
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Sofia Alexandra Silva Rosa | -- | 1092 | 2026-02-20 18:58:20 |
Video Upload Options
Visible-Near-Infrared (Vis-NIR) spectroscopy is an analytical technology that spans wavelengths from 400 to 2500 nm, enabling rapid, non-invasive, and reagent-free analysis of biological samples. Unlike traditional laboratory methods, Vis-NIR is compatible with portable, miniaturized devices, making it ideal for point-of-care (POC) diagnostics in clinical or field settings. By measuring how light interacts with samples such as blood or feces, the system captures spectral data related to molecular vibrations. When paired with Self-Learning Artificial Intelligence (SLAI) to filter technical interferences, Vis-NIR provides immediate biochemical results from micro-volumes of samples. This technology supports the One Health initiative by decentralizing diagnostics and accelerating clinical decision-making in both human and veterinary medicine.
1. Introduction
Modern medical technology demands high accuracy, rapid results, as the speed of data acquisition is often critical for therapeutic success. Consequently, diagnostics are shifting from centralized laboratories to point-of-care (POC) settings, integrating precision directly into primary or field-based workflows. In veterinary medicine, this shift is particularly vital, as clinicians must rely on objective, real-time physiological data to compensate for animal patients' inability to communicate symptoms [1]. This convergence of human and animal healthcare reflects the One Health framework, fostering multidisciplinary diagnostic innovation [2].
2. Technical Comparison with Other Spectroscopic Methods
Vis-NIR spectroscopy offers a superior balance of portability and analytical capability compared to other established veterinary techniques. Unlike Raman spectroscopy, which is hindered by background fluorescence [3][4], or FTIR, which suffers from strong water interference and bulky hardware [5][6], Vis-NIR is robust and easily miniaturized. Furthermore, while fluorescence spectroscopy often requires invasive probes or chemical labels [7][8], Vis-NIR remains non-invasive and reagent-free [9]. This allows for real-time monitoring of multiple physiological parameters without complex sample preparation, making it the most viable option for routine point-of-care adoption.
3. History and Concept
Vis-NIR spectroscopy is a natural evolution of near-infrared (NIR) spectroscopy. While William Herschel discovered infrared radiation in the 1800s, the analytical value of NIR emerged in 1960 when Karl Norris used it at the USDA for non-destructive moisture measurement in grains. By the late 1970s, it had expanded to forage and animal feed analysis. The integration of the visible spectrum (400–750 nm) led to modern Vis-NIR spectroscopy. Recent advances in fiber optics and LEDs have allowed these instruments to move from specialized labs into the field, where they can now estimate blood parameters in pets without reagents [1].
4. System Architecture and Performance
A typical Vis-NIR spectral detection system consists of a light source, a monochromator, a detector, and optical accessories (Figure 1) [10]. While tungsten-halogen lamps were traditionally used for their wide spectral coverage, they present limitations like excessive heat and short lifespans; consequently, LEDs have emerged as a superior alternative due to their higher efficiency, durability, and lack of radiation burn risk [11]. The interaction between light and sample, via reflectance, transmittance, or interactance, captures spectral information related to molecular vibrations and physical properties [11][12].
In practical POC IoT platforms, this technology is implemented using miniaturized spectrometer modules (e.g., Hamamatsu C12666MA or Ocean Insight STS-vis) and specialized plug-in capsules that require minimal sample volumes (~10 μL) for real-time registration [13]. This approach enables rapid, reagent-free, and decentralized diagnostics by capturing physical and molecular signals at distinctive wavelengths [9][14]. While the hardware captures raw multi-scale interference, the analytical core relies on Self-Learning Artificial Intelligence (SLAI) to unscramble these patterns and provide accurate quantitative parameters [1][13]. This convergence of Vis-NIR spectroscopy and POC testing facilitates rapid clinical decision-making and improves the diagnostic workflow in both human and veterinary medicine [15][16].
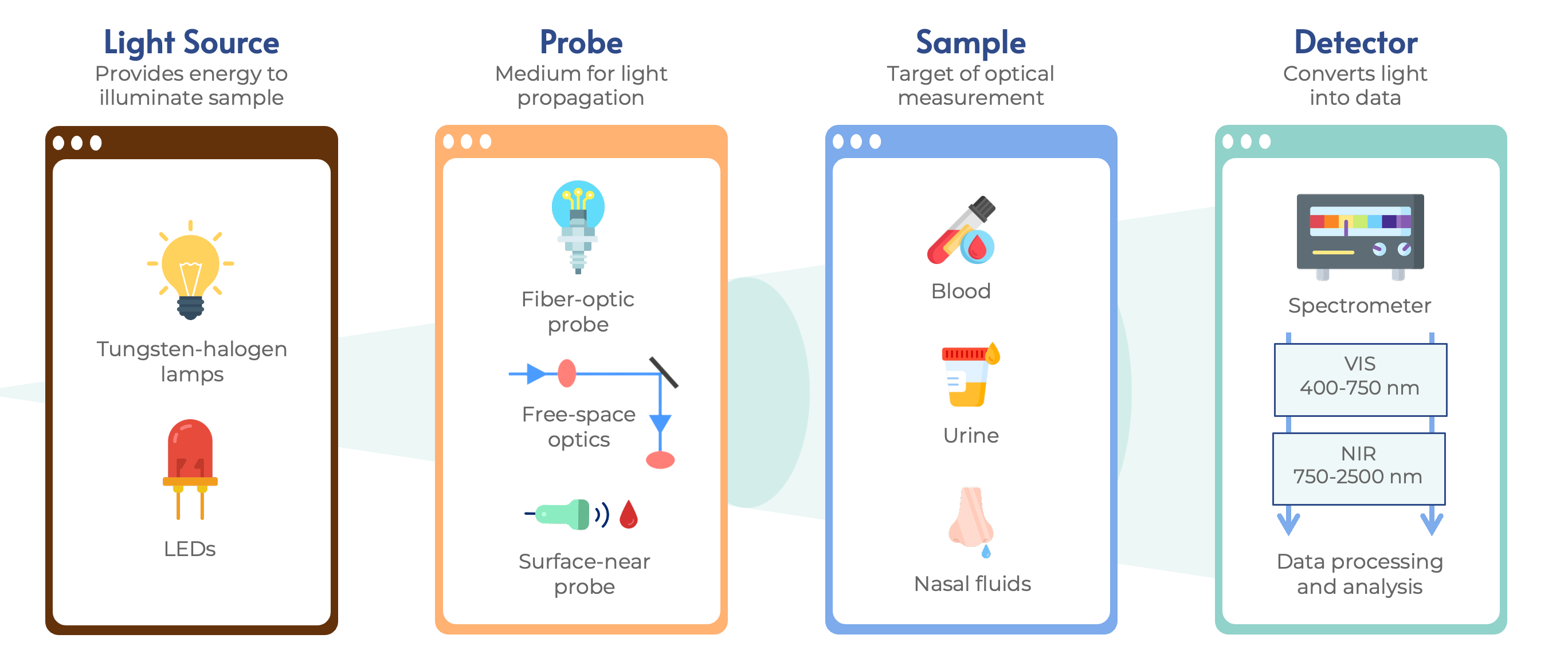
Figure 1. Overview of the Vis-NIR spectroscopy workflow: light emission (LEDs/Tungsten), propagation via probe, sample interaction (transmittance/reflectance), and final detection by the spectrometer.
5. Challenges and Limitations of Vis-NIR Spectroscopy
Despite its potential, Vis-NIR spectroscopy faces physical and analytical challenges, primarily the complex light-sample interactions where scattering effects (Rayleigh and Mie) are often dismissed as noise [1]. Physically, penetration depth is limited to 5–7 mm, and environmental factors like temperature or water absorption can mask critical signals [10][17][18].
Analytically, overlapping spectral bands hinder molecular specificity, particularly in blood, where dominant hemoglobin signals overwhelm less abundant elements, causing deviations from the Beer-Lambert law [1][13][19]. To overcome this, Self-Learning Artificial Intelligence (SLAI) uses covariance mode (CovM) search to transform these interferences into a diagnostic advantage [9][13]. Lastly, while miniaturization enables POC use, it involves constant trade-offs between portability, sensitivity, and cost [10][20].
6. One Health and Comparative Applications
Vis-NIR spectroscopy is a versatile tool with the potential to democratize access to health technologies by transitioning from high-cost laboratory equipment to affordable, robust devices capable of performing under field conditions. While centralized laboratories remain the gold standard, the time required for transport and processing often delays clinical decisions. POC technologies like Vis-NIR close this gap by enabling immediate, reagent-free analysis [15][16]. This shared potential aligns with the One Health perspective, fostering a comparative approach where insights from diverse physiological models contribute to a holistic diagnostic framework.
In human medicine, Vis-NIR has proven effective across various matrices, including hemoglobin quantification, glucose monitoring, and serum bilirubin prediction [21][22][23]. It has even been adapted to emerging challenges, such as assessing post-COVID conditions [24]. Similarly, in veterinary medicine, blood remains the most explored fluid for hemogram analysis in dogs, cats, and salmon [9][14][25], while fecal and milk samples have been used to screen for parasitic infections and dietary markers [26][27]. A summary of these diverse applications across both fields is presented in Table 1.
Table 1. Overview of Vis-NIR applications in human and veterinary medicine.
|
Field |
Matrix |
Main Applications |
Key References |
|
Human |
Blood, Nasal fluids |
Hemoglobin, glucose, bilirubin, Influenza A/B, post-COVID severity |
|
|
Animal |
Blood, Milk, Feces |
Hemograms (dog/cat/salmon), milk feeding systems, parasitic screening |
Despite these parallels, the wide variety of species in veterinary practice introduces unique challenges, such as species-specific differences in red blood cell shape and hemoglobin absorption. Furthermore, environmental variations like diet and stress add complexity to calibration models. However, when integrated with cloud-based AI, miniaturized Vis-NIR devices offer a non-invasive, affordable alternative that improves the standard of care for domestic and exotic species alike [13]. By facilitating both pre-diagnostic screening and post-diagnostic monitoring, this technology bridges the gap between human and animal healthcare, creating a collaborative space for diagnostic innovation.
7. Conclusions
Vis-NIR spectroscopy is a versatile tool that democratizes access to health technology by providing rapid, reagent-free results at the point of care. It complements centralized labs, allowing health professionals to act faster and with more confidence. Although challenges like environmental variation (diet, climate, stress) and spectral resolution persist, ongoing advancements in miniaturization and cloud-based machine learning promise to make these tools even more precise and accessible. Ultimately, this technology offers a bridge between fields, improving health outcomes for both people and animals alike.
References
- Barroso, T.G.; Queirós, C.; Monteiro-Silva, F.; Santos, F.; Gregório, A.H.; Martins, R.C. Reagentless Vis-NIR Spectroscopy Point-of-Care for Feline Total White Blood Cell Counts. Biosensors. 2024, 14, 53.
- Sarah J. Pitt; Alan Gunn; The One Health Concept. Br. J. Biomed. Sci.. 2024, 81, 12366.
- Acri Giuseppe; Falcone Annastella; Claudia Giannetto; Giudice Elisabetta; Piccione Giuseppe; Testagrossa Barbara; Luca Cicero; Giovanni Cassata; Di Pietro Simona; Preliminary study for the application of Raman spectroscopy for the identification of Leishmania infected dogs. Sci. Rep.. 2022, 12, 1-10.
- John L. Robertson; Nikolas Dervisis; John Rossmeisl; Marlie Nightengale; Daniel Fields; Cameron Dedrick; Lacey Ngo; Amr Sayed Issa; Georgi Guruli; Giuseppe Orlando; Ryan S. Senger; Cancer detection in dogs using rapid Raman molecular urinalysis. Front. Veter- Sci.. 2024, 11, 1328058.
- Gustavo Larios; Matheus Ribeiro; Carla Arruda; Samuel L. Oliveira; Thalita Canassa; Matthew J. Baker; Bruno Marangoni; Carlos Ramos; Cícero Cena; A new strategy for canine visceral leishmaniasis diagnosis based on FTIR spectroscopy and machine learning. J. Biophotonics. 2021, 14, e202100141.
- Sarah Malek; Federico Marini; Mark C. Rochat; Romain Béraud; Glenda M. Wright; Christopher B. Riley; Infrared spectroscopy of synovial fluid as a potential screening approach for the diagnosis of naturally occurring canine osteoarthritis associated with cranial cruciate ligament rupture. Osteoarthr. Cartil. Open. 2020, 2, 100120.
- Andrea D’agostino; Tommaso Di Palma; Stefano Cecchini Gualandi; Raffaele Boni; Fluorescence Spectroscopy for the Diagnosis of Endometritis in the Mare. Anim.. 2022, 12, 1157.
- Raffaele Boni; Alessandra Gallo; Elisabetta Tosti; Electrophysiology and Fluorescence Spectroscopy Approaches for Evaluating Gamete and Embryo Functionality in Animals and Humans. Biomol.. 2022, 12, 1685.
- Barroso, T.G.; Ribeiro, L.; Gregório, H.; Monteiro-Silva, F.; Neves Dos Santos, F.; Martins, R.C. Point-of-Care Using Vis-NIR Spectroscopy for White Blood Cell Count Analysis. Chemosensors. 2022, 10, 460.
- Krzysztof B. Beć; Justyna Grabska; Christian W. Huck; Principles and Applications of Miniaturized Near‐Infrared (NIR) Spectrometers. Chem. – A Eur. J.. 2020, 27, 1514-1532.
- Fu, X.; Wang, X.; Rao, X. An LED-based spectrally tuneable light source for visible and near-infrared spectroscopy analysis: A case study for sugar content estimation of citrus. Byosyst Eng. 2017, 163, 87-93.
- Poh, A.H.; Adikan, F.R.M.; Moghavvemi, M. The past, present, and prospective on UV–VIS–NIR skin photonics and spectroscopy—a wavelength guide. Med Biol Eng Comput. 2020, 58, 1159-1175.
- Barroso, T.G.; Ribeiro, L.; Gregório, H.; Santos, F.; Martins, R.C. Point-of-care Vis-SWNIR spectroscopy towards reagent-less hemogram analysis.. Sens Actuators B Chem . 2021, 343, 130138.
- Barroso, T.G.; Ribeiro, L.; Gregório, H.; Santos, F.; Martins, R.C. Visible–Near-Infrared Platelets Count: Towards Thrombocytosis Point-of-Care Diagnosis. Proceedings of the 1st International Electronic Conference on Chemical Sensors and Analytical Chemistry. 2021, MDPI , 78.
- Larkins, M.C.; Thombare, A. . Point-of-Care Testing ; StatPearls [Internet], Eds.; StatPearls Publishing : Treasure Island (FL) , 2023; pp. n.d..
- Manessis, G.; Gelasakis, A.I.; Bossis, I. Point-of-Care Diagnostics for Farm Animal Diseases: From Biosensors to Integrated Lab-on-Chip Devices. Biosensors. 2022, 12, 7.
- Nutte Teraphongphom; Christina S. Kong; Jason M. Warram; Eben L. Rosenthal; Specimen mapping in head and neck cancer using fluorescence imaging. Laryngoscope Investig. Otolaryngol.. 2017, 2, 447-452.
- Marcelo Blanco; Dámarih Valdés; Influence of Temperature on the Predictive Ability of near Infrared Spectroscopy Models. J. Near Infrared Spectrosc.. 2004, 12, 121-126.
- Yves Roggo; Pascal Chalus; Lene Maurer; Carmen Lema-Martinez; Aurélie Edmond; Nadine Jent; A review of near infrared spectroscopy and chemometrics in pharmaceutical technologies. J. Pharm. Biomed. Anal.. 2007, 44, 683-700.
- Christian W. Huck; Present and Future of Miniaturized NIR Spectrometers Combined with Challenging Data Management Strategies. Maced. Pharm. Bull.. 2022, 68, 9-10.
- Yadav, J.; Rani, A.; Singh, V.; Murari, B.M. Prospects and limitations of non-invasive blood glucose monitoring using near-infrared spectroscopy. . Biomed Signal Process Control . 2015, 18, 214-227.
- J. Todd Kuenstner; Karl H. Norris; Spectrophotometry of Human Hemoglobin in the near Infrared Region from 1000 to 2500 nm. J. Near Infrared Spectrosc.. 1994, 2, 59-65.
- Tan, H.; Liao, S.; Pan, T.; Zhang, J.; Chen, J. Rapid and simultaneous analysis of direct and indirect bilirubin indicators in serum through reagent-free visible-near-infrared spectroscopy combined with chemometrics. Spectrochim Acta A Mol Biomol Spectrosc. 2020, 233, 118215.
- Paula Antelo-Riveiro; Manuel Vázquez; María Jesús Domínguez-Santalla; Emilio Rodríguez-Ruiz; Ángel Piñeiro; Rebeca Garcia-Fandino; Rapid diagnosis and severity scale of post-COVID condition using advanced spectroscopy. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc.. 2024, 328, 125474.
- Eirik Svendsen; Lise L. Randeberg; Martin Føre; Bengt Finstad; Rolf Erik Olsen; Nina Bloecher; Jo Arve Alfredsen; Data for characterization of the optical properties of Atlantic salmon (Salmo salar) blood. J. Biophotonics. 2023, 16, e202300073.
- Kho, E.A.; Fernandes, J.N.; Kotze, A.C.; Fox, G.P.; Sikulu-Lord, M.T.; Beasley, A.M.; et al. Influence of environmental factors on the detection of blood in sheep faeces using visible–near-infrared spectroscopy as a measure of Haemonchus contortus infection. Parasit Vectors . 2020, 13, 591.
- A. Bahri; S. Nawar; H. Selmi; M. Amraoui; H. Rouissi; A. M. Mouazen; Application of visible and near-infrared spectroscopy for evaluation of ewes milk with different feeds. Anim. Prod. Sci.. 2018, 59, 1190-1200.
- Sakudo, A.; Baba, K.; Ikuta, K. Discrimination of influenza virus-infected nasal fluids by Vis-NIR spectroscopy. Clinica Chim Acta. 2012, 414, 130-134.
- Sakudo, A.; Baba, K.; Ikuta, K. Analysis of Vis–NIR spectra changes to measure the inflammatory response in the nasal mucosal region of influenza A and B virus-infected patients. J Clin Virol. 2012, 55, 334-338.





