
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Anna Wawrzyńska | + 4524 word(s) | 4524 | 2020-04-20 12:01:01 | | | |
| 2 | Rita Xu | -1783 word(s) | 2741 | 2020-04-21 09:15:02 | | | | |
| 3 | Rita Xu | -3 word(s) | 2738 | 2020-10-29 09:17:39 | | | | |
| 4 | Rita Xu | + 1636 word(s) | 4374 | 2020-11-06 10:12:47 | | |
Video Upload Options
Plants are able to synthesize all essential metabolites from minerals, water, and light to complete their life cycle. This plasticity comes at a high energy cost, therefore plants need to tightly allocate resources in order to control their economy. Being sessile, plants can only adapt to fluctuating environmental conditions, relying on quality control mechanisms. Remodeling of cellular components plays a crucial role, not only in response to stress, but also in normal plant development. Dynamic protein turnover is ensured through regulated protein synthesis and degradation processes. To effectively target a wide range of proteins for degradation, plants utilize ubiquitination as an essential signal of substrate recognition for the 26S proteasome. Recent progress has been made in understanding the cellular homeostasis of iron. In this review, we highlight the latest publications elucidating the role of Ubiquitin-Proteasome System (UPS) in control of iron metabolism during plant development, as well as environmental stresses.
1. Introduction
Because plants are sessile and often face biotic and abiotic stresses, including fluctuating availability of nutrients, drought, and diseases, their ability to thrive in a dynamic environment is a biochemical feat. Nonetheless, this plasticity comes at a high price. Plants are continuously pressured to recognize and allocate adequate resources to metabolic processes such as growth, development, and the assimilation of nutrients. They also need to determine when to slow or even stop development and growth if resources are low (i.e, nutrient restriction) and if they need to divert resources to support other essential processes, such as defense [1][2]. Plants have developed mechanisms to sense the availability of nutrients and, consequently, regulate their growth and development. They are also able to store excess nutrients and diminish their uptake if storage capacity is exceeded, to prevent the accumulation of nutrients to toxic levels (for reviews, see [3][4]). This is of particular importance for reactive elements such as iron and copper, which are required in relatively small amounts (micromolar range), but can become toxic at a relatively low level (submillimolar ranges). It has become clear in recent years that there is active crosstalk between networks regulating the uptake and use of nutrients [5][6][7].
Homeostasis is a term that has been used for nearly a century; it explains processes used by systems to sustain survival conditions [8]. Optimal growth and development are only achieved when nutrients are present at certain concentrations, i.e., when the demand meets the supply. Below such levels, the demand for a nutrient exceeds the supply, so plant growth is ultimately limited by a nutritional deficiency. Conversely, an excess of any nutrient is toxic to plants and also negatively affects plant growth and development. Plants can therefore experience transitions between deficiency or toxicity zones due to changes in the availability of nutrients and growth requirements, but they can return to homeostasis so long as the response is adequate and sufficient. Such adjustments may require time, but they are crucial for plant adaptation. A significant part of this adaptation relies on selective protein degradation (Figure 1).
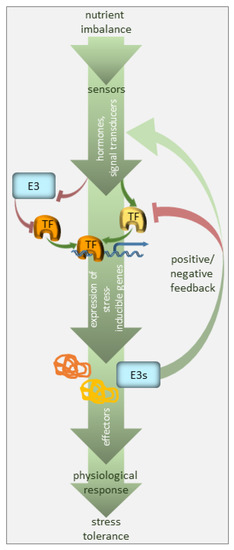
Figure 1. Regulation of nutrient deficiency stress by ubiquitination. The plant perceives nutrient deficiency via sensors (not defined in the model) and the signal is transduced via phytohormones, secondary messengers, and transcriptional regulators. The transcription factors (TF), many of which are stress-regulated, facilitate the expression of stress-inducible genes. E3 ubiquitin ligases target the components of the signaling pathway, i.e., mainly stress-responsive TFs, promoting their degradation, and thus, suppressing the pathway in the absence of the signal. E3 ubiquitin ligases may also serve as a feedback mechanism to enhance or silence the stress signal.
Protein turnover has been estimated to account for up to one-third of the total energy cost in fast-growing cells [9]. Consequently, an immediate cell reaction to various stresses, including nutrient starvation, is translation arrest. This thereby frees energy and resources for stress responses. A small, highly-conserved 76-amino-acid protein called ubiquitin is a tag marking soluble proteins for degradation. The process of ubiquitination requires the sequential action of three enzymes, i.e., E1, E2, and E3 [10] (Figure 2). In the first step of this enzymatic cascade, ubiquitin is bound to a cysteine residue of the activating enzyme (E1) through its C-terminal glycine, forming a thioester bond. Subsequently, activated ubiquitin is transferred to the conjugating enzyme (E2), also via a thioester bond. A member of the ubiquitin ligase family (E3) then recognizes the substrate for ubiquitination and catalyzes the transfer of ubiquitin from the E2 enzyme to the substrate protein. The cascade of ubiquitination has to be highly selective, and its accuracy depends on a sufficient number of E3 enzymes; hence, E3 ubiquitin ligases represent the largest protein family in plants. Plants use two mechanistically distinct but largely complementary systems to selectively target a wide range of proteins for degradation: the ubiquitin-proteasome system (UPS) and selective autophagy. The concerted action of the ubiquitination machinery in both systems ensures the targeted and tightly regulated degradation of cellular proteins. These degradation mechanisms can be perceived as a housecleaning process, where old, damaged, or no-longer-needed proteins, polypeptides, organelles, lipids, and other cellular components are recycled and reused. However, they are also of crucial importance when growth conditions get tough and metabolism remodeling is urgently needed. The UPS and autophagy are differentially regulated and usually degrade different protein sets. Proteins marked by K48-linked polyubiquitin are commonly believed to have a high affinity for the 26S proteasome [11], whereas those with a K63-polyubiquitin or monoubiquitin tag are preferred substrates for autophagy clearance [12][13][14] (Figure 2). The two systems of protein degradation are not fully independent; moreover, their functions must be coordinated, since both pathways are responsible for homeostasis of cellular proteins.
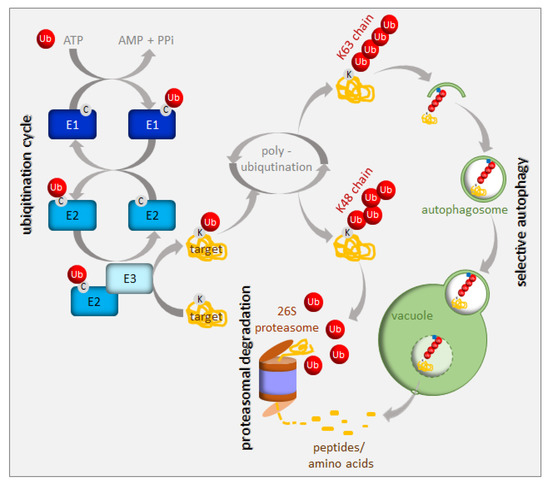
Figure 2. Ubiquitination and degradation of ubiquitinated proteins. Ubiquitin (Ub) is activated and conjugated to the target protein by a series of E1 (ubiquitin-activating enzyme), E2 (ubiquitin-conjugating enzyme), and E3 (ubiquitin ligase) activities. Additional ubiquitin molecules can be added to the monoubiquitinated target (polyubiquitination). The polyubiquitinated proteins are recruited either to the 26S proteasome or to the autophagy pathway depending on their polyubiquitin chain structure. See the text for more details.
The UPS is responsible for the rapid destruction of a wide range of intracellular regulators (usually short-lived proteins) via the 26S proteasome. Therefore, the UPS plays an important role in cell signaling, hormone biogenesis and responses, chromatin structure regulation and transcription, and even pathogenic infections (for a review see [15]). The 26S proteasome is a complex of ATP-dependent proteases found in the cytoplasm and the nucleus [16]. Autophagy is a distinct degradation pathway which transports a set of highly heterogeneous cargo to the vacuole in specific vesicles, called autophagosomes [17]. In plants, they are fused with the vacuole, where cargo becomes degraded and its primary molecules are recycled [18]. While general autophagy randomly engulfs parts of the cytosol, selective autophagy uses special cargo adaptors in response to various intra- or extra- cellular signals, to enrich the forming autophagosomes for a certain type of cargo [19][20][21]. Selective autophagy aims at a broad range of cargo, including long-lived proteins, protein complexes, organelles, and protein aggregates. The analysis of selective autophagy in plants is greatly facilitated by the functional and structural conservation of autophagy-related (ATG) proteins that were initially identified in yeast [22][23][24]. About 40 homologs of 18 yeast genes involved in the core autophagy machinery have been identified in Arabidopsis thaliana [19][20][21][25]. All autophagy-deficient plants exhibit hypersensitivity to carbon and nitrogen starvation, pointing to a central role for autophagy in nutrient recycling [26][27]. The upregulation of ATG genes during leaf senescence in Arabidopsis suggests a role for autophagy in nutrient recycling at the end of plant life [21][26][28][29][30]. Arabidopsis contains nine highly-conserved ATG8 proteins that, after processing, coat the autophagosomal membranes and serve as a docking platform for autophagy receptors that selectively recognize and bind the cargo designated for degradation [29][31]. Well-known examples of selective autophagy cargo receptors in mammals include p62 (also named SQSTM1 or sequestosome-1, A170, or ZIP) and NBR1 (neighbor of BRCA1 gene 1), which are both primarily involved in protein aggregate degradation [32][33][34][35]. The NBR1-like selective autophagy cargo receptors exist in plants as well [36][37], but not in yeast. The tobacco Joka2 and Arabidopsis NBR1 proteins are larger than their animal p62 or NBR1 counterparts, but they share a common domain structure, including the UBA domain at the C-terminus, which enable them to bind ubiquitinated proteins. The exact cargo for plant NBR1-like proteins is unknown, and their selectivity may be mediated by ubiquitin recognition and not by specific protein substrates, as is the case in mammals.
Since most of the metabolically-active iron is bound to sulfur in Fe–S clusters, the coordination between metabolisms of the two nutrients is strongly suggested [5][38]. There is physiological and molecular evidence for such crosstalk in different plant species, which additionally suggests that it seems to be species specific [38]. Grasses (Strategy II plants) use the chelating strategy for iron uptake requiring the synthesis of phytosiderophores [39]. Phytosiderophores are derived from nicotianamine synthesized from three S-adenosyl-methionine molecules; thus, there is a need for a well-balanced sulfur metabolism. Iron deficiency in wheat causes the induction of most of the genes of the sulfur assimilatory pathway despite sufficient sulfur supply, suggesting the connection between sulfur and iron metabolism and the necessity of upregulation of sulfur assimilation to increase the synthesis of phytosiderophores [40][41]. Similarly, under sulfur deficiency, the release of phytosiderophores was reduced; however, when barley plants were resupplied with sulfate, the release of phytosiderophores was enhanced [42]. In dicots, sulfur deficiency conditions render plants unable to fully induce their iron uptake machinery, while under iron limitation, the sulfite reduction is stopped [6][7]. Transcriptomic analyses of 5-week iron starved Arabidopsis roots indicated a downregulation of genes of sulfate assimilation [43]. Also, the vacuolar sulfate exporters were induced in leaves, which was interpreted as a necessity of rebalancing the sulfur metabolism under these conditions [44]. Zuchi et al. (2009) [45] showed that in tomato plants exposed to both sulfur and iron starvation, there is reduced activity of iron transporters, which suggests that sulfur deficiency prevents the typical responses to iron deficiency. However, it was also recently shown that iron limitation strongly reduced total sulfur content in both shoots and roots of tomato plants, leading to an increased transcription of sulfate transporters [6]. Altogether, these findings point to coregulation between the two pathways as one nutrient limitation affects the other’s uptake. Nonetheless, these results are based mostly on the changes in gene expression representing only one side of the coin. In both iron and sulfur metabolisms, there are many posttranscriptional regulatory mechanisms that can modulate the nutrient deficiency responses. In this review, we gathered examples of proteins involved in either iron or sulfur metabolism that are the targets of selective degradation. Those proteins have to be removed after specific intra- or extra- cellular cues to reprogram plant metabolism and sustain homeostasis. Although there are ample examples of such proteins for iron metabolism, data for sulfur metabolism is rather scarce.
2. UPS in the Regulation of Iron Homeostasis
Iron is an essential nutrient, facilitating photosynthesis, chlorophyll biosynthesis, and a variety of redox reactions. Conversely, homeostasis must be tightly controlled, between uptake, use, and storage of this nutrient, because iron excess inside living cells can be dangerous due to its redox properties. Iron is often present in soils as precipitates that are not readily available for plants. To solubilize iron, plants have evolved various mechanisms to increase iron availability. Arabidopsis responds to iron-limiting conditions by upregulating the expression of basic helix-loop-helix (bHLH) transcription factors, such as Fer-like Iron Deficiency-Induced Transcription Factor (FIT), Popeye (PYE), and PYE homolog IAA-Leu Resistant-3 (ILR3). These transcription factors induce the expression of genes that are needed to increase iron availability and sustain iron homeostasis [46][47][48][49][50]. PYE interacts with additional PYE-like proteins induced during iron-limiting conditions, including bHLH104, bHLH115, and ILR3 (bHLH105) [49][51]. PYE-like proteins are controlled by BRUTUS (BTS), which is an iron-binding E3 ligase found in green algae and plants, and is also induced under iron-limiting conditions [52][53][54] (Figure 1). Ubiquitination activity, together with susceptibility to proteasomal degradation, was proven for BTS [52]. In contrast to pye-1, a BTS partial loss of function mutation rendered plants more tolerant to iron deficiency, suggesting that BTS is a negative regulator of the iron deficiency response, and that it targets PYE-like proteins for degradation, thus damping the original response to iron deficiency. It should also be noted that the complete loss of function of BTS is lethal under normal growing conditions, which suggests that function of BTS is not limited to iron starvation [55]. Surprisingly, BTS does not interact with PYE directly, but instead, interacts with ILR3, a potential PYE dimerizing partner (Figure 1). During iron-deficient stress, BTS can influence the stability of ILR3, and consequently, indirectly influence PYE activity [49]. BTS is fascinating in its structure. In addition to being an E3 ligase, it also contains three hemerythrin (HHE) domains that can bind iron [53]. This domain is suggested to be critical for sensing whether the iron level is sufficiently restored after the induction of iron uptake machinery [54]. Once activated, BTS targets the positive regulators of iron deficiency response in order to avoid iron overload. Recently, an additional allele of BTS (bts-3) was characterized [56]. Bts-3 mutant overaccumulates iron and other transition elements, in roots, leaves, and seeds, due to constitutive activation of the iron regulon in roots, thus validating the role of BTS as a negative regulator of the response to iron deficiency. However, the constitutive activation of the iron regulon in bts-3 adds up to only one-fifth of the proper wild type response to iron limitation; more significantly, bts-3 was unable to activate the iron regulon under prolonged iron depletion [56]. These data suggest that there may be unidentified negative regulator(s), on top of the BTS network, controlling plant responses when iron becomes scarce. Similar regulation can be observed in rice, where the BTS ortholog is called Hemerythrin motif-containing Really interesting new gene and Zinc-finger protein 1 (OsHRZ1) [52]. OsHRZ1 is thought to negatively regulate the iron limitation response by controlling the activity of OsIRO3 (the rice PYE orthologue), which, in turn, coordinates the expression of the bHLH transcription factor gene, OsIRO2, and the synthesis of nicotianamine. Very recently, another factor from the bHLH family, OsbHLH058, was found to interact with OsHRZ1 and OsHRZ2; however, this interaction was not due to ubiquitination [57]. The phenotypes of overexpressors and knock-out lines proved that OsbHLH058 positively regulates major iron deficiency responses.
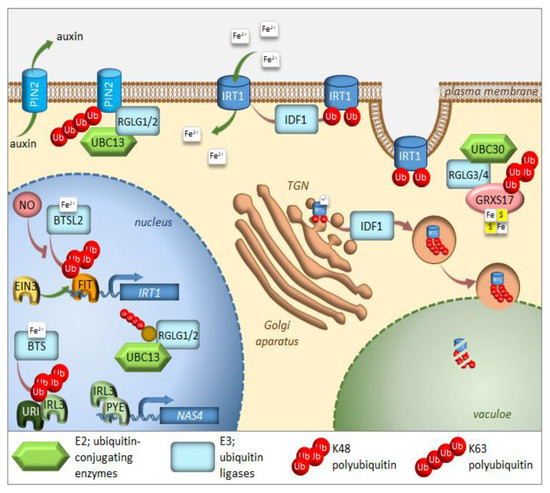
Figure 1. Regulation of iron homeostasis through selective protein degradation. During iron deficiency, FIT induces transcription of the IRT1 iron plasma membrane transporter. EIN3 interacts with FIT and stabilizes its function. IRL3 interacts with PYE to induce transcription of iron-deficiency induced genes, such as NAS4. RGLG1/2 ubiquitin ligases are kept within the nucleus, where they interact with UBC13 and target an unknown protein. Under iron-sufficient conditions, after sensing iron availability, two E3 ligases, BTS, and BTSL2 target IRL3 and FIT, respectively, for proteasomal degradation. Through heterodimerization with IRL3, the URI transcription factor responsible for the induction of many genes under iron deficiency, also undergoes proteasomal degradation. IDF1 monoubiquitinates IRT1 at the plasma membrane, causing its endocytosis and eventual targeting to the trans-Golgi network (TGN). There, IRT1, which is able to sense the intracellular iron availability, might be sent to the vacuole for degradation after K63 polyubiqutination via IDF1. RGLG1/2 ubiquitin ligases move from the nucleus to the cytoplasm of root cells, where they target the PIN2 auxin carrier for degradation, thus preventing branching of root hairs. RGLG3/4 ubiquitin ligases together with UBC30 cause K48 polyubiquitination of GRXS17, which takes part in the assembly of Fe–S clusters. Further details and full gene names can be found in the main text.
The transcription factor FIT constitutes the most upstream factor in the iron-deficiency signaling pathway. It directly induces the expression of Ferric Reduction Oxidase 2 (FRO2) and Iron-Regulated Transporter 1 (IRT1), the root iron uptake machinery genes [46]. The stability of the FIT protein appeared to be regulated in an iron-dependent manner, with iron deficiency triggering a roughly fivefold decrease in FIT stability [58]. Furthermore, it was proven that the FIT protein is degraded through UPS; however, the E3 ligase specifically recognizing FIT for ubiquitination is still missing. Very recently, FIT was shown to be a ubiquitination target of BRUTUS-like 2 (BTSL2) by an in vitro ubiquitination assay; however, this assay lacked proper controls [54] (Figure 1). The ubiquitination and degradation of FIT are thought necessary to maintain the activity of this regulator due to scavenging of the poorly functional older molecules of this protein from its target promoters [58]. A model was proposed where BTSL, predominantly expressed in the root epidermis and cortex, acts as a primary defense mechanism against excess iron uptake in the roots. The BTS expressed in the stele and leaf tissues, behind the Casparian strip, regulates a second defense mechanism against iron overload by targeting IRL3 [54]. Interestingly, nitric oxide was found as a signal that promoted not only the activation of FIT, but also its stabilization by counteracting proteasomal degradation [59]. Additionally, a link between the iron-deficiency stress response and ethylene signaling has shown UPS involvement in controlling FIT protein levels [50]. Inhibition of ethylene biosynthesis by aminoethoxyvinylglycin (AVG), during the treatment of iron deficiency stress, prevents FIT accumulation, indicating that ethylene signaling is required for stabilization of the FIT protein [50]. FIT interacts with the ethylene signaling transcription factors, Ethylene Insensitive 3 (EIN3) and EIN3-Like1 (EIL1) (Figure 1). This interaction has been shown to reduce FIT degradation and to maintain FIT in an active state, therefore, promoting iron uptake [50]. The loss of function of EIN3 and EIL1 led to a significant reduction of FIT accumulation, even under iron deficiency conditions. Ethylene stimulates FIT transcription, which can partly be caused by FIT itself because it is needed to induce its own gene [50][60][61]. Mediator, a large protein complex coordinating the transcription process, has been identified to be involved in the regulation of gene expression during iron deficiency [62]. One of the mediator subunits, MED16, interacts with FIT and recruits it to its promoters to enhance the expression of FIT-dependent genes. Additionally, MED16 was shown to associate with EIN3/EIL1 proteins through subunit MED25, which may play some roles in FIT stabilization [63]. Among other genes, FIT regulates the expression of transcription factor MYB72, which has been implicated in iron redistribution through the regulation of nicotianamine synthase 4 (NAS4) [58][64]. More recently, MYB72 has been shown to control the synthesis of another type of iron-chelating molecules, known as coumarins, more specifically scopoletin [65]. Interestingly, MYB72 was also found to interact physically with Sulfur LIMitation 1 (SLIM1) [66], one of the very few transcription factors known to regulate sulfur metabolism. How the interaction between MYB72 and SLIM1 impacts iron and/or sulfur homeostasis is currently unknown; however, one can envision a similar mechanism to the interaction observed in EIN3 and FIT. SLIM1 belongs to the same protein family as EIN3 and its degradation through proteasomes has recently been suggested [67].
A short time ago, another piece was added to the iron deficiency response puzzle [68]. Upstream Regulator of IRT1 (URI), basic helix-loop-helix transcription factor, appears to have a central role as an iron-dependent switch. When plants become iron deficient, a phosphorylated form of URI accumulates and induces numerous iron deficiency-induced genes. Under iron resupply, the phosphorylated URI undergoes proteasomal degradation due to heterodimer formation with PYE-like proteins, whose degradation is controlled by E3 ligase BTS. This feedback regulation prevents the overaccumulation of iron and maintains iron homeostasis. As the main iron importer and due to its affinity for other divalent metals, IRT1 is subjected to tight regulation at the transcriptional and posttranscriptional level [69][70][71] (Figure 3). It was surprising to find that IRT1, as a transmembrane protein, localizes predominantly to intracellular compartments, accumulating mainly at the trans-Golgi network (TGN) [72][73]. This demonstrates the importance of intracellular trafficking for iron uptake. In roots, the large cytosolic histidine-rich loop in IRT1 can detect the level of transition elements available for uptake, including iron [74]. Under iron sufficiency, IRT1 employs the machinery to trigger its monoubiquitination, followed by removal from the plasma membrane to be sent back to the TGN [72]. Variants of IRT1, where the ubiquitinatable lysine residues (K154 and K179) were mutated to arginines, caused stabilization of IRT1 and its overaccumulation at the plasma membrane, leading to uncontrolled metal uptake and, subsequently, to plant death [72][75]. The RING-type E3 ubiquitin ligase IRT1 Degradation Factor 1 (IDF1) ubiquitinates IRT1 and, as a result, is depleted from the plasma membrane [76] (Figure 1). Once internalized at the TGN, IRT1 is either reused and sent back to the plasma membrane or targeted for degradation. Interestingly, antibodies against IRT1, demonstrated on immunoblots, that four attached ubiquitin moieties exist in the predominant ubiquitinated form [76]. This suggests that monoubiquitination of K154 and K179 is the signal of IRT1 internalization and that at least two additional lysines might be marked by ubiquitin for further processing in the endomembrane system. The existence of pools of IRT1 decorated with different numbers of monoubiquitination residues may also represent a hierarchy in ubiquitinated residues. Ubiquitin-binding proteins may selectively recognize different monoubiquitinated regions of IRT1, thus controlling the destination of IRT1 along the endocytic pathway. Very recently, it was uncovered that IRT1 acts as a transporter and receptor (transceptor) and is able to directly sense the excess of non-iron metal substrates in the cytoplasm, and subsequently regulates its own degradation [77]. Direct metal binding to a histidine-rich stretch in IRT1 triggers its phosphorylation by the CIPK23 kinase. This facilitates the subsequent recruitment of the IDF1 for K63 polyubiquitination (Figure 1). Non-iron metal excess not only increased the pool of ubiquitinated IRT1 in the cell, but also resulted in a qualitative shift in the type of ubiquitination. Immunoblot analyses revealed that IRT1 is decorated with K63-linked ubiquitin chains that have previously been associated with endocytosis and autophagy [13][78][79]. The authors clearly showed that K63 polyubiquitinated IRT1 is trafficked to the vacuole (Figure 1). This feedback-mediated autoregulation of IRT1 represents a form of iron sensing and is definitely the first line of defense in avoiding an excess of iron or other IRT1 substrates like manganese, zinc, cobalt, and the non-essential element cadmium.
The E2 ubiquitin conjugase UBC13, the only E2 protein that is capable of catalyzing the formation of non-canonical K63-linked ubiquitin chains, has recently been linked to the response of Arabidopsis to iron deficiency. When adapting to reduced iron availability, Arabidopsis develops root hairs that are branched at the base [80]. Mutations in UBC13A abolished the branched root hair phenotype. Interestingly, mutations in RING Domain Ligase 1/2 genes (RGLG1/2), encoding UBC13 interacting E3 ubiquitin ligases, caused constitutive root hair branching [81]. A model for the function of K63-linked ubiquitination in root hair cells was proposed [82] (Figure 1). Under iron-sufficient conditions, RGLG1/2 targets a protein that acts as an inhibitor of proper root hair initiation, putatively via an auxin responsiveness reduction. This protein may act directly or indirectly on auxin distribution. In iron-deficient roots, RGLG1/2 moves from the plasma membrane to the nucleus, where it interacts with UBC13, thus releasing its targeted protein [83]. This may result in a decreased auxin concentration. The authors speculate further that that protein could be PIN-Formed 2 (PIN2), a plasma membrane-localized auxin carrier protein. RGLG proteins were actually shown to be involved in the proteolytic turnover of PIN2 via K63-linked ubiquitination [84] (Figure 1). The loss of PIN2 ubiquitination interferes with vacuolar targeting, stabilizes PIN2, decreases auxin levels in roots and, ultimately, leads to the branching of root hairs.
Other members of the RGLG family, RGLG3/4, were recently shown to target monothiol glutaredoxin, GRXS17, for proteasomal degradation [85]. The authors identified the cognate E2 enzyme as UBC30, which is closely related to human HsUbcH5b and, therefore, is probably involved in K48 polyubiquitination (Figure 1). GRXs are present in most organisms and their main function is to control the redox state of proteins. GRXS17 was shown to be necessary for temperature stress and auxin perception [86]. Although none of these functions directly link GRXS17 with iron homeostasis, multidomain GRXs are involved in iron signaling and distribution in yeast. Grx3 and Grx4, the yeast orthologs of GRXS17, can inhibit iron-responsive gene expression, by complexing with the iron-responsive transcription factor activator of ferrous transport 1, RCS1 [87]. The domain architecture of Arabidopsis GRXS17 resembles that of its closest human ortholog HsPICOT, which is involved in the regulation of iron homeostasis, as well as assembly and trafficking of iron-sulfur (Fe–S) clusters [88]. In another paper from this group, Inigo et al. proved that GRXS17 associates with most known cytosolic Fe–S assembly components, putatively to function as a [2Fe-2S] specific adaptor for this complex [89].
The COP9 signalosome (CSN) is an eight-subunit complex that regulates the activity of CULLIN-RING E3 ubiquitin ligases (CRLs) [90]. Recently, CSN has been proven to regulate the proteasome-mediated degradation of iron deficiency-inducible transcription factor, IDEF1, in rice [91]. The decrease in CSN activity followed the accumulation of IDEF1 in the early stages of iron deficiency. Bioinformatic analysis of existing microarray datasets for a set of iron-deficiency-responsive genes was conducted against the transcriptome of Arabidopsis mutants, which are lacking a functional CSN [92]. Hundreds of iron-deficiency-responsive genes appeared to be misregulated, underlying the fact that the correct transcriptional response to iron deficiency requires an intact COP9 signalosome in Arabidopsis and a properly functioning UPS.
In apple (Malus domestica), a bHLH transcription factor, MdbHLH104, has been found to function in iron acquisition by inducing the transcription of MdAHA8, thus modulating the activity of plasma membrane H+-ATPases that mediate rhizosphere acidification and iron uptake under iron-deficient conditions [93]. Two MdbHLH104-interacting BTB-TAZ proteins, MdBT1 and MdBT2, were next identified by screening with a yeast two-hybrid method [93]. Subsequently, their role in the ubiquitin-mediated degradation of the MdbHLH104 protein was proved, especially under conditions of iron surplus. MdBT proteins, that accumulate under oversupply of iron, interact with cullin MdCUL3 to bridge the formation of the E3 ligase complex and negatively regulate iron uptake. Conversely, MdbHLH104 is a direct target of the SUMO E3 ligase, MdSIZ1 [94]. Sumolyation, in most cases, serves to antagonize the effects of ubiquitination on target proteins [95]. MdbHLH104 was sumoylated at positions K139 and K153, especially under conditions of iron deficiency, and this modification was required for MdbHLH104 protein stability. Moreover, sumolyation seems to be superior to ubiquitination because MdSIZ1-mediated sumoylation of MdbHLH104 inhibited its ubiquitination. The sumoylation of MdbHLH104 promoted the expression of MdbHLH38 and MdbHLH39 and activated the MdFRO2 and MdIRT1 genes encoding iron transporters. Therefore, stabilized MdbHLH104 enhances H+-ATPase-mediated rhizosphere acidification, but also plays a role in the reduction and absorption of iron to improve tolerance toward iron deficiency in apple plants. The ubiquitination/sumolyation regulatory module of MdbHLH104 serves to sustain iron homeostasis.
References
- Katsir, L.; Chung, H.S.; Koo, A.J.; Howe, G.A. Jasmonate signaling: a conserved mechanism of hormone sensing. Curr. Opin. Plant Biol. 2008, 11, 428–435.
- Yang, D.L.; Yao, J.; Mei, C.S.; Tong, X.H.; Zeng, L.J.; Li, Q.; Xiao, L.T.; Sun, T.P.; Li, J.; Deng, X.W.; et al. Plant hormone jasmonate prioritizes defense over growth by interfering with gibberellin signaling cascade. Proc. Natl. Acad. Sci. USA 2012, 109, E1192–CE1200.
- Mendoza-Cozatl, D.G.; Jobe, T.O.; Hauser, F.; Schroeder, J.I. Long-distance transport, vacuolar sequestration, tolerance, and transcriptional responses induced by cadmium and arsenic. Curr. Opin. Plant Biol. 2011, 14, 554–562.
- Jeong, J.; Merkovich, A.; Clyne, M.; Connolly, E.L. Directing iron transport in dicots: regulation of iron acquisition and translocation. Curr. Opin. Plant Biol. 2017, 39, 106–113.
- Forieri, I.; Wirtz, M.; Hell, R. Toward new perspectives on the interaction of iron and sulfur metabolism in plants. Front. Plant Sci. 2013, 4, 357.
- Zuchi, S.; Watanabe, M.; Hubberten, H.M.; Bromke, M.; Osorio, S.; Fernie, A.R.; Celletti, S.; Paolacci, A.R.; Catarcione, G.; Ciaffi, M.; et al. The interplay between sulfur and iron nutrition in tomato. Plant Physiol. 2015, 169, 2624–2639.
- Hantzis, L.J.; Kroh, G.E.; Jahn, C.E.; Cantrell, M.; Peers, G.; Pilon, M.; Ravet, K. A program for iron economy during deficiency targets specific Fe proteins. Plant Physiol. 2018, 176, 596–610.
- Cannon, W.B. The wisdom of the body. W.W. Norton & Company: New York, USA, 1932; pp. 19-312.
- Lahtvee, P.J.; Seiman, A.; Arike, L.; Adamberg, K.; Vilu, R. Protein turnover forms one of the highest maintenance costs in Lactococcus lactis. Microbiology 2014, 160, 1501–1512.
- Lilienbaum, A. Relationship between the proteasomal system and autophagy. Int. J. Biochem Mol. Biol. 2013, 4, 1–26.
- Baboshina, O.V.; Haas, A.L. Novel multiubiquitin chain linkages catalyzed by the conjugating enzymes E2EPF and RAD6 are recognized by 26 S proteasome subunit 5. J. Biol. Chem. 1996, 271, 2823–2831.
- Tan, J.M.; Wong, E.S.; Kirkpatrick, D.S.; Pletnikova, O.; Ko, H.S.; Tay, S.P.; Ho, M.W.; Troncoso, J.; Gygi, S.P.; Lee, M.K.; et al. Lysine 63-linked ubiquitination promotes the formation and autophagic clearance of protein inclusions associated with neurodegenerative diseases. Hum. Mol. Genet. 2008, 17, 431–439.
- Ikeda, F.; Crosetto, N.; Dikic, I. What determines the specificity and outcomes of ubiquitin signaling? Cell 2010, 143, 677–681.
- Kim, H.P.; Wang, X.; Chen, Z.H.; Lee, S.J.; Huang, M.H.; Wang, Y.; Ryter, S.W.; Choi, A.M. Autophagic proteins regulate cigarette smoke-induced apoptosis: protective role of heme oxygenase-1. Autophagy 2008, 4, 887–895.
- Kurepa, J.; Wang, S.; Li, Y.; Smalle, J. Proteasome regulation, plant growth and stress tolerance. Plant Signal. Behav. 2009, 4, 924–927.
- Wojcik, C.; DeMartino, G.N. Intracellular localization of proteasomes. Int. J. Biochem. Cell Biol. 2003, 35, 579–589.
- Yorimitsu, T.; Klionsky, D.J. Atg11 links cargo to the vesicle-forming machinery in the cytoplasm to vacuole targeting pathway. Mol. Biol. Cell 2005, 16, 1593–1605.
- Klionsky, D.J. Autophagy. Curr. Biol. 2005, 15, R282–CR283.
- Yoshimoto, K.; Hanaoka, H.; Sato, S.; Kato, T.; Tabata, S.; Noda, T.; Ohsumi, Y. Processing of ATG8s, ubiquitin-like proteins, and their deconjugation by ATG4s are essential for plant autophagy. Plant Cell 2004, 16, 2967–2983.
- Hanaoka, H.; Noda, T.; Shirano, Y.; Kato, T.; Hayashi, H.; Shibata, D.; Tabata, S.; Ohsumi, Y. Leaf senescence and starvation-induced chlorosis are accelerated by the disruption of an Arabidopsis autophagy gene. Plant Physiol. 2002, 129, 1181–1193.
- Doelling, J.H.; Walker, J.M.; Friedman, E.M.; Thompson, A.R.; Vierstra, R.D. The APG8/12-activating enzyme APG7 is required for proper nutrient recycling and senescence in Arabidopsis thaliana. J. Biol. Chem. 2002, 277, 33105–33114.
- Reumann, S.; Voitsekhovskaja, O.; Lillo, C. From signal transduction to autophagy of plant cell organelles: lessons from yeast and mammals and plant-specific features. Protoplasma 2010, 247, 233–256.
- Diaz-Troya, S.; Perez-Perez, M.E.; Florencio, F.J.; Crespo, J.L. The role of TOR in autophagy regulation from yeast to plants and mammals. Autophagy 2008, 4, 851–865.
- Yoshimoto, K.; Takano, Y.; Sakai, Y. Autophagy in plants and phytopathogens. FEBS Lett. 2010, 584, 1350–1358.
- Xiong, Y.; Contento, A.L.; Bassham, D.C. AtATG18a is required for the formation of autophagosomes during nutrient stress and senescence in Arabidopsis thaliana. Plant J. 2005, 42, 535–546.
- Liu, Y.; Bassham, D.C. Autophagy: pathways for self-eating in plant cells. Annu Rev. Plant Biol. 2012, 63, 215–237.
- Guiboileau, A.; Avila-Ospina, L.; Yoshimoto, K.; Soulay, F.; Azzopardi, M.; Marmagne, A.; Lothier, J.; Masclaux-Daubresse, C. Physiological and metabolic consequences of autophagy deficiency for the management of nitrogen and protein resources in Arabidopsis leaves depending on nitrate availability. New Phytol. 2013, 199, 683–694.
- van der Graaff, E.; Schwacke, R.; Schneider, A.; Desimone, M.; Flugge, U.I.; Kunze, R. Transcription analysis of arabidopsis membrane transporters and hormone pathways during developmental and induced leaf senescence. Plant Physiol. 2006, 141, 776–792.
- Chung, T.; Phillips, A.R.; Vierstra, R.D. ATG8 lipidation and ATG8-mediated autophagy in Arabidopsis require ATG12 expressed from the differentially controlled ATG12A AND ATG12B loci. Plant J. 2010, 62, 483–493.
- Breeze, E.; Harrison, E.; McHattie, S.; Hughes, L.; Hickman, R.; Hill, C.; Kiddle, S.; Kim, Y.S.; Penfold, C.A.; Jenkins, D.; et al. High-resolution temporal profiling of transcripts during Arabidopsis leaf senescence reveals a distinct chronology of processes and regulation. Plant Cell 2011, 23, 873–894.
- Thompson, A.R.; Doelling, J.H.; Suttangkakul, A.; Vierstra, R.D. Autophagic nutrient recycling in Arabidopsis directed by the ATG8 and ATG12 conjugation pathways. Plant Physiol. 2005, 138, 2097–2110.
- Kirkin, V.; Lamark, T.; Johansen, T.; Dikic, I., NBR1 cooperates with p62 in selective autophagy of ubiquitinated targets. Autophagy 2009, 5, 732–733.
- Komatsu, M.; Waguri, S.; Koike, M.; Sou, Y.; Ueno, T.; Hara, T.; Mizushima, N.; Iwata, J.; Ezaki, J.; Murata, S.; et al. Homeostatic levels of p62 control cytoplasmic inclusion body formation in autophagy-deficient mice. Cell 2007, 131, 1149–1163.
- Pankiv, S.; Clausen, T.H.; Lamark, T.; Brech, A.; Bruun, J.A.; Outzen, H.; Overvatn, A.; Bjorkoy, G.; Johansen, T. p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J. Biol. Chem. 2007, 282, 24131–24145.
- Khaminets, A.; Beh, C.; Dikic, I. Ubiquitin-dependent and independent signals in selective autophagy. Trends Cell Biol. 2016, 26, 6–16.
- Svenning, S.; Lamark, T.; Krause, K.; Johansen, T. Plant NBR1 is a selective autophagy substrate and a functional hybrid of the mammalian autophagic adapters NBR1 and p62/SQSTM1. Autophagy 2011, 7, 993–1010.
- Zientara-Rytter, K.; Lukomska, J.; Moniuszko, G.; Gwozdecki, R.; Surowiecki, P.; Lewandowska, M.; Liszewska, F.; Wawrzynska, A.; Sirko, A. Identification and functional analysis of Joka2, a tobacco member of the family of selective autophagy cargo receptors. Autophagy 2011, 7, 1145–1158.
- Mendoza-Cozatl, D.G.; Gokul, A.; Carelse, M.F.; Jobe, T.O.; Long, T.A.; Keyster, M. Keep talking: crosstalk between iron and sulfur networks fine-tunes growth and development to promote survival under iron limitation. J. Exp. Bot. 2019, 70, 4197–4210.
- Kobayashi, T.; Nishizawa, N.K. Iron uptake, translocation, and regulation in higher plants. Annu. Rev. Plant Biol. 2012, 63, 131–52.
- Ciaffi, M.; Paolacci, A.R.; Celletti, S.; Catarcione, G.; Kopriva, S.; Astolfi, S. Transcriptional and physiological changes in the S assimilation pathway due to single or combined S and Fe deprivation in durum wheat (Triticum durum L.) seedlings. J. Exp. Bot. 2013, 64, 1663–1675.
- Zamboni, A.; Celletti, S.; Zenoni, S.; Astolfi, S.; Varanini, Z. Root physiological and transcriptional response to single and combined S and Fe deficiency in durum wheat. Environ. Exp. Bot. 2017, 143, 172–184.
- Astolfi, S.; Zuchi, S.; Hubberten, H.M.; Pinton, R.; Hoefgen, R. Supply of sulphur to S-deficient young barley seedlings restores their capability to cope with iron shortage. J. Exp. Bot. 2010, 61, 799–806.
- Forieri, I.; Sticht, C.; Reichelt, M.; Grerz, N.; Hawkesford, M.J.; Malagoli, M.; Wirtz, M.; Hell, R. System analysis of metabolism and the transcriptome in Arabidopsis thaliana roots reveals differential co-regulation upon iron, sulfur and potassium deficiency. Plant Cell Environ. 2017, 40, 95–107.
- Ivanov, R.; Brumbarova, T.; Bauer, P. Fitting into the harsh reality: Regulation of iron-deficiency responses in dicotyledonous plants. Mol. Plant 2012, 5, 27–42.
- Zuchi, S.; Cesco, S.; Varanini, Z.; Pinton, R.; Astolfi, S. Sulphur deprivation limits Fe-deficiency responses in tomato plants. Planta 2009, 230, 85–94.
- Elizabeth P. Colangelo; Mary Lou Guerinot; The Essential Basic Helix-Loop-Helix Protein FIT1 Is Required for the Iron Deficiency Response. The Plant Cell 2004, 16, 3400-3412, 10.1105/tpc.104.024315.
- Rebekah A. Rampey; Andrew Woodward; Brianne N. Hobbs; Megan P. Tierney; Brett Lahner; David E. Salt; Bonnie Bartel; An Arabidopsis Basic Helix-Loop-Helix Leucine Zipper Protein Modulates Metal Homeostasis and Auxin Conjugate Responsiveness. Genetics 2006, 174, 1841-1857, 10.1534/genetics.106.061044.
- Youxi Yuan; Huilan Wu; Ning Wang; Jie Li; Weina Zhao; Juan Du; Daowen Wang; Hong-Qing Ling; FIT interacts with AtbHLH38 and AtbHLH39 in regulating iron uptake gene expression for iron homeostasis in Arabidopsis. Cell Research 2008, 18, 385-397, 10.1038/cr.2008.26.
- Terri A. Long; Hironaka Tsukagoshi; Wolfgang Busch; Brett Lahner; David E. Salt; Philip N. Benfey; The bHLH transcription factor POPEYE regulates response to iron deficiency in Arabidopsis roots. The Plant Cell 2010, 22, 2219-36, 10.1105/tpc.110.074096.
- Sivasenkar Lingam; Julia Mohrbacher; Tzvetina Brumbarova; Thomas Potuschak; Claudia Fink-Straube; Eddy Blondet; Pascal Genschik; Petra Bauer; Interaction between the bHLH Transcription Factor FIT and ETHYLENE INSENSITIVE3/ETHYLENE INSENSITIVE3-LIKE1 Reveals Molecular Linkage between the Regulation of Iron Acquisition and Ethylene Signaling in Arabidopsis. The Plant Cell 2011, 23, 1815-1829, 10.1105/tpc.111.084715.
- Jie Zhang; Bing Liu; Mengshu Li; Dongru Feng; Honglei Jin; Peng Wang; Jun Liu; Feng Xiong; Jinfa Wang; Hong-Bin Wang; The bHLH Transcription Factor bHLH104 Interacts with IAA-LEUCINE RESISTANT3 and Modulates Iron Homeostasis in Arabidopsis. The Plant Cell 2015, 27, 787-805, 10.1105/tpc.114.132704.
- Takanori Kobayashi; Seiji Nagasaka; Takeshi Senoura; Reiko Nakanishi Itai; Hiromi Nakanishi; Naoko K. Nishizawa; Iron-binding haemerythrin RING ubiquitin ligases regulate plant iron responses and accumulation. Nature Communications 2013, 4, , 10.1038/ncomms3792.
- Devarshi Selote; Rozalynne Samira; Anna Matthiadis; Jeffrey W. Gillikin; Terri A. Long; Iron-binding E3 ligase mediates iron response in plants by targeting basic helix-loop-helix transcription factors. Plant Physiology 2015, 167, 273-86, 10.1104/pp.114.250837.
- Jorge Rodríguez-Celma; Hsuan Chou; Takanori Kobayashi; Terri A. Long; Janneke Balk; Hemerythrin E3 Ubiquitin Ligases as Negative Regulators of Iron Homeostasis in Plants. Frontiers in Plant Science 2019, 10, 98, 10.3389/fpls.2019.00098.
- J McElver; I Tzafrir; G Aux; R Rogers; C Ashby; K Smith; C Thomas; A Schetter; Q Zhou; M A Cushman; et al.J TossbergT NickleJ Z LevinM LawD MeinkeD Patton Insertional mutagenesis of genes required for seed development in Arabidopsis thaliana. Genetics 2001, 159, 1751-1763.
- Maria N. Hindt; Garo Z. Akmakjian; Kara Pivarski; Tracy Punshon; Ivan Baxter; David E. Salt; Mary Lou Guerinot; BRUTUS and its paralogs, BTS LIKE1 and BTS LIKE2, encode important negative regulators of the iron deficiency response in Arabidopsis thaliana. Metallomics 2017, 9, 876-890, 10.1039/c7mt00152e.
- Takanori Kobayashi; Asami Ozu; Subaru Kobayashi; Gynheung An; Jong-Seong Jeon; Naoko K. Nishizawa; OsbHLH058 and OsbHLH059 transcription factors positively regulate iron deficiency responses in rice. Plant Molecular Biology 2019, 101, 471-486, 10.1007/s11103-019-00917-8.
- Alicia Sivitz; Claudia Grinvalds; Marie Barberon; Catherine Curie; Grégory Vert; Proteasome-mediated turnover of the transcriptional activator FIT is required for plant iron-deficiency responses. The Plant Journal 2011, 66, 1044-1052, 10.1111/j.1365-313x.2011.04565.x.
- Johannes Meiser; Sivasenkar Lingam; Petra Bauer; Posttranslational regulation of the iron deficiency basic helix-loop-helix transcription factor FIT is affected by iron and nitric oxide. Plant Physiology 2011, 157, 2154-66, 10.1104/pp.111.183285.
- María J. García; Carlos Lucena; Francisco J. Romera; Esteban Alcántara; Rafael Pérez-Vicente; Ethylene and nitric oxide involvement in the up-regulation of key genes related to iron acquisition and homeostasis in Arabidopsis. Journal of Experimental Botany 2010, 61, 3885-3899, 10.1093/jxb/erq203.
- Hong-Yu Wang; Marco Klatte; Marc Jakoby; Helmut Bäumlein; Bernd Weisshaar; Petra Bauer; Iron deficiency-mediated stress regulation of four subgroup Ib BHLH genes in Arabidopsis thaliana. Planta 2007, 226, 897-908, 10.1007/s00425-007-0535-x.
- Yue Zhang; Huilan Wu; Ning Wang; Huajie Fan; Chunlin Chen; Yan Cui; Hongfei Liu; Hong-Qing Ling; Mediator subunit 16 functions in the regulation of iron uptake gene expression in Arabidopsis. New Phytologist 2014, 203, 770-783, 10.1111/nph.12860.
- Yan Yang; Bin Ou; Jinzhe Zhang; Wen Si; Hongya Gu; Genji Qin; Li-Jia Qu; The Arabidopsis Mediator subunit MED16 regulates iron homeostasis by associating with EIN3/EIL1 through subunit MED25. The Plant Journal 2014, 77, 838-851, 10.1111/tpj.12440.
- Christine Palmer; Maria N. Hindt; Holger Schmidt; Stephan Clemens; Mary Lou Guerinot; MYB10 and MYB72 Are Required for Growth under Iron-Limiting Conditions. PLOS Genetics 2013, 9, e1003953, 10.1371/journal.pgen.1003953.
- Ioannis A. Stringlis; Ke Yu; Kirstin Feussner; Ronnie De Jonge; Sietske Van Bentum; Marcel C. Van Verk; Roeland L. Berendsen; Peter A. H. M. Bakker; Ivo Feussner; Corné M. J. Pieterse; MYB72-dependent coumarin exudation shapes root microbiome assembly to promote plant health. Proceedings of the National Academy of Sciences 2018, 115, E5213-E5222, 10.1073/pnas.1722335115.
- Sjoerd Van Der Ent; Bas W.M. Verhagen; Ronald Van Doorn; Daniel Bakker; Maarten G. Verlaan; Michiel J.C. Pel; Ruth G. Joosten; Marcel C.G. Proveniers; L.C. Van Loon; Jurriaan Ton; et al.Corné M. J. Pieterse MYB72 Is Required in Early Signaling Steps of Rhizobacteria-Induced Systemic Resistance in Arabidopsis. Plant Physiology 2008, 146, 1293-1304, 10.1104/pp.107.113829.
- Wawrzynska, A.; Sirko, A. Proteasomal degradation of proteins is important for the proper transcriptional response to sulfur deficiency conditions in plants. Plant Cell Environ. 2020. under review.
- Kim, S.A.; LaCroix, I.S.; Gerber, S.A.; Guerinot, M.L; The iron deficiency response in Arabidopsis thaliana requires the phosphorylated transcription factor URI. Proc. Natl. Acad. Sci. USA 2019, 116, 24933–24942, 10.3410/f.736998790.793568363.
- Yulia O. Korshunova; David Eide; W. Gregg Clark; Mary Lou Guerinot; Himadri B. Pakrasi; The IRT1 protein from Arabidopsis thaliana is a metal transporter with a broad substrate range. Plant Molecular Biology 1999, 40, 37-44, 10.1023/A:1026438615520.
- Elizabeth E. Rogers; David J. Eide; Mary Lou Guerinot; Altered selectivity in an Arabidopsis metal transporter. Proceedings of the National Academy of Sciences 2000, 97, 12356-12360, 10.1073/pnas.210214197.
- Potocki, S.; Valensin, D.; Camponeschi, F.; Kozlowski, H. The extracellular loop of IRT1 ZIP protein—the chosen one for zinc? J. Inorg. Biochem. 2013, 127, 246–252.
- Marie Barberon; Enric Zelazny; Stéphanie Robert; Geneviève Conéjéro; Cathy Curie; Jìří Friml; Grégory Vert; Monoubiquitin-dependent endocytosis of the IRON-REGULATED TRANSPORTER 1 (IRT1) transporter controls iron uptake in plants. Proceedings of the National Academy of Sciences 2011, 108, E450-E458, 10.1073/pnas.1100659108.
- Rumen Ivanov; Tzvetina Brumbarova; Ailisa Blum; Anna-Maria Jantke; Claudia Fink-Straube; Petra Bauer; SORTING NEXIN1 Is Required for Modulating the Trafficking and Stability of the Arabidopsis IRON-REGULATED TRANSPORTER1. The Plant Cell 2014, 26, 1294-1307, 10.1105/tpc.113.116244.
- Wild, M.; Daviere, J.M.; Regnault, T.; Sakvarelidze-Achard, L.; Carrera, E.; Diaz, I.L.; Cayrel, A.; Dubeaux, G.; Vert, G.; Achard, P; et al. Tissue-specific regulation of gibberellin signaling fine-tunes Arabidopsis iron-deficiency responses. Dev. Cell 2016, 37, 190–200.
- Loubna Kerkeb; Indrani Mukherjee; Iera Chatterjee; Brett Lahner; David E. Salt; Erin L. Connolly; Iron-Induced Turnover of the Arabidopsis IRON-REGULATED TRANSPORTER1 Metal Transporter Requires Lysine Residues1. Plant Physiology 2008, 146, 1964-1973, 10.1104/pp.107.113282.
- Lung-Jiun Shin; Jing-Chi Lo; Guan-Hong Chen; Judy Callis; Hongyong Fu; Kuo‐Chen Yeh; IRT1 degradation factor1, a ring E3 ubiquitin ligase, regulates the degradation of iron-regulated transporter1 in Arabidopsis. The Plant Cell 2013, 25, 3039-51, 10.1105/tpc.113.115212.
- Dubeaux, G.; Neveu, J.; Zelazny, E.; Vert, G; Metal sensing by the IRT1 transporter-receptor orchestrates its own degradation and plant metal nutrition. Mol. Cell 2018, 69, 953–964, 10.3410/f.732844816.793543915.
- Martins, S.; Dohmann, E.M.N.; Cayrel, A.; Johnson, A.; Fischer, W.; Pojer, F.; Satiat-Jeunemaitre, B.; Jaillais, Y.; Chory, J.; Geldner, N.; et al. Internalization and vacuolar targeting of the brassinosteroid hormone receptor BRI1 are regulated by ubiquitination. Nat. Commun. 2015, 6.
- Tan, J.M.; Wong, E.S.; Kirkpatrick, D.S.; Pletnikova, O.; Ko, H.S.; Tay, S.P.; Ho, M.W.; Troncoso, J.; Gygi, S.P.; Lee, M.K; et al. Lysine 63-linked ubiquitination promotes the formation and autophagic clearance of protein inclusions associated with neurodegenerative diseases. Human Molecular Genetics 2008, 17, 431-439, 10.1093/hmg/ddm320.
- J. R. Dinneny; T. A. Long; J. Y. Wang; J. W. Jung; D. Mace; S. Pointer; C. Barron; Siobhan M. Brady; J. Schiefelbein; P. N. Benfey; Cell Identity Mediates the Response of Arabidopsis Roots to Abiotic Stress. Science 2008, 320, 942-945, 10.1126/science.1153795.
- Wenfeng Li; Wolfgang Schmidt; A lysine-63-linked ubiquitin chain-forming conjugase, UBC13, promotes the developmental responses to iron deficiency in Arabidopsis roots. The Plant Journal 2010, 62, 330-343, 10.1111/j.1365-313x.2010.04150.x.
- I-Chun Pan; Wolfgang Schmidt; Functional implications of K63-linked ubiquitination in the iron deficiency response of Arabidopsis roots. Frontiers in Plant Science 2014, 4, 542, 10.3389/fpls.2013.00542.
- Mei-Chun Cheng; En-Jung Hsieh; Jui-Hung Chen; Hsing-Yu Chen; Tsan-Piao Lin; Arabidopsis RGLG2, functioning as a RING E3 ligase, interacts with AtERF53 and negatively regulates the plant drought stress response. Plant Physiology 2012, 158, 363-375, 10.1104/pp.111.189738.
- Johannes Leitner; Katarzyna Retzer; Barbara Korbei; Christian Luschnig; Dynamics in PIN2 auxin carrier ubiquitylation in gravity-responding Arabidopsis roots. Plant Signaling & Behavior 2012, 7, 1271-1273, 10.4161/psb.21715.
- Astrid Nagels Durand; Sabrina Iñigo; Andres Ritter; Elisa Iniesto; Rebecca De Clercq; An Staes; Jelle Van Leene; Vicente Rubio; Kris Gevaert; Geert De Jaeger; Laurens Pauwels; Alain Goossens; The Arabidopsis Iron–Sulfur Protein GRXS17 is a Target of the Ubiquitin E3 Ligases RGLG3 and RGLG4. Plant And Cell Physiology 2016, 57, 1801-1813, 10.1093/pcp/pcw122.
- Cheng, N.H.; Liu, J.Z.; Liu, X.; Wu, Q.Y.; Thompson, S.M.; Lin, J.; Chang, J.; Whitham, S.A.; Park, S.; Cohen, J.D; et al. Arabidopsis Monothiol Glutaredoxin, AtGRXS17, Is Critical for Temperature-dependent Postembryonic Growth and Development via Modulating Auxin Response. Journal of Biological Chemistry 2011, 286, 20398-20406, 10.1074/jbc.M110.201707.
- Luis Ojeda; Greg Keller; Ulrich Muhlenhoff; Julian C. Rutherford; Roland Lill; Dennis R. Winge; Role of Glutaredoxin-3 and Glutaredoxin-4 in the Iron Regulation of the Aft1 Transcriptional Activator inSaccharomyces cerevisiae. Journal of Biological Chemistry 2006, 281, 17661-17669, 10.1074/jbc.m602165200.
- Petra Haunhorst; Eva-Maria Hanschmann; Lars Bräutigam; Oliver Stehling; Bastian Hoffmann; Ulrich Mühlenhoff; Roland Lill; Carsten Berndt; Christopher Horst Lillig; Crucial function of vertebrate glutaredoxin 3 (PICOT) in iron homeostasis and hemoglobin maturation. Molecular Biology of the Cell 2013, 24, 1895-1903, 10.1091/mbc.E12-09-0648.
- Inigo, S.; Durand, A.N.; Ritter, A.; Le Gall, S.; Termathe, M.; Klassen, R.; Tohge, T.; De Coninck, B.; Van Leene, J.; De Clercq, R; et al. Glutaredoxin GRXS17 Associates with the Cytosolic Iron-Sulfur Cluster Assembly Pathway. Plant Physiol. 2016, 172, 858-873, 10.1104/pp.16.00261.
- Emanuel Barth; Ron Hübler; Aria Baniahmad; Manja Marz; The Evolution of COP9 Signalosome in Unicellular and Multicellular Organisms. Genome Biology and Evolution 2016, 8, 1279-89, 10.1093/gbe/evw073.
- Tan, S.; Liu, F.; Pan, X.X.; Zang, Y.P.; Jin, F.; Zu, W.X.; Qi, X.T.; Xiao, W.; Yin, L.P; CSN6, a subunit of the COP9 signalosome, is involved in early response to iron deficiency in Oryza sativa. Scientific Reports 2016, 6, 25485, 10.1038/srep25485.
- Seckin Eroglu; Emre Aksoy; Genome-wide analysis of gene expression profiling revealed that COP9 signalosome is essential for correct expression of Fe homeostasis genes in Arabidopsis. Biometals 2017, 30, 685-698, 10.1007/s10534-017-0036-8.
- Qiang Zhao; Yi-Ran Ren; Qing-Jie Wang; Xiao-Fei Wang; Chun-Xiang You; Yu-Jin Hao; Ubiquitination-Related MdBT Scaffold Proteins Target a bHLH Transcription Factor for Iron Homeostasis.. Plant Physiology 2016, 172, 1973-1988, 10.1104/pp.16.01323.
- Li-Jie Zhou; Chun-Ling Zhang; Rui-Fen Zhang; Gui-Luan Wang; Yuan-Yuan Li; Yu-Jin Hao; The SUMO E3 Ligase MdSIZ1 Targets MdbHLH104 to Regulate Plasma Membrane H+-ATPase Activity and Iron Homeostasis. Plant Physiology 2018, 179, 88-106, 10.1104/pp.18.00289.
- Henning Ulrich; Mutual interactions between the SUMO and ubiquitin systems: a plea of no contest. Trends in Cell Biology 2005, 15, 525-532, 10.1016/j.tcb.2005.08.002.





