; Holmes, D.R., Jr. Sudden cardiac death: Epidemiology, circadian variation, and triggers. Curr. Probl. Cardiol. 2011, 36, 56–80.
- Wanner, C.; Krane, V.; Marz, W.; Olschewski, M.; Mann, J.F.; Ruf, G.; Ritz, E. Atorvastatin in patients with type 2 diabetes mellitus undergoing hemodialysis. N. Engl. J. Med. 2005, 353, 238–248.
- World Health Organization. International Classification of Disease 10 (ICD-10). 2010. Available online: https://www.who.int/ classifications/icd/ICD10Volume2_en_2010.pdf
- Alankus, G.; Lazar, A.; May, M.; Kelleher, C. Towards customizable games for stroke rehabilitation. In Proceedings of the CHI ’10: Proceedings of the 28th SIGCHI International Conference on Human Factors in Computing Systems, Atlanta, GA, USA, 10–15 April 2010; pp. 2113–2122.
- Fassbender, K.; Schmidt, R.; Mossner, R.; Daffertshofer, M.; Hennerici, M. Pattern of activation of the hypothalamic–pituitary– adrenal axis in acute stroke. Relation to acute confusional state, extent of brain damage, and clinical outcome. Stroke 1994, 25, 1105–1108.
- Barugh, A.J.; Gray, P.; Shenkin, S.D.; MacLullich, A.M.J.; Mead, G.E. Cortisol levels and the severity and outcomes of acute stroke: A systematic review. J. Neurol. 2014, 261, 533–545.
- Zi,W.J.;Shuai,J.CortisolasaPrognosticmarkerofshort-termoutcomeinChinesepatientswithacuteischemicstroke. PLoSONE 2013, 8, e72758.
- Olsson,T.;Marklund,N.;Gustafson,Y.;Näsman,B.Abnormalitiesatdifferentlevelsofthehypothalamic–pituitary–adrenocortical axis early after stroke. Stroke 1992, 23, 1573–1576.
- Slowik, A.; Turaj, W.; Pankiewicz, J.; Dziedzic, T.; Szermer, P.; Szczudlik, A. Hypercortisolemia in acute stroke is related to the inflammatory response. J. Neurol. Sci. 2002, 196, 27–32.
- Katan, M.; Elkind, M.S. Inflammatory and neuroendocrine biomarkers of prognosis after ischemic stroke. Expert. Rev. Neurother. 2011, 11, 225–239.
- Neidert, S.; Katan, M.; Schuetz, P.; Fluri, F.; Ernst, A.; Bingisser, R.; Kappos, L.; Engelter, S.T.; Steck, A.; Muller, B.; et al. Anterior pituitary axis hormones and outcome in acute ischaemic stroke. J. Intern. Med. 2011, 269, 420–432.
- Schoorlemmer,R.M.;Peeters,G.M.;vanSchoor,N.M.;Lips,P.Relationshipsbetweencortisollevel,mortalityandchronicdiseases in older persons. Clin. Endocrinol. 2009, 71, 779–786.