Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yang Jiale | -- | 1907 | 2024-03-01 02:14:45 | | | |
| 2 | Rita Xu | + 30 word(s) | 1937 | 2024-03-01 03:05:27 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Yang, J.; Zhu, X.; Feng, J. Lymphocyte Subpopulations during the Process of Sepsis. Encyclopedia. Available online: https://encyclopedia.pub/entry/55756 (accessed on 10 May 2026).
Yang J, Zhu X, Feng J. Lymphocyte Subpopulations during the Process of Sepsis. Encyclopedia. Available at: https://encyclopedia.pub/entry/55756. Accessed May 10, 2026.
Yang, Jiale, Xiaojian Zhu, Jun Feng. "Lymphocyte Subpopulations during the Process of Sepsis" Encyclopedia, https://encyclopedia.pub/entry/55756 (accessed May 10, 2026).
Yang, J., Zhu, X., & Feng, J. (2024, March 01). Lymphocyte Subpopulations during the Process of Sepsis. In Encyclopedia. https://encyclopedia.pub/entry/55756
Yang, Jiale, et al. "Lymphocyte Subpopulations during the Process of Sepsis." Encyclopedia. Web. 01 March, 2024.
Copy Citation
Sepsis remains a global challenge, especially in low- and middle-income countries, where there is an urgent need for easily accessible and cost-effective biomarkers to predict the occurrence and prognosis of sepsis. Lymphocyte counts are easy to measure clinically, and a large body of animal and clinical research has shown that lymphocyte counts are closely related to the incidence and prognosis of sepsis.
sepsis
lymphocyte depletion
revive
absolute lymphocyte count
1. Introduction
Sepsis is a common disease in the ICU and can easily lead to poor outcomes in patients due to multiple organ failure. In 2017, member states of the World Health Organization (WHO) declared improving the prevention, recognition, and treatment of sepsis a global health priority [1]. The latest epidemiological statistics show a decrease in the incidence rate of sepsis in recent years, but there is still a lack of effective data in low- and middle-income countries [2]. Another study from the same year shows that the incidence of sepsis has not declined in recent years [3]. This suggests that the incidence rate of sepsis may be underestimated while also indicating a lack of effective means for the early recognition and diagnosis of sepsis in low- and middle-income countries. In this scenario, there is an urgent need for highly adaptable biomarkers to identify sepsis, assess prognosis, and guide treatment plans. The pathophysiology of sepsis has deepened, there have been changes in the definition and treatment strategies of sepsis among clinical physicians. Initially, Bone’s definition emphasized sepsis as a systemic response to infection, characterized by an exaggerated inflammatory response. Clinical practice has found that during infection, patients’ blood contains many cytokines [4]. Subsequently, Clark and others [5] proposed the hypothesis that these cytokines may be the cause of tissue damage and symptoms such as shock observed during sepsis. This phenomenon is referred to as cytokine storm and has garnered significant attention from clinical physicians. Unfortunately, up until now, all clinical trials aimed at suppressing the inflammatory response or targeting cytokines have failed [6]. Faced with this disappointing reality and a further understanding of the pathophysiology of sepsis, many have turned their attention towards new directions. The new definition of sepsis (sepsis 3.0) introduced in 2016 highlights sepsis as a life-threatening organ dysfunction caused by a dysregulated host response to infection [7]. Clinical practice has found that patients who survive early sepsis often experience nosocomial infections, with most of the infections being caused by opportunistic pathogens. Additionally, latent viruses may be reactivated [8][9]. Based on this, clinicians speculate that after the early hyperinflammatory state, the body enters a state of low inflammation accompanied by persistent immune suppression [10]. This phenomenon is known as Compensatory Anti-Inflammatory Response Syndrome (CARS) (Figure 1) [11]. However, recent studies have shown that during the early stages of sepsis, both pro-inflammatory and anti-inflammatory responses occur simultaneously [12].
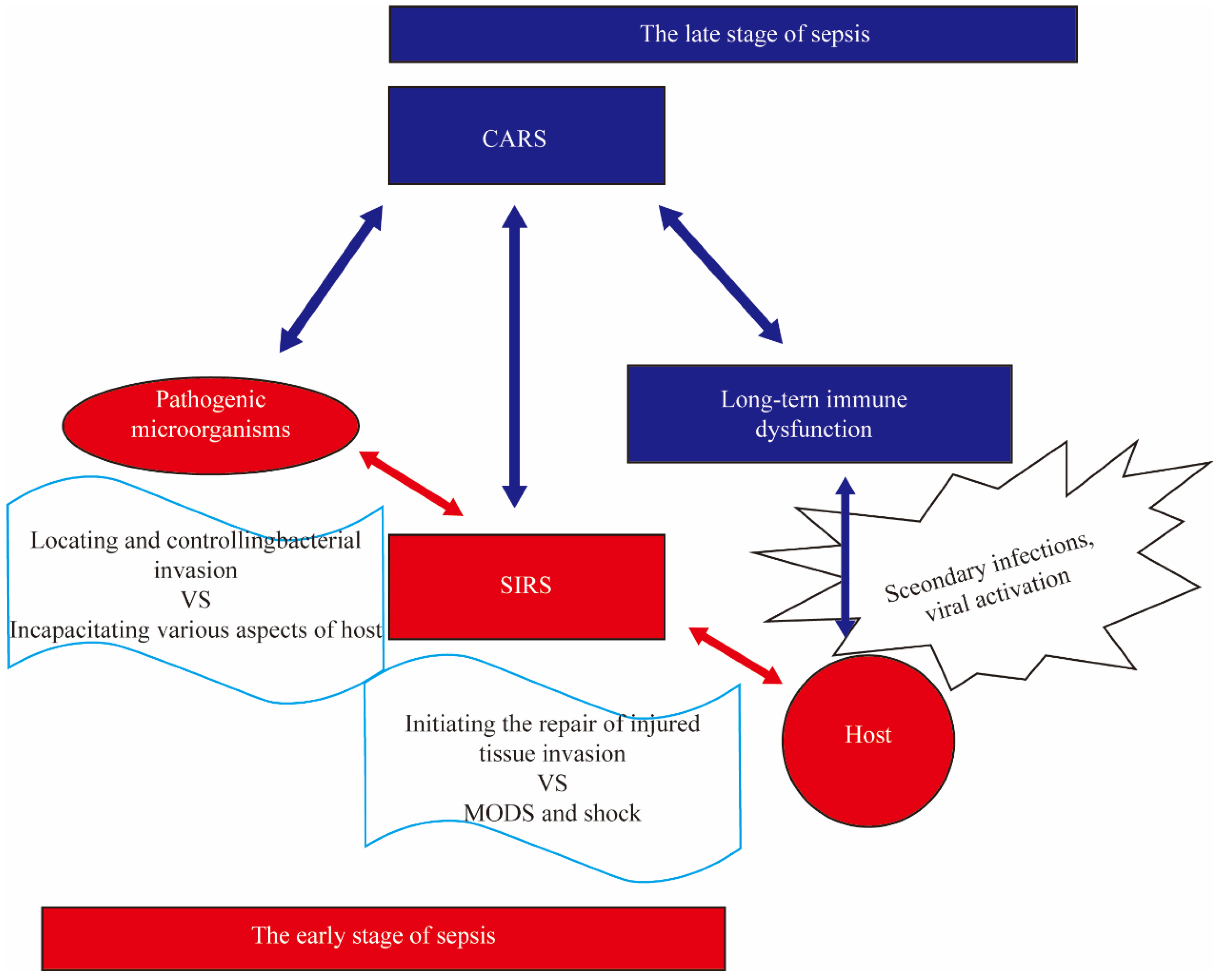
Figure 1. The Simplified Diagram of Compensatory Anti-Inflammatory Response Syndrome. The stimulation of pathogens and their products such as lipopolysaccharide (LPS) can induce systemic inflammatory response syndrome (SIRS) in the body, leading to multiple organ dysfunction syndrome (MODS), shock, and even death. The surviving patients during this stage will go through a compensatory anti-inflammatory response syndrome (CARS) stage, characterized by immunosuppression, which results in long-term immune dysfunction known as immune paralysis. Compared to patients without immune paralysis, those with immune paralysis are more prone to secondary infections, increased viral activation, and a decreased 5-year survival rate.
2. Lymphocytes and Their Subgroups
The immune system of the body is divided into innate immunity and adaptive immunity. Innate immunity primarily serves as the first line of defense through direct killing by phagocytic cells. Among them, innate lymphocyte subgroups (NK cells, γ-δ T cells, and CD4+CD25+ T cells) play an upstream regulatory role in the functions of macrophages, dendritic cells, and T lymphocytes [13]. Adaptive immunity targets specific antigens and involves cell-mediated immunity mediated by effector T lymphocytes (CD4+ T lymphocytes and CD8+ T lymphocytes) and humoral immunity mediated by B lymphocytes. It serves as the main component of host immune responses. In addition, dendritic cells (DCs) serve as crucial bridge cells between innate and adaptive immunity as antigen-presenting cells.
2.1. Absolute Lymphocyte Count
A total of 8 relevant articles were included [14][15][16][17][18][19][20][21]. Based on the common findings from the 8 articles, the following conclusions can be drawn: (a) the absolute lymphocyte count in septic patients is significantly lower compared to healthy adults; (b) the absolute lymphocyte count in septic patients who died is significantly lower compared to those who survived. These conclusions demonstrate the correlation between lymphocyte count and the mortality of sepsis, providing a potential indicator for predicting sepsis and its prognosis through absolute lymphocyte count. Furthermore, Francois et al. [22] conducted experiments that demonstrated the ability of human IL7 (CYT107) to reverse lymphocyte depletion in septic shock patients, resulting in a 3-to 4-fold increase in absolute lymphocyte count as well as circulating CD4+ and CD8+ T cell counts.
2.2. Congenital Lymphocyte Subsets
Congenital lymphocyte subsets include natural killer cells (NK cells), regulatory T cells, and γδ T cells (gamma delta T cells). These lymphocyte subsets are present during the early stages of human development. Unlike other lymphocyte subsets in the acquired immune system, their ability to combat pathogens is innate. They play a crucial role in the immune function of infants and other populations.
2.2.1. Gamma-Delta T Cells (γ-δT Cells)
γ-δT cells are members of the T lymphocyte subset [23], but due to their critical role in innate immunity [24], this research includes them in the description of congenital lymphocyte subsets. In early animal experiments, researchers observed that peripheral blood γ-δT cells can recognize heat shock proteins to initiate innate immunity after injury [25]. Meanwhile, γ-δT cells in mucosal tissues attract T lymphocytes by releasing chemotactic factors [26]. A subsequent retrospective clinical study revealed a decrease in the number of γ-δT cells in the peripheral blood of patients with trauma and sepsis [27]. Similar results were observed in a prospective clinical trial [28]. The research findings suggest a protective role of γ-δT cells in the human body, but the specific mechanisms are still unclear. There are reports suggesting that γ-δT cells have antigen-presenting cell (APC) functions [29][30], which has become a focal point in studying the protective mechanisms of γ-δT cells. The latest study [31] by Yang, Xunwei, and colleagues assessed the antigen-presenting function of γ-δT cells in sepsis patients. Peripheral blood γ-δT cells from sepsis patients were collected for cell-based assays. The experimental results showed that the ability of sepsis patient γ-δT cells to receive amplification signals after antigen stimulation was impaired. Additionally, the expression of APC markers did not increase after stimulation, adhesive capacity significantly decreased, and the ability to induce the proliferation of T lymphocytes was lost. All of the experimental results indicate that the peripheral blood γ-δT cells in sepsis patients have impaired APC function. Treating this impairment is theoretically feasible, but progress has been slow [32]. It is only recently that some achievements in immunotherapy based on γ-δT cells have emerged [33][34][35]. Prior to this, Yuan, Fawei, and colleagues [36] proposed a different viewpoint, suggesting that γ-δ1T cells play an immune suppressive role in sepsis. γ-δT cells can be divided into two subsets: γ-δ1T cells and γ-δ2T cells [37][38]. These two subsets have distinct spatial distributions and physiological functions. Previous experiments have demonstrated that γ-δ1T cells play an immunosuppressive role in autoimmune diseases and tumors [36]. Yuan, Fawei, and colleagues speculate that a similar phenomenon may exist in sepsis. Experimental results indicate that the proportion of peripheral blood γ-δ1T cells in sepsis patients is lower compared to the healthy control group. Furthermore, within the sepsis patient group, the proportion of peripheral blood γ-δ1T cells is even lower in shock patients compared to non-shock patients. Additionally, the experiment revealed that an increased proportion of peripheral blood γ-δ1T cells inhibits the proliferation of T lymphocytes and suppresses the secretion of IFN-γ by T lymphocytes, suggesting the immunosuppressive capability of γ-δ1T cells.
2.2.2. Regulatory T Cells
Regulatory T cells (Treg cells) are a subset of T lymphocytes characterized by the expression of CD4+CD25+ on their surface. Due to their specialized regulatory role in the immune system, they are classified as innate lymphoid cells. Under physiological conditions, Treg cells maintain normal immune homeostasis by secreting immunosuppressive cytokines such as IL-10 and Transforming Growth Factor Beta 1 (TGF-β1) [39][40]. Therefore, it is speculated that Treg cells participate in the development of immunosuppression in sepsis, and relevant experiments have validated this hypothesis [41]. Peripheral blood samples were collected from sepsis patients after burns, and Treg cells were isolated using a CD4+CD25+ regulatory T cell isolation kit. Flow cytometry was then used to measure the purity of Treg cells expressing Foxp-3 through staining with an anti-human Foxp-3-FITC antibody. Finally, the supernatant was collected to measure the levels of IL-10 and TGF-β1. The results showed significantly higher overall expression of Foxp-3 in the severe burn, sepsis, and death groups. Consistently, the cytokines IL-10 and TGF-β1 also showed elevated levels. Foxp-3 is crucial for the differentiation and function of Treg cells [42]. The above results seem to support the notion that regulatory T cells play a negative regulatory role in the process of sepsis, which may be harmful to the body. However, subsequent animal experiments have challenged this viewpoint. Kühlhorn, Franziska, and others [43] used DEREG mice (depletion of regulatory T cells) to create a cecal ligation and puncture (CLP) model of sepsis. The results showed that Treg depletion worsened late-stage survival, suggesting a beneficial role of Foxp-3+ Treg cells in severe sepsis. By collecting more clinical trial data [44][45], researchers have discovered that in the early stages of sepsis, the proportion of Foxp-3+ Treg cells is the same between the survivor group and the non-survivor group, but the absolute number is higher in the former. On the other hand, in the later stage of sepsis (three days later), the survivor group exhibits an increased absolute count of Foxp-3+ Treg cells, while the proportion is comparatively lower.
2.2.3. Natural Killer Cells (NK Cells)
NK cells are essential components of the innate immune system and are capable of killing target cells in their unactivated state [46]. Their role in combating infections and tumors has been widely recognized [47]. Through the study of changes in NK cells in collected cases of sepsis, researchers have observed two contradictory outcomes. One outcome supports the notion that NK cells are a risk factor for sepsis. Animal experiments conducted by Sherwood et al. [48] demonstrated that mice with depleted NK cells exhibited improved survival rates following cecal ligation and puncture. A prospective clinical trial conducted by David Andaluz-Ojeda et al. [49] showed that the absolute count and relative concentration of NK cells in survivors of sepsis were lower than those of deceased patients. According to the analysis of survival curves, it was observed that an NK cell count exceeding 83 cells/mm3 on the first day was linked to early mortality. De Pablo et al. [46] further supported these findings, indicating that patients with the highest NK cell counts in sepsis had the lowest probability of survival. However, another outcome supports the notion that NK cells are a protective factor in sepsis. Clinical trial results from Giamarellos-Bourboulis et al. [50] showed a significant increase in NK cells during the early stages of sepsis. Additionally, patients with NK cells accounting for ≥20% of the total lymphocyte count had longer survival times compared to patients with NK cells accounting for <20% of the total lymphocyte count. In addition to the previously mentioned clinical trials, studies conducted by Boomer et al. [48] and Holub et al. [49] also reported a substantial decrease in NK cells during the early stages of sepsis, specifically within 48 h. This decrease in NK cells may be associated with secondary nosocomial infections and adverse outcomes in patients.
References
- Reinhart, K.; Daniels, R.; Kissoon, N.; Machado, F.R.; Schachter, R.D.; Finfer, S. Recognizing Sepsis as a Global Health Priority—A WHO Resolution. N. Engl. J. Med. 2017, 377, 414–417.
- Fleischmann-Struzek, C.; Mellhammar, L.; Rose, N.; Cassini, A.; Rudd, K.E.; Schlattmann, P.; Allegranzi, B.; Reinhart, K. Incidence and mortality of hospital- and ICU-treated sepsis: Results from an updated and expanded systematic review and meta-analysis. Intensive Care Med. 2020, 46, 1552–1562.
- Rhee, C.; Klompas, M. Sepsis trends: Increasing incidence and decreasing mortality, or changing denominator? J. Thorac. Dis. 2020, 12, S89–S100.
- Clark, I.A.; Virelizier, J.L.; Carswell, E.A.; Wood, P.R. Possible importance of macrophage-derived mediators in acute malaria. Infect. Immun. 1981, 32, 1058–1066.
- Clark, I.A. Suggested importance of monokines in pathophysiology of endotoxin shock and malaria. Klin. Wochenschr. 1982, 60, 756–758.
- Chousterman, B.G.; Swirski, F.K.; Weber, G.F. Cytokine storm and sepsis disease pathogenesis. Semin. Immunopathol. 2017, 39, 517–528.
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.-D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801.
- Kollef, K.E.; Schramm, G.E.; Wills, A.R.; Reichley, R.M.; Micek, S.T.; Kollef, M.H. Predictors of 30-day mortality and hospital costs in patients with ventilator-associated pneumonia attributed to potentially antibiotic-resistant gram-negative bacteria. Chest 2008, 134, 281–287.
- Limaye, A.P.; Kirby, K.A.; Rubenfeld, G.D.; Leisenring, W.M.; Bulger, E.M.; Neff, M.J.; Gibran, N.S.; Huang, M.-L.; Santo Hayes, T.K.; Corey, L.; et al. Cytomegalovirus reactivation in critically ill immunocompetent patients. JAMA 2008, 300, 413–422.
- Schefold, J.C.; Hasper, D.; Reinke, P.; Monneret, G.; Volk, H.-D. Consider delayed immunosuppression into the concept of sepsis. Crit. Care Med. 2008, 36, 3118.
- Ward, N.S.; Casserly, B.; Ayala, A. The compensatory anti-inflammatory response syndrome (CARS) in critically ill patients. Clin. Chest Med. 2008, 29, 617–625, viii.
- Cavaillon, J.-M. During Sepsis and COVID-19, the Pro-Inflammatory and Anti-Inflammatory Responses Are Concomitant. Clin. Rev. Allergy Immunol. 2023, 65, 183–187.
- Schneider, D.F.; Glenn, C.H.; Faunce, D.E. Innate lymphocyte subsets and their immunoregulatory roles in burn injury and sepsis. J. Burn Care Res. Off. Publ. Am. Burn Assoc. 2007, 28, 365–379.
- Holub, M.; Klucková, Z.; Beneda, B.; Hobstová, J.; Huzicka, I.; Prazák, J.; Lobovská, A. Changes in lymphocyte subpopulations and CD3+/DR+ expression in sepsis. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2000, 6, 657–660.
- Boomer, J.S.; To, K.; Chang, K.C.; Takasu, O.; Osborne, D.F.; Walton, A.H.; Bricker, T.L.; Jarman, S.D.; Kreisel, D.; Krupnick, A.S.; et al. Immunosuppression in patients who die of sepsis and multiple organ failure. JAMA 2011, 306, 2594–2605.
- Drewry, A.M.; Samra, N.; Skrupky, L.P.; Fuller, B.M.; Compton, S.M.; Hotchkiss, R.S. Persistent lymphopenia after diagnosis of sepsis predicts mortality. Shock 2014, 42, 383–391.
- Sheikh Motahar Vahedi, H.; Bagheri, A.; Jahanshir, A.; Seyedhosseini, J.; Vahidi, E. Association of Lymphopenia with Short Term Outcomes of Sepsis Patients; a Brief Report. Arch. Acad. Emerg. Med. 2019, 7, e14.
- Jiang, J.; Du, H.; Su, Y.; Li, X.; Zhang, J.; Chen, M.; Ren, G.; He, F.; Niu, B. Nonviral infection-related lymphocytopenia for the prediction of adult sepsis and its persistence indicates a higher mortality. Medicine 2019, 98, e16535.
- Liu, S.; Li, Y.; She, F.; Zhao, X.; Yao, Y. Predictive value of immune cell counts and neutrophil-to-lymphocyte ratio for 28-day mortality in patients with sepsis caused by intra-abdominal infection. Burns Trauma 2021, 9, tkaa040.
- Cilloniz, C.; Peroni, H.J.; Gabarrús, A.; García-Vidal, C.; Pericàs, J.M.; Bermejo-Martin, J.; Torres, A. Lymphopenia Is Associated With Poor Outcomes of Patients With Community-Acquired Pneumonia and Sepsis. Open Forum Infect. Dis. 2021, 8, ofab169.
- Tang, H.; Qin, S.; Li, Z.; Gao, W.; Tang, M.; Dong, X. Early immune system alterations in patients with septic shock. Front. Immunol. 2023, 14, 1126874.
- Francois, B.; Jeannet, R.; Daix, T.; Walton, A.H.; Shotwell, M.S.; Unsinger, J.; Monneret, G.; Rimmelé, T.; Blood, T.; Morre, M.; et al. Interleukin-7 restores lymphocytes in septic shock: The IRIS-7 randomized clinical trial. JCI Insight 2018, 3, e98960.
- Haas, W.; Pereira, P.; Tonegawa, S. Gamma/delta cells. Annu. Rev. Immunol. 1993, 11, 637–685.
- Hayday, A.; Tigelaar, R. Immunoregulation in the tissues by gammadelta T cells. Nat. Rev. Immunol. 2003, 3, 233–242.
- Heng, M.K.; Heng, M.C. Heat-shock protein 65 and activated gamma/delta T cells in injured arteries. Lancet 1994, 344, 921–923.
- Boismenu, R.; Feng, L.; Xia, Y.Y.; Chang, J.C.; Havran, W.L. Chemokine expression by intraepithelial gamma delta T cells. Implications for the recruitment of inflammatory cells to damaged epithelia. J. Immunol. 1996, 157, 985–992.
- Matsushima, A.; Ogura, H.; Fujita, K.; Koh, T.; Tanaka, H.; Sumi, Y.; Yoshiya, K.; Hosotsubo, H.; Kuwagata, Y.; Shimazu, T.; et al. Early activation of gammadelta T lymphocytes in patients with severe systemic inflammatory response syndrome. Shock 2004, 22, 11–15.
- Venet, F.; Bohé, J.; Debard, A.-L.; Bienvenu, J.; Lepape, A.; Monneret, G. Both percentage of gammadelta T lymphocytes and CD3 expression are reduced during septic shock. Crit. Care Med. 2005, 33, 2836–2840.
- Brandes, M.; Willimann, K.; Moser, B. Professional antigen-presentation function by human gammadelta T Cells. Science 2005, 309, 264–268.
- Brandes, M.; Willimann, K.; Bioley, G.; Lévy, N.; Eberl, M.; Luo, M.; Tampé, R.; Lévy, F.; Romero, P.; Moser, B. Cross-presenting human gammadelta T cells induce robust CD8+ alphabeta T cell responses. Proc. Natl. Acad. Sci. USA 2009, 106, 2307–2312.
- Yang, X.-W.; Li, H.; Feng, T.; Zhang, W.; Song, X.-R.; Ma, C.-Y.; Nie, M.; Wang, L.; Tan, X.; Kang, Y.; et al. Impairment of antigen-presenting function of peripheral γδ T cells in patients with sepsis. Clin. Exp. Immunol. 2022, 207, 104–112.
- Zheng, J.; Liu, Y.; Lau, Y.-L.; Tu, W. γδ-T cells: An unpolished sword in human anti-infection immunity. Cell. Mol. Immunol. 2013, 10, 50–57.
- Xu, Y.; Xiang, Z.; Alnaggar, M.; Kouakanou, L.; Li, J.; He, J.; Yang, J.; Hu, Y.; Chen, Y.; Lin, L.; et al. Clinical investigations of allogenic Vγ9Vδ2 T cell therapeutics for hepatocellular carcinoma. In Proceedings of the Abstracts of IUIS 2019 Beijing—17th International Congress of Immunology (V) 1, Beijing, China, 19 October 2019.
- Liang, J.; Fu, L.; Li, M.; Chen, Y.; Wang, Y.; Lin, Y.; Zhang, H.; Xu, Y.; Qin, L.; Liu, J.; et al. Allogeneic Vγ9Vδ2 T-Cell Therapy Promotes Pulmonary Lesion Repair: An Open-Label, Single-Arm Pilot Study in Patients with Multidrug-Resistant Tuberculosis. Front. Immunol. 2021, 12, 756495.
- Hu, Y.; Hu, Q.; Li, Y.; Lu, L.; Xiang, Z.; Yin, Z.; Kabelitz, D.; Wu, Y. γδ T cells: Origin and fate, subsets, diseases and immunotherapy. Signal Transduct. Target. Ther. 2023, 8, 434.
- Yuan, F.; Yin, H.; Tan, J.; Zheng, K.; Mei, X.; Yuan, L. The Proportion of Vδ1T Cells in Peripheral Blood Correlated with Prognosis of Sepsis. Iran. J. Immunol. 2022, 19, 232–242.
- Hayday, A.C. γδ cells: A right time and a right place for a conserved third way of protection. Annu. Rev. Immunol. 2000, 18, 975–1026.
- Harly, C.; Peyrat, M.-A.; Netzer, S.; Déchanet-Merville, J.; Bonneville, M.; Scotet, E. Up-regulation of cytolytic functions of human Vδ2-γ T lymphocytes through engagement of ILT2 expressed by tumor target cells. Blood 2011, 117, 2864–2873.
- Sakaguchi, S. Naturally arising CD4+ regulatory t cells for immunologic self-tolerance and negative control of immune responses. Annu. Rev. Immunol. 2004, 22, 531–562.
- Klein, L.; Robey, E.A.; Hsieh, C.-S. Central CD4+ T cell tolerance: Deletion versus regulatory T cell differentiation. Nat. Rev. Immunol. 2019, 19, 7–18.
- Monneret, G.; Debard, A.-L.; Venet, F.; Bohe, J.; Hequet, O.; Bienvenu, J.; Lepape, A. Marked elevation of human circulating CD4+CD25+ regulatory T cells in sepsis-induced immunoparalysis. Crit. Care Med. 2003, 31, 2068–2071.
- Ramsdell, F.; Rudensky, A.Y. Foxp3: A genetic foundation for regulatory T cell differentiation and function. Nat. Immunol. 2020, 21, 708–709.
- Kühlhorn, F.; Rath, M.; Schmoeckel, K.; Cziupka, K.; Nguyen, H.H.; Hildebrandt, P.; Hünig, T.; Sparwasser, T.; Huehn, J.; Pötschke, C.; et al. Foxp3+ regulatory T cells are required for recovery from severe sepsis. PLoS ONE 2013, 8, e65109.
- Bomans, K.; Schenz, J.; Sztwiertnia, I.; Schaack, D.; Weigand, M.A.; Uhle, F. Sepsis Induces a Long-Lasting State of Trained Immunity in Bone Marrow Monocytes. Front. Immunol. 2018, 9, 2685.
- Liu, Q.; Lu, Y.; An, L.; Li, C.-S. B- and T-Lymphocyte Attenuator Expression on Regulatory T-Cells in Patients with Severe Sepsis. Chin. Med. J. 2018, 131, 2637–2639.
- Guo, Y.; Patil, N.K.; Luan, L.; Bohannon, J.K.; Sherwood, E.R. The biology of natural killer cells during sepsis. Immunology 2018, 153, 190–202.
- Kucuksezer, U.C.; Aktas Cetin, E.; Esen, F.; Tahrali, I.; Akdeniz, N.; Gelmez, M.Y.; Deniz, G. The Role of Natural Killer Cells in Autoimmune Diseases. Front. Immunol. 2021, 12, 622306.
- Sherwood, E.R.; Enoh, V.T.; Murphey, E.D.; Lin, C.Y. Mice depleted of CD8+ T and NK cells are resistant to injury caused by cecal ligation and puncture. Lab. Investig. J. Tech. Methods Pathol. 2004, 84, 1655–1665.
- Andaluz-Ojeda, D.; Iglesias, V.; Bobillo, F.; Almansa, R.; Rico, L.; Gandía, F.; Loma, A.M.; Nieto, C.; Diego, R.; Ramos, E.; et al. Early natural killer cell counts in blood predict mortality in severe sepsis. Crit. Care 2011, 15, R243.
- Giamarellos-Bourboulis, E.J.; Tsaganos, T.; Spyridaki, E.; Mouktaroudi, M.; Plachouras, D.; Vaki, I.; Karagianni, V.; Antonopoulou, A.; Veloni, V.; Giamarellou, H. Early changes of CD4-positive lymphocytes and NK cells in patients with severe Gram-negative sepsis. Crit. Care 2006, 10, R166.
More
Information
Subjects:
Critical Care Medicine
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
621
Revisions:
2 times
(View History)
Update Date:
01 Mar 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





