Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Richard Kozarek | -- | 2706 | 2024-03-06 18:15:55 | | | |
| 2 | Lindsay Dong | Meta information modification | 2706 | 2024-03-07 02:38:43 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Masood, M.; Low, D.E.; Deal, S.B.; Kozarek, R.A. Gastroesophageal Reflux Disease following Sleeve Gastrectomy. Encyclopedia. Available online: https://encyclopedia.pub/entry/55932 (accessed on 28 March 2026).
Masood M, Low DE, Deal SB, Kozarek RA. Gastroesophageal Reflux Disease following Sleeve Gastrectomy. Encyclopedia. Available at: https://encyclopedia.pub/entry/55932. Accessed March 28, 2026.
Masood, Muaaz, Donald E. Low, Shanley B. Deal, Richard A. Kozarek. "Gastroesophageal Reflux Disease following Sleeve Gastrectomy" Encyclopedia, https://encyclopedia.pub/entry/55932 (accessed March 28, 2026).
Masood, M., Low, D.E., Deal, S.B., & Kozarek, R.A. (2024, March 06). Gastroesophageal Reflux Disease following Sleeve Gastrectomy. In Encyclopedia. https://encyclopedia.pub/entry/55932
Masood, Muaaz, et al. "Gastroesophageal Reflux Disease following Sleeve Gastrectomy." Encyclopedia. Web. 06 March, 2024.
Copy Citation
Obesity is associated with serious comorbidities and economic implications. Bariatric surgery, most commonly Roux-en-Y gastric bypass and sleeve gastrectomy, are effective options for weight loss and the improvement of obesity-related comorbidities. With the growing obesity epidemic, there has been a concomitant rise in bariatric surgeries, particularly in sleeve gastrectomy. Gastroesophageal reflux disease (GERD) is highly prevalent in obese individuals, can significantly impact quality of life and may lead to serious complications. Obesity and GERD both improve with weight loss.
gastroesophageal
reflux
esophagitis
obesity
bariatric
gastric
1. Introduction
Obesity, defined as a body mass index (BMI) > 30 kg/m2, affects approximately four out of ten individuals in the United States and has serious health and economic implications [1]. The World Obesity Foundation has projected that over 4 billion people, or half of the world’s current population, will be overweight or obese by 2035 (World Obesity Federation, World Obesity Atlas, 2023). Obesity is associated with several chronic conditions including diabetes, hypertension, hyperlipidemia, obstructive sleep apnea, metabolic dysfunction-associated steatotic liver disease and malignancy [2][3][4]. Coinciding with the growing obesity epidemic, there has been an increase in the rate of bariatric surgeries over the last two decades [3]. Roux-en-Y gastric bypass (RYGB) and sleeve gastrectomy (SG) are the most commonly performed bariatric surgeries [3]. Sleeve gastrectomy surpassed RYGB in 2013 and continues to be the most widely performed bariatric surgery to date [3][5]. A shift towards minimally invasive bariatric surgery compared to open bariatric surgery has also been evident over the past 20 years [3].
Gastroesophageal reflux disease (GERD) is a chronic condition which is highly prevalent in obese individuals [6][7]. GERD manifests as heartburn and regurgitation although atypical or extraesophageal symptoms, in particular cough and chest pain, may be present in up to one-third of patients [8]. GERD has been documented in 62.4–73% of candidates for bariatric surgery [9][10]. Obesity and GERD both improve with weight loss [11][12]. However, as the incidence of minimally invasive SG increases, emerging data have revealed a risk of worsening GERD post-SG [5][13][14][15][16][17].
2. Gastroesophageal Reflux Disease following Sleeve Gastrectomy
2.1. What Is the Evidence for Worsening GERD following SG?
Sleeve gastrectomy remains one of the most effective options for weight loss and the improvement of obesity-related comorbidities, namely diabetes, hypertension, hyperlipidemia and obstructive sleep apnea [2][3][4]. Sleeve gastrectomy also has a slightly lower postoperative complication rate when compared to RYGB [18]. As the incidence of SG rises, however, GERD following SG has become a notable concern. A multitude of studies have associated sleeve gastrectomy with the exacerbation of pre-existing GERD. A retrospective review of 28 patients suggested that LSG may increase in the prevalence of GERD despite adequate weight loss [15]. Matar et al. retrospectively reviewed 517 patients and noted a higher prevalence of erosive esophagitis (EE) (37.9% vs. 17.6%, p = 0.0001), including severe EE, with SG vs. RYGB [16]. A decreased resting lower esophageal sphincter was also noted in the SG group [16]. A prospective, nonrandomized, open-label study of 30 patients noted GERD in two-thirds of patients following LSG [19].
2.2. Is De Novo GERD a Topic of Concern following Sleeve Gastrectomy?
In addition to worsening pre-existing GERD, new-onset or de novo GERD has been widely reported after SG. In a recent meta-analysis by Znamirowski et al., the pooled odds ratio (OR) for overall GERD occurrence post-LSG across eight studies and 599 patients was 3.61 (95% confidence interval: 1.92–6.79; p < 0.001) [20]. The combined percentage of patients with de novo GERD across five studies was 50.8% [20]. Across seven studies, the combined percentages of patients with LA Grade C and LA Grade D esophagitis were 4.3% and 3.3%, respectively [20]. However, significant heterogeneity was noted in the study [20]. In addition, there was variability in how GERD was documented, as studies used patients’ symptoms, PPI usage, endoscopic findings, pH testing, esophageal manometry or a combination of these parameters.
2.3. What Are the Underlying Mechanisms for De Novo or Increased GERD following a Sleeve Gastrectomy?
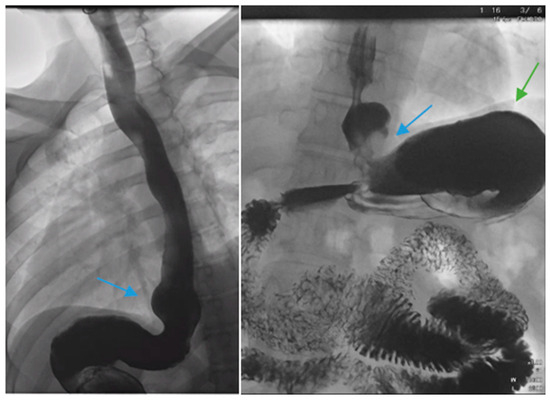
There are factors which may contribute to alleviating GERD after SG including weight loss leading to decreased abdominal pressure, reduced gastric volume and possibly accelerated gastric emptying [21][22]. In contrast, several pathophysiological mechanisms for de novo or increased GERD following SG have been described. Anatomic disruption of the antireflux barrier, including the esophagogastric junction, gastroesophageal flap valve, the angle of His, the gastric sling fibers and the fundus, may result in an increasing propensity for the reflux of gastric contents. Others discuss that caution must be made when positioning the staple line during SG in a manner that will preserve the antireflux barrier [23]. In contrast to RYGB, SG may be more likely to disrupt the angle of His and gastroesophageal junction attachments. There may be a functional impairment at the gastroesophageal junction such as with a hiatal hernia (Figure 1 and Figure 2).

Figure 1. Upper gastrointestinal series in a patient post-sleeve gastrectomy with early satiety and reflux reveals the following: a small, hiatal hernia and gastroesophageal reflux (blue arrow, left) in addition to abnormal angulation (blue arrow, right) and dilation of the gastric sleeve (green arrow, right). The patient underwent conversion to Roux-en-Y gastric bypass with marked clinical improvement.

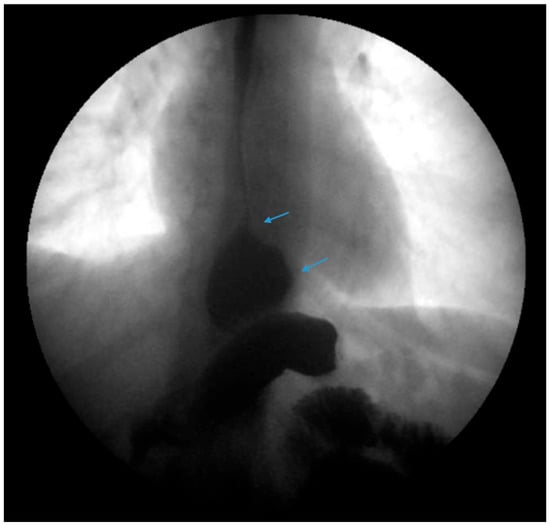
Figure 2. An upper gastrointestinal series reveals a moderate, sliding type-one hiatal hernia (bottom arrow) in addition to gastroesophageal reflux disease (top arrow) in a patient with morbid obesity.
Conditions in which there is an increase in intragastric pressure may lead to reflux. Greenan et al. explored gastric pressurization using high-resolution manometry and reflux using pH impedance in patients post-SG and post-RYGB as well as in symptomatic and asymptomatic control groups. Patients in the SG group had higher acid exposure time (median 6.0% vs. 0.2%), reflux episode numbers (median 63.0 vs. 37.5) and baseline intragastric pressure (median 17.3 mmHg vs. 13.1 mmHg) compared to the RYGB group, p < 0.001 [24].
2.4. The Current Management and Treatment Paradigms of GERD following SG
2.4.1. Lifestyle Modifications
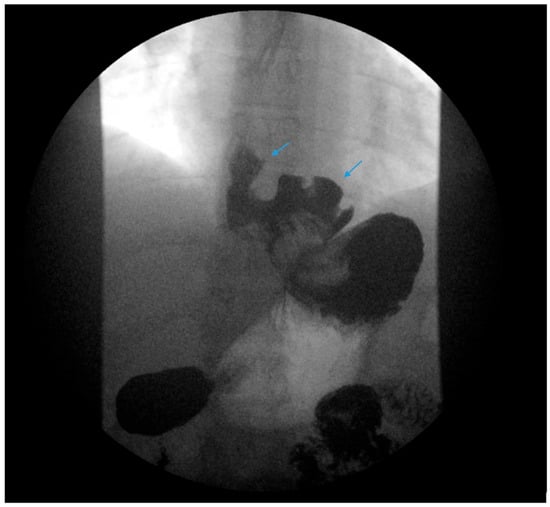
A comprehensive, stepwise approach to the management and treatment of GERD following SG has recently been published by Masood et al. [25]. Lifestyle modifications are initially recommended following SG as part of a comprehensive bariatric surgery center’s pre- and postoperative education and long-term follow-up plan with patients. Dietary education should emphasize the gradual progression of the postoperative diet from liquids to eventually healthy, protein-rich meals. Small portion sizes, typically ½ to 1 cup or 4 to 6 ounces, are suggested. Overfilling the stomach with a large portion may exacerbate reflux and induce regurgitation or food stasis in the distal esophagus, which may also be interpreted as GERD-like symptoms or induce stasis esophagitis if overfilling occurs repeatedly (Figure 3). Counseling regarding exercise, nutritional supplementation, the treatment of constipation and the avoidance of underlying risk factors of GERD, i.e., alcohol, tobacco, caffeine, chocolate and postprandial supination, should be provided.

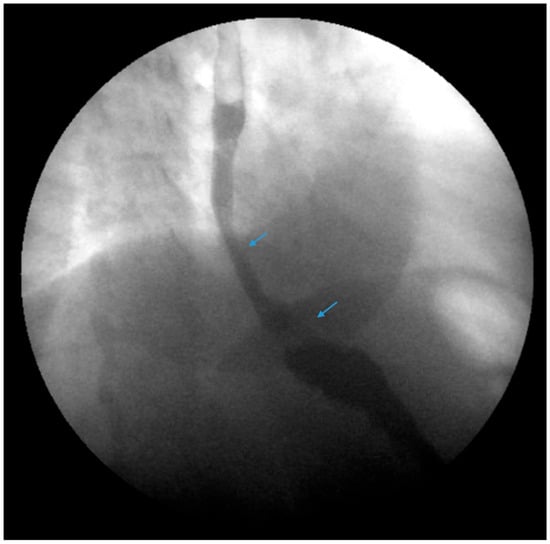
Figure 3. An upper gastrointestinal series reveals contrast filling of sleeve with reflux of a column of contrast into the distal esophagus (blue arrow) in a patient following sleeve gastrectomy.
2.4.2. Optimization of Medication Regimens
Patients are often prescribed a PPI or histamine-2 receptor antagonist (H2RA) to prevent ulcer formation and reflux. In patients with post-SG GERD, PPI and H2RA regimens should be reviewed and optimized accordingly. Of note, the novel potassium-competitive acid blocker, vonoprazan, has shown promising results in PPI-refractory GERD, although its use has not been assessed in the bariatric surgery population [26][27][28].
2.4.3. Diagnostic Work-Up
The authors recommend obtaining an esophagram, EGD, manometry and pH testing in selected patients presenting with GERD following SG for a further anatomic and physiologic evaluation of symptoms. Post-SG complications should be promptly identified and corrected. Stenosis, angulation and kinking of the gastric sleeve are associated with increased intragastric pressure and reflux (Figure 3) [29]. Ulcers and gastric sleeve leaks should be addressed appropriately.
2.4.4. Treatment of Underlying Postoperative Complications: Gastric Sleeve Stenosis
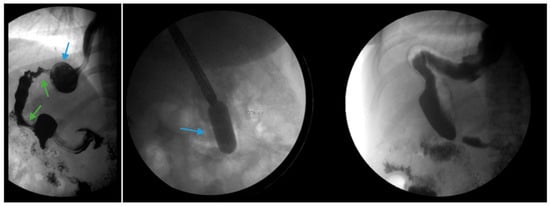
Endoscopic balloon dilation (EBD) has been shown to be a safe and efficacious modality for gastric sleeve stenosis (GSS) (Figure 4) [30][31]. A meta-analysis by Chang et al. of 18 studies involving a total of 426 patients revealed an overall success rate of 76% and an average of 1.8 dilations per patient [32]. Proximal GSS had a higher efficacy rate of 90% compared to distal GSS which had an efficacy rate of 70% [32]. Studies in the meta-analysis utilized through-the-scope, controlled radial expansion balloons, pneumatic balloons used for achalasia or a combination of the two [32]. The optimal balloon type, balloon size and the number of dilations required remains unclear [33]. Fully covered self-expanding metal stents (FCSEMS) have been reported to be effective in 70% of cases in which EBD failed though stent migration remains a concern [32][34].

Figure 4. (Left) An upper gastrointestinal series in a patient post-sleeve gastrectomy with recurrent dysphagia reveals dilation of the gastric sleeve at the cardia (blue arrow) with narrowing and delayed passage of contrast through the middle and distal portions (green arrows) of the gastric sleeve. (Middle) The patient underwent serial endoscopic through-the-scope balloon dilation to 20 mm (blue arrow) with improvement in sleeve stenosis (Right) and dysphagia.
2.4.5. Treatment of Underlying Postoperative Complications: Gastric Sleeve Leak
It is noteworthy to mention gastric sleeve leak (GSL), a serious, life-threatening complication of SG, which involves the leak of gastrointestinal contents from the staple line into the abdominal cavity. GSL may occur at any point along the staple line from the gastroesophageal junction to the antrum, although they most commonly occur at the angle of His. GSL confers a high risk of morbidity and mortality. The leak rate has been reported to be between 1% and 5% for patients who undergo SG as a primary procedure. The leak rate is >10% for patients who underwent SG as a revisional procedure, after failed laparoscopic adjustable gastric banding or vertical banded gastroplasty, likely due to increased dissection, prior staple lines, vascular integrity and the overall quality of tissue adhesions which may be encountered during a reoperation [35][36][37][38].
Csendes and Burgos proposed a classification of GSL according to the timing, location and severity of the leak [36][39]. Type I (subclinical) leaks are well-defined without septic complications or dissemination into the abdominal or pleural cavity. Type II leaks (clinical) are outlined by septic complications with generalized dissemination into the abdominal or pleural cavity. Leaks may be classified based on timing as early, intermediate and late if they occur 1–3 days, 4–7 days and ≥8 days following surgery, respectively. Leaks may also be classified using a combination of clinical and radiologic data as follows: Type A are microperforations without clinical or radiologic evidence of leak; Type B are leaks noted on radiographic studies without a clinical finding; and Type C are leaks with both radiologic and clinical evidence [40]. GSL may manifest as sepsis, abdominal pain, chest/shoulder pain and dizziness or it can be asymptomatic. Radiographic imaging, including upper gastrointestinal series and CT with contrast, are typically helpful in establishing the diagnosis.
2.4.6. Discussion of Risks Related to GERD with Potential Candidates for Sleeve Gastrectomy
Due to the association of SG with an increased risk of GERD, it is paramount for clinicians to discuss the risk of exacerbation of pre-existing GERD and the development of new-onset GERD during the shared decision-making process with potential candidates for SG (Figure 5). Data regarding the long-term risk of PPI use and the risk of cardiovascular adverse events should be considered as they relate to the treatment of GERD post-SG.

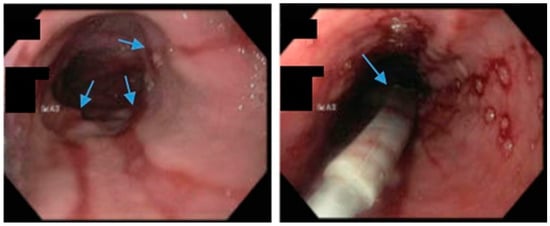
Figure 5. Upper endoscopy demonstrates Los Angeles Grade C esophagitis in a patient following sleeve gastrectomy (arrows, left). A Bravo™ capsule was placed endoscopically for pH monitoring (Medtronic, Minneapolis, Minnesota, USA) (arrow, right).
An international expert panel on sleeve gastrectomy reported that 52.5% of general surgeons and 23.3% of bariatric surgeons considered GERD as a contraindication to SG [41]. According to a multi-society consensus statement, SG should not be performed as an antireflux procedure [42]. Data suggest that patients with a BMI > 35 and medically refractory GERD should be considered for RYGB rather than fundoplication due to the following: an increased risk of hiatal hernia and GERD recurrence with fundoplication in patients with BMI > 35, as well as an additional weight loss and metabolic benefit conferred by RYGB to address a myriad of comorbidities [42][43][44][45][46].
2.4.7. Conversion to Roux-en-Y Gastric Bypass
Conversion from SG to RYGB is ultimately necessary for some patients with refractory GERD post-SG (Figure 6). There are robust data that conversion to RYGB is very effective for the treatment of GERD, in addition to its beneficial effects on weight loss and other comorbidities (i.e., diabetes, hypertension, dyslipidemia). In a recent retrospective study by MacVicar et al., 4412 patients required revisional surgery due to GERD, which comprises 24% of all conversion procedures [47]. SG was the original surgery in 80.1% of cases and Roux-en-Y was the revisional surgery in 84.4% of cases [47]. However, conversion from SG to RYGB is not without additional risk. In a recent study with matched cohorts of 5912 patients, an increase in re-admissions, intervention, conversion to open surgery and operative time was documented in the group which underwent conversion from SG to RYGB compared to primary RYGB. There were no significant differences in mortality or complications noted between the two groups.

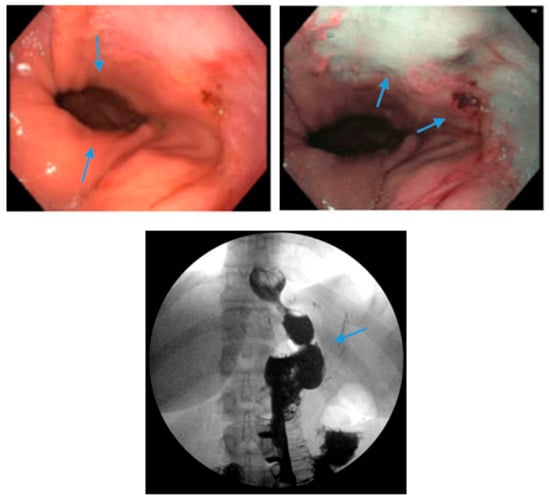
Figure 6. Endoscopic images reveal a 3 cm hiatal hernia (arrows, top left) and Los Angeles Grade B esophagitis (arrows, top right) in the lower third of the esophagus with narrow-band imaging in a patient post-sleeve gastrectomy with symptoms of gastroesophageal reflux disease. The patient eventually required Roux-en-Y gastric bypass (arrow, bottom) and concomitant hiatal hernia repair, as demonstrated on upper gastrointestinal series.
2.4.8. Sleeve Gastrectomy with Fundoplication or Hiatal Hernia Repair
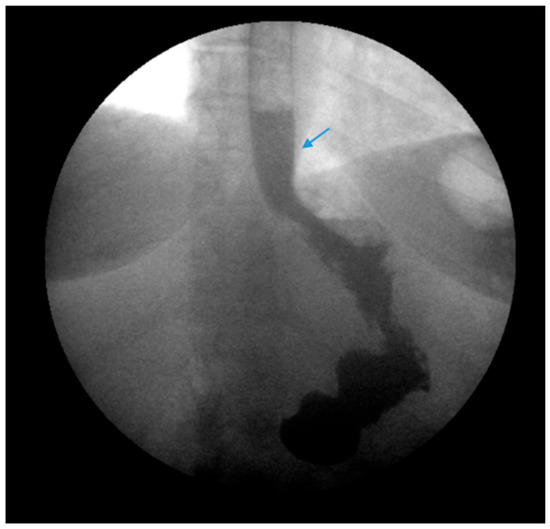
Due to the prevalence of hiatal hernias in the bariatric population, several studies have explored GERD outcomes when SG is combined with either fundoplication or hiatal hernia repair (Figure 7 and Figure 8) [48][49][50][51][52][53][54][55][56][57][58][59][60]. The authors acknowledge that the published studies on SG combined with either fundoplication or hiatal hernia repair are investigational, with a limited sample size and variable follow-up.

Figure 7. An upper gastrointestinal series reveals a small, sliding hiatal hernia (bottom arrow) in addition to gastroesophageal reflux disease (top arrow) in a patient following sleeve gastrectomy.

Figure 8. An upper gastrointestinal series reveals a small, sliding hiatal hernia (bottom arrow) with spontaneous gastroesophageal reflux (top arrow) in a patient following sleeve gastrectomy.
A randomized clinical trial comparing SG with SG and Rosetti fundoplication (RF) by Olmi et al. concluded that there was a significant reduction in PPI use and esophagitis in the SG with RF group compared to the SG group [54]. Wrap perforation occurred in 4.3% of patients and reportedly improved with the surgeon’s learning curve [54].
2.4.9. The Use of Magnetic Sphincter Augmentation Devices
Magnetic sphincter augmentation (MSA) devices, such as the LINX© system, have also been used as an experimental approach to mitigate post-SG GERD. MSA devices utilize titanium beads with a magnetic core connected with titanium wires to form a ring shape, which is laparoscopically placed around the lower esophageal sphincter as a reinforcement. Some studies have revealed favorable results [61][62][63][64][65]. A meta-analysis of three studies with a total of 33 patients revealed a pooled mean difference between preoperative and postoperative GERD-Heath-Related Quality of Life Questionnaire (GERD-HRQL) scores to be 17.5 following MSA [66]. However, data are limited. Further studies are warranted to establish the safety and efficacy of MSA devices following SG.
2.4.10. Endoscopic Sleeve Gastroplasty
Of note, a recent technique known as endoscopic sleeve gastroplasty (ESG), or endoluminal vertical gastroplasty (EVG), has been reported to be a safe and effective primary weight loss procedure or revisional procedure for weight recidivism in the setting of SG and has also shown promising results with regard to GERD improvement [67][68][69][70]. According to a case-matched study by Fayad et al., new-onset GERD was significantly lower in the ESG group compared with the LSG group (1.9% vs. 14.5%, p < 0.05), which may be due to sparing of both the fundus of the stomach and attachments to the gastroesophageal junction [71]. In a multicenter study of 18 patients, the Transoral Gastric Volume Reduction as Intervention for Weight Management (TRIM) trial, which evaluated outcomes in transoral gastric volume reduction surgery using an endoscopic suturing system, reported a reduction in patients who reported reflux from 8 of 14 patients prior to the procedure to 5 out of 14 at 1-year follow-up [72]. Further studies are needed to determine the effect of ESG on GERD.
2.4.11. Surveillance for Barrett’s Esophagus Post-Sleeve Gastrectomy
It is important to note that emerging data have revealed an association of Barrett’s esophagus (BE) following SG. According to two recent meta-analyses by Yeung and Qumseya, the prevalence of BE following SG is approximately 8% and 11.6%, respectively [73][74]. While the current data regarding BE following SG have limitations, they suggest that endoscopic surveillance is important following SG. The American Society for Metabolic and Bariatric Surgery (ASMBS) has recommended to offer screening for BE in SG patients three or more years post-SG, regardless of the presence of GERD symptoms, in addition to the standard screening indications for GERD and Barrett’s esophagus [75].
References
- Stierman, B.; Afful, J.; Carroll, M.D.; Chen, T.-C.; Davy, O.; Fink, S.; Fryar, C.D.; Gu, Q.; Hales, C.M.; Hughes, J.P. National health and nutrition examination survey 2017–March 2020 prepandemic data files development of files and prevalence estimates for selected health outcomes. In National Health Statistics Reports; CDC: Atlanta, GA, USA, 2021.
- Kloock, S.; Ziegler, C.G.; Dischinger, U. Obesity and its comorbidities, current treatment options and future perspectives: Challenging bariatric surgery? Pharmacol. Ther. 2023, 251, 108549.
- Alalwan, A.A.; Friedman, J.; Park, H.; Segal, R.; Brumback, B.A.; Hartzema, A.G. US national trends in bariatric surgery: A decade of study. Surgery 2021, 170, 13–17.
- Buchwald, H.; Avidor, Y.; Braunwald, E.; Jensen, M.D.; Pories, W.; Fahrbach, K.; Schoelles, K. Bariatric surgery: A systematic review and meta-analysis. JAMA 2004, 292, 1724–1737.
- Clapp, B.; Ponce, J.; DeMaria, E.; Ghanem, O.; Hutter, M.; Kothari, S.; LaMasters, T.; Kurian, M.; English, W. American Society for Metabolic and Bariatric Surgery 2020 estimate of metabolic and bariatric procedures performed in the United States. Surg. Obes. Relat. Dis. 2022, 18, 1134–1140.
- Delshad, S.D.; Almario, C.V.; Chey, W.D.; Spiegel, B.M.R. Prevalence of Gastroesophageal Reflux Disease and Proton Pump Inhibitor-Refractory Symptoms. Gastroenterology 2020, 158, 1250–1261.e1252.
- El-Serag, H.B.; Sweet, S.; Winchester, C.C.; Dent, J. Update on the epidemiology of gastro-oesophageal reflux disease: A systematic review. Gut 2014, 63, 871–880.
- Durazzo, M.; Lupi, G.; Cicerchia, F.; Ferro, A.; Barutta, F.; Beccuti, G.; Gruden, G.; Pellicano, R. Extra-Esophageal Presentation of Gastroesophageal Reflux Disease: 2020 Update. J. Clin. Med. 2020, 9, 2559.
- Sharara, A.I.; Rustom, L.B.O.; Bou Daher, H.; Rimmani, H.H.; Shayto, R.H.; Minhem, M.; Ichkhanian, Y.; Aridi, H.; Al-Abbas, A.; Shaib, Y.; et al. Prevalence of gastroesophageal reflux and risk factors for erosive esophagitis in obese patients considered for bariatric surgery. Dig. Liver Dis. 2019, 51, 1375–1379.
- Merrouche, M.; Sabaté, J.M.; Jouet, P.; Harnois, F.; Scaringi, S.; Coffin, B.; Msika, S. Gastro-esophageal reflux and esophageal motility disorders in morbidly obese patients before and after bariatric surgery. Obes. Surg. 2007, 17, 894–900.
- El-Serag, H.B.; Graham, D.Y.; Satia, J.A.; Rabeneck, L. Obesity is an independent risk factor for GERD symptoms and erosive esophagitis. Am. J. Gastroenterol. 2005, 100, 1243–1250.
- Hampel, H.; Abraham, N.S.; El-Serag, H.B. Meta-analysis: Obesity and the risk for gastroesophageal reflux disease and its complications. Ann. Intern. Med. 2005, 143, 199–211.
- Sheppard, C.E.; Sadowski, D.C.; de Gara, C.J.; Karmali, S.; Birch, D.W. Rates of reflux before and after laparoscopic sleeve gastrectomy for severe obesity. Obes. Surg. 2015, 25, 763–768.
- Tai, C.M.; Huang, C.K.; Lee, Y.C.; Chang, C.Y.; Lee, C.T.; Lin, J.T. Increase in gastroesophageal reflux disease symptoms and erosive esophagitis 1 year after laparoscopic sleeve gastrectomy among obese adults. Surg. Endosc. 2013, 27, 1260–1266.
- Howard, D.D.; Caban, A.M.; Cendan, J.C.; Ben-David, K. Gastroesophageal reflux after sleeve gastrectomy in morbidly obese patients. Surg. Obes. Relat. Dis. 2011, 7, 709–713.
- Matar, R.; Maselli, D.; Vargas, E.; Veeravich, J.; Bazerbachi, F.; Beran, A.; Storm, A.C.; Kellogg, T.; Abu Dayyeh, B.K. Esophagitis After Bariatric Surgery: Large Cross-sectional Assessment of an Endoscopic Database. Obes. Surg. 2020, 30, 161–168.
- Gu, L.; Chen, B.; Du, N.; Fu, R.; Huang, X.; Mao, F.; Khadaroo, P.A.; Zhao, S. Relationship Between Bariatric Surgery and Gastroesophageal Reflux Disease: A Systematic Review and Meta-analysis. Obes. Surg. 2019, 29, 4105–4113.
- Birkmeyer, N.J.O.; Dimick, J.B.; Share, D.; Hawasli, A.; English, W.J.; Genaw, J.; Finks, J.F.; Carlin, A.M.; Birkmeyer, J.D.; for the Michigan Bariatric Surgery Collaborative. Hospital Complication Rates with Bariatric Surgery in Michigan. JAMA 2010, 304, 435–442.
- Raj, P.P.; Bhattacharya, S.; Misra, S.; Kumar, S.S.; Khan, M.J.; Gunasekaran, S.C.; Palanivelu, C. Gastroesophageal reflux–related physiologic changes after sleeve gastrectomy and Roux-en-Y gastric bypass: A prospective comparative study. Surg. Obes. Relat. Dis. 2019, 15, 1261–1269.
- Znamirowski, P.; Kołomańska, M.; Mazurkiewicz, R.; Tymchyshyn, O.; Nawacki, Ł. GERD as a Complication of Laparoscopic Sleeve Gastrectomy for the Treatment of Obesity: A Systematic Review and Meta-Analysis. J. Pers. Med. 2023, 13, 1243.
- Melissas, J.; Koukouraki, S.; Askoxylakis, J.; Stathaki, M.; Daskalakis, M.; Perisinakis, K.; Karkavitsas, N. Sleeve gastrectomy: A restrictive procedure? Obes. Surg. 2007, 17, 57–62.
- Melissas, J.; Daskalakis, M.; Koukouraki, S.; Askoxylakis, I.; Metaxari, M.; Dimitriadis, E.; Stathaki, M.; Papadakis, J.A. Sleeve gastrectomy—A “food limiting” operation. Obes. Surg. 2008, 18, 1251–1256.
- Felinska, E.; Billeter, A.; Nickel, F.; Contin, P.; Berlth, F.; Chand, B.; Grimminger, P.; Mikami, D.; Schoppmann, S.F.; Müller-Stich, B. Do we understand the pathophysiology of GERD after sleeve gastrectomy? Ann. N. Y. Acad. Sci. 2020, 1482, 26–35.
- Greenan, G.; Rogers, B.D.; Gyawali, C.P. Proximal Gastric Pressurization After Sleeve Gastrectomy Associates with Gastroesophageal Reflux. Am. J. Gastroenterol. 2023, 118, 2148–2156.
- Masood, M.; Low, D.; Deal, S.B.; Kozarek, R.A. Gastroesophageal Reflux Disease in Obesity: Bariatric Surgery as Both the Cause and the Cure in the Morbidly Obese Population. J. Clin. Med. 2023, 12, 5543.
- Laine, L.; DeVault, K.; Katz, P.; Mitev, S.; Lowe, J.; Hunt, B.; Spechler, S. Vonoprazan Versus Lansoprazole for Healing and Maintenance of Healing of Erosive Esophagitis: A Randomized Trial. Gastroenterology 2023, 164, 61–71.
- Shinozaki, S.; Osawa, H.; Hayashi, Y.; Miura, Y.; Lefor, A.K.; Yamamoto, H. Long-term vonoprazan therapy is effective for controlling symptomatic proton pump inhibitor-resistant gastroesophageal reflux disease. Biomed. Rep. 2021, 14, 32.
- Niikura, R.; Yamada, A.; Hirata, Y.; Hayakawa, Y.; Takahashi, A.; Shinozaki, T.; Takeuchi, Y.; Fujishiro, M.; Koike, K. Efficacy of Vonoprazan for Gastroesophageal Reflux Symptoms in Patients with Proton Pump Inhibitor-resistant Non-erosive Reflux Disease. Intern. Med. 2018, 57, 2443–2450.
- Keidar, A.; Appelbaum, L.; Schweiger, C.; Elazary, R.; Baltasar, A. Dilated upper sleeve can be associated with severe postoperative gastroesophageal dysmotility and reflux. Obes. Surg. 2010, 20, 140–147.
- Mazer, L.; Yu, J.X.; Bhalla, S.; Platt, K.; Watts, L.; Volk, S.; Schulman, A.R. Pneumatic Balloon Dilation of Gastric Sleeve Stenosis Is Not Associated with Weight Regain. Obes. Surg. 2022, 32, 1–6.
- Dhorepatil, A.S.; Cottam, D.; Surve, A.; Medlin, W.; Zaveri, H.; Richards, C.; Cottam, A. Is pneumatic balloon dilation safe and effective primary modality of treatment for post-sleeve gastrectomy strictures? A retrospective study. BMC Surg. 2018, 18, 52.
- Chang, S.H.; Popov, V.B.; Thompson, C.C. Endoscopic balloon dilation for treatment of sleeve gastrectomy stenosis: A systematic review and meta-analysis. Gastrointest. Endosc. 2020, 91, 989–1002.e4.
- Kurien, R.; Menon, S. Balloon dilation in sleeve gastrectomy stenosis: A simple solution to an occasionally tricky problem. Gastrointest. Endosc. 2020, 91, 1003–1004.
- Joo, M.K. Endoscopic Approach for Major Complications of Bariatric Surgery. Clin. Endosc. 2017, 50, 31–41.
- Gagner, M.; Kemmeter, P. Comparison of laparoscopic sleeve gastrectomy leak rates in five staple-line reinforcement options: A systematic review. Surg. Endosc. 2020, 34, 396–407.
- Burgos, A.M.; Braghetto, I.; Csendes, A.; Maluenda, F.; Korn, O.; Yarmuch, J.; Gutierrez, L. Gastric leak after laparoscopic-sleeve gastrectomy for obesity. Obes. Surg. 2009, 19, 1672–1677.
- Bekheit, M.; Katri, K.M.; Nabil, W.; Sharaan, M.A.; El Kayal, E.S.A. Earliest signs and management of leakage after bariatric surgeries: Single institute experience. Alex. J. Med. 2013, 49, 29–33.
- Abou Rached, A.; Basile, M.; El Masri, H. Gastric leaks post sleeve gastrectomy: Review of its prevention and management. World J. Gastroenterol. 2014, 20, 13904–13910.
- Csendes, A.; Díaz, J.C.; Burdiles, P.; Braghetto, I.; Maluenda, F.; Nava, O.; Korn, O. Classification and treatment of anastomotic leakage after extended total gastrectomy in gastric carcinoma. Hepatogastroenterology 1990, 37 (Suppl. 2), 174–177.
- Welsch, T.; von Frankenberg, M.; Schmidt, J.; Büchler, M.W. Diagnosis and definition of anastomotic leakage from the surgeon’s perspective. Chirurg 2011, 82, 48–55.
- Gagner, M.; Hutchinson, C.; Rosenthal, R. Fifth International Consensus Conference: Current status of sleeve gastrectomy. Surg. Obes. Relat. Dis. 2016, 12, 750–756.
- Slater, B.J.; Collings, A.; Dirks, R.; Gould, J.C.; Qureshi, A.P.; Juza, R.; Rodríguez-Luna, M.R.; Wunker, C.; Kohn, G.P.; Kothari, S.; et al. Multi-society consensus conference and guideline on the treatment of gastroesophageal reflux disease (GERD). Surg. Endosc. 2023, 37, 781–806.
- Perez, A.R.; Moncure, A.C.; Rattner, D.W. Obesity adversely affects the outcome of antireflux operations. Surg. Endosc. 2001, 15, 986–989.
- Morgenthal, C.B.; Lin, E.; Shane, M.D.; Hunter, J.G.; Smith, C.D. Who will fail laparoscopic Nissen fundoplication? Preoperative prediction of long-term outcomes. Surg. Endosc. 2007, 21, 1978–1984.
- Kellogg, T.A.; Andrade, R.; Maddaus, M.; Slusarek, B.; Buchwald, H.; Ikramuddin, S. Anatomic findings and outcomes after antireflux procedures in morbidly obese patients undergoing laparoscopic conversion to Roux-en-Y gastric bypass. Surg. Obes. Relat. Dis. 2007, 3, 52–57, discussion 58–59.
- Prachand, V.N.; Alverdy, J.C. Gastroesophageal reflux disease and severe obesity: Fundoplication or bariatric surgery? World J. Gastroenterol. 2010, 16, 3757–3761.
- MacVicar, S.; Mocanu, V.; Jogiat, U.; Verhoeff, K.; Dang, J.; Birch, D.; Karmali, S.; Switzer, N. Revisional bariatric surgery for gastroesophageal reflux disease: Characterizing patient and procedural factors and 30-day outcomes for a retrospective cohort of 4412 patients. Surg Endosc. 2024, 38, 75–84.
- Castagneto-Gissey, L.; Russo, M.F.; D’Andrea, V.; Genco, A.; Casella, G. Efficacy of Sleeve Gastrectomy with Concomitant Hiatal Hernia Repair versus Sleeve-Fundoplication on Gastroesophageal Reflux Disease Resolution: Systematic Review and Meta-Analysis. J. Clin. Med. 2023, 12, 3323.
- Carandina, S.; Zulian, V.; Nedelcu, A.; Danan, M.; Vilallonga, R.; Nocca, D.; Nedelcu, M. Is It Safe to Combine a Fundoplication to Sleeve Gastrectomy? Review of Literature. Medicina 2021, 57, 392.
- Amor, I.B.; Casanova, V.; Vanbiervliet, G.; Bereder, J.M.; Habitan, R.; Kassir, R.; Gugenheim, J. The Nissen-Sleeve (N-Sleeve): Results of a Cohort Study. Obes. Surg. 2020, 30, 3267–3272.
- Moon, R.C.; Teixeira, A.F.; Jawad, M.A. Safety and effectiveness of anterior fundoplication sleeve gastrectomy in patients with severe reflux. Surg. Obes. Relat. Dis. 2017, 13, 547–552.
- Lasnibat, J.P.; Braghetto, I.; Gutierrez, L.; Sanchez, F. Sleeve Gastrectomy and Fundoplication as a Single Procedure in Patients with Obesity and Gastroesophageal Reflux. Arq. Bras. Cir. Dig. 2017, 30, 216–221.
- Le Page, P.A.; Martin, D. Laparoscopic partial sleeve gastrectomy with fundoplication for gastroesophageal reflux and delayed gastric emptying. World J. Surg. 2015, 39, 1460–1464.
- Olmi, S.; Cesana, G.; Gambioli, A.; Bonaldi, M.; Ferrari, D.; Uccelli, M.; Ciccarese, F.; Stefano, C.; Riccardo, G.; Lorenzo, M. Effect of laparoscopic sleeve gastrectomy vs laparoscopic sleeve + Rossetti fundoplication on weight loss and de novo GERD in patients affected by morbid obesity: A randomized clinical study. Obes. Surg. 2022, 32, 1451–1458.
- Olmi, S.; Uccelli, M.; Cesana, G.C.; Ciccarese, F.; Oldani, A.; Giorgi, R.; De Carli, S.M.; Villa, R. Modified laparoscopic sleeve gastrectomy with Rossetti antireflux fundoplication: Results after 220 procedures with 24-month follow-up. Surg. Obes. Relat. Dis. 2020, 16, 1202–1211.
- Olmi, S.; Cesana, G.; D’Angiolella, L.; Bonaldi, M.; Uccelli, M.; Mantovani, L. Sleeve gastrectomy with tailored 360° fundoplication according to Rossetti in patients affected by obesity and gastroesophageal reflux: A prospective observational study. Surg. Obes. Relat. Dis. 2021, 17, 1057–1065.
- da Silva, L.E.; Alves, M.M.; El-Ajouz, T.K.; Ribeiro, P.C.; Cruz, R.J., Jr. Laparoscopic Sleeve-Collis-Nissen Gastroplasty: A Safe Alternative for Morbidly Obese Patients with Gastroesophageal Reflux Disease. Obes. Surg. 2015, 25, 1217–1222.
- Del Genio, G.; Tolone, S.; Gambardella, C.; Brusciano, L.; Volpe, M.L.; Gualtieri, G.; Del Genio, F.; Docimo, L. Sleeve Gastrectomy and Anterior Fundoplication (D-SLEEVE) Prevents Gastroesophageal Reflux in Symptomatic GERD. Obes. Surg. 2020, 30, 1642–1652.
- Di Capua, F.; Cesana, G.C.; Uccelli, M.; De Carli, S.M.; Giorgi, R.; Ferrari, D.; Olmi, S. Sleeve Gastrectomy with Rossetti Fundoplication Increases Lower Esophageal Sphincter Tone Preventing Gastroesophageal Reflux Disease: High-Resolution Manometry Assessment. J. Laparoendosc. Adv. Surg. Tech. A 2023, 33, 44–51.
- Uccelli, M.; Cesana, G.C.; Ciccarese, F.; Oldani, A.; Giorgi, R.; De Carli, S.M.; Villa, R.; Zanoni, A.A.G.; Ismail, A.; Di Capua, F.; et al. Laparoscopic sleeve gastrectomy with Rossetti fundoplication: Long-term (5-year) follow-up. Surg. Obes. Relat. Dis. 2022, 18, 1199–1205.
- Riva, C.G.; Asti, E.; Lazzari, V.; Aquilino, K.; Siboni, S.; Bonavina, L. Magnetic Sphincter Augmentation After Gastric Surgery. JSLS 2019, 23, e2019.00035.
- Hawasli, A.; Sadoun, M.; Meguid, A.; Dean, M.; Sahly, M.; Hawasli, B. Laparoscopic placement of the LINX(®) system in management of severe reflux after sleeve gastrectomy. Am. J. Surg. 2019, 217, 496–499.
- Desart, K.; Rossidis, G.; Michel, M.; Lux, T.; Ben-David, K. Gastroesophageal Reflux Management with the LINX® System for Gastroesophageal Reflux Disease Following Laparoscopic Sleeve Gastrectomy. J. Gastrointest. Surg. 2015, 19, 1782–1786.
- Broderick, R.C.; Smith, C.D.; Cheverie, J.N.; Omelanczuk, P.; Lee, A.M.; Dominguez-Profeta, R.; Cubas, R.; Jacobsen, G.R.; Sandler, B.J.; Fuchs, K.H.; et al. Magnetic sphincter augmentation: A viable rescue therapy for symptomatic reflux following bariatric surgery. Surg. Endosc. 2020, 34, 3211–3215.
- Khaitan, L.; Hill, M.; Michel, M.; Chiasson, P.; Woodworth, P.; Bell, R.; Sadek, R.; Hoffman, A.; Loing, K.; Veldhuis, P.; et al. Feasibility and Efficacy of Magnetic Sphincter Augmentation for the Management of Gastroesophageal Reflux Disease Post-Sleeve Gastrectomy for Obesity. Obes. Surg. 2023, 33, 387–396.
- Rausa, E.; Manfredi, R.; Kelly, M.E.; Bianco, F.; Aiolfi, A.; Bonitta, G.; Lucianetti, A.; Zappa, M.A. Magnetic Sphincter Augmentation Placement for Recalcitrant Gastroesophageal Reflux Disease Following Bariatric Procedures: A Systematic Review and Bayesian Meta-Analysis. J. Laparoendosc. Adv. Surg. Technol. A 2021, 31, 1034–1039.
- Novikov, A.A.; Afaneh, C.; Saumoy, M.; Parra, V.; Shukla, A.; Dakin, G.F.; Pomp, A.; Dawod, E.; Shah, S.; Aronne, L.J.; et al. Endoscopic Sleeve Gastroplasty, Laparoscopic Sleeve Gastrectomy, and Laparoscopic Band for Weight Loss: How Do They Compare? J. Gastrointest. Surg. 2018, 22, 267–273.
- Sharaiha, R.Z.; Kedia, P.; Kumta, N.; Aronne, L.J.; Kahaleh, M. Endoscopic sleeve plication for revision of sleeve gastrectomy. Gastrointest. Endosc. 2015, 81, 1004.
- Maselli, D.B.; Alqahtani, A.R.; Abu Dayyeh, B.K.; Elahmedi, M.; Storm, A.C.; Matar, R.; Nieto, J.; Teixeira, A.; Al Khatry, M.; Neto, M.G.; et al. Revisional endoscopic sleeve gastroplasty of laparoscopic sleeve gastrectomy: An international, multicenter study. Gastrointest. Endosc. 2021, 93, 122–130.
- Nduma, B.N.; Mofor, K.A.; Tatang, J.T.; Ekhator, C.; Ambe, S.; Fonkem, E. Endoscopic Gastric Sleeve: A Review of Literature. Cureus 2023, 15, e36353.
- Fayad, L.; Adam, A.; Schweitzer, M.; Cheskin, L.J.; Ajayi, T.; Dunlap, M.; Badurdeen, D.S.; Hill, C.; Paranji, N.; Lalezari, S.; et al. Endoscopic sleeve gastroplasty versus laparoscopic sleeve gastrectomy: A case-matched study. Gastrointest. Endosc. 2019, 89, 782–788.
- Brethauer, S.A.; Chand, B.; Schauer, P.R.; Thompson, C.C. Transoral gastric volume reduction as intervention for weight management: 12-month follow-up of TRIM trial. Surg. Obes. Relat. Dis. 2012, 8, 296–303.
- Yeung, K.T.D.; Penney, N.; Ashrafian, L.; Darzi, A.; Ashrafian, H. Does Sleeve Gastrectomy Expose the Distal Esophagus to Severe Reflux?: A Systematic Review and Meta-analysis. Ann. Surg. 2020, 271, 257–265.
- Qumseya, B.J.; Qumsiyeh, Y.; Ponniah, S.A.; Estores, D.; Yang, D.; Johnson-Mann, C.N.; Friedman, J.; Ayzengart, A.; Draganov, P.V. Barrett’s esophagus after sleeve gastrectomy: A systematic review and meta-analysis. Gastrointest. Endosc. 2021, 93, 343–352.e342.
- Campos, G.M.; Mazzini, G.S.; Altieri, M.S.; Docimo, S., Jr.; DeMaria, E.J.; Rogers, A.M. ASMBS position statement on the rationale for performance of upper gastrointestinal endoscopy before and after metabolic and bariatric surgery. Surg. Obes. Relat. Dis. 2021, 17, 837–847.
More
Information
Subjects:
Gastroenterology & Hepatology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
401
Revisions:
2 times
(View History)
Update Date:
07 Mar 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





