
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | James C. L. Chow | -- | 937 | 2026-03-03 03:30:44 | | | |
| 2 | Jeremy Duan | Meta information modification | 937 | 2026-03-03 03:34:16 | | |
Video Upload Options
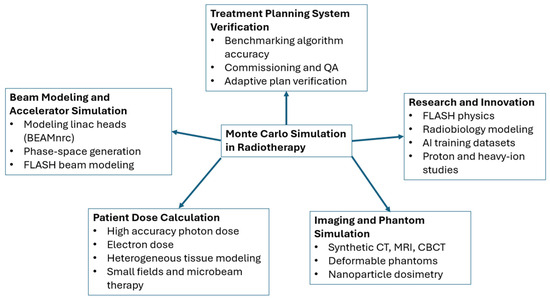
Hybrid artificial intelligence (AI) and Monte Carlo (MC) methods for radiotherapy dose calculation refer to computational approaches that integrate machine learning models with physics-based MC simulation to achieve both fast and accurate estimation of radiation dose distributions. These methods use AI to approximate complex MC dose calculations with greatly reduced computation time, while retaining MC simulation as the standard for physical fidelity and validation. The hybrid strategy supports real-time or near-real-time dose evaluation, enables adaptive treatment workflows, and allows accurate modeling of photon and electron beams in heterogeneous patient anatomy. By combining the strengths of data-driven prediction and physics-based simulation, hybrid AI and MC methods provide a pathway toward efficient and high-precision dose calculation in modern radiotherapy.
References
- Andreo, P. Monte Carlo simulations in radiotherapy dosimetry. Radiat. Oncol. 2018, 13, 121.
- Chow, J.C. Monte Carlo simulations in nanomedicine: Advancing cancer imaging and therapy. Nanomaterials 2025, 15, 117.
- Seco, J.; Verhaegen, F. Monte Carlo Techniques in Radiation Therapy; CRC Press: Boca Raton, FL, USA, 2013.
- Bielajew, A. The Monte Carlo simulation of radiation transport. In Handbook of Radiotherapy Physics: Theory and Practice; Hodder Arnold: London, UK, 2007; pp. 75–87.
- Brualla, L.; Rodriguez, M.; Lallena, A.M. Monte Carlo systems used for treatment planning and dose verification. Strahlenther. Onkol. 2017, 193, 243–259.
- Lopez-Tarjuelo, J.; Garcia-Molla, R.; Juan-Senabre, X.J.; Quirós-Higueras, J.D.; Santos-Serra, A.; de Marco-Blancas, N.; Calzada-Feliu, S. Acceptance and commissioning of a treatment planning system based on Monte Carlo calculations. Technol. Cancer Res. Treat. 2014, 13, 129–138.
- Can, S.; Karaçetin, D.; Meriç, N. Beam modeling and commissioning for Monte Carlo photon beam on an Elekta Versa HD LINAC. Appl. Radiat. Isot. 2022, 180, 110054.
- Chow, J.C.; Ruda, H.E. Impact of scattering foil composition on electron energy distribution in a clinical linear accelerator modified for FLASH radiotherapy: A Monte Carlo study. Materials 2024, 17, 3355.
- Reis, C.Q.; Nicolucci, P.; Fortes, S.S.; Silva, L.P. Effects of heterogeneities in dose distributions under nonreference conditions: Monte Carlo simulation vs dose calculation algorithms. Med. Dosim. 2019, 44, 74–82.
- Pokhrel, D.; McClinton, C.; Sood, S.; Badkul, R.; Saleh, H.; Jiang, H.; Lominska, C. Monte Carlo evaluation of tissue heterogeneities corrections in the treatment of head and neck cancer patients using stereotactic radiotherapy. J. Appl. Clin. Med. Phys. 2016, 17, 258–270.
- Paganetti, H.; Jiang, H.; Adams, J.A.; Chen, G.T.; Rietzel, E. Monte Carlo simulations with time-dependent geometries to investigate effects of organ motion with high temporal resolution. Int. J. Radiat. Oncol. Biol. Phys. 2004, 60, 942–950.
- Galeone, C.; Nakas, A.; Donetti, M.; Martire, M.C.; Milian, F.M.; Pella, A.; Paganelli, C.; Sacchi, R.; Vignati, A.; Durante, M.; et al. Real-time motion modeling and treatment verification for irregular motion in carbon ion therapy: A feasibility study. Phys. Med. Biol. 2025, 70, 165009.
- Kowatsch, M.; Partoll, E.; Künzler, T.; Attenberger, C.; Szeverinski, P.; Clemens, P.; Tschann, P. Quality assurance of beam base data in modern radiotherapy: A Monte Carlo simulation approach. Biomed. Phys. Eng. Express 2025, 11, 045010.
- Chow, J.C. A performance evaluation on Monte Carlo simulation for radiation dosimetry using cell processor. J. Comput. Methods Sci. Eng. 2011, 11, 1–12.
- Liu, Y.; Yang, J.; Lee, P.; Horowitz, D.; Thomas, T.O. Adaptive radiation therapy. Adv. Oncol. 2025, 5, 1–13.
- Chow, J.C. Quantum computing and machine learning in medical decision-making: A comprehensive review. Algorithms 2025, 18, 156.
- Webster, M.; Podgorsak, A.; Li, F.; Zhou, Y.; Jung, H.; Yoon, J.; Dona Lemus, O.; Zheng, D. New approaches in radiotherapy. Cancers 2025, 17, 1980.
- Rabe, M.; Kurz, C.; Thummerer, A.; Landry, G. Artificial intelligence for treatment delivery: Image guided radiotherapy. Strahlenther. Onkol. 2025, 201, 283–297.
- Chow, J.C. Nanomaterial-based molecular imaging in cancer: Advances in simulation and AI integration. Biomolecules 2025, 15, 444.





