
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Flora De Conto | -- | 2163 | 2024-03-27 17:55:59 | | | |
| 2 | Mona Zou | Meta information modification | 2163 | 2024-04-15 09:24:55 | | |
Video Upload Options
Influenza is one of the most prevalent causes of death worldwide. Influenza A viruses (IAVs) naturally infect various avian and mammalian hosts, causing seasonal epidemics and periodic pandemics with high morbidity and mortality. The recent SARS-CoV-2 pandemic showed how an animal virus strain could unpredictably acquire the ability to infect humans with high infection transmissibility. Importantly, highly pathogenic avian influenza A viruses (AIVs) may cause human infections with exceptionally high mortality. Because these latter infections pose a pandemic potential, analyzing the ecology and evolution features of host expansion helps to identify new broad-range therapeutic strategies. Although IAVs are the prototypic example of molecular strategies that capitalize on their coding potential, the outcome of infection depends strictly on the complex interactions between viral and host cell factors. Most of the studies have focused on the influenza virus, while the contribution of host factors remains largely unknown. Therefore, a comprehensive understanding of mammals’ host response to AIV infection is crucial.
1. Avian Influenza A Virus Infection Transmission to Humans
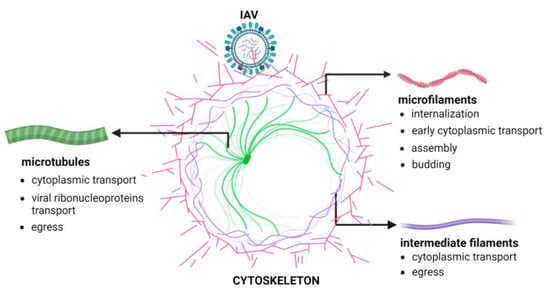
2. The Cellular Cytoskeleton
3. The Involvement of the Cellular Cytoskeleton in Influenza Virus Infection
References
- Swayne, D.E.; Suarez, D.L. Highly pathogenic avian influenza. Rev. Sci. Tech. 2000, 19, 463–482.
- Chen, J.; Lee, K.H.; Steinhauer, D.A.; Stevens, D.J.; Skehel, J.J.; Wiley, D.C. Structure of the hemagglutinin precursor cleavage site, a determinant of influenza pathogenicity and the origin of the labile conformation. Cell 1998, 95, 409–417.
- Escalera-Zamudio, M.; Golden, M.; Gutiérrez, B.; Thézé, J.; Keown, J.R.; Carrique, L.; Bowden, T.A.; Pybus, O.G. Parallel evolution in the emergence of highly pathogenic avian influenza A viruses. Nat Commun 2020, 11, 5511.
- Centers for Disease Control and Prevention. Available online: https://www.cdc.gov/flu/avianflu/avian-in-humans.htm (accessed on 31 January 2024).
- World Health Organization. Influenza Fact Sheet. Available online: https://www.who.int/en/news-room/fact-sheets/detail/influenza-(seasonal) (accessed on 3 October 2023).
- Gao, R.; Cao, B.; Hu, Y.; Feng, Z.; Wang, D.; Hu, W.; Chen, J.; Jie, Z.; Qiu, H.; Xu, K.; et al. Human infection with a novel avian-origin influenza A (H7N9) virus. N. Engl. J. Med. 2013, 368, 1888–1897.
- Jiang, L.; Chen, H.; Li, C. Advances in deciphering the interactions between viral proteins of influenza A virus and host cellular proteins. Cell Insight 2023, 2, 100079.
- Webster, R.G. Predictions for future human influenza pandemics. J. Infect. Dis. 1997, 176, S14–S19.
- Webster, R.G. Influenza virus: Transmission between species and relevance to emergence of the next human pandemic. Arch. Virol. Suppl. 1997, 13, 105–113.
- Webby, R.J.; Webster, R.G. Are we ready for pandemic influenza? Science 2003, 302, 1519–1522.
- de Graaf, M.; Fouchier, R.A. Role of receptor binding specificity in influenza A virus transmission and pathogenesis. EMBO J. 2014, 33, 823–841.
- Steel, J.; Lowen, A.C.; Mubareka, S.; Palese, P. Transmission of influenza virus in a mammalian host is increased by PB2 amino acids 627K or 627E/701N. PLoS Pathog. 2009, 5, e1000252.
- Huang, X.; Zheng, M.; Wang, P.; Mok, B.W.; Liu, S.; Lau, S.Y.; Chen, P.; Liu, Y.C.; Liu, H.; Chen, Y.; et al. An NS-segment exonic splicing enhancer regulates influenza A virus replication in mammalian cells. Nat. Commun. 2017, 8, 14751.
- Du, W.; de Vries, E.; van Kuppeveld, F.J.M.; Matrosovich, M.; de Haan, C.A.M. Second sialic acid-binding site of influenza A virus neuraminidase: Binding receptors for efficient release. FEBS J. 2021, 288, 5598–5612.
- Vourc’h, G.; Moutou, F.; Morand, S.; Jourdain, E. Zoonoses the Ties that Bind Humans to Animals ; Éditions Quae: Versailles, France, 2022.
- Chen, H.; Smith, G.J.; Li, K.S.; Wang, J.; Fan, X.H.; Rayner, J.M.; Vijaykrishna, D.; Zhang, J.X.; Zhang, L.J.; Guo, C.T.; et al. Establishment of multiple sublineages of H5N1 influenza virus in Asia: Implications for pandemic control. Proc. Natl. Acad. Sci. USA 2006, 103, 2845–2850.
- Yuen, K.Y.; Chan, P.K.; Peiris, M.; Tsang, D.N.; Que, T.L.; Shortridge, K.F.; Cheung, P.T.; To, W.K.; Ho, E.T.; Sung, R.; et al. Clinical features and rapid viral diagnosis of human disease associated with avian influenza A H5N1 virus. Lancet 1998, 351, 467–471.
- Claas, E.C.; de Jong, J.C.; van Beek, R.; Rimmelzwaan, G.F.; Osterhaus, A.D. Human influenza virus A/HongKong/156/97 (H5N1) infection. Vaccine 1998, 16, 977–978.
- Bridges, C.B.; Lim, W.; Hu-Primmer, J.; Sims, L.; Fukuda, K.; Mak, K.H.; Rowe, T.; Thompson, W.W.; Conn, L.; Lu, X.; et al. Risk of influenza A (H5N1) infection among poultry workers, Hong Kong, 1997–1998. J. Infect. Dis. 2002, 185, 1005–1010.
- Katz, J.M.; Lim, W.; Bridges, C.B.; Rowe, T.; Hu-Primmer, J.; Lu, X.; Abernathy, R.A.; Clarke, M.; Conn, L.; Kwong, H.; et al. Antibody response in individuals infected with avian influenza A (H5N1) viruses and detection of anti-H5 antibody among household and social contacts. J. Infect. Dis. 1999, 180, 1763–1770.
- Bahgat, M.M.; Kutkat, M.A.; Nasraa, M.H.; Mostafa, A.; Webby, R.; Bahgat, I.M.; Ali, M.A. Characterization of an avian influenza virus H5N1 Egyptian isolate. J. Virol. Methods 2009, 159, 244–250.
- World Health Organization. Cumulative Number of Confirmed Human Cases for Avian Influenza A (H5N1) Reported to WHO, 2003–2023. 2023. Available online: https://www.who.int/publications/m/item/cumulative-number-of-confirmed-human-cases-for-avian-influenza-a(h5n1)-reported-to-who-2003-2022-5-jan-2023 (accessed on 26 January 2024).
- Xie, R.; Edwards, K.M.; Wille, M.; Wei, X.; Wong, S.S.; Zanin, M.; El-Shesheny, R.; Ducatez, M.; Poon, L.L.M.; Kayali, G.; et al. The episodic resurgence of highly pathogenic avian influenza H5 virus. Nature 2023, 622, 810–817.
- Monne, I.; Yamage, M.; Dauphin, G.; Claes, F.; Ahmed, G.; Giasuddin, M.; Salviato, A.; Ormelli, S.; Bonfante, F.; Schivo, A.; et al. Reassortant avian influenza A (H5N1) viruses with H9N2-PB1 gene in poultry, Bangladesh. Emerg. Infect. Dis. 2013, 19, 1630–1634.
- Gerloff, N.A.; Khan, S.U.; Balish, A.; Shanta, I.S.; Simpson, N.; Berman, L.; Haider, N.; Poh, M.K.; Islam, A.; Gurley, E.; et al. Multiple reassortment events among highly pathogenic avian influenza A(H5N1) viruses detected in Bangladesh. Virology 2014, 450–451, 297–307.
- Li, Y.; Shi, J.; Zhong, G.; Deng, G.; Tian, G.; Ge, J.; Zeng, X.; Song, J.; Zhao, D.; Liu, L.; et al. Continued evolution of H5N1 influenza viruses in wild birds, domestic poultry, and humans in China from 2004 to 2009. J. Virol. 2010, 84, 8389–8397.
- Li, Z.; Chen, H.; Jiao, P.; Deng, G.; Tian, G.; Li, Y.; Hoffmann, E.; Webster, R.G.; Matsuoka, Y.; Yu, K. Molecular basis of replication of duck H5N1 influenza viruses in a mammalian mouse model. J. Virol. 2005, 79, 12058–12064.
- Hatta, M.; Gao, P.; Halfmann, P.; Kawaoka, Y. Molecular basis for high virulence of Hong Kong H5N1 influenza A viruses. Science 2001, 293, 1840–1842.
- Pan, M.; Gao, R.; Lv, Q.; Huang, S.; Zhou, Z.; Yang, L.; Li, X.; Zhao, X.; Zou, X.; Tong, W.; et al. Human infection with a novel, highly pathogenic avian influenza A (H5N6) virus: Virological and clinical findings. J. Infect. 2016, 72, 52–59.
- Matrosovich, M.N.; Krauss, S.; Webster, R.G. H9N2 influenza A viruses from poultry in Asia have human virus-like receptor specificity. Virology 2001, 281, 156–162.
- Liu, N.; Song, W.; Wang, P.; Lee, K.; Chan, W.; Chen, H.; Cai, Z. Proteomics analysis of differential expression of cellular proteins in response to avian H9N2 virus infection in human cells. Proteomics 2008, 8, 1851–1858.
- Butt, K.M.; Smith, G.J.; Chen, H.; Zhang, L.J.; Leung, Y.H.; Xu, K.M.; Lim, W.; Webster, R.G.; Yuen, K.Y.; Peiris, J.S.; et al. Human infection with an avian H9N2 influenza A virus in Hong Kong in 2003. J. Clin. Microbiol. 2005, 43, 5760–5767.
- Ha, Y.; Stevens, D.J.; Skehel, J.J.; Wiley, D.C. X-ray structures of H5 avian and H9 swine influenza virus hemagglutinins bound to avian and human receptor analogs. Proc. Natl. Acad. Sci. USA 2001, 98, 11181–11186.
- Huang, R.; Liu, J.; Liang, W.; Wang, A.; Liu, Z.; Yang, Y.; Lv, J.; Bao, Y.; Gao, Y.; Miao, Z.; et al. Response profiles of cytokines and chemokines against avian H9N2 influenza virus within the mouse lung. Med. Microbiol. Immunol. 2014, 203, 109–114.
- Ungchusak, K.; Auewarakul, P.; Dowell, S.F.; Kitphati, R.; Auwanit, W.; Puthavathana, P.; Uiprasertkul, M.; Boonnak, K.; Pittayawonganon, C.; Cox, N.J.; et al. Probable person-to-person transmission of avian influenza A (H5N1). N. Engl. J. Med. 2005, 352, 333–340.
- Kandun, I.N.; Wibisono, H.; Sedyaningsih, E.R.; Yusharmen; Hadisoedarsuno, W.; Purba, W.; Santoso, H.; Septiawati, C.; Tresnaningsih, E.; Heriyanto, B.; et al. Three Indonesian clusters of H5N1 virus infection in 2005. N. Engl. J. Med. 2006, 355, 2186–2194.
- Wang, H.; Feng, Z.; Shu, Y.; Yu, H.; Zhou, L.; Zu, R.; Huai, Y.; Dong, J.; Bao, C.; Wen, L.; et al. Probable limited person-to-person transmission of highly pathogenic avian influenza A (H5N1) virus in China. Lancet 2008, 371, 1427–1434.
- Li, Q.; Zhou, L.; Zhou, M.; Chen, Z.; Li, F.; Wu, H.; Xiang, N.; Chen, E.; Tang, F.; Wang, D.; et al. Epidemiology of human infections with avian influenza A(H7N9) virus in China. N. Engl. J. Med. 2014, 370, 520–532.
- Zhang, Z.H.; Meng, L.S.; Kong, D.H.; Liu, J.; Li, S.Z.; Zhou, C.; Sun, J.; Song, R.J.; Wu, J.J. A Suspected Person-to-person Transmission of Avian Influenza A (H7N9) Case in Ward. Chin. Med. J. 2017, 130, 1255–1256.
- Du Ry van Beest Holle, M.; Meijer, A.; Koopmans, M.; de Jager, C.M. Human-to-human transmission of avian influenza A/H7N7, The Netherlands, 2003. Eurosurveillance 2005, 10, 3–4.
- Parry, J. H7N9 avian flu infects humans for the first time. BMJ 2013, 346, f2151.
- To, K.K.; Tsang, A.K.; Chan, J.F.; Cheng, V.C.; Chen, H.; Yuen, K.Y. Emergence in China of human disease due to avian influenza A(H10N8)—Cause for concern? J. Infect. 2014, 68, 205–215.
- Shi, W.; Shi, Y.; Wu, Y.; Liu, D.; Gao, G.F. Origin and molecular characterization of the human-infecting H6N1 influenza virus in Taiwan. Protein Cell 2013, 4, 846–853.
- Wu, A.; Su, C.; Wang, D.; Peng, Y.; Liu, M.; Hua, S.; Li, T.; Gao, G.F.; Tang, H.; Chen, J.; et al. Sequential reassortments underlie diverse influenza H7N9 genotypes in China. Cell Host Microbe 2013, 14, 446–452.
- Qi, W.; Zhou, X.; Shi, W.; Huang, L.; Xia, W.; Liu, D.; Li, H.; Chen, S.; Lei, F.; Cao, L.; et al. Genesis of the novel human-infecting influenza A(H10N8) virus and potential genetic diversity of the virus in poultry, China. Eurosurveillance 2014, 19, 20841.
- Li, M.; Peng, D.; Cao, H.; Yang, X.; Li, S.; Qiu, H.J.; Li, L.F. The Host Cytoskeleton Functions as a Pleiotropic Scaffold: Orchestrating Regulation of the Viral Life Cycle and Mediating Host Antiviral Innate Immune Responses. Viruses 2023, 15, 1354.
- Mostowy, S.; Cossart, P. Septins: The fourth component of the cytoskeleton. Nat. Rev. Mol. Cell Biol. 2012, 13, 183–194.
- Liu, L.; Luo, Q.; Sun, J.; Song, G. Nucleus and nucleus-cytoskeleton connections in 3D cell migration. Exp. Cell Res. 2016, 348, 56–65.
- Fuchs, E.; Cleveland, D.W. A structural scaffolding of intermediate filaments in health and disease. Science 1998, 279, 514–519.
- Słońska, A.; Polowy, R.; Golke, A.; Cymerys, J. Role of cytoskeletal motor proteins in viral infection. Postepy Hig. Med. Dosw. 2012, 66, 810–817.
- MacTaggart, B.; Kashina, A. Posttranslational modifications of the cytoskeleton. Cytoskeleton 2021, 78, 142–173.
- Buxboim, A.; Kronenberg-Tenga, R.; Salajkova, S.; Avidan, N.; Shahak, H.; Thurston, A.; Medalia, O. Scaffold, mechanics and functions of nuclear lamins. FEBS Lett. 2023, 597, 2791–2805.
- Buchwalter, A. Intermediate, but not average: The unusual lives of the nuclear lamin proteins. Curr. Opin. Cell Biol. 2023, 84, 102220.
- Hohmann, T.; Dehghani, F. The Cytoskeleton-A Complex Interacting Meshwork. Cells 2019, 8, 362.
- Pollard, T.D.; Cooper, J.A. Actin and actin-binding proteins. A critical evaluation of mechanisms and functions. Annu. Rev. Biochem. 1986, 55, 987–1035.
- Carlier, M.F.; Shekhar, S. Global treadmilling coordinates actin turnover and controls the size of actin networks. Nat. Rev. Mol. Cell Biol. 2017, 18, 389–401.
- Davidson, P.M.; Cadot, B. Actin on and around the Nucleus. Trends Cell Biol. 2021, 31, 211–223.
- Kloc, M.; Wilk, K.; Vargas, D.; Shirato, Y.; Bilinski, S.; Etkin, L.D. Potential structural role of non-coding and coding RNAs in the organization of the cytoskeleton at the vegetal cortex of Xenopus oocytes. Development 2005, 132, 3445–3457.
- Simpson, C.; Yamauchi, Y. Microtubules in Influenza Virus Entry and Egress. Viruses 2020, 12, 117.
- Rieder, C.L.; Faruki, S.; Khodjakov, A. The centrosome in vertebrates: More than a microtubule-organizing center. Trends Cell Biol. 2001, 11, 413–419.
- Gudimchuk, N.B.; McIntosh, J.R. Regulation of microtubule dynamics, mechanics and function through the growing tip. Nat. Rev. Mol. Cell. Biol. 2021, 22, 777–795.
- Guru, A.; Saravanan, S.; Sharma, D.; Narasimha, M. The microtubule end-binding proteins EB1 and Patronin modulate the spatiotemporal dynamics of myosin and pattern pulsed apical constriction. Development 2022, 149, dev199759.
- Ramaekers, F.C.; Bosman, F.T. The cytoskeleton and disease. J. Pathol. 2004, 204, 351–354.
- Wu, H.; Shen, Y.; Sivagurunathan, S.; Weber, M.S.; Adam, S.A.; Shin, J.H.; Fredberg, J.J.; Medalia, O.; Goldman, R.; Weitz, D.A. Vimentin intermediate filaments and filamentous actin form unexpected interpenetrating networks that redefine the cell cortex. Proc. Natl. Acad. Sci. USA 2022, 119, e2115217119.
- Redmond, C.J.; Coulombe, P.A. Intermediate filaments as effectors of differentiation. Curr. Opin. Cell Biol. 2021, 68, 155–162.
- Odell, J.; Lammerding, J. Lamins as structural nuclear elements through evolution. Curr. Opin. Cell Biol. 2023, 85, 102267.
- Aebi, U.; Cohn, J.; Buhle, L.; Gerace, L. The nuclear lamina is a meshwork of intermediate-type filaments. Nature 1986, 323, 560–564.
- Maly, I.V.; Hofmann, W.A. Myosins in the Nucleus. Adv. Exp. Med. Biol. 2020, 1239, 199–231.
- da Silva, E.S.; Naghavi, M.H. Microtubules and viral infection. Adv. Virus Res. 2023, 115, 87–134.
- Horníková, L.; Bruštíková, K.; Huérfano, S.; Forstová, J. Nuclear Cytoskeleton in Virus Infection. Int. J. Mol. Sci. 2022, J23, 578.
- Wang, I.H.; Burckhardt, C.J.; Yakimovich, A.; Greber, U.F. Imaging, Tracking and Computational Analyses of Virus Entry and Egress with the Cytoskeleton. Viruses 2018, 10, 166.
- Radtke, K.; Döhner, K.; Sodeik, B. Viral interactions with the cytoskeleton: A hitchhiker’s guide to the cell. Cell. Microbiol. 2006, 8, 387–400.
- Sun, X.; Whittaker, G.R. Role of the actin cytoskeleton during influenza virus internalization into polarized epithelial cells. Cell. Microbiol. 2007, 9, 1672–1682.
- Wu, W.; Panté, N. Vimentin plays a role in the release of the influenza A viral genome from endosomes. Virology 2016, 497, 41–52.
- De Conto, F.; Di Lonardo, E.; Arcangeletti, M.C.; Chezzi, C.; Medici, M.C.; Calderaro, A. Highly dynamic microtubules improve the effectiveness of early stages of human influenza A/NWS/33 virus infection in LLC-MK2 cells. PLoS ONE 2012, 7, e41207.
- Lakadamyali, M.; Rust, M.J.; Babcock, H.P.; Zhuang, X. Visualizing infection of individual influenza viruses. Proc. Natl. Acad. Sci. USA 2003, 100, 9280–9285.
- de Vries, E.; Tscherne, D.M.; Wienholts, M.J.; Cobos-Jiménez, V.; Scholte, F.; García-Sastre, A.; Rottier, P.J.; de Haan, C.A. Dissection of the influenza A virus endocytic routes reveals macropinocytosis as an alternative entry pathway. PLoS Pathog. 2011, 7, e1001329.
- Huang, X.; Yin, G.; Zhou, B.; Cai, Y.; Hu, J.; Huang, J.; Chen, Z.; Liu, Q.; Feng, X. KRT10 plays an important role in the release of viral genome from endosomes during H9N2 subtype AIV replication in HeLa cells. Vet. Microbiol. 2023, 284, 109824.
- Zhang, L.J.; Xia, L.; Liu, S.L.; Sun, E.Z.; Wu, Q.M.; Wen, L.; Zhang, Z.L.; Pang, D.W. A “Driver Switchover” Mechanism of Influenza Virus Transport from Microfilaments to Microtubules. ACS Nano 2018, 12, 474–484.
- Greber, U.F.; Way, M. A superhighway to virus infection. Cell 2006, 124, 741–754.
- Rust, M.J.; Lakadamyali, M.; Zhang, F.; Zhuang, X. Assembly of endocytic machinery around individual influenza viruses during viral entry. Nat. Struct. Mol. Biol. 2004, 11, 567–573.
- Hodgson, L.; Verkade, P.; Yamauchi, Y. Correlative Light and Electron Microscopy of Influenza Virus Entry and Budding. Methods Mol. Biol. 2018, 1836, 237–260.
- Ramos, I.; Stamatakis, K.; Oeste, C.L.; Pérez-Sala, D. Vimentin as a Multifaceted Player and Potential Therapeutic Target in Viral Infections. Int. J. Mol. Sci. 2020, 21, 4675.
- Lu, A.; Yang, J.; Huang, X.; Huang, X.; Yin, G.; Cai, Y.; Feng, X.; Zhang, X.; Li, Y.; Liu, Q. The Function behind the Relation between Lipid Metabolism and Vimentin on H9N2 Subtype AIV Replication. Viruses 2022, 14, 1814.
- Arcangeletti, M.C.; De Conto, F.; Ferraglia, F.; Pinardi, F.; Gatti, R.; Orlandini, G.; Covan, S.; Motta, F.; Rodighiero, I.; Dettori, G.; et al. Host-cell-dependent role of actin cytoskeleton during the replication of a human strain of influenza A virus. Arch. Virol. 2008, 153, 1209–1221.
- Simpson-Holley, M.; Ellis, D.; Fisher, D.; Elton, D.; McCauley, J.; Digard, P. A functional link between the actin cytoskeleton and lipid rafts during budding of filamentous influenza virions. Virology 2002, 301, 212–225.
- Kumakura, M.; Kawaguchi, A.; Nagata, K. Actin-myosin network is required for proper assembly of influenza virus particles. Virology 2015, 476, 141–150.
- De Conto, F.; Fazzi, A.; Razin, S.V.; Arcangeletti, M.C.; Medici, M.C.; Belletti, S.; Chezzi, C.; Calderaro, A. Mammalian Diaphanous-related formin-1 restricts early phases of influenza A/NWS/33 virus (H1N1) infection in LLC-MK2 cells by affecting cytoskeleton dynamics. Mol. Cell. Biochem. 2018, 437, 185–201.
- Bedi, S.; Ono, A. Friend or Foe: The Role of the Cytoskeleton in Influenza A Virus Assembly. Viruses 2019, 11, 46.
- De Conto, F.; Conversano, F.; Razin, S.V.; Belletti, S.; Arcangeletti, M.C.; Chezzi, C.; Calderaro, A. Host-cell dependent role of phosphorylated keratin 8 during influenza A/NWS/33 virus (H1N1) infection in mammalian cells. Virus Res. 2021, 295, 198333.
- Zhang, C.; Wu, Y.; Xuan, Z.; Zhang, S.; Wang, X.; Hao, Y.; Wu, J.; Zhang, S. p38MAPK, Rho/ROCK and PKC pathways are involved in influenza-induced cytoskeletal rearrangement and hyperpermeability in PMVEC via phosphorylating ERM. Virus Res. 2014, 192, 6–15.





