| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alejandro Beceiro | + 2901 word(s) | 2901 | 2020-12-10 08:59:30 |
Video Upload Options
Carbapenem resistance is a major global health problem that seriously compromises the treatment of infections caused by nosocomial pathogens. Production of carbapenemases (carbapenem-hydrolyzing enzymes) is the most important mechanism of carbapenem resistance. A new generation of promising carbapenemase inhibitors, together with the recently approved avibactam, relebactam and vaborbactam are being tested in clinical and pre-clinical trials.This review summarizes the main, most promising carbapenemase inhibitors synthesized to date.
1. Introduction
β-lactams are the most diverse and widely used group of antibiotics in clinical practice. The mechanism of action of β-lactams is based on binding and blocking the penicillin binding proteins (PBPs), which are involved in the final steps of cell wall synthesis [1][2][3]. Carbapenems differ structurally from penicillins, cephalosporins and monobactams and have a wider spectrum of action and stability against β-lactamase enzymes. The longest established carbapenems are imipenem, meropenem and ertapenem, while more recently developed examples include doripenem, biapenem, panipenem, razupenem and tomopenem [4]. The use of carbapenems in clinical settings increased in the 2000s due to the emergence and spread of extended spectrum β-lactamases (ESBLs). However, the massive use/overuse of these agents led to the emergence of resistance, as had previously occurred with other groups of antibiotics [5].
In 2017 the World Health Organization (WHO) published a list of priority pathogens for which new treatments are required. The pathogens included in the highest category of urgency are carbapenem-resistant Acinetobacter baumannii, carbapenem-resistant Pseudomonas aeruginosa and carbapenem- and third generation cephalosporin-resistant Enterobacterales [6]. This situation highlights the importance of β-lactam antibiotics, especially carbapenems, in the treatment of infections caused by nosocomial pathogens. Production of carbapenemases (carbapenem-hydrolyzing enzymes) is the most important mechanism of carbapenem resistance [7], with examples in the four classes of β-lactamases, categorised according to the Ambler classification [8][9]. Among the carbapenemases identified, the following are the most important: (i) class A carbapenemases, especially those coded in plasmids, such as KPC and GES. The KPC group is the most widely distributed worldwide and the constituents are predominantly found in Klebsiella pneumoniae [8][9][10]; (ii) class B carbapenemases (also known as metallo-β-lactamases, MBLs) are usually found in pathogens such as P. aeruginosa, A. baumannii and Enterobacterales,and their prevalence has increased in recent years [8][11]. The most common groups of MBLs are VIM, IMP and NDM [11]; (iii) class C carbapenemases are not numerous and have been identified recently. Although, class C β-lactamases production does not offer carbapenem resistance, exceptionally five enzymes in this group are capable of hydrolyzing carbapenems (ACT-1, DHA-1, CMY-2, CMY-10 and ADC-68) [9], and (iv) class D carbapenemases (also known as OXAs); although discovered many years ago, the rapid spread of carbapenem hydrolyzing class D β-lactamase (CHDLs) is recent [12][13]. OXA-48-like is widely disseminated in Enterobacterales, while the groups OXA-23-like, OXA-24/40-like, OXA-58-like, OXA-143-like and OXA-235-like are mainly responsible for resistance to carbapenems in A. baumannii [13][14](Table 1).
Table 1. Most clinically significant carbapenemases.
|
Mechanism of Action |
Class |
Carbapenemase |
More Common Enzymes |
|||
|
Serine-β-lactamases |
A |
KPC |
KPC-2 |
KPC-3 |
|
|
|
GES |
GES-2 |
GES-5 |
GES-6 |
|
||
|
D |
OXA |
OXA-23 |
OXA-24/40 |
OXA-58 |
OXA-48 |
|
|
Metallo-β-lactamases |
B |
IMP |
IMP-1 |
IMP-6 |
IMP-7 |
|
|
VIM |
VIM-1 |
VIM-2 |
|
|
||
|
NDM |
NDM-1 |
NDM-4 |
NDM-5 |
|
||
Although carbapenemase activity is the main cause of carbapenem resistance, other elements are also involved. Porins have been shown to be associated with the development of resistance to carbapenems in synergy with hyperexpression of AmpC and/or ESBLs [15][16][17]. Similarly, efflux pumps [15] and mutations in PBPs (PBP2 orPBP3) have also been implicated in resistance to carbapenems [18][19].
One of the main strategies used to restore the effectiveness of β-lactam antibiotics is to use β-lactamase inhibitors (molecules that are able to bind to the active site of the enzyme) to prevent the antibiotic being hydrolysed by the enzyme [20][21]. The first β-lactamase inhibitor discovered (in 1972) was clavulanic acid, followed by sulbactam (in 1978) and tazobactam (in 1984). These are the so-called classical β-lactamase inhibitors [20][21][22][23]. In recent years new groups of inhibitors have appeared, and some have already been approved by regulatory agencies and are now available in the clinical setting, thus extending and recovering the antimicrobial activity of some β-lactam antibiotics. The main new groups are diazobicyclooctanes (DBOs) (avibactam and relebactam have been approved by the FDA) and boronic acid derivatives (vaborbactam is currently the only inhibitor approved) [24][25] (Figure 1).
2. New Carbapenemase Inhibitors in Development
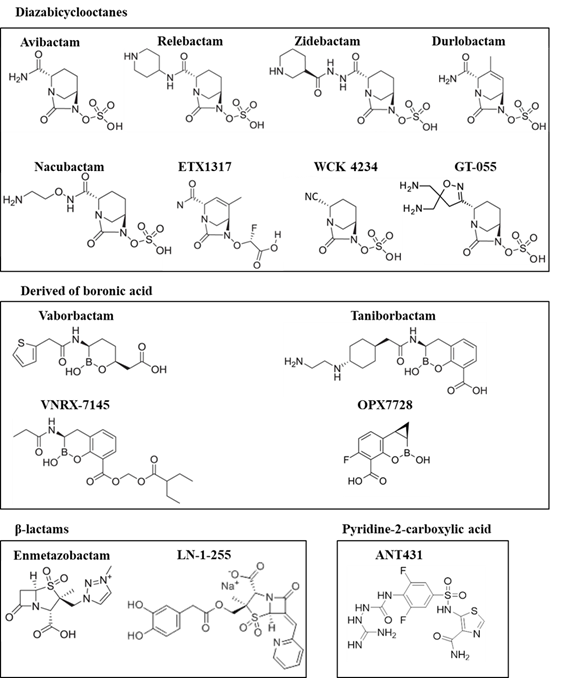
This review focuses on the recent studies of new carbapenemase inhibitors at preclinical or clinical stages of development (Figure 1, Tables 2 and 3).
Figure 1. Structures of the recent carbapenemase inhibitors
2.1. Diazabicyclooctane Derived Inhibitors
In the last few years the chemical scaffolds of the diazabicyclooctane class of β-lactamase inhibitors such as avibactam and relebactam have been modified to enhance the activity of these compounds [26]. Although DBOs do not exert inhibitory activity against class B β-lactamases, they can be combined with a monobactam, such as aztreonam, as these are stable to MBLs and need only to be protected from ESBL or AmpC enzymes [27]. Indeed, some second generation developmental DBO inhibitors, such as durlobactam, zidebactam and nacubactam are “dual action inhibitors” and have high affinity for the PBP2 of many Gram-negative species, thus exerting an antimicrobial effect. These DBOs thus exhibit “β-lactam enhancer” activity, potentiating partner β-lactams, and the new combination displays activity against carbapenemase-producing pathogens, including MBLs.
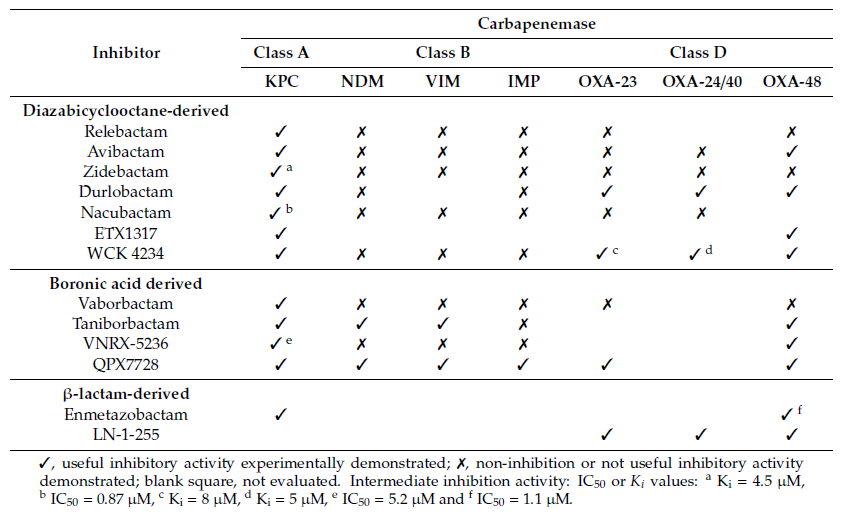
Table 2. Inhibition of major carbapenemases by new carbapenemase inhibitors
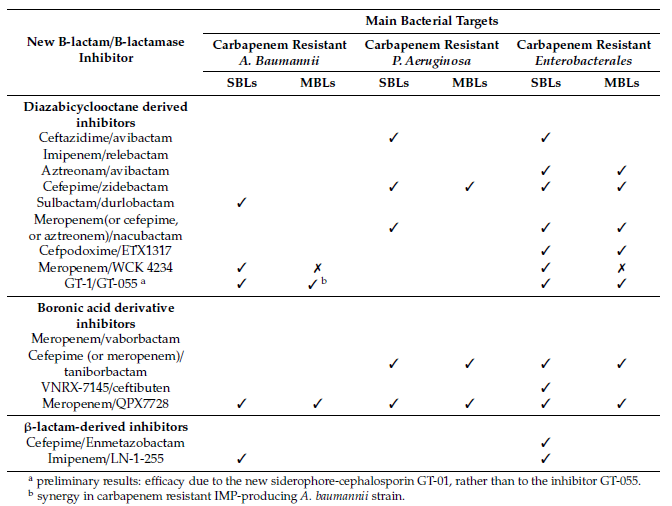
Table 3. Potential therapies aimed at treating infections caused by priority carbapenem-resistant pathogens for which new drugs are urgently needed
2.1.1 Avibactam (aztreonam/avibactam)
Avibactam is capable of inhibiting ESBLs, AmpCs and the carbapenemases produced by Enterobacterales, such as KPC, GES, IMI, SME or OXA-48, but not MBLs [28][29][30]. A new combination is now being developed, aimed at MBLs, selecting the only monobactam approved to date, aztreonam.
2.1.2 Zidebactam (cefepime/zidebactam)
Zidebactam (formerly WCK 5107, Wockhardt, India), as well as other new generation DBOs such as nacubactam and durlobactam, inhibit PBPs, β-lactamases and display synergy with β-lactams [31]. Zidebactam is a new bicyclo-acyl hydrazide which displays activity against of Gram-negative bacteria such as Enterobacterales, P. aeruginosa and A. baumannii, including strains producing different class A, C and D β-lactamases, such as CTX-M-like, AmpC or OXA-48 [32][33][34]. Although zidebactam is derived from a DBO scaffold, it has been designed with the objective of augmenting PBP2 binding rather than increasing the β-lactamase-inhibitory activity of the compound, thus enhancing β-lactam activity; however, it also displays moderate activity as a β-lactam inhibitor [31].
2.1.3 Durlobactam (sulbactam/durlobactam)
Durlobactam (previously ETX2514, Entasis Therapeutics, USA), a diazabicyclooctenone β-lactamase inhibitor of class A, C and D β-lactamases [35], displays some intrinsic antimicrobial activity against some Enterobacterales [36]. It is being tested in combination with sulbactam, another β-lactamase inhibitor also able to inhibit class A and C β-lactamases. Sulbactam, commercially available in combination with ampicillin, displays antimicrobial activity against Acinetobacter spp. [37].
2.1.4 Nacubactam (meropenem/nacubactam)
Similarly to zidebactam, nacubactam (formerly RG6080/OP0595, Meiji Seika Pharma, Japan; Fedora Pharmaceuticals, Canada and Roche, Switzerland) is a next generation DBO which also acts in different ways, by inhibiting class A and C β-lactamases, and as an antibiotic by inhibiting PBP2, thus enhancing the activity of the β-lactam partner [38]. It has a high affinity for class A carbapenemases such as KPC, but not for class D carbapenemases such as OXA-23 and OXA-24/40, displaying highest activity against class A β-lactamases such as TEM and CTX-M. To our knowledge, data on the inhibitory activity of nacubactam against OXA-48 is lacking. In combination with meropenem, nacubactam shows good synergy against carbapenem-resistant KPC-producing Enterobacterales [38][39].
2.1.5 ETX1317 (Cefpodoxime/ETX1317)
ETX1317 (Entasis Therapeutics) is a new DBO that displays broad spectrum activity against class A and C serine-β-lactamases (SBLs). The ester prodrug of ETX1317 is the compound ETX0282, used in combination with cefpodoxime (an oral third-generation cephalosporin), and it is currently in development for treatment of multidrug-resistant and carbapenem-resistant Enterobacterales infections. This combination exhibits good antimicrobial activity in Enterobacterales isolates with multiple β-lactamases, including KPC-producing types [40]. As the high polarity and low pKa of DBOs are well-suited to intravenous administration, but lead to low oral bioavailability, the main advantage of ETX0282 is the oral dosing availability [41].
2.1.6 WCK 4234 (meropenem/WCK 4234)
WCK 4234 (Wockhardt) displays potent inhibitory activity against class A and D carbapenemases and class C enzymes. It does not show antibacterial activity, unlike other DBOs considered in this review. It can enhance the activity of carbapenems against Enterobacterales producing OXA-48 or KPC carbapenemases, but not against MBLs-producing Enterobacterales. Importantly, the meropenem-WCK 4234 combination displays activity against OXA carbapenemases produced by A. baumannii, such as OXA-23, OXA-24/40 and hyperproduced OXA-51. WCK4234 did not display any activity against a collection of carbapenem-resistant isolates of P. aeruginosa [31][42][43].
2.1.7 GT-055 (GT-1/GT-055)
The diazabicyclooctane inhibitor GT-055 (also referred to as LCB18-055, LegoChem Biosciences) exhibits intrinsic activity against many Enterobacterales isolates, which bind tightly to PBP2. It is being tested in combination with GT-1 (also known as LCB10-0200), a novel siderophore-dihydroxypyridone and a modified aminothiazolylglycyl cephalosporin, which exploits bacterial iron-uptake systems to enhance entry into Gram-negative pathogens using a “Trojan-horse” strategy. GT-055 is able to enhance the in vitro activity of GT-1 against many GT-1–resistant strains [42].
2.2. Boronic Acid Derived Inhibitors
2.2.1. Taniborbactam (Cefepime/Taniborbactam)
Taniborbactam (formerly VNRX-5133, Everest Medicines, New York, USA) is a cyclic boronate with broad spectrum β-lactamase inhibitory activity against KPC, OXA-48 and MBLs (such as VIM and NDM, but not IMP) [44][45]. It was probably the first inhibitor showing direct inhibitory activity against Ambler class A, B, C, and D enzymes [46]. Taniborbactam uses distinct mechanisms to inhibit both SBLs and MBLs. It inhibits SBLs with slow dissociation, while in MBLs, it behaves as a reversible competitive inhibitor, with low inhibitor constant (Ki) values and rapid dissociation [46].
Taniborbactam is being developed for use in combination with cefepime and meropenem to treat complicated infections caused by MDR pathogens such as carbapenem-resistant Enterobacterales and carbapenem-resistant P. aeruginosa, including strains expressing serine carbapenemases and metallo-β-lactamases. Several phase I clinical trials have been undertaken to evaluate its PK/PD and safety. A phase III clinical trial is being conducted with cefepime/taniborbactam to evaluate the safety and efficacy of this combination against complicated urinary tract infections, including infections caused by MBL-producing strains.
2.2.2. VNRX-5236 (Ceftibuten/VNRX-7145)
VNRX-7145 (VenatoRx Pharmaceuticals, Malvern, PA, USA) is a novel cyclic boronate β-lactamase inhibitor with good oral bioavailability. In vivo, VNRX-7145 undergoes biotransformation to the active VNRX-5236, which covalently and reversibly binds the active serine site of Ambler class A and D β-lactamases, including those that hydrolyze carbapenems, such as KPC and OXA-48. VNRX-7145 is being developed in combination with ceftibuten, because of the good oral bioavailability of the cephalosporin [47][48].
2.2.3. QPX7728 (Meropenem/QPX7728)
QPX7728 (QPEX Biopharma) is an ultra-broad-spectrum cyclic boronic acid β-lactamase inhibitor with activity against both SBLs and MBLs [30]. In comparison with other β-lactamase inhibitor combinations (such as meropenem-vaborbactam, ceftazidime-avibactam and imipenem-relebactam), meropenem plus QPX7728 was found to be the most potent β-lactam-β-lactamase inhibitor combination tested against all groups of carbapenem resistant Enterobacterales with multiple resistance mechanisms, including KPC and MBLs carbapenemases [49]. Similarly, the meropenem/QPX7728 combination showed antimicrobial activity against a collection of carbapenem-resistant A. baumannii isolates producing CHDLs, NDM and KPC carbapenemases [49], and it was also being active against KPC-producing P. aeruginosa strains [50].
2.3. β-Lactam-Derived Inhibitors. Penicillin Sulfones
Penicillin-based sulfones, such as clavulanic acid, sulbactam and tazobactam, were the first inhibitors to be discovered. Unfortunately, none of these three compounds can inhibit carbapenemases. In recent years, within the group of penicillin sulfones, several molecules have emerged with higher or broader activity than the classical inhibitors, but only enmetazobactam and LN-1-255 have exhibited a relevant activity against carbapenemases.
2.3.1 Enmetazobactam (cefepime/enmetazobactam)
Enmetazobactam (formerly AAI101, Allecra Therapeutics), a novel penicillanic acid sulfone ESBLs inhibitor, also displays slight activity against some class C and D carbapenemases and also against some class A carbapenemases from Enterobacterales strains [51][52][53][54]. Enmetazobactam differs from tazobactam in the presence of a methyl group in the triazole moiety, which causes the compound to have a net neutral charge that enhances its activity [55]. The cefepime/enmetazobactam combination produced better results against MDR ESBLs-producing Enterobacterales strains than recently approved treatments such as ceftazidime/avibactam, ceftolozane/tazobactam and imipenem/relebactam [56]. Enmetazobactam is a good inhibitor of all class A β-lactamases, including the carbapenemases KPC-2 and KPC-3, with IC50 values in the nanomolar range, and it presents higher levels of inhibition than tazobactam and similar levels to those displayed by avibactam. However, the levels of inhibition observed against class C and D are significantly lower for enmetazobactam than for the other two compounds [55].
2.3.2 LN-1-255 (imipenem or meropenem/LN-1-255)
A group of penicillin sulfones with activity against class A, C and D β-lactamases have been synthesized by John D. Buynak and collaborators [57][58][59]. Among these, the LN-1-255 molecule is the most widely studied due to its ability to inhibit CHDLs, yielding good results in combination with carbapenems. LN-1-255 displayed a significant ability to inhibit the carbapenemases typically produced by A. baumannii (OXA-23, OXA-24/40, OXA-51, OXA-58, OXA-143 and OXA-235) and the OXA-48 carbapenemase produced by Enterobacterales [60][61][62][63]. LN-1-255 possesses a catechol group that is responsible for its effectiveness due to its ability to enhance internalization of the compound in the bacteria through iron uptake systems [25]. In vivo models of murine pneumonia showed a reduction in the bacterial load in mice treated with imipenem/LN-1-255, of between 1.7-4.5 logs, in lungs infected with CHDL-producing A. baumannii strains [64]. In 2020, Rodriguez and collaborators designed a series of six 6-arylmethylidene penicillin-based sulfones, derived from LN-1-255, through modifications in the pyridine ring. Some of these compounds improved the efficiency of action against CHDLs [65].
2.4. Other Promising MBL Inhibitors
MBLs are Zn(II)-dependent enzymes with the ability to hydrolyze β-lactam antibiotics. No clinically useful inhibitors of these enzymes have yet been approved. In the last few years, new structures focused specifically on inhibiting MBLs have been developed, with the compound ANT2681 (Antabio) probably representing one of the most important examples. This novel thiazole-carboxylate inhibitor, optimized from ANT431, is the result of a medicinal chemistry hit-to-lead program starting with pyridine-2-carboxylic acid, and it is a preclinical candidate with potential for clinical development as a specific inhibitor of MBLs [66]. ANT2681 inhibits the activity of MBLs through interaction with the dinuclear zinc ion cluster present in the active site of these enzymes [67]. The inhibitor displays the highest affinity for NDM-1, lower affinity for VIM-1 and very poor affinity for IMP-1. This inhibitor has shown efficacy in a mouse thigh model with an NDM-1-producing clinical isolate of K. pneumoniae. Although meropenem was ineffective at reducing tissue burden, its coadministration with ANT2681 yielded a statistically significant (1.8 log) reduction in colony forming units. Thus, ANT2681 is undergoing preclinical development with the intention of combining it with meropenem as a new treatment for serious infections caused by MBL-producing CRE.
In addition to the above-mentioned β-lactam-derived inhibitors, other compounds with a β-lactam core are capable of inhibiting carbapenemases, but which are at early stages of development. Thus, within the group of penicillin sulfones, C-6 substitutions affect the specificity of inhibition, displaying good activity against some class B β-lactamase [68]. In the group of penems, fusion of different bicyclic and tricyclic heterocycles with 6-methylidine penem yielded activity against class B carbapenemases [69][70][71]. CcrA and IMP-1, among others, were inhibited by J-110,441, a 1β-methylcarbapenem with a benzothenyl moiety at the C-2 position [72]. J-111,225, another new 1β-methylcarbapenem (with a trans-3,5 disubstituted pyrrolidinylthio moiety in C-2), inhibited IMP-1 [73] and showed bactericidal activity per se against S. aureus and P. aeruginosa [74]. Reverse hydroxamates (cephalosporins derived molecules) have exhibited activity against the MBL GIM-1 [75], or bisthiazolidines, also able to inhibit MBL enzymes [76].
3. Major Challenges in the Development of New Carbapenemase Inhibitors
As discussed above, a new generation of carbapenemase inhibitors is being developed. Development of inhibitors of MBL type and A. baumannii CHDL enzymes is perhaps the most difficult challenge. One of the main difficulties in designing inhibitors of class B β-lactamases is the wide genetic diversity among these enzymes. Thus, e.g. taniborbactam can inhibit NDM and VIM but not IMP enzymes. On the other hand, small molecules able to bind and chelate zinc ions have been reported to inhibit MBLs; however, they also inhibit human metalloenzymes and they may therefore be toxic to living tissues. Preclinical assays are also complicated to perform, due to the lack of zinc needed for appropriate behaviour of the MBLs at the infection sites [11][77]. In vitro conditions used for determining antibiotic susceptibility are very different from those that actually occur during infection [78]. It is therefore challenging to design and evaluate specific inhibitors for MBLs, and further research is necessary.
CHDLs, especially those produced by A. baumannii, are resistant to the action of most classical inhibitors [13]. The moderate capacity of CHDLs to hydrolyse carbapenems, combined with low permeability of A. baumannii, generates a high level of resistance to these antibiotics, which are considered the first choice for treating A. baumannii. There is an urgent need to develop new compounds capable of restoring the susceptibility to carbapenems in CHDL-producing strains of A. baumannii. So far, two compounds have exhibited useful activity against these enzymes: durlobactam [79] and LN-1-255 [80]. In both cases the key factor was the development of highly permeable compounds. Another important challenge is the coexistence of different β-lactams in the same pathogen. Thus, it is common to find numerous β-lactamases in Enterobacterales, for example strains co-producing blaNDM-1, blaOXA-48, blaCTX-M-15, blaTEM-1 and blaSHV-182 [81], often expressing two or more different carbapenemases or even MBLs.
Future inhibitors must be very potent and able to inhibit different classes of β-lactamases at the simultaneously, which requires complex structural and biochemical development [82]. Likewise, research must continue in order to develop new compounds that are effective against the main enzymatic resistance mechanism of this multi-drug resistant pathogen.
References
- Karen Bush; Patricia A. Bradford; β-Lactams and β-Lactamase Inhibitors: An Overview. Cold Spring Harbor Perspectives in Medicine 2016, 6, a025247, 10.1101/cshperspect.a025247.
- Catherine L. Tooke; Philip Hinchliffe; Eilis C. Bragginton; Charlotte K. Colenso; Viivi H.A. Hirvonen; Yuiko Takebayashi; Jim Spencer; β-Lactamases and β-Lactamase Inhibitors in the 21st Century. Journal of Molecular Biology 2019, 431, 3472-3500, 10.1016/j.jmb.2019.04.002.
- Kohanski, M.A.; Dwyer, D.J.; Collins, J.J.; How antibiotics kill bacteria: from targets to networks.. Nat. Rev. Microbiol. 2010, 8, 423-35, 10.1038/nrmicro2333..
- Mohammed I. El-Gamal; Imen Brahim; Noorhan Hisham; Rand Aladdin; Haneen Mohammed; Amany Bahaaeldin; Recent updates of carbapenem antibiotics. European Journal of Medicinal Chemistry 2017, 131, 185-195, 10.1016/j.ejmech.2017.03.022.
- Jessica M. A. Blair; Mark A. Webber; Alison J. Baylay; David O. Ogbolu; Laura J. V. Piddock; Molecular mechanisms of antibiotic resistance. Nature Reviews Genetics 2014, 13, 42-51, 10.1038/nrmicro3380.
- Global priority list of antibiotic-resistant bacteria to guide research, discovery, and development of new antibiotics. . World Health Organization.. Retrieved 2020-12-21
- Patrice Nordmann; Laurent Poirel; Epidemiology and Diagnostics of Carbapenem Resistance in Gram-negative Bacteria. Clinical Infectious Diseases 2019, 69, S521-S528, 10.1093/cid/ciz824.
- Anne Marie Queenan; Karen Bush; Carbapenemases: the Versatile β-Lactamases. Clinical Microbiology Reviews 2007, 20, 440-458, 10.1128/cmr.00001-07.
- Jeong Ho Jeon; Jung Hun Lee; Jae Jin Lee; Kwang Seung Park; Asad Mustafa Karim; Chang-Ro Lee; Byeong-Chul Jeong; Sang Hee Lee; Structural Basis for Carbapenem-Hydrolyzing Mechanisms of Carbapenemases Conferring Antibiotic Resistance. International Journal of Molecular Sciences 2015, 16, 9654-9692, 10.3390/ijms16059654.
- Laurent Dortet; Bogdan I Iorga; Structural and Functional Aspects of Class A Carbapenemases. Current Drug Targets 2016, 17, 1006-1028, 10.2174/1389450117666160310144501.
- Sara E. Boyd; David M. Livermore; David C. Hooper; William Hope; Metallo-β-Lactamases: Structure, Function, Epidemiology, Treatment Options, and the Development Pipeline. Antimicrobial Agents and Chemotherapy 2020, 64, e00397-20, 10.1128/aac.00397-20.
- Laurent Poirel; Thierry Naas; Patrice Nordmann; Diversity, Epidemiology, and Genetics of Class D β-Lactamases. Antimicrobial Agents and Chemotherapy 2009, 54, 24-38, 10.1128/aac.01512-08.
- Benjamin A. Evans; Sebastian G. B. Amyes; OXA -Lactamases. Clinical Microbiology Reviews 2014, 27, 241-263, 10.1128/cmr.00117-13.
- Sarah M. Drawz; Krisztina M. Papp-Wallace; Robert A. Bonomo; New β-Lactamase Inhibitors: a Therapeutic Renaissance in an MDR World. Antimicrobial Agents and Chemotherapy 2013, 58, 1835-1846, 10.1128/aac.00826-13.
- Lucía Fernández; Robert E. W. Hancock; Adaptive and Mutational Resistance: Role of Porins and Efflux Pumps in Drug Resistance. Clinical Microbiology Reviews 2012, 25, 661-681, 10.1128/cmr.00043-12.
- Hui Li; Yi-Feng Luo; Bryan J. Williams; Timothy S. Blackwell; Can-Mao Xie; Structure and function of OprD protein in Pseudomonas aeruginosa: From antibiotic resistance to novel therapies. International Journal of Medical Microbiology 2012, 302, 63-68, 10.1016/j.ijmm.2011.10.001.
- Carole Ayoub Moubareck; Dalal Hammoudi Halat; Insights into Acinetobacter baumannii: A Review of Microbiological, Virulence, and Resistance Traits in a Threatening Nosocomial Pathogen. Antibiotics 2020, 9, 119, 10.3390/antibiotics9030119.
- Bartolomé Moyá; Alejandro Beceiro; Gabriel Cabot; Carlos Juan; Laura Zamorano; Sebastián Alberti; Antonio Oliver; Pan-β-Lactam Resistance Development in Pseudomonas aeruginosa Clinical Strains: Molecular Mechanisms, Penicillin-Binding Protein Profiles, and Binding Affinities. Antimicrobial Agents and Chemotherapy 2012, 56, 4771-4778, 10.1128/aac.00680-12.
- Shinichiro Yamachika; Chika Sugihara; Yasuki Kamai; Makoto Yamashita; Correlation between penicillin-binding protein 2 mutations and carbapenem resistance in Escherichia coli. Journal of Medical Microbiology 2013, 62, 429-436, 10.1099/jmm.0.051631-0.
- Sarah M. Drawz; Robert A. Bonomo; Three Decades of β-Lactamase Inhibitors. Clinical Microbiology Reviews 2010, 23, 160-201, 10.1128/cmr.00037-09.
- Mohammed Shahid; F. Sobia; A. Singh; A. Malik; H. M. Khan; D. Jonas; P. M. Hawkey; Beta-lactams and Beta-lactamase-inhibitors in current- or potential-clinical practice: A comprehensive update. Critical Reviews in Microbiology 2009, 35, 81-108, 10.1080/10408410902733979.
- Francisco J Pérez-Llarena; Francisco Jose Perez-Llarena And German Bou; β-Lactamase Inhibitors: The Story so Far. Current Medicinal Chemistry 2009, 16, 3740-3765, 10.2174/092986709789104957.
- Kimberly A. Toussaint; Jason C. Gallagher; β-Lactam/β-Lactamase Inhibitor Combinations. Annals of Pharmacotherapy 2014, 49, 86-98, 10.1177/1060028014556652.
- Kamaleddin Haj Mohammad Ebrahim Tehrani; Nathaniel I. Martin; β-lactam/β-lactamase inhibitor combinations: an update. MedChemComm 2018, 9, 1439-1456, 10.1039/c8md00342d.
- Concepción González-Bello; Diana Rodríguez; Marina Pernas; Ángela Rodríguez; Esther Colchón; β-Lactamase Inhibitors To Restore the Efficacy of Antibiotics against Superbugs. Journal of Medicinal Chemistry 2019, 63, 1859-1881, 10.1021/acs.jmedchem.9b01279.
- Ryan K Shields; Yohei Doi; Aztreonam Combination Therapy: An Answer to Metallo-β-Lactamase–Producing Gram-Negative Bacteria?. Clinical Infectious Diseases 2019, 71, 1099-1101, 10.1093/cid/ciz1159.
- Lee, M.; Abbey, T.; Biagi, M.; Wenzler, E.; Activity of aztreonam in combination with ceftazidime-avibactam against serine- and metallo-β-lactamase-producing Pseudomonas aeruginosa.. Diagn. Microbiol. Infect. Dis. 2020, 99, 115227, 10.1016/j.diagmicrobio.2020.115227.
- David M Livermore; Danièle Meunier; Katie L Hopkins; Michel Doumith; Robert Hill; Rachel Pike; Peter Staves; Neil Woodford; Activity of ceftazidime/avibactam against problem Enterobacteriaceae and Pseudomonas aeruginosa in the UK, 2015–16. Journal of Antimicrobial Chemotherapy 2017, 73, 648-657, 10.1093/jac/dkx438.
- David E. Ehmann; Haris Jahić; Philip L. Ross; Rong-Fang Gu; Jun Hu; Thomas F. Durand-Réville; Sushmita Lahiri; Jason Thresher; Stephania Livchak; Ning Gao; et al.Tiffany PalmerGrant K. WalkupStewart L. Fisher Kinetics of Avibactam Inhibition against Class A, C, and D β-Lactamases. Journal of Biological Chemistry 2013, 288, 27960-27971, 10.1074/jbc.m113.485979.
- Ruslan Tsivkovski; Maxim Totrov; Olga Lomovskaya; Biochemical Characterization of QPX7728, a New Ultrabroad-Spectrum Beta-Lactamase Inhibitor of Serine and Metallo-Beta-Lactamases. Antimicrobial Agents and Chemotherapy 2020, 64, e00130-20, 10.1128/aac.00130-20.
- Krisztina M. Papp-Wallace; Nhu Q. Nguyen; Michael R. Jacobs; Christopher R. Bethel; Melissa D. Barnes; Vijay Kumar; Saralee Bajaksouzian; Susan D. Rudin; Philip N. Rather; Satish Bhavsar; et al.Tadiparthi RavikumarPrasad K. DeshpandeVijay PatilRavindra YeoleSachin S. BhagwatMahesh V. PatelFocco Van Den AkkerRobert A. Bonomo Strategic Approaches to Overcome Resistance against Gram-Negative Pathogens Using β-Lactamase Inhibitors and β-Lactam Enhancers: Activity of Three Novel Diazabicyclooctanes WCK 5153, Zidebactam (WCK 5107), and WCK 4234. Journal of Medicinal Chemistry 2018, 61, 4067-4086, 10.1021/acs.jmedchem.8b00091.
- David M. Livermore; Shazad Mushtaq; Marina Warner; Anna Vickers; Neil Woodford; In vitro activity of cefepime/zidebactam (WCK 5222) against Gram-negative bacteria. Journal of Antimicrobial Chemotherapy 2017, 72, 1373-1385, 10.1093/jac/dkw593.
- Bartolome Moya; Isabel M. Barcelo; Sachin Bhagwat; Mahesh Patel; German Bou; Krisztina M. Papp-Wallace; Robert A. Bonomo; Antonio Oliver; WCK 5107 (Zidebactam) and WCK 5153 Are Novel Inhibitors of PBP2 Showing Potent “β-Lactam Enhancer” Activity against Pseudomonas aeruginosa, Including Multidrug-Resistant Metallo-β-Lactamase-Producing High-Risk Clones. Antimicrobial Agents and Chemotherapy 2017, 61, e02529-16, 10.1128/aac.02529-16.
- James A. Karlowsky; Meredith A. Hackel; Samuel K. Bouchillon; Daniel F. Sahm; In Vitro Activity of WCK 5222 (Cefepime-Zidebactam) against Worldwide Collected Gram-Negative Bacilli Not Susceptible to Carbapenems. Antimicrobial Agents and Chemotherapy 2020, 64, e01432-20, 10.1128/aac.01432-20.
- Adam B. Shapiro; Ning Gao; Haris Jahić; Nicole M. Carter; April Chen; Alita A. Miller; Reversibility of Covalent, Broad-Spectrum Serine β-Lactamase Inhibition by the Diazabicyclooctenone ETX2514. ACS Infectious Diseases 2017, 3, 833-844, 10.1021/acsinfecdis.7b00113.
- Sarah M. McLeod; Adam B Shapiro; Samir H. Moussa; Michele Johnstone; Robert E. McLaughlin; Boudewijn L. M. De Jonge; Alita A. Miller; Frequency and Mechanism of Spontaneous Resistance to Sulbactam Combined with the Novel β-Lactamase Inhibitor ETX2514 in Clinical Isolates of Acinetobacter baumannii. Antimicrobial Agents and Chemotherapy 2017, 62, e01576-17, 10.1128/aac.01576-17.
- Paul G. Higgins; Hilmar Wisplinghoff; Danuta Stefanik; Harald Seifert; In Vitro Activities of the β-Lactamase Inhibitors Clavulanic Acid, Sulbactam, and Tazobactam Alone or in Combination with β-Lactams against Epidemiologically Characterized Multidrug-Resistant Acinetobacter baumannii Strains. Antimicrobial Agents and Chemotherapy 2004, 48, 1586-1592, 10.1128/aac.48.5.1586-1592.2004.
- Akihiro Morinaka; Yuko Tsutsumi; Mototsugu Yamada; Kenji Suzuki; Takashi Watanabe; Takao Abe; Takeshi Furuuchi; Seiichi Inamura; Yoshiaki Sakamaki; Nakako Mitsuhashi; et al.Takashi IdaDavid M. Livermore OP0595, a new diazabicyclooctane: mode of action as a serine β-lactamase inhibitor, antibiotic and β-lactam ‘enhancer’. Journal of Antimicrobial Chemotherapy 2015, 70, 2779-2786, 10.1093/jac/dkv166.
- Melissa D. Barnes; Magdalena A. Taracila; Caryn E. Good; Saralee Bajaksouzian; Laura J. Rojas; David Van Duin; Barry N. Kreiswirth; Michael R. Jacobs; Andreas Haldimann; Krisztina M. Papp-Wallace; et al.Robert A. Bonomo Nacubactam Enhances Meropenem Activity against Carbapenem-Resistant Klebsiella pneumoniae Producing KPC. Antimicrobial Agents and Chemotherapy 2019, 63, e00432-19, 10.1128/aac.00432-19.
- Alita A. Miller; Adam B. Shapiro; Sarah M. McLeod; Nicole Marie Carter; Samir H. Moussa; Ruben Tommasi; John P. Mueller; In Vitro Characterization of ETX1317, a Broad-Spectrum β-Lactamase Inhibitor That Restores and Enhances β-Lactam Activity against Multi-Drug-Resistant Enterobacteriales, Including Carbapenem-Resistant Strains. ACS Infectious Diseases 2020, 6, 1389-1397, 10.1021/acsinfecdis.0c00020.
- Thomas F. Durand-Réville; Janelle Comita-Prevoir; Jing Zhang; Xiaoyun Wu; Tricia L. May-Dracka; Jan Antoinette C. Romero; Frank Wu; April Chen; Adam B Shapiro; Nicole M. Carter; et al.Sarah M. McLeodRobert A. GiacobbeJeroen C. VerheijenSushmita D. LahiriMichael D. SaccoYu ChenJohn P. O’DonnellAlita A. MillerJohn P. MuellerRuben Tommasi Discovery of an Orally Available Diazabicyclooctane Inhibitor (ETX0282) of Class A, C, and D Serine β-Lactamases. Journal of Medicinal Chemistry 2020, 63, 12511-12525, 10.1021/acs.jmedchem.0c00579.
- Le Phuong Nguyen; Naina Adren Pinto; Thao Nguyen Vu; Hyunsook Lee; Young Lag Cho; Jung-Hyun Byun; Roshan D’Souza; Dongeun Yong; In Vitro Activity of a Novel Siderophore-Cephalosporin, GT-1 and Serine-Type β-Lactamase Inhibitor, GT-055, against Escherichia coli, Klebsiella pneumoniae and Acinetobacter spp. Panel Strains. Antibiotics 2020, 9, 267, 10.3390/antibiotics9050267.
- Alejandro Iregui; Zeb Khan; David Landman; John Quale; Activity of Meropenem with a Novel Broader-Spectrum β-Lactamase Inhibitor, WCK 4234, against Gram-Negative Pathogens Endemic to New York City. Antimicrobial Agents and Chemotherapy 2019, 64, e01666-19, 10.1128/aac.01666-19.
- Alen Krajnc; Jürgen Brem; Philip Hinchliffe; Karina Calvopiña; Tharindi D. Panduwawala; Pauline A. Lang; Jos J. A. G. Kamps; Jonathan M. Tyrrell; Emma Widlake; Benjamin G. Saward; et al.Timothy R. WalshJames SpencerChristopher J. Schofield Bicyclic Boronate VNRX-5133 Inhibits Metallo- and Serine-β-Lactamases. Journal of Medicinal Chemistry 2019, 62, 8544-8556, 10.1021/acs.jmedchem.9b00911.
- Xiaojuan Wang; ChunJiang Zhao; Qi Wang; Zhanwei Wang; Xinyue Liang; Feifei Zhang; Yawei Zhang; Han Meng; HongBin Chen; Shuguang Li; et al.Chengcheng ZhouHenan LiHui Wang In vitro activity of the novel β-lactamase inhibitor taniborbactam (VNRX-5133), in combination with cefepime or meropenem, against MDR Gram-negative bacterial isolates from China. Journal of Antimicrobial Chemotherapy 2020, 75, 1850-1858, 10.1093/jac/dkaa053.
- Jodie C. Hamrick; Jean-Denis Docquier; Tsuyoshi Uehara; Cullen L. Myers; David A. Six; Cassandra L. Chatwin; Kaitlyn J. John; Salvador F. Vernacchio; Susan M. Cusick; Robert E. L. Trout; et al.Cecilia PozziFilomena De LucaManuela BenvenutiStefano ManganiBin LiuRandy W. JacksonGreg MoeckLuigi XerriChristopher J. BurnsDaniel C. PevearDenis M. Daigle VNRX-5133 (Taniborbactam), a Broad-Spectrum Inhibitor of Serine- and Metallo-β-Lactamases, Restores Activity of Cefepime in Enterobacterales and Pseudomonas aeruginosa. Antimicrobial Agents and Chemotherapy 2019, 64, e01963-19, 10.1128/aac.01963-19.
- John, K.J.; Chatwin, C.L.; Hamrick, J.C.; Moeck, G.; Pevear, D.C. Rescue of Ceftibuten Activity by the Oral β-Lactamase Inhibitor VNRX-7145 against Enterobacteriaceae Expressing Class A, C and/or D β-Lactamases. In Proceedings of the ASM 2019; San Francisco, CA, 2019.
- Hamrick, J.; Chatwin, Cassandra John, K.; Burns, C.; Xerri, L.; Moeck, G.; Pevear, D.C. Selection of ceftibuten as the partner antibiotic for the oral beta-lactamase inhibitor VNRX-7145. In Proceedings of the ECCMID 2019; Amsterdam, 2019
- Mojgan Sabet; Ziad Tarazi; David C. Griffith; In Vivo Activity of QPX7728, an Ultrabroad-Spectrum Beta-Lactamase Inhibitor, In Combination with Beta-Lactams Against Carbapenem-Resistant K. pneumoniae. Antimicrobial Agents and Chemotherapy 2020, 64, e01267-20, 10.1128/aac.01267-20.
- Rubio-Aparicio, D.; Nelson, K.; Griffith, D.C.; Dudley, M.N.; Lomovskaya, O. QPX7728: In Vitro Activity in Combination with Oral Beta-Lactam Antibiotics against Enterobacteriaceae. In Proceedings of the ASM 2019; San Francisco, CA, 2019
- Lucas Tselepis; Gareth W. Langley; Ali F. Aboklaish; Emma Widlake; Dana E. Jackson; Timothy R. Walsh; T.R. Walsha C.J. Schofield; Jürgen Brem; Jonathan M. Tyrrell; In vitro efficacy of imipenem-relebactam and cefepime-AAI101 against a global collection of ESBL-positive and carbapenemase-producing Enterobacteriaceae. International Journal of Antimicrobial Agents 2020, 56, 105925, 10.1016/j.ijantimicag.2020.105925.
- Jared L. Crandon; David P. Nicolau; In Vitro Activity of Cefepime/AAI101 and Comparators against Cefepime Non-susceptible Enterobacteriaceae. Pathogens 2015, 4, 620-625, 10.3390/pathogens4030620.
- Mushtaq, S.; Chaudhry, A.; Adkin, R.; Woodford, N.; Benedict, N.; Pypstra, R.; Shapiro, S. In-vitro activity of diverse β-lactam/aai101 combinations vs. Multidrug-resistant gram-negative clinical strains. In Proceedings of the ECCMID 2014; Barcelona, 2014.
- Nordmann, P.; Girlich, D.; Benedict, N.; Pypstra, R.; Shapiro, S. Characterization of b-lactamase inhibition by aai101. In Proceedings of the ECCMID 2014; Barcelona, 2014.
- Krisztina M. Papp-Wallace; Christopher R. Bethel; Jocelyne Caillon; Melissa D. Barnes; Gilles Potel; Saralee Bajaksouzian; Joseph D. Rutter; Amokrane Reghal; Stuart Shapiro; Magdalena A. Taracila; et al.Michael R. JacobsRobert A. BonomoCedric Jacqueline Beyond Piperacillin-Tazobactam: Cefepime and AAI101 as a Potent β-Lactam−β-Lactamase Inhibitor Combination. Antimicrobial Agents and Chemotherapy 2019, 63, e00105-19, 10.1128/aac.00105-19.
- Ian Morrissey; Sophie Magnet; Stephen Hawser; Stuart Shapiro; Philipp Knechtle; In Vitro Activity of Cefepime-Enmetazobactam against Gram-Negative Isolates Collected from U.S. and European Hospitals during 2014–2015. Antimicrobial Agents and Chemotherapy 2019, 63, e00514-19, 10.1128/aac.00514-19.
- John D. Buynak; A.Srinivasa Rao; Venkata Ramana Doppalapudi; Greg Adam; Peter J. Petersen; Sirishkumar D. Nidamarthy; The synthesis and evaluation of 6-alkylidene-2'β-substituted penam sulfones as β-lactamase inhibitors. Bioorganic & Medicinal Chemistry Letters 1999, 9, 1997-2002, 10.1016/s0960-894x(99)00325-x.
- Matthew Kalp; Anjaneyulu Sheri; John D. Buynak; Christopher R. Bethel; Robert A. Bonomo; Paul R. Carey; Efficient Inhibition of Class A and Class D β-Lactamases by Michaelis Complexes. Journal of Biological Chemistry 2007, 282, 21588-21591, 10.1074/jbc.c700080200.
- John D. Buynak; The Discovery and Development of Modified Penicillin- and Cephalosporin- Derived β-Lactamase Inhibitors. Current Medicinal Chemistry 2004, 11, 1951-1964, 10.2174/0929867043364847.
- Sarah M. Drawz; Christopher R. Bethel; Venkata R. Doppalapudi; Anjaneyulu Sheri; Sundar Ram Reddy Pagadala; Andrea M. Hujer; Marion J. Skalweit; Vernon E. Anderson; Shu G. Chen; John D. Buynak; et al.Robert A. Bonomo Penicillin Sulfone Inhibitors of Class D β-Lactamases. Antimicrobial Agents and Chemotherapy 2010, 54, 1414-1424, 10.1128/aac.00743-09.
- Germán Bou; Elena Santillana; Anjaneyulu Sheri; Alejandro Beceiro; Jared M. Sampson; Matthew Kalp; Christopher R. Bethel; Anne M. Distler; Sarah M. Drawz; Sundar Ram Reddy Pagadala; et al.Focco Van Den AkkerRobert A. BonomoAntonio RomeroJohn D. Buynak Design, Synthesis, and Crystal Structures of 6-Alkylidene-2′-Substituted Penicillanic Acid Sulfones as Potent Inhibitors ofAcinetobacter baumanniiOXA-24 Carbapenemase. Journal of the American Chemical Society 2010, 132, 13320-13331, 10.1021/ja104092z.
- Juan C. Vázquez-Ucha; María Maneiro; Marta Martínez-Guitián; John Buynak; Christopher R. Bethel; Robert A. Bonomo; Germán Bou; Margarita Poza; Concepción González-Bello; Alejandro Beceiro; et al. Activity of the β-Lactamase Inhibitor LN-1-255 against Carbapenem-Hydrolyzing Class D β-Lactamases from Acinetobacter baumannii. Antimicrobial Agents and Chemotherapy 2017, 61, e01172-17, 10.1128/aac.01172-17.
- Juan A. Vallejo; Marta Martínez-Guitián; Juan Carlos Vázquez-Ucha; Concepción González-Bello; Margarita Poza; John D. Buynak; Christopher R. Bethel; Robert A. Bonomo; Germán Bou; Alejandro Beceiro; et al. LN-1-255, a penicillanic acid sulfone able to inhibit the class D carbapenemase OXA-48. Journal of Antimicrobial Chemotherapy 2016, 71, 2171-2180, 10.1093/jac/dkw105.
- Juan Carlos Vázquez-Ucha; Marta Martínez-Guitián; María Maneiro; Kelly Conde-Pérez; Laura Álvarez-Fraga; Gabriel Torrens; Antonio Oliver; John D. Buynak; Robert A. Bonomo; Germán Bou; et al.Concepción González-BelloMargarita PozaAlejandro Beceiro Therapeutic Efficacy of LN-1-255 in Combination with Imipenem in Severe Infection Caused by Carbapenem-Resistant Acinetobacter baumannii. Antimicrobial Agents and Chemotherapy 2019, 63, e01092-19, 10.1128/aac.01092-19.
- Diana Rodríguez; Maria Maneiro; Juan Carlos Vázquez-Ucha; Alejandro Beceiro; Concepción González-Bello; 6-Arylmethylidene Penicillin-Based Sulfone Inhibitors for Repurposing Antibiotic Efficiency in Priority Pathogens. Journal of Medicinal Chemistry 2020, 63, 3737-3755, 10.1021/acs.jmedchem.0c00127.
- David T. Davies; Simon Leiris; Nicolas Sprynski; Jérôme Castandet; Clarisse Lozano; Justine Bousquet; Magdalena Zalacain; Srinivas Vasa; Praveen Kumar Dasari; Ramesh Pattipati; et al.Naresh VempalaSwetha GujjewarSyamkumar GodiRaju JallalaRajashekar Reddy SathyapNarasimha A. DarshanojuVengala R. RavuRamakrishna Reddy JuventhalaNarender PottabathiniSomesh SharmaSrinivasu PothukanuriKirsty HoldenPeter WarnFrancesca MarcocciaManuela BenvenutiCecilia PozziStefano ManganiJean-Denis DocquierMarc LemonnierMartin Everett ANT2681: SAR Studies Leading to the Identification of a Metallo-β-lactamase Inhibitor with Potential for Clinical Use in Combination with Meropenem for the Treatment of Infections Caused by NDM-Producing Enterobacteriaceae. ACS Infectious Diseases 2020, 6, 2419-2430, 10.1021/acsinfecdis.0c00207.
- Martin Everett; Nicolas Sprynski; Alicia Coelho; Jérôme Castandet; Maëlle Bayet; Juliette Bougnon; Clarisse Lozano; David T. Davies; Simon Leiris; Magdalena Zalacain; et al.Ian MorrisseySophie MagnetKirsty HoldenPeter WarnFilomena De LucaJean-Denis DocquierMarc Lemonnier Discovery of a Novel Metallo-β-Lactamase Inhibitor That Potentiates Meropenem Activity against Carbapenem-Resistant Enterobacteriaceae. Antimicrobial Agents and Chemotherapy 2018, 62, e00074-18, 10.1128/aac.00074-18.
- John D. Buynak; Hansong Chen; Lakshminaryana Vogeti; Venkat Rao Gadhachanda; Christine A. Buchanan; Timothy Palzkill; Robert W. Shaw; James Spencer; Timothy R. Walsh; Penicillin-derived inhibitors that simultaneously target both metallo- and serine-β-lactamases. Bioorganic & Medicinal Chemistry Letters 2004, 14, 1299-1304, 10.1016/j.bmcl.2003.12.037.
- † Aranapakam M. Venkatesan; † Yansong Gu; † Osvaldo Dos Santos; ‡ Takao Abe; † Atul Agarwal; † Youjun Yang; † Peter J. Petersen; † William J. Weiss; Tarek S. Mansour; § Michiyoshi Nukaga; et al.‖ Andrea M. Hujer‖ And Robert A. Bonomo§ James R. Knox Structure−Activity Relationship of 6-Methylidene Penems Bearing Tricyclic Heterocycles as Broad-Spectrum β-Lactamase Inhibitors: Crystallographic Structures Show Unexpected Binding of 1,4-Thiazepine Intermediates. Journal of Medicinal Chemistry 2004, 47, 6556-6568, 10.1021/jm049680x.
- Aranapakam M. Venkatesan; Atul Agarwal; Takao Abe; Hideki Ushirogochi; Itsuka Yamamura; Mihira Ado; Takasaki Tsuyoshi; Osvaldo Dos Santos; Yansong Gu; Fuk-Wah Sum; et al.Zhong LiGerry FranciscoYang-I LinPeter J. PetersenYouJun YangToshio KumagaiWilliam J. WeissDavid M. ShlaesJames R. KnoxTarek S. Mansour Structure-Activity Relationship of 6-Methylidene Penems Bearing 6,5 Bicyclic Heterocycles as Broad-Spectrum β-Lactamase Inhibitors: Evidence for 1,4-Thiazepine Intermediates with C7 R Stereochemistry by Computational Methods. Journal of Medicinal Chemistry 2006, 49, 4623-4637, 10.1021/jm060021p.
- William J. Weiss; Peter J. Petersen; Timothy M. Murphy; Luanna Tardio; YouJun Yang; Patricia A. Bradford; Aranapakam M. Venkatesan; Takao Abe; Takeshi Isoda; Ado Mihira; et al.Hideki UshirogochiTsuyoshi TakasakeSteve ProjanJohn O'connellTarek S. Mansour In Vitro and In Vivo Activities of Novel 6-Methylidene Penems as β-Lactamase Inhibitors. Antimicrobial Agents and Chemotherapy 2004, 48, 4589-4596, 10.1128/aac.48.12.4589-4596.2004.
- Rie Nagano; Yuka Adachi; Hideaki Imamura; Koji Yamada; Terutaka Hashizume; Hajime Morishima; Carbapenem Derivatives as Potential Inhibitors of Various β-Lactamases, Including Class B Metallo-β-Lactamases. Antimicrobial Agents and Chemotherapy 1999, 43, 2497-2503, 10.1128/aac.43.10.2497.
- Rie Nagano; Yuka Adachi; Terutaka Hashizume; Hajime Morishima; In vitro antibacterial activity and mechanism of action of J-111,225, a novel 1β-methylcarbapenem, against transferable IMP-1 metallo-β-lactamase producers. Journal of Antimicrobial Chemotherapy 2000, 45, 271-276, 10.1093/jac/45.3.271.
- Rie Nagano; Kaneyoshi Shibata; Yuka Adachi; Hideaki Imamura; Terutaka Hashizume; Hajime Morishima; In Vitro Activities of Noveltrans-3,5-Disubstituted Pyrrolidinylthio-1β-Methylcarbapenems with Potent Activities against Methicillin-Resistant Staphylococcus aureus andPseudomonas aeruginosa. Antimicrobial Agents and Chemotherapy 2000, 44, 489-495, 10.1128/aac.44.3.489-495.2000.
- Sudhakar Reddy Ganta; Senthil Perumal; Sundar Ram Reddy Pagadala; Ørjan Samuelsen; James Spencer; R. F. Pratt; John D. Buynak; Approaches to the simultaneous inactivation of metallo- and serine-β-lactamases. Bioorganic & Medicinal Chemistry Letters 2009, 19, 1618-1622, 10.1016/j.bmcl.2009.02.018.
- Mariano M. González; Magda Kosmopoulou; Maria F. Mojica; Valerie Castillo; Philip Hinchliffe; Ilaria Pettinati; Jürgen Brem; Christopher J. Schofield; S. Graciela Mahler; Robert A. Bonomo; et al.Leticia I. LlarrullJames SpencerAlejandro J. Vila Bisthiazolidines: A Substrate-Mimicking Scaffold as an Inhibitor of the NDM-1 Carbapenemase. ACS Infectious Diseases 2015, 1, 544-554, 10.1021/acsinfecdis.5b00046.
- Anou M. Somboro; John Osei Sekyere; Daniel Gyamfi Amoako; Hezekiel M. Kumalo; René Khan; Linda A. Bester; Sabiha Y. Essack; In vitro potentiation of carbapenems with tannic acid against carbapenemase-producing enterobacteriaceae: exploring natural products as potential carbapenemase inhibitors. Journal of Applied Microbiology 2018, 126, 452-467, 10.1111/jam.14141.
- Tomefa E Asempa; Kamilia Abdelraouf; David P Nicolau; Metallo-β-lactamase resistance in Enterobacteriaceae is an artefact of currently utilized antimicrobial susceptibility testing methods. Journal of Antimicrobial Chemotherapy 2020, 75, 997-1005, 10.1093/jac/dkz532.
- Thomas F. Durand-Réville; Satenig Guler; Janelle Comita-Prevoir; Brendan Chen; Neil Bifulco; Hoan Huynh; Sushmita Lahiri; Adam B. Shapiro; Sarah M. McLeod; Nicole M. Carter; et al.Samir H MoussaCamilo Velez-VegaNelson B. OlivierRobert McLaughlinNing GaoJason ThresherTiffany PalmerBeth AndrewsRobert A. GiacobbeJoseph V. NewmanDavid E. EhmannBoudewijn De JongeJohn O'donnellJohn P. MuellerRuben TommasiAlita A. Miller ETX2514 is a broad-spectrum β-lactamase inhibitor for the treatment of drug-resistant Gram-negative bacteria including Acinetobacter baumannii. Nature Microbiology 2017, 2, 17104-17104, 10.1038/nmicrobiol.2017.104.
- Juan Carlos Vázquez-Ucha; Marta Martínez-Guitián; María Maneiro; Kelly Conde-Pérez; Laura Álvarez-Fraga; Gabriel Torrens; Antonio Oliver; John D. Buynak; Robert A. Bonomo; Germán Bou; et al.Concepción González-BelloMargarita PozaAlejandro Beceiro Therapeutic Efficacy of LN-1-255 in Combination with Imipenem in Severe Infection Caused by Carbapenem-Resistant Acinetobacter baumannii. Antimicrobial Agents and Chemotherapy 2019, 63, e01092-19, 10.1128/aac.01092-19.
- Helio S. Sader; David J. Farrell; Robert K. Flamm; Ronald N. Jones; Variation in Potency and Spectrum of Tigecycline Activity against Bacterial Strains from U.S. Medical Centers since Its Approval for Clinical Use (2006 to 2012). Antimicrobial Agents and Chemotherapy 2014, 58, 2274-2280, 10.1128/aac.02684-13.
- Silvia Corcione; Tommaso Lupia; Alberto Enrico Maraolo; Simone Mornese Pinna; Ivan Gentile; Francesco G. De Rosa; Carbapenem-sparing strategy. Current Opinion in Infectious Diseases 2019, 32, 663-673, 10.1097/qco.0000000000000598.
- David M Livermore; Danièle Meunier; Katie L Hopkins; Michel Doumith; Robert Hill; Rachel Pike; Peter Staves; Neil Woodford; Activity of ceftazidime/avibactam against problem Enterobacteriaceae and Pseudomonas aeruginosa in the UK, 2015–16. Journal of Antimicrobial Chemotherapy 2017, 73, 648-657, 10.1093/jac/dkx438.
- David E. Ehmann; Haris Jahić; Philip L. Ross; Rong-Fang Gu; Jun Hu; Thomas F. Durand-Réville; Sushmita Lahiri; Jason Thresher; Stephania Livchak; Ning Gao; et al.Tiffany PalmerGrant K. WalkupStewart L. Fisher Kinetics of Avibactam Inhibition against Class A, C, and D β-Lactamases. Journal of Biological Chemistry 2013, 288, 27960-27971, 10.1074/jbc.m113.485979.
- Ruslan Tsivkovski; Maxim Totrov; Olga Lomovskaya; Biochemical Characterization of QPX7728, a New Ultrabroad-Spectrum Beta-Lactamase Inhibitor of Serine and Metallo-Beta-Lactamases. Antimicrobial Agents and Chemotherapy 2020, 64, e00130-20, 10.1128/aac.00130-20.
- Krisztina M. Papp-Wallace; Nhu Q. Nguyen; Michael R. Jacobs; Christopher R. Bethel; Melissa D. Barnes; Vijay Kumar; Saralee Bajaksouzian; Susan D. Rudin; Philip N. Rather; Satish Bhavsar; et al.Tadiparthi RavikumarPrasad K. DeshpandeVijay PatilRavindra YeoleSachin S. BhagwatMahesh V. PatelFocco Van Den AkkerRobert A. Bonomo Strategic Approaches to Overcome Resistance against Gram-Negative Pathogens Using β-Lactamase Inhibitors and β-Lactam Enhancers: Activity of Three Novel Diazabicyclooctanes WCK 5153, Zidebactam (WCK 5107), and WCK 4234. Journal of Medicinal Chemistry 2018, 61, 4067-4086, 10.1021/acs.jmedchem.8b00091.
- David M. Livermore; Shazad Mushtaq; Marina Warner; Anna Vickers; Neil Woodford; In vitro activity of cefepime/zidebactam (WCK 5222) against Gram-negative bacteria. Journal of Antimicrobial Chemotherapy 2017, 72, 1373-1385, 10.1093/jac/dkw593.
- Bartolome Moya; Isabel M. Barcelo; Sachin Bhagwat; Mahesh Patel; German Bou; Krisztina M. Papp-Wallace; Robert A. Bonomo; Antonio Oliver; WCK 5107 (Zidebactam) and WCK 5153 Are Novel Inhibitors of PBP2 Showing Potent “β-Lactam Enhancer” Activity against Pseudomonas aeruginosa, Including Multidrug-Resistant Metallo-β-Lactamase-Producing High-Risk Clones. Antimicrobial Agents and Chemotherapy 2017, 61, e02529-16, 10.1128/aac.02529-16.
- James A. Karlowsky; Meredith A. Hackel; Samuel K. Bouchillon; Daniel F. Sahm; In Vitro Activity of WCK 5222 (Cefepime-Zidebactam) against Worldwide Collected Gram-Negative Bacilli Not Susceptible to Carbapenems. Antimicrobial Agents and Chemotherapy 2020, 64, e01432-20, 10.1128/aac.01432-20.
- Adam B. Shapiro; Ning Gao; Haris Jahić; Nicole M. Carter; April Chen; Alita A. Miller; Reversibility of Covalent, Broad-Spectrum Serine β-Lactamase Inhibition by the Diazabicyclooctenone ETX2514. ACS Infectious Diseases 2017, 3, 833-844, 10.1021/acsinfecdis.7b00113.
- Sarah M. McLeod; Adam B Shapiro; Samir H. Moussa; Michele Johnstone; Robert E. McLaughlin; Boudewijn L. M. De Jonge; Alita A. Miller; Frequency and Mechanism of Spontaneous Resistance to Sulbactam Combined with the Novel β-Lactamase Inhibitor ETX2514 in Clinical Isolates of Acinetobacter baumannii. Antimicrobial Agents and Chemotherapy 2017, 62, e01576-17, 10.1128/aac.01576-17.
- Paul G. Higgins; Hilmar Wisplinghoff; Danuta Stefanik; Harald Seifert; In Vitro Activities of the β-Lactamase Inhibitors Clavulanic Acid, Sulbactam, and Tazobactam Alone or in Combination with β-Lactams against Epidemiologically Characterized Multidrug-Resistant Acinetobacter baumannii Strains. Antimicrobial Agents and Chemotherapy 2004, 48, 1586-1592, 10.1128/aac.48.5.1586-1592.2004.
- Akihiro Morinaka; Yuko Tsutsumi; Mototsugu Yamada; Kenji Suzuki; Takashi Watanabe; Takao Abe; Takeshi Furuuchi; Seiichi Inamura; Yoshiaki Sakamaki; Nakako Mitsuhashi; et al.Takashi IdaDavid M. Livermore OP0595, a new diazabicyclooctane: mode of action as a serine β-lactamase inhibitor, antibiotic and β-lactam ‘enhancer’. Journal of Antimicrobial Chemotherapy 2015, 70, 2779-2786, 10.1093/jac/dkv166.
- Melissa D. Barnes; Magdalena A. Taracila; Caryn E. Good; Saralee Bajaksouzian; Laura J. Rojas; David Van Duin; Barry N. Kreiswirth; Michael R. Jacobs; Andreas Haldimann; Krisztina M. Papp-Wallace; et al.Robert A. Bonomo Nacubactam Enhances Meropenem Activity against Carbapenem-Resistant Klebsiella pneumoniae Producing KPC. Antimicrobial Agents and Chemotherapy 2019, 63, e00432-19, 10.1128/aac.00432-19.
- Alita A. Miller; Adam B. Shapiro; Sarah M. McLeod; Nicole Marie Carter; Samir H. Moussa; Ruben Tommasi; John P. Mueller; In Vitro Characterization of ETX1317, a Broad-Spectrum β-Lactamase Inhibitor That Restores and Enhances β-Lactam Activity against Multi-Drug-Resistant Enterobacteriales, Including Carbapenem-Resistant Strains. ACS Infectious Diseases 2020, 6, 1389-1397, 10.1021/acsinfecdis.0c00020.
- Thomas F. Durand-Réville; Janelle Comita-Prevoir; Jing Zhang; Xiaoyun Wu; Tricia L. May-Dracka; Jan Antoinette C. Romero; Frank Wu; April Chen; Adam B Shapiro; Nicole M. Carter; et al.Sarah M. McLeodRobert A. GiacobbeJeroen C. VerheijenSushmita D. LahiriMichael D. SaccoYu ChenJohn P. O’DonnellAlita A. MillerJohn P. MuellerRuben Tommasi Discovery of an Orally Available Diazabicyclooctane Inhibitor (ETX0282) of Class A, C, and D Serine β-Lactamases. Journal of Medicinal Chemistry 2020, 63, 12511-12525, 10.1021/acs.jmedchem.0c00579.
- Shazad Mushtaq; Anna Vickers; Neil Woodford; David M Livermore; WCK 4234, a novel diazabicyclooctane potentiating carbapenems against Enterobacteriaceae, Pseudomonas and Acinetobacter with class A, C and D β-lactamases. Journal of Antimicrobial Chemotherapy 2017, 72, 1688-1695, 10.1093/jac/dkx035.
- Le Phuong Nguyen; Naina Adren Pinto; Thao Nguyen Vu; Hyunsook Lee; Young Lag Cho; Jung-Hyun Byun; Roshan D’Souza; Dongeun Yong; In Vitro Activity of a Novel Siderophore-Cephalosporin, GT-1 and Serine-Type β-Lactamase Inhibitor, GT-055, against Escherichia coli, Klebsiella pneumoniae and Acinetobacter spp. Panel Strains. Antibiotics 2020, 9, 267, 10.3390/antibiotics9050267.
- Alen Krajnc; Jürgen Brem; Philip Hinchliffe; Karina Calvopiña; Tharindi D. Panduwawala; Pauline A. Lang; Jos J. A. G. Kamps; Jonathan M. Tyrrell; Emma Widlake; Benjamin G. Saward; et al.Timothy R. WalshJames SpencerChristopher J. Schofield Bicyclic Boronate VNRX-5133 Inhibits Metallo- and Serine-β-Lactamases. Journal of Medicinal Chemistry 2019, 62, 8544-8556, 10.1021/acs.jmedchem.9b00911.
- Xiaojuan Wang; ChunJiang Zhao; Qi Wang; Zhanwei Wang; Xinyue Liang; Feifei Zhang; Yawei Zhang; Han Meng; HongBin Chen; Shuguang Li; et al.Chengcheng ZhouHenan LiHui Wang In vitro activity of the novel β-lactamase inhibitor taniborbactam (VNRX-5133), in combination with cefepime or meropenem, against MDR Gram-negative bacterial isolates from China. Journal of Antimicrobial Chemotherapy 2020, 75, 1850-1858, 10.1093/jac/dkaa053.
- Jodie C. Hamrick; Jean-Denis Docquier; Tsuyoshi Uehara; Cullen L. Myers; David A. Six; Cassandra L. Chatwin; Kaitlyn J. John; Salvador F. Vernacchio; Susan M. Cusick; Robert E. L. Trout; et al.Cecilia PozziFilomena De LucaManuela BenvenutiStefano ManganiBin LiuRandy W. JacksonGreg MoeckLuigi XerriChristopher J. BurnsDaniel C. PevearDenis M. Daigle VNRX-5133 (Taniborbactam), a Broad-Spectrum Inhibitor of Serine- and Metallo-β-Lactamases, Restores Activity of Cefepime in Enterobacterales and Pseudomonas aeruginosa. Antimicrobial Agents and Chemotherapy 2019, 64, e01963-19, 10.1128/aac.01963-19.
- John, K.J.; Chatwin, C.L.; Hamrick, J.C.; Moeck, G.; Pevear, D.C. Rescue of Ceftibuten Activity by the Oral β-Lactamase Inhibitor VNRX-7145 against Enterobacteriaceae Expressing Class A, C and/or D β-Lactamases. In Proceedings of the ASM 2019; San Francisco, CA, 2019.
- Hamrick, J.; Chatwin, Cassandra John, K.; Burns, C.; Xerri, L.; Moeck, G.; Pevear, D.C. Selection of ceftibuten as the partner antibiotic for the oral beta-lactamase inhibitor VNRX-7145. In Proceedings of the ECCMID 2019; Amsterdam, 2019
- Mojgan Sabet; Ziad Tarazi; David C. Griffith; In Vivo Activity of QPX7728, an Ultrabroad-Spectrum Beta-Lactamase Inhibitor, In Combination with Beta-Lactams Against Carbapenem-Resistant K. pneumoniae. Antimicrobial Agents and Chemotherapy 2020, 64, e01267-20, 10.1128/aac.01267-20.
- Rubio-Aparicio, D.; Nelson, K.; Griffith, D.C.; Dudley, M.N.; Lomovskaya, O. QPX7728: In Vitro Activity in Combination with Oral Beta-Lactam Antibiotics against Enterobacteriaceae. In Proceedings of the ASM 2019; San Francisco, CA, 2019
- Lucas Tselepis; Gareth W. Langley; Ali F. Aboklaish; Emma Widlake; Dana E. Jackson; Timothy R. Walsh; T.R. Walsha C.J. Schofield; Jürgen Brem; Jonathan M. Tyrrell; In vitro efficacy of imipenem-relebactam and cefepime-AAI101 against a global collection of ESBL-positive and carbapenemase-producing Enterobacteriaceae. International Journal of Antimicrobial Agents 2020, 56, 105925, 10.1016/j.ijantimicag.2020.105925.
- Jared L. Crandon; David P. Nicolau; In Vitro Activity of Cefepime/AAI101 and Comparators against Cefepime Non-susceptible Enterobacteriaceae. Pathogens 2015, 4, 620-625, 10.3390/pathogens4030620.
- Mushtaq, S.; Chaudhry, A.; Adkin, R.; Woodford, N.; Benedict, N.; Pypstra, R.; Shapiro, S. In-vitro activity of diverse β-lactam/aai101 combinations vs. Multidrug-resistant gram-negative clinical strains. In Proceedings of the ECCMID 2014; Barcelona, 2014.
- Nordmann, P.; Girlich, D.; Benedict, N.; Pypstra, R.; Shapiro, S. Characterization of b-lactamase inhibition by aai101. In Proceedings of the ECCMID 2014; Barcelona, 2014.
- Krisztina M. Papp-Wallace; Christopher R. Bethel; Jocelyne Caillon; Melissa D. Barnes; Gilles Potel; Saralee Bajaksouzian; Joseph D. Rutter; Amokrane Reghal; Stuart Shapiro; Magdalena A. Taracila; et al.Michael R. JacobsRobert A. BonomoCedric Jacqueline Beyond Piperacillin-Tazobactam: Cefepime and AAI101 as a Potent β-Lactam−β-Lactamase Inhibitor Combination. Antimicrobial Agents and Chemotherapy 2019, 63, e00105-19, 10.1128/aac.00105-19.
- Ian Morrissey; Sophie Magnet; Stephen Hawser; Stuart Shapiro; Philipp Knechtle; In Vitro Activity of Cefepime-Enmetazobactam against Gram-Negative Isolates Collected from U.S. and European Hospitals during 2014–2015. Antimicrobial Agents and Chemotherapy 2019, 63, e00514-19, 10.1128/aac.00514-19.
- John D. Buynak; A.Srinivasa Rao; Venkata Ramana Doppalapudi; Greg Adam; Peter J. Petersen; Sirishkumar D. Nidamarthy; The synthesis and evaluation of 6-alkylidene-2'β-substituted penam sulfones as β-lactamase inhibitors. Bioorganic & Medicinal Chemistry Letters 1999, 9, 1997-2002, 10.1016/s0960-894x(99)00325-x.
- Matthew Kalp; Anjaneyulu Sheri; John D. Buynak; Christopher R. Bethel; Robert A. Bonomo; Paul R. Carey; Efficient Inhibition of Class A and Class D β-Lactamases by Michaelis Complexes. Journal of Biological Chemistry 2007, 282, 21588-21591, 10.1074/jbc.c700080200.
- John D. Buynak; The Discovery and Development of Modified Penicillin- and Cephalosporin- Derived β-Lactamase Inhibitors. Current Medicinal Chemistry 2004, 11, 1951-1964, 10.2174/0929867043364847.
- Sarah M. Drawz; Christopher R. Bethel; Venkata R. Doppalapudi; Anjaneyulu Sheri; Sundar Ram Reddy Pagadala; Andrea M. Hujer; Marion J. Skalweit; Vernon E. Anderson; Shu G. Chen; John D. Buynak; et al.Robert A. Bonomo Penicillin Sulfone Inhibitors of Class D β-Lactamases. Antimicrobial Agents and Chemotherapy 2010, 54, 1414-1424, 10.1128/aac.00743-09.
- Germán Bou; Elena Santillana; Anjaneyulu Sheri; Alejandro Beceiro; Jared M. Sampson; Matthew Kalp; Christopher R. Bethel; Anne M. Distler; Sarah M. Drawz; Sundar Ram Reddy Pagadala; et al.Focco Van Den AkkerRobert A. BonomoAntonio RomeroJohn D. Buynak Design, Synthesis, and Crystal Structures of 6-Alkylidene-2′-Substituted Penicillanic Acid Sulfones as Potent Inhibitors ofAcinetobacter baumanniiOXA-24 Carbapenemase. Journal of the American Chemical Society 2010, 132, 13320-13331, 10.1021/ja104092z.
- Juan C. Vázquez-Ucha; María Maneiro; Marta Martínez-Guitián; John Buynak; Christopher R. Bethel; Robert A. Bonomo; Germán Bou; Margarita Poza; Concepción González-Bello; Alejandro Beceiro; et al. Activity of the β-Lactamase Inhibitor LN-1-255 against Carbapenem-Hydrolyzing Class D β-Lactamases from Acinetobacter baumannii. Antimicrobial Agents and Chemotherapy 2017, 61, e01172-17, 10.1128/aac.01172-17.
- Juan A. Vallejo; Marta Martínez-Guitián; Juan Carlos Vázquez-Ucha; Concepción González-Bello; Margarita Poza; John D. Buynak; Christopher R. Bethel; Robert A. Bonomo; Germán Bou; Alejandro Beceiro; et al. LN-1-255, a penicillanic acid sulfone able to inhibit the class D carbapenemase OXA-48. Journal of Antimicrobial Chemotherapy 2016, 71, 2171-2180, 10.1093/jac/dkw105.
- Juan Carlos Vázquez-Ucha; Marta Martínez-Guitián; María Maneiro; Kelly Conde-Pérez; Laura Álvarez-Fraga; Gabriel Torrens; Antonio Oliver; John D. Buynak; Robert A. Bonomo; Germán Bou; et al.Concepción González-BelloMargarita PozaAlejandro Beceiro Therapeutic Efficacy of LN-1-255 in Combination with Imipenem in Severe Infection Caused by Carbapenem-Resistant Acinetobacter baumannii. Antimicrobial Agents and Chemotherapy 2019, 63, e01092-19, 10.1128/aac.01092-19.
- Diana Rodríguez; Maria Maneiro; Juan Carlos Vázquez-Ucha; Alejandro Beceiro; Concepción González-Bello; 6-Arylmethylidene Penicillin-Based Sulfone Inhibitors for Repurposing Antibiotic Efficiency in Priority Pathogens. Journal of Medicinal Chemistry 2020, 63, 3737-3755, 10.1021/acs.jmedchem.0c00127.
- David T. Davies; Simon Leiris; Nicolas Sprynski; Jérôme Castandet; Clarisse Lozano; Justine Bousquet; Magdalena Zalacain; Srinivas Vasa; Praveen Kumar Dasari; Ramesh Pattipati; et al.Naresh VempalaSwetha GujjewarSyamkumar GodiRaju JallalaRajashekar Reddy SathyapNarasimha A. DarshanojuVengala R. RavuRamakrishna Reddy JuventhalaNarender PottabathiniSomesh SharmaSrinivasu PothukanuriKirsty HoldenPeter WarnFrancesca MarcocciaManuela BenvenutiCecilia PozziStefano ManganiJean-Denis DocquierMarc LemonnierMartin Everett ANT2681: SAR Studies Leading to the Identification of a Metallo-β-lactamase Inhibitor with Potential for Clinical Use in Combination with Meropenem for the Treatment of Infections Caused by NDM-Producing Enterobacteriaceae. ACS Infectious Diseases 2020, 6, 2419-2430, 10.1021/acsinfecdis.0c00207.
- Martin Everett; Nicolas Sprynski; Alicia Coelho; Jérôme Castandet; Maëlle Bayet; Juliette Bougnon; Clarisse Lozano; David T. Davies; Simon Leiris; Magdalena Zalacain; et al.Ian MorrisseySophie MagnetKirsty HoldenPeter WarnFilomena De LucaJean-Denis DocquierMarc Lemonnier Discovery of a Novel Metallo-β-Lactamase Inhibitor That Potentiates Meropenem Activity against Carbapenem-Resistant Enterobacteriaceae. Antimicrobial Agents and Chemotherapy 2018, 62, e00074-18, 10.1128/aac.00074-18.
- John D. Buynak; Hansong Chen; Lakshminaryana Vogeti; Venkat Rao Gadhachanda; Christine A. Buchanan; Timothy Palzkill; Robert W. Shaw; James Spencer; Timothy R. Walsh; Penicillin-derived inhibitors that simultaneously target both metallo- and serine-β-lactamases. Bioorganic & Medicinal Chemistry Letters 2004, 14, 1299-1304, 10.1016/j.bmcl.2003.12.037.
- † Aranapakam M. Venkatesan; † Yansong Gu; † Osvaldo Dos Santos; ‡ Takao Abe; † Atul Agarwal; † Youjun Yang; † Peter J. Petersen; † William J. Weiss; Tarek S. Mansour; § Michiyoshi Nukaga; et al.‖ Andrea M. Hujer‖ And Robert A. Bonomo§ James R. Knox Structure−Activity Relationship of 6-Methylidene Penems Bearing Tricyclic Heterocycles as Broad-Spectrum β-Lactamase Inhibitors: Crystallographic Structures Show Unexpected Binding of 1,4-Thiazepine Intermediates. Journal of Medicinal Chemistry 2004, 47, 6556-6568, 10.1021/jm049680x.
- Aranapakam M. Venkatesan; Atul Agarwal; Takao Abe; Hideki Ushirogochi; Itsuka Yamamura; Mihira Ado; Takasaki Tsuyoshi; Osvaldo Dos Santos; Yansong Gu; Fuk-Wah Sum; et al.Zhong LiGerry FranciscoYang-I LinPeter J. PetersenYouJun YangToshio KumagaiWilliam J. WeissDavid M. ShlaesJames R. KnoxTarek S. Mansour Structure-Activity Relationship of 6-Methylidene Penems Bearing 6,5 Bicyclic Heterocycles as Broad-Spectrum β-Lactamase Inhibitors: Evidence for 1,4-Thiazepine Intermediates with C7 R Stereochemistry by Computational Methods. Journal of Medicinal Chemistry 2006, 49, 4623-4637, 10.1021/jm060021p.
- William J. Weiss; Peter J. Petersen; Timothy M. Murphy; Luanna Tardio; YouJun Yang; Patricia A. Bradford; Aranapakam M. Venkatesan; Takao Abe; Takeshi Isoda; Ado Mihira; et al.Hideki UshirogochiTsuyoshi TakasakeSteve ProjanJohn O'connellTarek S. Mansour In Vitro and In Vivo Activities of Novel 6-Methylidene Penems as β-Lactamase Inhibitors. Antimicrobial Agents and Chemotherapy 2004, 48, 4589-4596, 10.1128/aac.48.12.4589-4596.2004.
- Rie Nagano; Yuka Adachi; Hideaki Imamura; Koji Yamada; Terutaka Hashizume; Hajime Morishima; Carbapenem Derivatives as Potential Inhibitors of Various β-Lactamases, Including Class B Metallo-β-Lactamases. Antimicrobial Agents and Chemotherapy 1999, 43, 2497-2503, 10.1128/aac.43.10.2497.
- Rie Nagano; Yuka Adachi; Terutaka Hashizume; Hajime Morishima; In vitro antibacterial activity and mechanism of action of J-111,225, a novel 1β-methylcarbapenem, against transferable IMP-1 metallo-β-lactamase producers. Journal of Antimicrobial Chemotherapy 2000, 45, 271-276, 10.1093/jac/45.3.271.
- Rie Nagano; Kaneyoshi Shibata; Yuka Adachi; Hideaki Imamura; Terutaka Hashizume; Hajime Morishima; In Vitro Activities of Noveltrans-3,5-Disubstituted Pyrrolidinylthio-1β-Methylcarbapenems with Potent Activities against Methicillin-Resistant Staphylococcus aureus andPseudomonas aeruginosa. Antimicrobial Agents and Chemotherapy 2000, 44, 489-495, 10.1128/aac.44.3.489-495.2000.
- Sudhakar Reddy Ganta; Senthil Perumal; Sundar Ram Reddy Pagadala; Ørjan Samuelsen; James Spencer; R. F. Pratt; John D. Buynak; Approaches to the simultaneous inactivation of metallo- and serine-β-lactamases. Bioorganic & Medicinal Chemistry Letters 2009, 19, 1618-1622, 10.1016/j.bmcl.2009.02.018.
- Mariano M. González; Magda Kosmopoulou; Maria F. Mojica; Valerie Castillo; Philip Hinchliffe; Ilaria Pettinati; Jürgen Brem; Christopher J. Schofield; S. Graciela Mahler; Robert A. Bonomo; et al.Leticia I. LlarrullJames SpencerAlejandro J. Vila Bisthiazolidines: A Substrate-Mimicking Scaffold as an Inhibitor of the NDM-1 Carbapenemase. ACS Infectious Diseases 2015, 1, 544-554, 10.1021/acsinfecdis.5b00046.
- Thomas F. Durand-Réville; Satenig Guler; Janelle Comita-Prevoir; Brendan Chen; Neil Bifulco; Hoan Huynh; Sushmita Lahiri; Adam B. Shapiro; Sarah M. McLeod; Nicole M. Carter; et al.Samir H MoussaCamilo Velez-VegaNelson B. OlivierRobert McLaughlinNing GaoJason ThresherTiffany PalmerBeth AndrewsRobert A. GiacobbeJoseph V. NewmanDavid E. EhmannBoudewijn De JongeJohn O'donnellJohn P. MuellerRuben TommasiAlita A. Miller ETX2514 is a broad-spectrum β-lactamase inhibitor for the treatment of drug-resistant Gram-negative bacteria including Acinetobacter baumannii. Nature Microbiology 2017, 2, 17104-17104, 10.1038/nmicrobiol.2017.104.
- Juan Carlos Vázquez-Ucha; Marta Martínez-Guitián; María Maneiro; Kelly Conde-Pérez; Laura Álvarez-Fraga; Gabriel Torrens; Antonio Oliver; John D. Buynak; Robert A. Bonomo; Germán Bou; et al.Concepción González-BelloMargarita PozaAlejandro Beceiro Therapeutic Efficacy of LN-1-255 in Combination with Imipenem in Severe Infection Caused by Carbapenem-Resistant Acinetobacter baumannii. Antimicrobial Agents and Chemotherapy 2019, 63, e01092-19, 10.1128/aac.01092-19.
- Helio S. Sader; David J. Farrell; Robert K. Flamm; Ronald N. Jones; Variation in Potency and Spectrum of Tigecycline Activity against Bacterial Strains from U.S. Medical Centers since Its Approval for Clinical Use (2006 to 2012). Antimicrobial Agents and Chemotherapy 2014, 58, 2274-2280, 10.1128/aac.02684-13.
- Silvia Corcione; Tommaso Lupia; Alberto Enrico Maraolo; Simone Mornese Pinna; Ivan Gentile; Francesco G. De Rosa; Carbapenem-sparing strategy. Current Opinion in Infectious Diseases 2019, 32, 663-673, 10.1097/qco.0000000000000598.
- Alejandro Iregui; Zeb Khan; David Landman; John Quale; Activity of Meropenem with a Novel Broader-Spectrum β-Lactamase Inhibitor, WCK 4234, against Gram-Negative Pathogens Endemic to New York City. Antimicrobial Agents and Chemotherapy 2019, 64, e01666-19, 10.1128/aac.01666-19.
- Anou M. Somboro; John Osei Sekyere; Daniel Gyamfi Amoako; Hezekiel M. Kumalo; René Khan; Linda A. Bester; Sabiha Y. Essack; In vitro potentiation of carbapenems with tannic acid against carbapenemase-producing enterobacteriaceae: exploring natural products as potential carbapenemase inhibitors. Journal of Applied Microbiology 2018, 126, 452-467, 10.1111/jam.14141.
- Tomefa E Asempa; Kamilia Abdelraouf; David P Nicolau; Metallo-β-lactamase resistance in Enterobacteriaceae is an artefact of currently utilized antimicrobial susceptibility testing methods. Journal of Antimicrobial Chemotherapy 2020, 75, 997-1005, 10.1093/jac/dkz532.