Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Natália Cruz-Martins | -- | 3074 | 2024-03-04 11:14:42 | | | |
| 2 | Peter Tang | Meta information modification | 3074 | 2024-03-05 04:17:05 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Kumar, H.; Bhardwaj, K.; Cruz-Martins, N.; Sharma, R.; Siddiqui, S.A.; Dhanjal, D.S.; Singh, R.; Chopra, C.; Dantas, A.; Verma, R.; et al. Phyto-Enrichment of Yogurt to Control Hypercholesterolemia. Encyclopedia. Available online: https://encyclopedia.pub/entry/55820 (accessed on 07 May 2026).
Kumar H, Bhardwaj K, Cruz-Martins N, Sharma R, Siddiqui SA, Dhanjal DS, et al. Phyto-Enrichment of Yogurt to Control Hypercholesterolemia. Encyclopedia. Available at: https://encyclopedia.pub/entry/55820. Accessed May 07, 2026.
Kumar, Harsh, Kanchan Bhardwaj, Natália Cruz-Martins, Ruchi Sharma, Shahida Anusha Siddiqui, Daljeet Singh Dhanjal, Reena Singh, Chirag Chopra, Adriana Dantas, Rachna Verma, et al. "Phyto-Enrichment of Yogurt to Control Hypercholesterolemia" Encyclopedia, https://encyclopedia.pub/entry/55820 (accessed May 07, 2026).
Kumar, H., Bhardwaj, K., Cruz-Martins, N., Sharma, R., Siddiqui, S.A., Dhanjal, D.S., Singh, R., Chopra, C., Dantas, A., Verma, R., Dosoky, N.S., & Kumar, D. (2024, March 04). Phyto-Enrichment of Yogurt to Control Hypercholesterolemia. In Encyclopedia. https://encyclopedia.pub/entry/55820
Kumar, Harsh, et al. "Phyto-Enrichment of Yogurt to Control Hypercholesterolemia." Encyclopedia. Web. 04 March, 2024.
Copy Citation
Cholesterol is essential for normal human health, but elevations in its serum levels have led to the development of various complications, including hypercholesterolemia (HC). Cholesterol accumulation in blood circulation formsplaques on artery walls and worsens the individuals’ health. To overcome this complication, different pharmacological and non-pharmacological approaches are employed to reduce elevated blood cholesterol levels. Atorvastatin and rosuvastatin are the most commonly used drugs, but their prolonged use leads to several acute side effects. The potential benefit of ingesting yogurt on lipid profile has attracted the interest of researchers and medical professionals worldwide.
functional foods
yogurt
phyto-enrichment
hypercholesterolemia
1. Introduction
Low-density lipoprotein (LDL) cholesterol and serum total cholesterol (TC) levels are both linked to a high risk of ischemic heart disease found in people at a young age and people at low risk of coronary heart disease [1][2][3][4]. With the increasing incidence of hypercholesterolemia and associated cardiovascular risk, safe, effective, and inexpensive therapeutic approaches were developed to manage such affections [5]. However, over time, it is necessary to understand the extent of blood lipid-induced complications and how they can be managed, which will allow a clear understanding of coronary risk factors among populations [4][5][6][7]. Such studies will also help to identify opportunities to reduce coronary heart risk [8]. According to the World Health Organization (WHO) guidelines, in Kazakhstan, the age-standardized prevalence of total cholesterol increase (≥6.2 mmol/L) was estimated at 12%, which is close to the Russian Federation (15%), but much higher than other Central Asian countries. For instance, Turkmenistan estimated the age-standardized prevalence of total cholesterol increase as the highest, i.e., 8%, and the minimum was 5% in Tajikistan. However, in Central Asian countries, it was estimated to be much lower than in other developed countries, such as Germany (25%) and England (22%) [9].
Regarding blood cholesterol management and distribution, national population-based studies have been conducted in some countries [10][11][12][13][14][15]. From 1999 to 2003, hypercholesterolemia (HC) in Western Europe contributed to heart attacks by about 45%, and in Central and Eastern Europe, it accounted for approximately 35% of heart attack cases [16][17]. However, the risk of heart attack is three times greater in people with high cholesterol levels than those with normal blood lipid profiles. WHO has also demarcated some unhealthy diets, leading to increased cardiovascular risk, e.g., free sugar, high-fat diets, salt, and sugars low in complex carbohydrates [18].
Nowadays, functional foods are referred to as “superfoods”, and, when consumed as part of the daily diets, are believed to have the potential to reduce the risk of several diseases [19]. There are a variety of food products amid the superfoods, including fruits, vegetables, nuts, seeds, and dairy products such as kefir and yogurt [20].
Milk and its products represent an essential part of a healthy balanced diet, rich in proteins and micronutrients [21][22]. Nonetheless, there was a misconception regarding dairy products consumption in the past, linked to the premise that they are high-risk factors for cardiovascular diseases and create adverse effects on human health due to the presence of a marked content of saturated fatty acids [23]. Though, this claim was proved wrong by the fact that dairy products have functional components (calcium, milk proteins, and phospholipids) of high nutritional value, which are efficient in lowering the risk of cardiovascular disease. Their health benefit is attributed to their probiotic effects and the ability to modulate the lipoproteins metabolism [24][25].
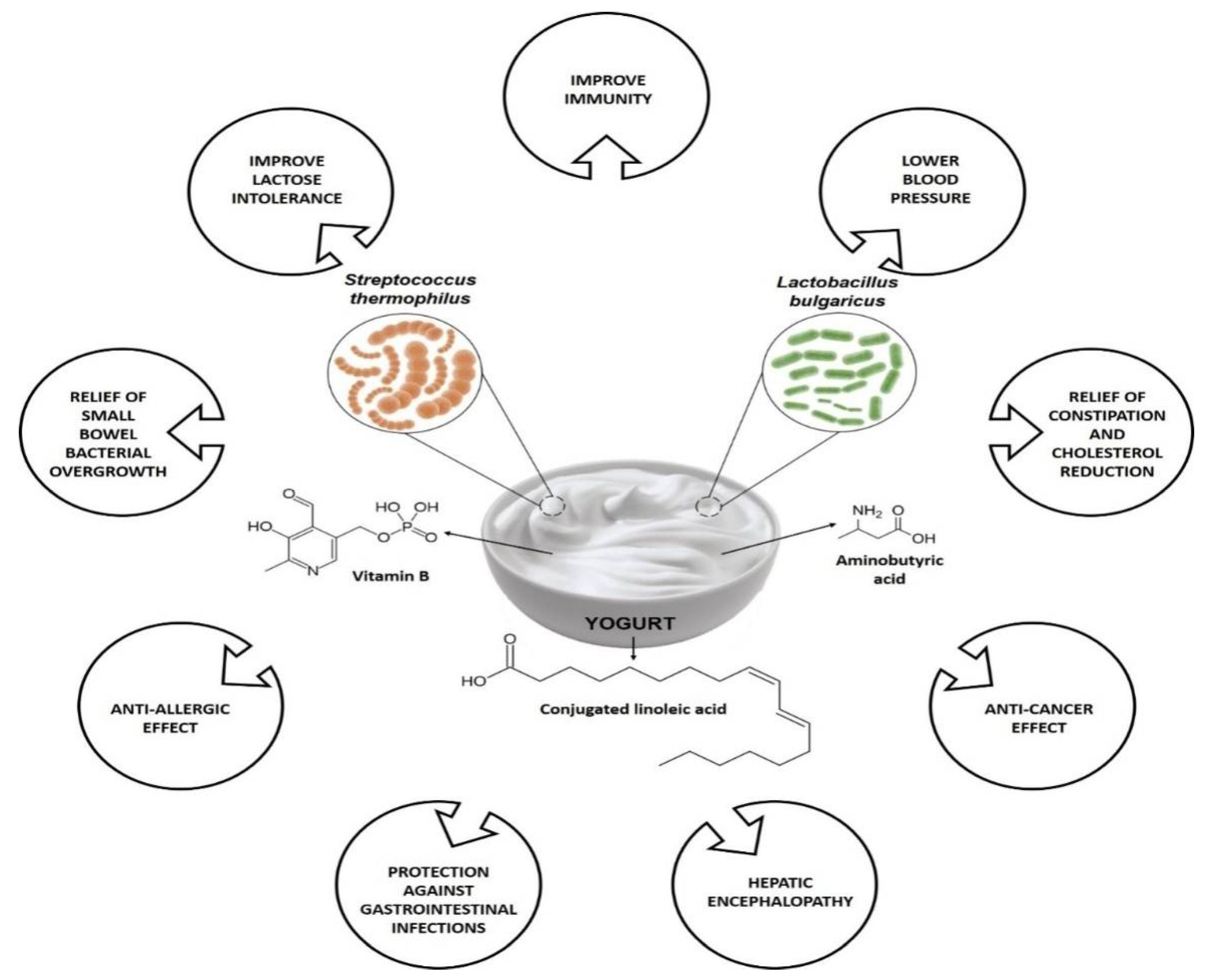
2. Hypercholesterolemia
2.1. Low-Density Lipoprotein-Cholesterol and the Concept of Hypercholesterolemia
Hypercholesterolemia (HC) is related to an elevated level of plasma low-density lipoprotein-cholesterol (LDL-C), as plasma LDL is the chief carrier of cholesterol [26]. The gut absorbs dietary cholesterol primarily packed in chylomicrons(triglyceride-rich); these chylomicrons first break down with the help of lipoprotein lipase (LPL), then the fatty acids and monoglycerides released are further supplied to the adipose tissue and muscles. In contrast, the remnant of chylomicron passes through the liver. Then liver produces very-low-density lipoproteins (VLDL), from which they are exuded into the bloodstream.VLDL becomes enlarged due to the presence of triglycerides in it. As these triglycerides are unable to accommodate themselves in VLDL, instead they start to aggregate themselves in the liver resulting in the development of fatty liver disease.LPL metabolizes VLDL and produces intermediate-density lipoproteins (IDL). Later, it becomesconverted to LDL. The liver also produces high-density lipoprotein (HDL) particles from cholesterol, incorporating cholesteryl esters released from peripheral tissues. In this way, HDL becomesinvolved in the reverse transport of cholesterol from peripheral tissues to the liver. Cholesteryl ester-laden HDL particles are moved to LDLs via cholesteryl ester transfer protein (CETP). Then, these cholesteryl esters are carted via LDL particles, which are absorbed by the liver and peripheral tissues (to a lesser extent). The LDL uptakes, as well as degradation in the liver, rely on LDL particles’ binding onto cell surface receptors (LDL-receptors) of hepatocytes. These receptors are particularly connected to apolipoprotein B (apoB) on the LDL particle. Then, the LDL: LDL-receptor complex is internalized via endocytosis. The whole internalization process is reconciled through the LDL receptor adaptor protein. These receptor molecules becomerecycled, and the LDL particles undergo lysosomal degradation. A protein expressed in hepatocytes, the proprotein convertase subtilisin/kexin type 9 (PCSK9), is believed to play a vital role in the catabolism of LDL-receptors [27].
2.2. Epidemiology
Among the Indian population, the prevalence of HC differs between rural (10–15%) and urban (25–30%) populations [28]. To date, different data-based studies were performed on the Indian population, including the India Migration Study (n = 1983), India Heart Watch (n = 6123), Indian Industrial Population Surveillance Study (n = 10,442), Indian Women Health Study (n = 4624), Indian Council of Medical Research (ICMR) Integrated Disease Surveillance Project (urban n = 15,223, slum/periurbann = 15,751, rural n = 13,517), a nationwide industry-sponsored FitHeart Study (n = 46,919) and the INDIAB study (n = 2042) [29][30][31][32][33][34]. Among those, Al-Kharj of Saudi Arabia showed a positive association between the prevalence of HC and increasing age in the general population [35].
2.3. Investigation and Laboratory Assessment
Currently, there are various therapeutic options to manage HC; however, before starting the treatment for HC, clinical examination, detailed history, and basic laboratory tests are needed to properly diagnose the causes of secondary HC, other complications related to atherosclerosis, and primary manifestations of lipoprotein changes [36]. In this sense, a family as well as occupational history, including the history of alcohol consumption, smoking, and dietary preferences, will help to determine the level to which these factors contribute to the risk of cardiovascular disease and/or HC and determine the patient’s ability to change lifestyle [36]. Regarding physical examination, it should include waist circumference, body weight, blood pressure, height, and a search for the presence of xanthoma [36]. Finally, family history is also an important factor in helping cascade testing at the primary stage of HC [37].
Moreover, during HC screening, it is advisable to obtain blood samples post-fasting for a minimum period of 10 h to avoid the postprandial contribution of plasma triglycerides [36]. If the plasma triglycerides range is in the limit of up to 4.5 mmol/L, the LDL cholesterol concentration can be calculated using the Friedewald formula:
Low density lipoprotein cholesterol = total serum cholesterol—(HDL cholesterol + (serum triglycerides/2.2)).
Non-HDL cholesterol = total cholesterol − HDL cholesterol.
Non-HDL cholesterol may act as a target for statin treatment if LDL-C cannot be measured owing to hypertriglyceridemia. The HDL cholesterol and total cholesterol: HDL ratio values help evaluate the risk of cardiovascular disease, but LDL cholesterol can be used as a therapeutic target whenever possible. Non-HDL cholesterol targets are 0.75 mmol/L more than the LDL cholesterol target. In addition, to assess the risk of cardiovascular disease, it is also important to determine fasting glucose concentration, and before starting lipid-lowering treatment, some parameters must be measured, such as creatine kinase activity, liver function tests, dipstick urine protein, and serum creatinine [36].
For routine assessment, many testing laboratories use handheld point-of-care testing (POCT) devices and automated analyzers depends on enzymatic assays a commercialised cholesterol quantitation assay kits [38][39]. Point-of-care testing (POCT) gives quick results, with simplefunction, making it more suitable for people screening tests. POCT clinical application has been found efficient in raising awareness regarding significance of lipid levels to prevent future stroke events and CVD [40]. The CardioChek PA analyzer (PTS Diagnostics) is a handy whole blood test system including a single test strip to determinetriglycerides (TG)HDL cholesterol (HDL-C) and total cholesterol (TC) [41]. In Brazil, the CardioChek PA analyzer used by health professional workers is well recommended for the proposed screening programs; the diagnostic performance is appropriate for use as part of national health services, providing quick and accurate results.
2.4. Treatment
2.4.1. Non-Pharmacological Therapy
There are various non-pharmacological options to manage HC, and dietary therapy is one of the most important ones. Among others, this approach’s primary goals are to progressively lessen the intake of total fat, saturated fats (saturated fatty acids), cholesterol and attain desirable body weight. For example, it is advisable to reduce the intake of saturated fat daily up to 7% of calories, the daily intake of total fat to 25 to 35% of calories, to limit the dietary cholesterol quantity to no more than 200 mg/day; incite the intake of soluble fiber to 20 to 30 g/day, widely abundant in oats, beans, peas, and in some specific fruits [42]. Dietary fibers containing insoluble and soluble fibers attained from plants involvinglignin and non-digestible carbohydrates. Soluble fibers consist of viscous fibers such asmucilage, gum, β-glucans, pectin, fructans (inulin, fructooligosaccharides), and hemicellulose (non-viscous fibers). The insoluble fibers consist of some lignin, cellulose, and hemicellulose [43]. While both insoluble and soluble fibers are not digestible and can be fermented by bacteria with the help of enzymes to hydrolyze the fiber, soluble fibers are more easily fermented by the gut bacteria and have prebiotic functions providing a short-chain fatty acids source. As such, short-chain fatty acids are quickly absorbed by the large intestine, can be oxidized, and used for the production of energy. Short-chain fatty acids suchas propionic acid absorption have been found to reducethe synthesis of cholesterol in the liver, leading to lessening the blood cholesterol and enhanced water and sodium absorption into the colonic mucosal cells [44][45]. Alternatively, functional fibers are nondigestible carbohydrates that are either synthesized or extracted (isolated) and manufactured, to confer valuable health effects. Functional fibers are chitosan, β-glucans, chitins, cellulose, gums, fructans, resistant dextrins, lignin, polydextrose, pectin, and resistant starches, polyols, and psylliums [46].
There are numerous studiescarried outonmetabolism diseases to check the effect of oat β-glucan [47]. A meta-analysis that included 17 randomized controlled trials (RCTs) (916 hypercholesterolemic patients) displayed that consumption of β-glucan significantly reduced LDL-C (−0.21 mmol/L (8.1 mg/dL)) [48]. In a randomized, single-blind, wheat bran–controlled study, it revealed that within 8 h if 11 g oat bran β-glucantaken it nearly doubled the plasma secretion of bile acids and reduces serum cholesterol by measuring the metabolite 7-hydroxy-4-cholesten-3-one in the plasma [49].
It is also expected the inclusion of plant stanols or sterols in the diet (2 to 3 g daily) plus vegetable oils. Other target food items used in reducing total cholesterol include cold-water fishes, such as salmon, sardines, and mackerel, which have rich omega-3 fatty acid contents, widely recognized for lowering triglyceride levels. Similarly, soybeans present in soy, nuts, tofu, and various meat items have a potent antioxidant effect that can help in lowering LDL-C [42].
2.4.2. Pharmacological Therapy
Among the various drugs available to manage HC, HMG-CoA reductase inhibitors, bile acid resins, fibric acids, niacin, and ezetimibe, are the most often used.
2.4.3. Herbal Treatment
It is widely recognized that Mother Nature has a plethora of potential. With a broad spectrum of activity, a systematic review has suggested that medicinal plants are also rich sources of compounds with anti-HC action rather than saturated fat; plant sterols containing drinks and margarine or stanols can reduce the plasma cholesterol up to 10% [50].
3. Yogurt
3.1. Concept, History, and Health Effects
Yogurt (also spelled “yoghourt” or “yoghurt”) is a fermented milk-based product believed by some regulatory agencies worldwide as lactose-free and defined explicitly by containing Streptococcus thermophilus and Lactobacillus bulgaricus viable bacterial strains [51]. In ancient times, the use of yogurt for food purposes was recognized by other names over the millennia: cuajada (Spain), mast (Iran), katyk (Armenia), matsoni (Georgia, Russia, and Japan),roba (Sudan), zabadi (Egypt), laban (Iraq and Lebanon), dahi (India), coalhada (Portugal), leben raib (Saudi Arabia), iogurte (Brazil) and dovga (Azerbaijan). Indeed, it has been reported that milk-based products were added to the human diet near around 10,000–5000 BC, with the help of domestic milk-producing animals (such asgoats, cows, yaks, sheep, and camels, as well as buffalo and horses) [52].
Indian Ayurveda dated back approx. 6000 BC, about the health-related benefits of having fermented milk-based items [51]. These days, around 700 different cheeses and yogurt items are consumed as part of Indian food. Yogurt was well known in the ROman and Greek empires, and the Greeks were the first to mention it in written references in 100 BC, noting the use of yogurt by barbarous nations [51]. In the book Bible, Abraham owed his long life and fecundity to consuming yogurt, as is referenced in the description of the “Land of Milk and Honey,” which several historians claim refers to yogurt [51]. Figure 1 summarizes some of the health benefits associated with yogurt consumption. Both yogurts’ bacterial and non-bacterial components are believed to play health-promoting effects in human health, specifically on the host’s immune system. Among other constituents, bioactive peptides, conjugated linoleic acid (CLA), group B vitamins, c-aminobutyric acid are a few metabolites are found in yogurt, and where the dairy products matrix, bioactive metabolites (i.e., exopolysaccharides and peptides) secreted throughout fermentation along with the active enzymes, fermentation process, are responsible for the health benefits of yogurt [53][54]. Based on fat content, the three most important types of yogurts currently available are low-fat, regular, and non-fat yogurts. Low-fat yogurts are made from partially skim or low-fat milk, whereas regular yogurt is formed from full-fat milk. Skim milk is also used for non-fat yogurt [55].

Figure 1. Variety of health benefits of yogurt consumption.
3.2. Concept of Food Enrichment
An enriched food refers to the food where nutrients have been added to supplement the product with nutrients that are usually present in the original form, but were removed during processing. For example, white bread is a commonly consumed enriched food where some specific vitamins are added because the bleaching process, depletes them [56].
In a broad sense, despite both enrichment and fortification refer to the addition of nutrients in food, the accurate definition varies. The WHO and the Food and Agricultural Organization of the United Nations (FAO) identifies the term fortification as “the practicing deliberately increasing the essential micronutrient content, i.e., minerals (including trace elements) and vitamins in food irrespective of whether the nutritional components were primarily present before processing in food or not, so this is to enhance the quality of the nutrients food supply and to avail public health benefit with least health risk”, whereas the term enrichment is defined as “synonymous with fortification and refers to the adding micronutrients in food which are removed during the time of processing” [56].
3.3. Phyto-Enrichment of Yogurt
Plants produce a broad pool of secondary metabolites that, among other functions, help them to adapt to various types of harsh environments, and defend them from different pests and microbial attacks, and resist all kinds of abiotic and biotic stresses. In this broad group of secondary metabolites, phenolic compounds have gained significant attention these days because of their anti-mutagenic, antioxidant, anti-clotting, and anti-inflammatory potential, which has been directly linked to their ability to decrease the risk of cardiovascular diseases and cancer development [57][58][59].
Phenolic compounds are the primary dietary source found in fruits [60]. It has been increasingly underlined that fruit juices, extracts, and powders show different biological activities, owing to which they have been used as functional ingredients in the food industry, such as dairy food industries [61][62]. Nonetheless, it is also worthy to note that the seasonal variation in vegetables and fruit production and the high demand for fresh fruits in the market have led researchers to explore alternative sources and strategies for the bioproduction of naturally-occurring bioactive compounds similar to phenolic acids and anthocyanins [63].
In this way, several foodstuffs have also been used, and specifically, yogurt is a well renowned fermented dairy-based product. Despite its nutritional features and significance in the human diet, it is not being used as a chief source of phenolic compounds [64]. In dairy food products, the amount of phenolics is minimal due to cattle feed, including a low quantity of phenolics given, contamination of food production equipment with other sanitizing agents, and bacterial decomposition in milk proteins. Hence, some plant-derived additives have been incorporated to raise the phenolic content of yogurt, including the addition of four types of grape callus and grape varieties as functional ingredients [64][65].
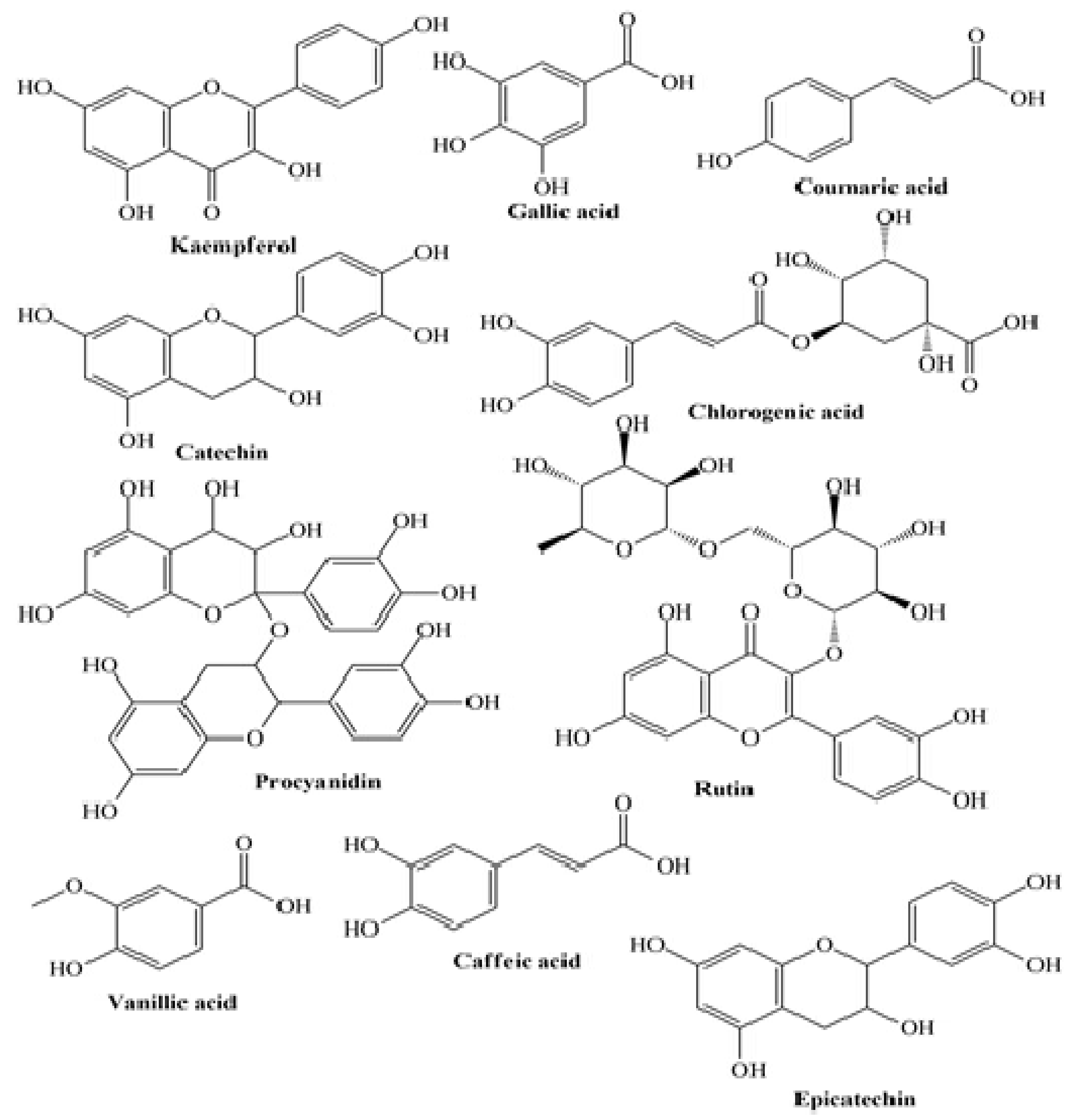
Jaster et al. revealed that anthocyanins present in strawberries-enriched yogurts were directly correlated with a higher antioxidant activity (ABTS) on the 1st and 7th day of storage for control and fortified yogurt, respectively [66]. A similar trend was stated using the DPPH method, indicating a strong correlation between the antioxidant activity and the anthocyanins content for control and fortified yogurts on the 1st and 7th day, respectively. Marand et al. revealed that after 14 days of storage, the total phenolic content in flaxseed enriched yogurt was higher than that of control yogurt, attributed to the phenolic compounds: ρ-coumaric acid, lignans, ferulic acid, and hydroxycinnamic acid derivatives [67] (Figure 2).

Figure 2. Some plant-derived phytocompounds present in enriched yogurt.
4. Phyto-Enriched Yogurt and Hypercholesterolemia Management: Clinical Evidence
There has been an increasing interest in addressing the impact of phyto-enriched yogurt in hypercholesterolemia management in clinical studies. For example, Buyuktuncer et al., in a double-blind, placebo-controlled randomized study for 4 weeks, given to the control group (n = 35) a placebo yogurt and the intervention (n = 35) group consumed low-fat yogurt (115 g) with 1.9 g/day plant stanols as esters [68]. The main findings were a marked decrease in LDL cholesterol (6.3%), serum total cholesterol (4.6%), and non-HDL cholesterol (6.2%) levels from baseline in the intervention group as compared to the control. Insignificant changes were observed at the anthropometric level during the intervention. Similarly, Párraga-Martínez et al. conducted a double-blind, placebo-controlled study involving 182 adults diagnosed with HC [69]. The authors defined two study groups: the intervention group orally received 2 g of plant stanols containing yogurt drink (n = 91), and the control group received an un-supplemented yogurt (n = 91). At 12 months, the lipid profile, defined as the primary endpoint, was changed. In addition, the authors found a >10% reduction in LDL-C in the intervention group (RR 1.7; 95% CI 1.1–2.7). Similarly, Vásquez-Trespalacios et al. revealed in their study that the consumption of a yogurt drink (regular) supplemented with 4 g stanols (plant origin) added as esters compared to yogurt drink (Benecol®, Colanta) promoted a significant reduction in LDL-C and total cholesterol [70]. Doornbos et al. revealed that as compared to a placebo, a single-dose yogurt drink (100 g) taken with a meal regardless of its fat content led to a marked decrease in LDL-C [71].
References
- Verschuren, W.M.; Jacobs, D.R.; Bloemberg, B.P.; Kromhout, D.; Menotti, A.; Aravanis, C.; Toshima, H. Serum total cholesterol and long-term coronary heart disease mortality in different cultures: Twenty-five-year follow-up of the seven countries study. JAMA 1995, 274, 131–136.
- Stamler, J.; Daviglus, M.L.; Garside, D.B.; Dyer, A.R.; Greenland, P.; Neaton, J.D. Relationship of baseline serum cholesterol levels in 3 large cohorts of younger men to long-term coronary, cardiovascular, and all-cause mortality and to longevity. JAMA 2000, 284, 311–318.
- Baigent, C.; Keech, A.; Kearney, P.M.; Blackwell, L.; Buck, G.; Pollicino, C. Efficacy and safety of cholesterol-lowering treatment: Prospective meta-analysis of data from 90056 participants in 14 randomised trials of statins. Lancet 2005, 366, 1267–1278.
- Guallar-Castillón, P.; Gil-Montero, M.; León-Muñoz, L.M.; Graciani, A.; Bayán-Bravo, A.; Taboada, J.M.; Rodríguez-Artalejo, F. Magnitude and management of hypercholesterolemia in the adult population of Spain, 2008–2010: The ENRICA Study. Rev. Esp. Cardiol. 2012, 65, 551–558.
- Catapano, A.L.; Reiner, Ž.; De Backer, G.; Graham, I.; Taskinen, M.R.; Wiklund, O.; Wood, D. ESC/EAS Guidelines for the management of dyslipidaemias: The Task Force for the management of dyslipidaemias of the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS). Atherosclerosis 2011, 217, 1–44.
- Kuulasmaa, K.; Tunstall-Pedoe, H.; Dobson, A.; Fortmann, S.; Sans, S.; Tolonen, H.; Ferrario, M. Estimation of contribution of changes in classic risk factors to trends in coronary-event rates across the WHO MONICA Project populations. Lancet 2000, 355, 675–687.
- Flores-Mateo, G.; Grau, M.; O’Flaherty, M.; Ramos, R.; Elosua, R.; Violan-Fors, C.; Capewell, S. Analyzing the coronary heart disease mortality decline in a Mediterranean population: Spain 1988-2005. Rev. Esp. Cardiol. 2011, 64, 988–996.
- Rodŕiguez-Artalejo, F.; Guallar-Castillon, P.; Banegas, J.R. Critical review and proposals for improvement of the health information systems on cardiovascular diseases in Spain. Med. Clin. 2008, 131, 302–311.
- Supiyev, A.; Nurgozhin, T.; Zhumadilov, Z.; Peasey, A.; Hubacek, J.A.; Bobak, M. Prevalence, awareness, treatment and control of dyslipidemia in older persons in urban and rural population in the Astana region, Kazakhstan. BMC Public Health 2017, 17, 1–9.
- He, J.; Gu, D.; Reynolds, K.; Wu, X.; Muntner, P.; Zhao, J.; Whelton, P.K. Serum total and lipoprotein cholesterol levels and awareness, treatment, and control of hypercholesterolemia in China. Circulation 2004, 110, 405–411.
- Carroll, M.D.; Lacher, D.A.; Sorlie, P.D.; Cleeman, J.I.; Gordon, D.J.; Wolz, M.; Johnson, C.L. Trends in serum lipids and lipoproteins of adults, 1960–2002. JAMA 2005, 294, 1773–1781.
- Primatesta, P.; Poulter, N.R. Levels of dyslipidaemia and improvement in its management in England: Results from the Health Survey for England 2003. Clin. Endocrinol. 2006, 64, 292–298.
- Carroll, M.D.; Lacher, D.A.; Wolz, M.; Sorlie, P.D. 30-year trends in serum lipids among United States adults: Results from the national health and nutrition examination surveys II, III, and 1999–2006. Am. J. Cardiol. 2011, 107, 1868–1870.
- Centers for Disease Control and Prevention (CDC). Vital signs: Prevalence, treatment, and control of high levels of low-density lipoprotein cholesterol—United States, 1999–2002 and 2005–2008. MMWR Morb. Mortal. Wkly Rep. 2011, 60, 109–114.
- Roth, G.A.; Fihn, S.D.; Mokdad, A.H.; Aekplakorn, W.; Hasegawa, T.; Lim, S.S. High total serum cholesterol, medication coverage and therapeutic control: An analysis of national health examination survey data from eight countries. Bull. World Health Organ. 2011, 89, 92–101.
- Yusuf, S.; Hawken, S.; Ôunpuu, S.; Dans, T.; Avezum, A.; Lanas, F.; INTERHEART Study Investigators. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): Case-control study. Lancet 2004, 364, 937–952.
- Kumar, M.; Nagpal, R.; Kumar, R.; Hemalatha, R.; Verma, V.; Kumar, A.; Yadav, H. Cholesterol-lowering probiotics as potential biotherapeutics for metabolic diseases. Exp. Diabetes Res. 2012, 2012, 902717.
- Anandharaj, M.; Sivasankari, B.; Parveen Rani, R. Effects of probiotics, prebiotics, and synbiotics on hypercholesterolemia: A review. Chin. J. Biol. 2014, 572754, 1–7.
- Proestos, C. Superfoods: Recent data on their role in the prevention of diseases. Curr. Res. Nutr. Food Sci. J. 2018, 6, 576–593.
- van den Driessche, J.J.; Plat, J.; Mensink, R.P. Effects of superfoods on risk factors of metabolic syndrome: A systematic review of human intervention trials. Food Funct. 2018, 9, 1944–1966.
- Astrup, A. Yogurt and dairy product consumption to prevent cardiometabolic diseases: Epidemiologic and experimental studies. Am. J. Clin. Nutr. 2014, 99, 1235S–1242S.
- Fernandez, M.A.; Panahi, S.; Daniel, N.; Tremblay, A.; Marette, A. Yogurt and cardiometabolic diseases: A critical review of potential mechanisms. Adv. Nutr. 2017, 8, 812–829.
- Lordan, R.; Tsoupras, A.; Mitra, B.; Zabetakis, I. Dairy fats and cardiovascular disease: Do we really need to be concerned? Foods 2018, 7, 29.
- Şanlier, N.; Gökcen, B.B.; Sezgin, A.C. Health benefits of fermented foods. Crit. Rev. Food Sci. Nutr. 2019, 59, 506–527.
- Wang, H.; Livingston, K.A.; Fox, C.S.; Meigs, J.B.; Jacques, P.F. Yogurt consumption is associated with better diet quality and metabolic profile in American men and women. Nutr. Res. 2013, 33, 18–26.
- Paththinige, C.S.; Sirisena, N.D.; Dissanayake, V.H.W. Genetic determinants of inherited susceptibility to hypercholesterolemia–a comprehensive literature review. Lipids Health Dis. 2017, 16, 1–22.
- Soutar, A.K.; Naoumova, R.P. Mechanisms of disease: Genetic causes of familial hypercholesterolemia. Nat. Clin. Pract. Cardiovasc. Med. 2007, 4, 214–225.
- Gupta, R.; Rao, R.S.; Misra, A.; Sharma, S.K. Recent trends in epidemiology of dyslipidemias in India. Indian Heart J. 2017, 69, 382–392.
- Reddy, K.S.; Prabhakaran, D.; Chaturvedi, V.; Jeemon, P.; Thankappan, K.R.; Ramakrishnan, L.; Jaison, T.M. Methods for establishing a surveillance system for cardiovascular diseases in Indian industrial populations. Bull. World Health Organ. 2006, 84, 461–469.
- Kinra, S.; Bowen, L.J.; Lyngdoh, T.; Prabhakaran, D.; Reddy, K.S.; Ramakrishnan, L.; Ebrahim, S. Sociodemographic patterning of non-communicable disease risk factors in rural India: A cross sectional study. BMJ 2010, 341, c4974.
- Shah, B.; Mathur, P. Surveillance of cardiovascular disease risk factors in India: The need & scope. Indian J. Med. Res. 2010, 132, 634–642.
- Pandey, R.M.; Gupta, R.; Misra, A.; Misra, P.; Singh, V.; Agrawal, A.; Sharma, V. Determinants of urban–rural differences in cardiovascular risk factors in middle-aged women in India: A cross-sectional study. Int. J. Cardio. 2013, 163, 157–162.
- Guptha, S.; Gupta, R.; Deedwania, P.; Bhansali, A.; Maheshwari, A.; Gupta, A.; Sharma, K.K. Cholesterol lipoproteins and prevalence of dyslipidemias in urban Asian Indians: A cross sectional study. Indian Heart J. 2014, 66, 280–288.
- Joshi, S.R.; Anjana, R.M.; Deepa, M.; Pradeepa, R.; Bhansali, A.; Dhandania, V.K.; ICMR–INDIAB Collaborative Study Group. Prevalence of dyslipidemia in urban and rural India: The ICMR–INDIAB study. PLoS ONE 2014, 9, e96808.
- Al-Zahrani, J.; Shubair, M.M.; Al-Ghamdi, S.; Alrasheed, A.A.; Alduraywish, A.A.; Alreshidi, F.S.; Aldossari, K.K. The prevalence of hypercholesterolemia and associated risk factors in Al-Kharj population, Saudi Arabia: A cross-sectional survey. BMC Cardiovas. Disord. 2021, 21, 1–8.
- Bhatnagar, D.; Soran, H.; Durrington, P.N. Hypercholesterolaemia and its management. BMJ 2008, 337, a993.
- Bhatnagar, D.; Morgan, J.; Siddiq, S.; Mackness, M.I.; Miller, J.P.; Durrington, P.N. Outcome of case finding among relatives of patients with known heterozygous familial hypercholesterolemia. BMJ 2000, 321, 1497.
- Ferreira, C.E.D.S.; França, C.N.; Correr, C.J.; Zucker, M.L.; Andriolo, A.; Scartezini, M. Clinical correlation between a point-of-care testing system and laboratory automation for lipid profile. Clin. Chim. Acta 2015, 446, 263–266.
- Owen, W.E.; Thatcher, M.L.; Crabtree, K.J.; Greer, R.W.; Strathmann, F.G.; Straseski, J.A.; Genzen, J.R. Body fluid matrix evaluation on a Roche cobas 8000 system. Clin. Biochem. 2015, 48, 911–914.
- Singh, A.; Dubey, A.; Sonker, A.; Chaudhary, R. Evaluation of various methods of point-of care testing of haemoglobin concentration in blood donors. Blood Transfus. 2014, 29, 1–7.
- Matteucci, E.; Della Bartola, L.; Rossi, L.; Pellegrini, G.; Giampietro, O. Improving CardioCheck PA analytical performance: Three-year study. Clin. Chem. Lab. Med. 2014, 52, 1291–12966.
- Gupta, M.; Vandana, S.; Sidharth, M. Hyperlipidemia: An Updated Review. Int. J. Biopharm. Toxicol. Res. 2011, 1, 81–89.
- Soliman, G.A. Dietary fiber, atherosclerosis, and cardiovascular disease. Nutrients 2019, 11, 1155.
- Anderson, J.W. Dietary fiber, lipids and atherosclerosis. Am. J. Cardiol. 1987, 60, 17–22.
- Anderson, J.W.; Baird, P.; Davis, R.H., Jr.; Ferreri, S.; Knudtson, M.; Koraym, A.; Waters, V.; Williams, C.L. Health benefits of dietary fiber. Nutr. Rev. 2009, 67, 188–205.
- Gropper, S.S.; Smith, J. Advanced Nutrition and Human Metabolism, 6th ed.; Wadsworth Publishing: Belmont, CA, USA, 2013.
- Nie, Y.; Luo, F. Dietary fiber: An opportunity for a global control of hyperlipidemia. Oxid. Med. Cell Longev. 2021, 2021, 5542342.
- Zhu, X.; Sun, X.; Wang, M.; Zhang, C.; Cao, Y.; Mo, G.; Liang, J.; Zhu, S. Quantitative assessment of the effects of beta-glucan consumption on serum lipid profile and glucose level in hypercholesterolemic subjects. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 714–723.
- Andersson, M.; Ellegård, L.; Andersson, H. Oat bran stimulates bile acid synthesis within 8 h as measured by 7α-hydroxy-4-cholesten-3-one. Am. J. Clin. Nutr. 2002, 76, 1111–1116.
- Dhaliya, S.A.; Surya, A.S.; Dawn, V.T.; Betty, C.; Arun, K.; Sunil, C. A review of hyperlipidemia and medicinal plants. Int. JA PS BMS 2013, 2, 219–237.
- Fisberg, M.; Machado, R. History of yogurt and current patterns of consumption. Nutr. Rev. 2015, 73, 4–7.
- Aznar, L.A.M.; Ral, P.C.; Ortega Anta, R.M.; Díaz Martín, J.J.; Baladia, E.; Basulto, J.; Salas-Salvado, J. Scientific evidence about the role of yogurt and other fermented milks in the healthy diet for the spanish population. Nutr. Hosp. 2013, 28, 2039–2089.
- Pei, R.; Martin, D.A.; DiMarco, D.M.; Bolling, B.W. Evidence for the effects of yogurt on gut health and obesity. Crit. Rev. Food Sci. Nutr. 2017, 57, 1569–1583.
- Fernandez, M.A.; Marette, A. Novel perspectives on fermented milks and cardiometabolic health with a focus on type 2 diabetes. Nutr. Rev. 2018, 76, 16–28.
- Weerathilake, W.A.D.V.; Rasika, D.M.D.; Ruwanmali, J.K.U.; Munasinghe, M.A.D.D. The evolution, processing, varieties and health benefits of yogurt. Int. J. Sci. Res. Publ. 2014, 4, 1–10.
- Kumar, H.; Bhardwaj, K.; Kuča, K.; Sharifi-Rad, J.; Verma, R.; Machado, M.; Kumar, D.; Cruz-Martins, N. Edible mushrooms’ enrichment in food and feed: A mini review. Int. J. Food Sci. Tech. 2022, 57, 1386–1398.
- Fresco, P.; Borges, F.; Marques, M.P.M.; Diniz, C. The anticancer properties of dietary polyphenols and its relation with apoptosis. Curr. Pharm. Des. 2010, 16, 114–134.
- Loke, W.M.; Proudfoot, J.M.; Hodgson, J.M.; McKinley, A.J.; Hime, N.; Magat, M.; Croft, K.D. Specific dietary polyphenols attenuate atherosclerosis in apolipoprotein E–knockout mice by alleviating inflammation and endothelial dysfunction. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 749–757.
- Ostertag, L.M.; O’Kennedy, N.; Kroon, P.A.; Duthie, G.G.; De Roos, B. Impact of dietary polyphenols on human platelet function–a critical review of controlled dietary intervention studies. Mol. Nutr. Food Res. 2010, 54, 60–81.
- Record, I.R.; Dreosti, I.E.; McInerney, J.K. Changes in plasma antioxidant status following consumption of diets high or low in fruit and vegetables or following dietary supplementation with an antioxidant mixture. Br. J. Nutr. 2001, 85, 459–464.
- Coïsson, J.D.; Travaglia, F.; Piana, G.; Capasso, M.; Arlorio, M. Euterpe oleracea juice as a functional pigment for yogurt. Food Res. Int. 2005, 38, 893–897.
- Wallace, T.C.; Giusti, M.M. Determination of color, pigment, and phenolic stability in yogurt systems colored with nonacylated anthocyanins from Berberis boliviana L. as compared to other natural/synthetic colorants. J. Food Sci. 2008, 73, C241–C248.
- Blando, F.; Gerardi, C.; Nicoletti, I. Sour cherry (Prunus cerasus L.) anthocyanins as ingredients for functional foods. J. Biomed. Biotechnol. 2004, 2004, 253.
- O’connell, J.E.; Fox, P.F. Significance and applications of phenolic compounds in the production and quality of milk and dairy products: A review. Int. Dairy J. 2001, 11, 103–120.
- Karaaslan, M.; Ozden, M.; Vardin, H.; Turkoglu, H. Phenolic fortification of yogurt using grape and callus extracts. LWT 2011, 44, 1065–1072.
- Jaster, H.; Arend, G.D.; Rezzadori, K.; Chaves, V.C.; Reginatto, F.H.; Petrus, J.C.C. Enhancement of antioxidant activity and physicochemical properties of yogurt enriched with concentrated strawberry pulp obtained by block freeze concentration. Food Res. Int. 2018, 104, 119–125.
- Marand, M.A.; Amjadi, S.; Marand, M.A.; Roufegarinejad, L.; Jafari, S.M. Fortification of yogurt with flaxseed powder and evaluation of its fatty acid profile, physicochemical, antioxidant, and sensory properties. Powder Technol. 2020, 359, 76–84.
- Buyuktuncer, Z.; Fisunoğlu, M.; Guven, G.S.; Unal, S.; Besler, H.T. The cholesterol lowering efficacy of plant stanol ester yoghurt in a Turkish population: A double-blind, placebo-controlled trial. Lipids Health Dis. 2013, 12, 1–8.
- Párraga-Martínez, I.; López-Torres-Hidalgo, J.D.; Del Campo, J.M.; Galdón-Blesa, M.P.; Precioso-Yáñez, J.C.; Rabanales-Sotos, J.; Lloret-Callejo, Á. Long-term effects of plant stanols on the lipid profile of patients with hypercholesterolemia. A randomized clinical trial. Rev. Esp. Cardiol. 2015, 68, 665–671.
- Vásquez-Trespalacios, E.M.; Romero-Palacio, J. Efficacy of yogurt drink with added plant stanol esters (Benecol®, Colanta) in reducing total and LDL cholesterol in subjects with moderate hypercholesterolemia: A randomized placebo-controlled crossover trial NCT01461798. Lipids Health Dis. 2014, 13, 1–7.
- Doornbos, A.M.E.; Meynen, E.M.; Duchateau, G.S.M.J.E.; Van der Knaap, H.C.M.; Trautwein, E.A. Intake occasion affects the serum cholesterol lowering of a plant sterol-enriched single-dose yoghurt drink in mildly hypercholesterolaemic subjects. Eur. J. Clin. Nutr. 2006, 60, 325–333.
More
Information
Subjects:
Food Science & Technology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
732
Revisions:
2 times
(View History)
Update Date:
05 Mar 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





