
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jianhua He | + 1103 word(s) | 1103 | 2021-01-13 04:15:43 | | | |
| 2 | Vicky Zhou | -1 word(s) | 1102 | 2021-01-13 08:36:04 | | | | |
| 3 | Vicky Zhou | + 47 word(s) | 1149 | 2021-01-13 08:36:44 | | | | |
| 4 | Catherine Yang | Meta information modification | 1149 | 2021-09-30 03:27:06 | | |
Video Upload Options
Resveratrol (3,4′,5-trihy- droxystilbene), a natural phytoalexin polyphenol, exhibits anti-oxidative, anti-inflammatory, and anti-carcinogenic properties. This phytoalexin is well-absorbed, rapidly and extensively metabolized in the body.
1. Introduction
In 1976, resveratrol (3,4′,5-trihy- droxystilbene) was thought to be just a phytoalexin [1], one of the polyphenolic compounds produced by plants in response to environmental stress [2]. Subsequently, resveratrol was found to exhibit multiple bioactivities, including anti-oxidative [3], anti-inflammatory [4], cardiovascular protective [5], and anti-aging [6] properties, in animals. This phytoalexin has been found in at least 72 plant species, such as mulberries, peanuts, and grapes (Table 1). As a nonflavonoid polyphenol, resveratrol exists as two geometric isomers, trans- and cis- (Figure 1), and their glucosides, trans- and cis- piceids [7].
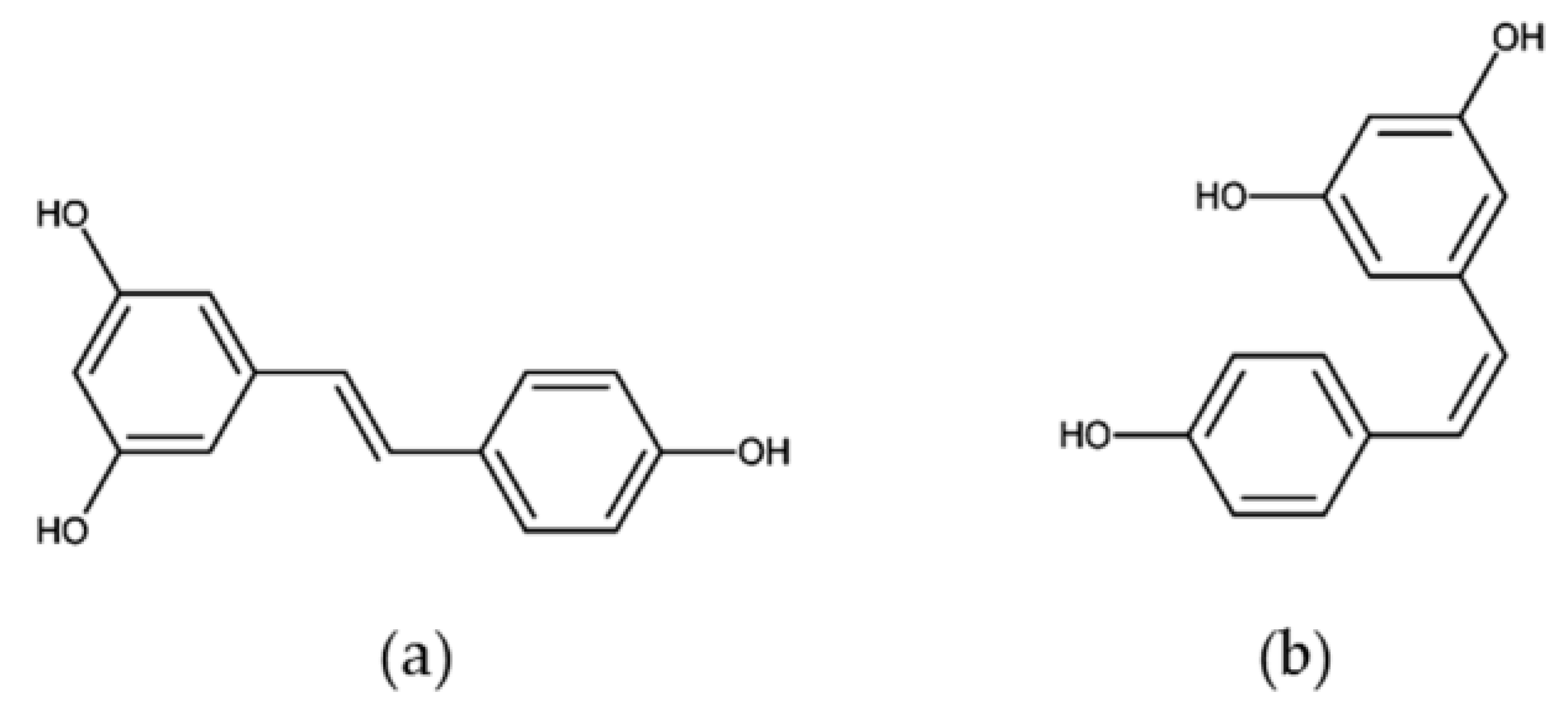
Table 1. The content of resveratrol in some food products.
| Plants | Content |
|---|---|
| Mulberries | 5 mg of resveratrol per 100 g |
| Lingonberries | 3 mg per 100 g |
| Cranberries | 1.92 mg of resveratrol per 100 g |
| Red currants | 1.57 mg of resveratrol per 100 g |
| Bilberries | 0.67 mg of resveratrol per 100 g |
| Blueberries | 0.383 mg per 100 g |
| Peanuts | 1.12 mg of resveratrol per 100 g |
| Pistachios | 0.11 mg of resveratrol per 100 g |
| Fresh grapes | 0.24 to 1.25 mg per cup (160 g) |
| Red grape juice | 0.5 mg per liter |
2. The Anti-Inflammatory Activity of Resveratrol
Inflammatory response is a multi-stage process involving multiple cell types as well as mediator signals [8]. Inflammation is an adaptive response, which can be triggered by various danger signals, such as invasion by microorganisms or tissue injury [9]. The exogenous and endogenous signaling molecules are known as pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs), respectively [8][9][10].
Both PAMPs and DAMPs are recognized by various pattern recognition receptors (PRRs), such as Toll-like receptors (TLRs) [11][12][13]. The activation of PRR induces intracellular signaling cascades, such as kinases and transcription factors [12][14]. The signaling pathways mentioned above can promote the production of a variety of inflammatory mediators (such as cytokines) for inflammation development.
Multiple lines of evidence from laboratory studies, both in vivo and in vitro, have shown that the anti-inflammatory properties of resveratrol may be explained though inhibiting the production of anti-inflammatory factors. For example, resveratrol has been stated to suppress the proliferation of spleen cells induced by concanavalin A (ConA), interleukin (IL)-2, or allo-antigens, and to more effectively prevent lymphocytes from producing IL-2 and interferon-gamma (IFN-γ) and macrophages from producing tumor necrosis factor alpha (TNF-α) or IL-12 [15]. Resveratrol has been found to induce a dose-dependent suppression of the production of IL-1α, IL-6, and TNF-α and down-regulate both mRNA expression and protein secretion of IL-17 in vitro [16]. Dietary resveratrol supplementation is capable of improving tight junction protein zonula occludens-1, Occludin, and claudin-1 expression to reduce intestinal permeability in vivo [17][18]. Resveratrol treatment also reduced the expression of the inflammatory factors, glycation end product receptor (RAGE), NF-кB (P65) and nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 4 (NOX4) and improved the renal pathological structure [19]. In addition, as the natural precursor of resveratrol, polydatin (a resveratrol glycoside isolated from Polygonum cuspidatum) significantly downregulated IL-6, IL-1β, and TNF-α expression induced by Mycoplasma gallisepticum both in vivo and in vitro, suggested that polydatin also has anti-inflammatory effect [20].
The anti-inflammatory activity of this compound was also demonstrated in a rat model of carrageenan-induced paw edema [21]. Additionally, it has been reported that resveratrol preconditioning modulates inflammatory response of hippocampus after global cerebral ischemia in rat [22]. In addition, resveratrol has been documented to be able to suppress neuro-inflammation mediated by microglia, protect neurons from inflammatory damage, and relieve inflammation of the airway caused by asthma and airway remodeling [23][24].
Heat stress may induce reactive oxygen species (ROS) production, cause antioxidant system disorders, and cause damage to the immune organs in vivo [25]. In a recent report, our research group observed that dietary resveratrol supplementation in broilers was successful in partially alleviating the detrimental effects of heat stress on the function of the intestinal barrier, by restoring the damaged villus-crypt structure, altering the mRNA expression of intestinal heat-shock proteins, secreted immunoglobulin A, and close junction-related genes and inhibiting pro-inflammation secretion [26]. Zhang et al. [27] reported that dietary supplementation with resveratrol in broilers could partly reverse the adverse effects of heat stress on the growth of immune organ by reestablishing redox status and inhibiting apoptosis. Importantly, our recent study demonstrated that resveratrol could alleviate heat stress-induced innate immunity and inflammatory response by inhibiting the activation of PRRs signaling in the spleen of broilers [28].
Lipopolysaccharides (LPS) is an essential glycolipid component of Gram-negative bacterial endotoxin which can trigger inflammatory responses to the host [29]. The addition of resveratrol has also led to decreases in inflammatory mediator expression, including prostaglandin (PG)E2, COX-2, IL-1β, IL-8, TNF-α, and monocyte chemoattractant protein-1 in BV-2 or monocyte LPS stimulation cells [30][31]. Additionally, Toll-like receptor-4 (TLR-4) expression in LPS-stimulated cells was attenuated after resveratrol pre-treatment [32]. Long-term treatment with this compound is able to increase brain defenses in aged animals against acute LPS pro-inflammatory stimuli [33]. Moreover, palmitate-induced IL-6 and TNF-α at mRNA and protein levels in C2C12 cells can also be substantially prevented by resveratrol pretreatment [34].
On the other hand, resveratrol can induce anti-inflammatory properties by suppressing the production of ROS and nitric oxide (NO). Oxidative stress caused by the accumulation of ROS plays a role in promoting inflammation in a wide spectrum of diseases, such as chronic inflammation and cancer [35]. It was found that resveratrol was able to suppress strongly the generation of NO in activated macrophages, as well as decrease strongly the amount of cytosolic inducible nitric oxide synthase (iNOS) protein and steady state mRNA levels [36]. Dietary resveratrol supplementation also can effectively eliminate free radicals, enhance the activities of SOD, CAT, and GPX [25][37]. Babu et al. [38] showed that the cytoprotective effect of resveratrol is predominantly due to mitigation of mitochondrial ROS. A recent study conducted by Kortam et al. [39] showed that resveratrol increased the liver’s antioxidant and anti-inflammatory activity against chronic unpredictable mild stress induced depression in the animal model, as explained by the normalization of total antioxidant ability, glutathione, malondialdehyde (MDA), NF-кB, TNF-α, and myeloperoxidase. Additionally, resveratrol down-regulated the expression of iNOS mRNA and protein expression in the LPS-stimulated intestinal cells in a dose-dependent manner, resulting in a decreased production of NO [32]. Similarly, resveratrol dose-dependently inhibited the expression of iNOS and IL-6 in LPS-treated RAW264.7 cells, therefore, suppressed the production of NO and the secretion of IL-6 [40].
3. Summary and Perspectives
Multiple lines of compelling evidence indicate that resveratrol has a promising role in the prevention and treatment of many autoimmune and inflammatory chronic diseases, such as inflammatory, neurological, and multiple cancers. This phytoalexin was demonstrated to modulate many cellular and molecular mediators of inflammation, but the molecular mechanisms of polyphenol are complex and involve multiple signal transduction pathways, and have not been fully elucidated.
Therefore, future research should focus on: (1) further evaluating the compound during clinical trials and improving oral absorption efficiency; and (2) elucidating the underlying mechanisms of resveratrol action in several physiological conditions, in order to make this compound a cutting-edge therapeutic strategy for the prevention and treatment of a wide variety of chronic diseases.
References
- Langcake, P.; Pryce, R.J. The production of resveratrol by Vitis vinifera and other members of the Vitaceae as a response to infection or injury. Physiol. Plant Pathol. 1976, 9, 77–86.
- Lastra, C.A.; Villegas, I. Resveratrol as an antioxidant and pro-oxidant agent: Mechanisms and clinical implications. Biochem. Soc. Trans. 2007, 35, 1156–1160.
- Meng, Q.; Guo, T.; Li, G.; Sun, S.; He, S.; Cheng, B.; Shi, B.; Shan, A. Dietary resveratrol improves antioxidant status of sows and piglets and regulates antioxidant gene expression in placenta by Keap1-Nrf2 pathway and Sirt1. J. Anim. Sci. Biotechnol. 2018, 9, 34.
- Nunes, S.; Danesi, F.; Del Rio, D.; Silva, P. Resveratrol and inflammatory bowel disease: The evidence so far. Nutr. Res. Rev. 2018, 31, 85–97.
- Duthie, G.G.; Duthie, S.J.; Kyle, J.A.M. Plant polyphenols in cancer and heart disease: Implications as nutritional antioxidants. Nutr. Res. Rev. 2000, 13, 79–106.
- Lastra, C.A.; Villegas, I. Resveratrol as an anti-inflammatory and anti-aging agent: Mechanisms and clinical implications. Mol. Nutr. Food Res. 2005, 49, 405–430.
- Signorelli, P.; Ghidoni, R. Resveratrol as an anticancer nutrient: Molecular basis, open questions and promises. J. Nutr. Biochem. 2005, 16, 449–466.
- Lugrin, J.; Rosenblatt-Velin, N.; Parapanov, R.; Liaudet, L. The role of oxidative stress during inflammatory processes. Biol. Chem. 2014, 395, 203–230.
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435.
- Iwasaki, A.; Medzhitov, R. Control of adaptive immunity by the innate immune system. Nat. Immunol. 2015, 16, 343–353.
- Nathan, C. Points of control in inflammation. Nature 2002, 420, 846–852.
- Liu, J.; Cao, X. Cellular and molecular regulation of innate inflammatory responses. Cell Mol. Immunol. 2016, 13, 711–721.
- Fitzgerald, K.A.; Kagan, J.C. Toll-like Receptors and the Control of Immunity. Cell 2020, 180, 1044–1066.
- Singh, A.; Yau, Y.F.; Leung, K.S.; El-Nezami, H.; Lee, J.C.-Y. Interaction of Polyphenols as Antioxidant and Anti-Inflammatory Compounds in Brain–Liver–Gut Axis. Antioxidants 2020, 9, 669.
- Gao, X.; Xu, Y.X.; Janakiraman, N.; Chapman, R.A.; Gautam, S.C. Immunomodulatory activity of resveratrol: Suppression of lymphocyte proliferation, development of cell-mediated cytotoxicity, and cytokine production. Biochem. Pharmacol. 2001, 62, 1299–1308.
- Fuggetta, M.P.; Bordignon, V.; Cottarelli, A.; Macchi, B.; Frezza, C.; Cordiali-Fei, P.; Ensoli, F.; Ciafre, S.; Marino-Merlo, F.; Mastino, A.; et al. Downregulation of proinflammatory cytokines in HTLV-1-infected T cells by Resveratrol. J. Exp. Clin. Cancer Res. 2016, 35, 118.
- Zhang, C.; Zhao, X.H.; Yang, L.; Chen, X.Y.; Jiang, R.S.; Jin, S.H.; Geng, Z.Y. Resveratrol alleviates heat stress-induced impairment of intestinal morphology, microflora, and barrier integrity in broilers. Poult. Sci. 2017, 96, 4325–4332.
- Zhang, H.; Chen, Y.; Chen, Y.; Ji, S.; Jia, P.; Li, Y.; Wang, T. Comparison of the protective effects of resveratrol and pterostilbene against intestinal damage and redox imbalance in weanling piglets. J. Anim. Sci. Biotechnol. 2020, 11, 52.
- Xian, Y.; Gao, Y.; Lv, W.; Ma, X.; Hu, J.; Chi, J.; Wang, W.; Wang, Y. Resveratrol prevents diabetic nephropathy by reducing chronic inflammation and improving the blood glucose memory effect in non-obese diabetic mice. Naunyn Schmiedebergs Arch. Pharmacol. 2020, 393, 2009–2017.
- Zou, M.; Yang, W.; Niu, L.; Sun, Y.; Luo, R.; Wang, Y.; Peng, X. Polydatin attenuates Mycoplasma gallisepticum (HS strain)-induced inflammation injury via inhibiting the TLR6/ MyD88/NF-kappaB pathway. Microb. Pathog. 2020, 149, 104552.
- Jang, M.; Cai, L.; Udeani, G.O.; Slowing, K.V.; Thomas, C.F.; Beecher, C.W.; Fong, H.H.; Farnsworth, N.R.; Kinghorn, A.D.; Mehta, R.G.; et al. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science 1997, 275, 218–220.
- Simao, F.; Matte, A.; Pagnussat, A.S.; Netto, C.A.; Salbego, C.G. Resveratrol preconditioning modulates inflammatory response in the rat hippocampus following global cerebral ischemia. Neurochem. Inter. 2012, 61, 659–665.
- Hou, Y.; Zhang, Y.; Mi, Y.; Wang, J.; Zhang, H.; Xu, J.; Yang, Y.; Liu, J.; Ding, L.; Yang, J.; et al. A Novel Quinolyl-Substituted Analogue of Resveratrol Inhibits LPS-Induced Inflammatory Responses in Microglial Cells by Blocking the NF-κB/MAPK Signaling Pathways. Mol. Nutr. Food. Res. 2019, 63, e1801380.
- Jiang, H.; Duan, J.; Xu, K.; Zhang, W. Resveratrol protects against asthma-induced airway inflammation and remodeling by inhibiting the HMGB1/TLR4/NF-kappa B pathway. Exp. Ther. Med. 2019, 18, 459–466.
- Liu, L.L.; He, J.H.; Xie, H.B.; Yang, Y.S.; Li, J.C.; Zou, Y. Resveratrol induces antioxidant and heat shock protein mRNA expression in response to heat stress in black-boned chickens. Poult. Sci. 2014, 93, 54–62.
- He, S.; Chen, L.; He, Y.; Chen, F.; Ma, Y.; Xiao, D.; He, J. Resveratrol alleviates heat stress-induced impairment of intestinal morphology, barrier integrity and inflammation in yellow-feather broilers. Anim. Prod. Sci. 2020, 60, 1547.
- Zhang, C.; Chen, K.; Zhao, X.; Geng, Z. Protective effects of resveratrol against high ambient temperature-induced spleen dysplasia in broilers through modulating splenic redox status and apoptosis. J. Sci. Food Agric. 2018, 98, 5409–5417.
- He, S.; Yu, Q.; He, Y.; Hu, R.; Xia, S.; He, J. Dietary resveratrol supplementation inhibits heat stress-induced high-activated innate immunity and inflammatory response in spleen of yellow-feather broilers. Poult. Sci. 2019, 98, 6378–6387.
- Rietschel, E.T.; Kirikae, T.; Schade, F.U.; Mamat, U.; Schmidt, G.; Loppnow, H.; Ulmer, A.J.; Zahringer, U.; Seydel, U.; Di Padova, F.; et al. Bacterial endotoxin: Molecular relationships of structure to activity and function. FASEB J. 1994, 8, 217–225.
- Zhong, L.M.; Zong, Y.; Sun, L.; Guo, J.Z.; Zhang, W.; He, Y.; Song, R.; Wang, W.M.; Xiao, C.J.; Lu, D. Resveratrol inhibits inflammatory responses via the mammalian target of rapamycin signaling pathway in cultured LPS-stimulated microglial cells. PLoS ONE 2012, 7, e32195.
- Lopes Pinheiro, D.M.; Sales de Oliveira, A.H.; Coutinho, L.G.; Fontes, F.L.; de Medeiros Oliveira, R.K.; Oliveira, T.T.; Fonseca Faustino, A.L.; da Silva, V.L.; Araujo de Melo Campos, J.T.; Petta Lajus, T.B.; et al. Resveratrol decreases the expression of genes involved in inflammation through transcriptional regulation. Free Radic. Biol. Med. 2019, 130, 8–22.
- Panaro, M.A.; Carofiglio, V.; Acquafredda, A.; Cavallo, P.; Cianciulli, A. Anti-inflammatory effects of resveratrol occur via inhibition of lipopolysaccharide-induced NF-κB activation in Caco-2 and SW480 human colon cancer cells. Br. J. Nutr. 2012, 108, 1623–1632.
- Palomera-Avalos, V.; Grinan-Ferre, C.; Izquierdo, V.; Camins, A.; Sanfeliu, C.; Canudas, A.M.; Pallas, M. Resveratrol modulates response against acute inflammatory stimuli in aged mouse brain. Exp. Gerontol. 2018, 102, 3–11.
- Sadeghi, A.; Ebrahimi, S.S.S.; Golestani, A.; Meshkani, R. Resveratrol Ameliorates Palmitate-Induced Inflammation in Skeletal Muscle Cells by Attenuating Oxidative Stress and JNK/NF-kappa B Pathway in a SIRT1-Independent Mechanism. J. Cell. Biochem. 2017, 118, 2654–2663.
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616.
- Tsai, S.H.; Lin-Shiau, S.Y.; Lin, J.K. Suppression of nitric oxide synthase and the down- regulation of the activation of NF-kappa B in macrophages by resveratrol. Br. J. Pharmacol. 1999, 126, 673–680.
- Das, A. Heat stress-induced hepatotoxicity and its prevention by resveratrol in rats. Toxicol. Mech. Methods 2011, 21, 393–399.
- Babu, D.; Leclercq, G.; Goossens, V.; Remijsen, Q.; Vandenabeele, P.; Motterlini, R.; Lefebvre, R.A. Antioxidant potential of CORM-A1 and resveratrol during TNF-alpha/cycloheximide-induced oxidative stress and apoptosis in murine intestinal epithelial MODE-K cells. Toxicol. Appl. Pharmacol. 2015, 288, 161–178.
- Kortam, M.A.; Ali, B.M.; Fathy, N. The deleterious effect of stress-induced depression on rat liver: Protective role of resveratrol and dimethyl fumarate via inhibiting the MAPK/ERK/JNK pathway. J. Biochem. Mol. Toxicol. 2020, 1–11.
- Ma, C.; Wang, Y.; Dong, L.; Li, M.; Cai, W. Anti-inflammatory effect of resveratrol through the suppression of NF-kappa B and JAK/STAT signaling pathways. Acta Biochim. Biophys. Sin. 2015, 47, 207–213.





