Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Everton Freitas de Morais | -- | 4370 | 2024-02-28 13:37:10 | | | |
| 2 | Sirius Huang | Meta information modification | 4370 | 2024-02-29 01:43:41 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Siquara Da Rocha, L.D.O.; De Morais, E.F.; De Oliveira, L.Q.R.; Barbosa, A.V.; Lambert, D.W.; Gurgel Rocha, C.A.; Coletta, R.D. Cell Death Pathways in Oral Cancer. Encyclopedia. Available online: https://encyclopedia.pub/entry/55662 (accessed on 06 June 2026).
Siquara Da Rocha LDO, De Morais EF, De Oliveira LQR, Barbosa AV, Lambert DW, Gurgel Rocha CA, et al. Cell Death Pathways in Oral Cancer. Encyclopedia. Available at: https://encyclopedia.pub/entry/55662. Accessed June 06, 2026.
Siquara Da Rocha, Leonardo De Oliveira, Everton Freitas De Morais, Lilianny Querino Rocha De Oliveira, Andressa Vollono Barbosa, Daniel W. Lambert, Clarissa A. Gurgel Rocha, Ricardo D. Coletta. "Cell Death Pathways in Oral Cancer" Encyclopedia, https://encyclopedia.pub/entry/55662 (accessed June 06, 2026).
Siquara Da Rocha, L.D.O., De Morais, E.F., De Oliveira, L.Q.R., Barbosa, A.V., Lambert, D.W., Gurgel Rocha, C.A., & Coletta, R.D. (2024, February 28). Cell Death Pathways in Oral Cancer. In Encyclopedia. https://encyclopedia.pub/entry/55662
Siquara Da Rocha, Leonardo De Oliveira, et al. "Cell Death Pathways in Oral Cancer." Encyclopedia. Web. 28 February, 2024.
Copy Citation
Oral squamous cell carcinoma (OSCC) is the most common and lethal type of head and neck cancer in the world. Variable response and acquisition of resistance to traditional therapies show that it is essential to develop novel strategies that can provide better outcomes for the patient. Activation of cell death pathways, such as the emerging forms of non-apoptotic programmed cell death, including ferroptosis, pyroptosis, necroptosis, NETosis, parthanatos, mitoptosis and paraptosis, may represent clinically relevant novel therapeutic opportunities.
emerging types of cell death
tumor microenvironment
oral cancer
ferroptosis
pyroptosis
necroptosis
1. Introduction
Among the hallmarks of cancer, the acquisition of resistance to cell death plays an important role in cancer initiation and progression to high-grade malignant states, which are frequently unresponsive to conventional anti-cancer therapies [1]. In recent years, there has been a significant improvement in the understanding of different mechanisms of cell death beyond the traditionally observed apoptosis and necrosis [2][3]. Alternative cell death pathways, collectively called programmed cell death (PCD), have been identified in a variety of pathological processes, including oral cancer [4]. These pathways not only contribute to the understanding of cancer pathophysiology [5] but also reveal an intricate balance between molecules involved in cell survival and death, with significant implications, for example, as prognostic biomarkers and in the development of new therapeutic strategies that are beginning to be explored [6].
While malignant cells develop strategies to escape or limit conventional cell death pathways, understanding of their ability to escape death by other mechanisms is much more limited [5]. Since 2018, the Nomenclature Committee on Cellular Death has classified PCD into 12 subtypes of death that differ in molecular mechanisms but may share small similarities in their morphological characteristics, ranging from a necrotic profile, that is, unprogrammed and with a disordered appearance, to an apoptotic profile with an organized profile [2][7]. However, an update proposed by Yan, Elbadawi and Efferth [3] divided cell deaths into three large groups based on activation of specific signaling pathways: (1) Non-programmed cell death (NPCD) or necrosis, (2) apoptotic programmed cell death (APCD) and (3) non-apoptotic programmed cell death (NAPCD), which differs from the apoptotic form because it does not maintain the integrity of the cell membrane and is independent of caspases. Similar to apoptosis, NAPCD has a highly regulated molecular machinery that can be targeted or modulated by molecular strategies [2].
The most studied emerging NAPCD types in oncology are ferroptosis, pyroptosis and necroptosis [8]. Ferroptosis involves the accumulation of lipid peroxides due to disrupted cellular antioxidant defenses, leading to oxidative stress-induced cell death [9]. Cancer cells can escape ferroptosis by enhancing antioxidant defenses and modifying lipid metabolism, enabling them to survive and proliferate despite conditions that typically trigger ferroptotic cell death [10]. Meanwhile, during pyroptosis and necroptosis, the intracellular content is expelled from the cell through membrane pores formed by proteins such as those from the Gasdermin family (GSDM) in pyroptosis and mixed lineage kinase domain-like (MLKL) in necroptosis, resulting in recruitment of inflammatory cells [11][12]. The pro-inflammatory environment induced by the extravasation of intracellular contents may promote the transformation and progression of tumor cells [13][14]. However, the role of NAPCD in cancer progression is only partially understood, and the literature is conflicting.
2. Ferroptosis
Ferroptosis is a distinguished type of NAPCD characterized by iron-dependent lipid peroxide accumulation, particularly of polyunsaturated fatty acids [15][16]. The research landscape of this type of cell death and its implications in diseases is relatively recent [17][18] but promising for increased knowledge on mechanisms and therapeutic strategies in cancer [17][19]. Ferroptosis can occur through two major pathways: the extrinsic pathway or transporter-dependent, and the intrinsic or enzyme-regulated pathway [17][20] (Figure 1). Despite being distinct pathways, it is important to highlight that one can influence the other, as both rely on iron metabolism and glutathione (GSH)-dependent antioxidant mechanisms [21]. The extrinsic mechanism depends on the balance of iron and amino acid transport across the cell membrane [20]. A higher intracellular iron level increases the production of reactive oxygen species (ROS) through the Fenton reaction [22]. Moreover, inhibition of the Xc-system reduces cystine uptake, which is essential for synthesizing GSH [20]. The depletion of GSH reduces the cell’s antioxidant defenses and favors lipid peroxidation. In turn, the intrinsic pathway is mainly induced by inhibiting glutathione peroxidase 4 (GPX4), an enzyme that plays a pivotal role in reducing lipid hydroperoxides to non-toxic lipid alcohols using GSH as a cofactor [23]. The inhibition of GPX4 activity occurs in an unchecked accumulation of lipid hydroperoxides, leading to cellular damage and eventual ferroptosis cell death [20][23].
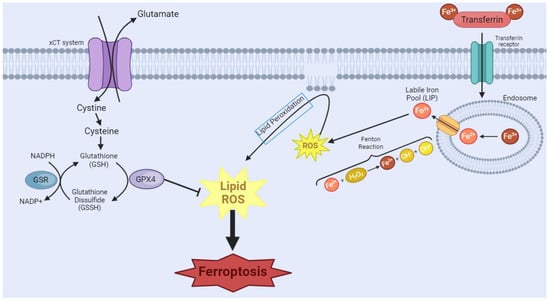
Figure 1. Ferroptosis manifests through two primary pathways. In the first pathway, transferrin (TRF1), a crucial player in iron homeostasis, binds to Fe3+, forming a complex, which then binds to the transferrin receptor. Endocytosis of this complex occurs, leading to the formation of the endosome. Within the endosome, there will be acidification of the environment, causing the dissociation of the complex and the reduction of Fe3+ to Fe2+. This Fe2+ is released through the endosomal membrane. However, surplus free iron ions form a labile iron pool (LIP), which partakes in Fenton’s reactions, causing the generation of the reactive oxygen species (ROS) free radical hydroxyl. This cascade of events culminates in the initiation of lipoperoxidation, a critical step in the ferroptotic process. In the second pathway, System Xc-, a cystine/glutamate antiporter, imports extracellular cystine. Intracellularly, cystine undergoes conversion into glutathione (GSH), a vital antioxidant. The availability of GSH is integral to the function of glutathione peroxidase 4 (GPX4), which tackles intracellular lipid peroxides, preventing ferroptosis. However, the inhibition of GPX4 leads to a disruption in this protective mechanism, resulting in an augmented presence of ROS and ultimately contributing to the progression of ferroptosis. [Image was created using Biorender.com (accessed on 16 November 2023)].
Tumor cells are more susceptible to ferroptosis due to altered metabolism and increased iron uptake [16]. Although ferroptosis is expected to be associated with tumor suppression [17], evidence demonstrates that ferroptosis regulatory pathways may promote tumor growth or progression [24]. The p53 gene can promote ferroptosis by repression of solute carrier family 7 member 11 (SLC7A11), a component of the cystine/glutamate antiporter, reducing GSH and increasing cellular oxidative stress and lipid peroxidation [25]. In addition, tumor cells display increased iron uptake, which can increase the Fenton reaction, producing reactive oxygen species and lipid peroxidation [26]. In other circumstances, the oxidative stress, a prominent feature of ferroptosis, can activate the nuclear erythroid factor 2-related factor 2 (NRF2), a well-known transcription factor activated in response to oxidative stress [27]. Although NRF2 may be cytoprotective, chronic NRF2 activation in cancer cells supports proliferation, metabolic reprogramming and resistance to therapy [28][29]. The hypoxia inducible factor 1 subunit alpha (HIF-1α) has also been implicated in increasing iron storage and transport proteins, influencing cellular propensity for ferroptosis [30]. While HIF-1α is primarily known for its role in the cellular response to hypoxia, oxidative stress can also stabilize HIF-1α, resulting in the transcription of genes involved in angiogenesis, glucose metabolism, and cell survival, and supporting tumor growth, angiogenesis, and metastasis [31]. The tumor microenvironment is also influenced by ferroptosis once tumor cells release damage-associated molecular patterns (DAMPs) that recruit and activate immune cells such as dendritic cells and macrophages, initiating an antitumor immune response [32]. Increased lipid peroxidation resulting from ferroptosis may also increase T cell-mediated cytotoxicity against tumor cells [33].
Targeting ferroptosis in OSCC may offer a new path in tumor treatment [34], especially those resistant to traditional therapy [17], and holds the potential to be integrated into combination therapies for enhanced efficacy [35]. Cancer cells, due to their high metabolic activity and dependency on iron and lipid metabolism [36][37], are vulnerable targets for ferroptosis induction, which could inhibit their growth and proliferation. This is further enhanced by targeting CSCs, which are often responsible for tumor initiation and recurrence [38][39]. Iron metabolism plays a crucial role in CSC maintenance and survival [40][41]. These cells exhibit an “iron addiction” by showing a higher concentration of iron compared to the non-stem cell population in the tumor [26]. By hijacking iron metabolism in these cells, ferroptosis becomes a potential tumor suppressor agent to control a significantly powerful population of cells within cancer. Several strategies have been described to induce ferroptosis in this manner, such as manipulation of tumor-suppressor p53 [42][43], cysteine deprivation [44], and inhibition of ferritinophagy, a selective autophagic process capable of repressing accumulation of iron and lipid ROS [45]. In a similar manner, many studies use the collectively termed “ferroptosis inducers” in OSCC as a potential therapeutic approach, essentially by interfering with intracellular iron levels or ROS accumulation [46]. This has been evaluated both in vitro and in vivo by tampering with the signaling cascades that trigger ferroptosis or silencing of ferroptosis inhibitors, which can be achieved through direct genetic modifications or chemical compounds. Some of the compounds used to achieve such effects are erastin [47][48][49], carnosic acid [50], piperlongumine [51] and Disulfiram [52].
Some studies used strategies to induce ferroptosis in OSCC by silencing target genes. One study silenced the circular RNA FNDC3B (circFNDC3B) with interference RNA, which inhibited GPX4 and SLC7A11 expression (negative regulators of ferroptosis), inducing intracellular ROS and iron accumulation [53]. Another study silenced eukaryotic translation initiation factor 3 subunit B (EIF3B), commonly associated with unfavorable head and neck squamous cell carcinoma (HNSCC) prognosis [54]. EIF3B knockdown resulted in decreased invasion and migration in OSCC, as well as induction of cell death [54]. The knockdowns of enhancer of zeste 2 polycomb repressive complex 2 subunit (EZH2) and SLC7A11, both highly expressed in OSCC, resulted in ferroptosis induction [55]. Other target genes whose knockdown promoted ferroptosis were heat shock protein family A (Hsp70) member 5 (HSPA5) [56], adipocyte enhancer-binding protein 1 (AEBP1) [57], glutaredoxin 5 (GLRX5) [58], and miR-7-5p [59]. AEBP1 silencing was especially effective after sulfasalazine treatment, which significantly increased levels of ROS and free intracellular iron [57]. Another study targeting GPX4 with circular RNA showed increased levels of ROS and intracellular iron, meanwhile repressing tumor growth in OSCC cells [53]. Inhibition of GPX4 in Erlotinib-tolerant persisted cancer cells (erPCC) was also effective in increasing sensitivity to ferroptosis [60].
Many studies evaluated the potential of compounds or nanoparticles in controlling tumor progression by inducing ferroptosis. The recent study by Wu et al. [61] reported the effect of A-GSP (aqueous-soluble sporoderm-removed G. lucidum spore power) in tumor suppression by activating ferroptosis, which was confirmed by the assessment of GSH, malondialdehyde (MDA) and ROS levels, as well as ferroptosis-marker expression and mitochondrial morphological alterations—key in confirming the occurrence of this pathway [62]. Additionally, the authors evaluated this compound in vivo in a tumorigenesis assay, and their results showed that there was a decrease in tumor growth among the treated groups while maintaining low toxicity to the treated animals. These results strongly suggest the effectiveness and specificity of this compound in suppressing the tumor through ferroptosis induction. Another study achieved similar ferroptosis induction using nanoparticles termed ginseng-based carbon dots [63]. Another interesting study was performed by Huang et al. [64] using on-oxidized zero-valent iron (ZVI) nanoparticles. The authors treated several oral cancer cell lines with ZVI nanoparticles, and cell death was observed, together with ROS accumulation and mitochondrial damage. However, some cell lines managed to acquire resistance to treatment, and further examination revealed that ferroptosis-related genes were associated with this resistance. Glutathione reductase replenishes cellular GSH stock and circumvents ferroptotic cell death [65]. Therefore, by targeting these cells with ferroptosis-inducers, the authors were able to overcome treatment resistance without affecting the viability of other non-tumoral cells in vitro [64]. These results provide further evidence for the role of ferroptosis as a target while minimizing toxic side effects.
Ferroptosis induction is especially powerful in treatment-resistant cell lines, as was shown in a study that overcame Cetuximab resistance in oral cancer by inducing ferroptosis [66]. It was noticed that the utilization of Cetuximab in resistant cells was not enough to suppress tumor growth or reduce viability. However, when Cetuximab was associated with RSL3, a ferroptosis-inducer agent, there was mitochondrial damage and increased cellular sensitivity to ferroptosis. RSL3 acts by depletion of GTX4, which is responsible for regulating a GTX4 protein depletor, which is, in turn, responsible for reducing the amount of intracellular lipid peroxide [67]. However, the study does not suggest through which mechanism this chemical synergy occurs, despite this effect being described in other studies [68][69]. Ferroptosis can also be induced by hyperbaric oxygen and X-ray radiation in a synergic effect [70]. It has been evaluated as a mechanism to overcome radiotherapy resistance by oral cancer cells, as has been done in other cancers [71]. Another interesting approach is the development of carrier particles to increase intracellular iron and trigger cell death [72]. OSCC-specific nanoprobes carrying iron can be internalized by endocytosis, much like “Trojan horses”. The acidic lysosomal conditions lead to the release of iron from the nanoprobes, inducing ferroptosis [72]. Non-thermal plasma treatment also showed an effect in oral squamous cell carcinoma cells by promoting lipid peroxidation and, ultimately, cell death by ferroptosis [73]. Moreover, studies show that regulation of ferroptosis inhibitor proteins is a promising therapeutic approach, as is the case of caveolin 1 (CAV1) downregulation [74]. CAV1 is known for its ferroptosis-inhibiting capabilities [74]. By knocking down CAV1, authors increased ROS and intracellular iron levels while reducing tumor cell growth in vitro. One of the main obstacles to inducing ferroptosis is assuring target specificity in order to avoid non-tumoral cell toxicity and side effects. In most studies, this has been achieved with a certain degree of success, such as the use of A-GSP, which was able to promote ferroptosis in oral cancer cells by inducing Fe2+ influx and GSH depletion, as well as lipid peroxide and ROS accumulation, while not producing significant toxic side effects [61].
In summary, several studies have shown promising results in using the existent ferroptosis-related cellular machinery as potential therapeutic strategies. Both knockdown strategies targeting key proteins involved in promoting/regulating this type of cell death and application of compounds with activity to induce ferroptosis of the OSCC cells were used. Additionally, the characterization of several gene signatures of ferroptosis-associated genes for both prognostication and response to treatment is relevant, but further verification in large-scale clinical studies is required. Thereafter, the ongoing exploration of ferroptosis in oral cancer not only deepens our understanding of cancer mechanisms but also holds the potential to translate this knowledge to improve patient outcomes.
3. Pyroptosis
Pyroptosis, an inflammatory type of caspase-mediated cell death, can modulate the immunogenic potential of specific cancers [75]. The role of pyroptosis in OSCC is an area of ongoing research, and the mechanisms and implications are not fully understood, but there is evidence to suggest that pyroptosis may play a role in both development and progression of OSCC [75][76][77][78][79][80][81][82]. Pyroptosis derives its name from the combination of “pyro” and “ptosis”. “Pyro” signifies fire, highlighting its inflammatory properties, while “ptosis” refers to falling, which aligns with other forms of programmed cell death [83]. There are notable similarities between pyroptosis and apoptosis, including features like DNA damage and chromatin condensation [84]. Interestingly, pyroptotic cells exhibit swelling and numerous bubble-like protrusions on the cellular membrane before rupture, a phenomenon reminiscent of membrane blebbing observed in apoptosis. Pyroptosis was officially defined as Gasdermin-mediated programmed cell death in 2015 [83]. The Gasdermin superfamily in humans includes Gasdermin A/B/C/D (GSDMA/B/C/D), Gasdermin E (GSDME, also known as DFNA5), and DFNB59 (Pejvakin, PJVK). Inflammasomes are responsible for initiating pyroptosis via two distinct pathways (Figure 2). The canonical inflammasome pathway is reliant on the activation of caspase-1, and the noncanonical inflammasome pathway involves the activation of caspase-4, caspase-5, or caspase-11 [85]. Furthermore, certain studies have demonstrated that proapoptotic caspase-3 activation can also initiate pyroptosis by cleaving GSDME [86][87].
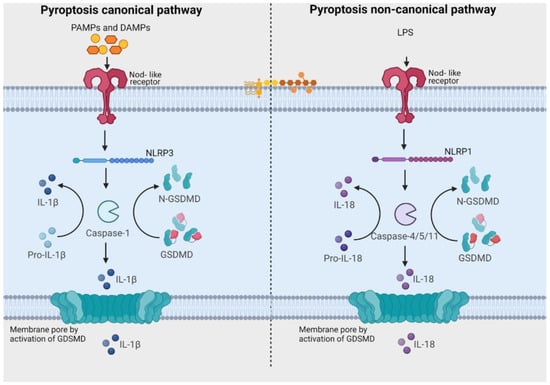
Figure 2. Pathways related to activation of pyroptosis. In the canonical signaling pathway, intracellular sensors Nod-like receptor family, pyrin domain containing 1 (NLRP1), 3 (NLRP3), 4 (NLRC4), absent in melanoma 2 (AIM2) and other inflammasome sensors are responsible for detecting microbial signals. Upon detection, they initiate a response by recruiting the adaptor protein ASC (apoptosis-associated speck-like protein containing a C-terminal caspase recruitment domain), which subsequently recruits pro-caspase-1. Once activated, caspase-1 cleaves Gasdermin D (GSDMD), generating GSDMD-NT fragments. These GSDMD-NT fragments create pores in the plasma membrane that are associated with phosphoinositides. Simultaneously, caspase-1 itself undergoes cleavage, giving rise to caspase-1 P10/P20 and P33/P10 tetramers. These tetramers play a crucial role in the maturation of pro-interleukin-18 (IL-18) and pro-interleukin-1β (IL-1β) into their active forms, IL-18 and IL-1β. These mature cytokines are subsequently released into the extracellular matrix, leading to the initiation of inflammatory responses. In the noncanonical pathway, the presence of lipopolysaccharides (LPS) from Gram-negative bacteria triggers the activation of caspase-4 and caspase-5 (in humans) or caspase-11 (in mice). These caspases, in turn, cleave GSDMD, forming pores in the plasma membrane. These GSDMD pores permit the release of potassium ions, which further activate the NLRP3 inflammasome and contribute to the maturation of IL-1β and IL-18. Additionally, GSDMD pores release mature cytokines, ultimately leading to pyroptosis. [Image was created using Biorender.com (accessed on 13 December 2023)].
Inflammation is a critical component of tumor progression [88], and inflammation intensified by chemotherapy can lead to therapy failure and metastasis [89]. In the Nod-like receptor family, pyrin domain containing 3 (NLRP3) inflammasome is one of the critical components of the innate immune system and plays an important role in cancer [90][91]. Many factors can activate NLRP3 inflammasomes, including potassium efflux, intracellular calcium, endoplasmic reticulum (ER) stress and ROS [91]. Chronic inflammation has the potential to impact every phase of the carcinogenic process, increasing the risk of tumorigenesis with prolonged exposure to an inflammatory milieu [92]. Pyroptosis, as a form of lytic cell death, amplifies the release of mature interleukin-1 (IL-1) and interleukin-18 (IL-18), potentially influencing the development of cancer [93]. Furthermore, pyroptosis serves as the mechanism for inflammatory cell death in cancer cells, thereby restraining the proliferation and migration of these cancer cells [78][79][94][95][96][97][98]. Consequently, pyroptosis assumes a dual role, both promoting and inhibiting tumorigenesis [99]. Previous findings revealed that 5-fluorouracil (5-FU) treatment increased NLRP3 expression in OSCC, which mediated drug resistance. It was also proven that NLRP3 could promote tumor growth and metastasis in OSCC [97][100][101]. Activation of pyroptosis has also been directly associated with increased chemoresistance to cisplatin and 5-FU treatment [100], and inhibition of pyroptosis has been associated with increased sensitivity of neoplastic cells to cisplatin treatment [102].
Methods of pyroptosis inhibition are gaining the attention of the scientific community [97][103][104][105]. Yang et al. [97] highlighted that extracellular vesicles derived from bitter melon led to a significant decrease in the expression of NLRP3, reducing OSCC resistance to 5-FU treatment. The study developed by Yue et al. [103] evaluated the effect of anthocyanin on OSCC. Anthocyanin reduced the viability of OSCC cells and inhibited migration and invasion capacity, concomitantly increasing pyroptosis. Simultaneously, activation of pyroptosis was associated with increased expression of NLRP3, caspase-1 and interleukin-1β (IL-1β). After administration of caspase-1 inhibitors, anthocyanin-activated pyroptosis was suppressed, and cell viability, migration and invasion rates increased concomitantly. In vitro studies in monoculture do not show the real dimension of the role of pyroptosis in OSCC. Therefore, conflicting results can be seen depending on the methodology used in different studies. The poor prognosis of pyroptosis is associated with its ability to activate inflammation; however, the development of in vitro and in vivo studies capable of more broadly evaluating the tumor microenvironment and all mechanisms triggered by the activation of pyroptosis should be encouraged. The inflammation associated with pyroptosis can lead to the recruitment of immune cells and other factors that support tumor growth and metastasis [105]. The pro-inflammatory environment can also contribute to resistance to therapy and promote angiogenesis, which is the formation of new blood vessels that supply nutrients to the tumor. The study developed by Xin et al. [104] used The Cancer Genome Atlas (TCGA) dataset to investigate the predictive value of pyroptosis-related lncRNAs in the prognosis of OSCC. The authors identified eight pyroptosis-related lncRNAs associated with overall survival in patients with OSCC by multivariate regression analysis.
Taken together, the analysis indicates that the number of studies exploring pyroptosis in OSCC is still restricted, limiting our knowledge of the mechanisms of how molecules related to this type of cell death affect OSCC cells. However, the studies highlighted the potential of pyroptosis in the control of OSCC development and progression and described drugs and molecules related to both blockage and induction of it. Moreover, two studies demonstrated that pyroptosis gene clusters are correlated with clinical characteristics, infiltration of immune cells, susceptibility to chemotherapy and immunotherapy, and prognosis of patients with OSCC [104][106]. It will be important to validate these risk models and to explore potential drugs that could induce cell death through pyroptosis and concomitantly inhibit the pro-tumor inflammatory response in OSCCs.
4. Necroptosis
Necroptosis is considered a programmed form of necrosis mediated by receptor-interacting protein kinase 1 (RIPK1) and receptor-interacting protein kinase 3 (RIPK3) [107][108]. The stimulus is initiated by tumor necrosis factor (TNF) binding to its receptors [109][110]. The interaction of ligand and receptor leads to the formation of a signaling complex, which may include adaptor proteins such as Fas-associated protein with death domain (FADD) and TNF receptor-associated death domain (TRADD). RIPK1 is recruited to the signaling complex and is activated by phosphorylation. Depending on cellular conditions, RIPK1 can associate with caspases, promoting apoptosis, or with RIPK3, initiating the necroptosis pathway. If the necroptosis pathway is activated, RIPK1 recruits RIPK3 and the protein MLKL, forming the necrosome complex. RIPK3 phosphorylates MLKL, activating it. Activated MLKL oligomerizes and translocates to the plasma membrane, where it causes damage, leading to membrane rupture and necroptosis (Figure 3) [111].
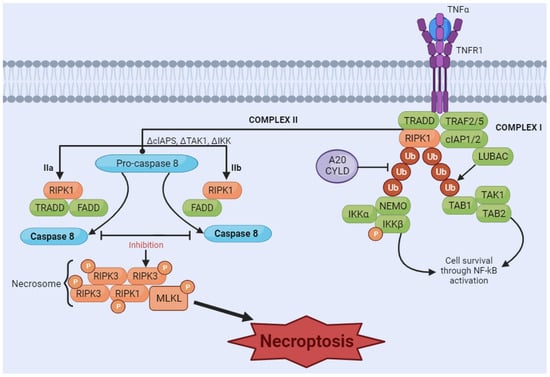
Figure 3. Necroptosis signaling pathway. The binding of tumor necrosis factor (TNF) induces the formation of the membrane-associated complex I, composed of TNF receptor-associated death domain (TRADD), TNF receptor-associated factor 2 (TRAF2), cellular inhibitor of apoptosis protein 1 and 2 (cIAP1/2), receptor-interacting serine/threonine kinase 1 (RIPK1), and LUBAC, an E3 ubiquitin ligase. cIAP1/2 and LUBAC induce the polyubiquitination of RIPK1, recruiting the IκB kinase (IkappaB kinase) complex (IKKa, IKKB, and NEMO) and TGF-β-activated kinase 1 (TAK1) complex (TAK1, TAB1, and TAB2). These two complexes can eventually lead to the activation of the NF-κB pathway and cell survival. A20CYLD promotes the deubiquitination of RIPK1, inducing the dissociation of TRADD and RIPK1 from TNFR1 and leading to the formation of complex IIa or complex IIb. Complex IIa, consisting of TRADD and Fas-associated protein with death domain (FADD), activates caspase-8 and induces apoptosis through cleavage. Inhibition of RIPK1 ubiquitination results in the induction of complex IIb, composed of RIPK1, FADD and caspase-8. When caspase-8 is inhibited (in either complex IIa or IIb), RIPK1 and RIPK3 form necrosome complexes, activating MLKL through a phosphorylation cascade. Phosphorylated MLKL undergoes oligomerization and migrates to the membrane, which induces necroptosis by membrane rupture or regulating ion flow. [Image was created using Biorender.com (accessed on 16 November 2023)].
The formation of an MLKL oligomer opens a pore in the membrane, allowing the entrance of ROS and DAMPs [112]. In this manner, death by necroptosis, despite being activated by specific signals, leads to a similar end as necrosis and ferroptosis, triggering the entrance of ROS in the cell and leading to membrane damage [112][113]. To allow for necroptosis to occur, the apoptotic pathway must be impaired or damaged [107][114][115], which can be quite common in cancer since the inhibition of healthy apoptosis as a control allows for the unchecked reproduction of damaged cells [116]. This phenomenon holds significant implications for oral cancer, where dysregulation of cell death mechanisms can tip the balance in favor of tumor progression.
In this context, necroptosis, which is characterized by a regulated inflammatory response, becomes a last-resort mechanism to eliminate aberrant cells [117][118]. However, cancer cells can hijack this mechanism to promote their survival and evade the body’s natural defenses, using pro-tumoral inflammation to their advantage [117]. Additionally, necroptosis can generate an immunosuppressive tumor microenvironment, which may further contribute to cancer cell survival and progression [119]. It comes as no surprise that the expression level of key mediators of necroptosis is elevated in cancer [117][120], indicating that necroptosis may play a role in promoting oncogenesis and cancer metastasis [121]. However, it is possible to interfere in this process by targeting necroptosis as an ally in halting cancer progression and survival, as has been evaluated in oral cancer studies, by targeting focal adhesion molecules [122][123] or even caspase-8 itself, which is responsible for deciding the pathway outcome of apoptosis versus necrosis [122][124]. In the context of OSCC, studies have explored the induction of necroptosis using different agents, such as Obatoclax [125]. This agent targets members of the BCL-2 family, specifically the myeloid cell leukemia sequence 1 (MCL-1). The study suggests that Obatoclax induces cell death in OSCC cells through autophagy-dependent necroptosis [126], with mitochondrial stress and dysfunction as detectable upstream events. Additionally, capsaicin was found to inhibit cell proliferation and induce endoplasmic reticulum stress and autophagy in oral cancer [127]. This mechanism negatively regulates ribophorin II, impairing P-glycoprotein functions and sensitizing cells to anticancer therapy [128]. The association of capsaicin with anticancer agents promotes necroptosis rather than apoptosis, showcasing a unique pathway for inhibiting OSCC cell viability. Similarly, chelerythrine chloride (CS) demonstrated necroptosis induction in OSCC, impairing cell proliferation and inducing morphological alterations in a dose-dependent manner, such as membrane rupture, and dose-dependent cell death [127]. Moreover, the development of targeted delivery systems such as PLGA-Dtx (poly-lactic-co-glycolic acid nanoparticles containing docetaxel) has shown enhanced efficacy in inhibiting cancer cell proliferation. This strategy induced both apoptosis and necroptosis in oral cancer cells [129]. These results altogether suggest a potential role for these compounds in triggering necroptosis in OSCC cells.
Studies on necroptosis-related genes may reveal potential targets, either enhancers or inhibitors of this NAPCD. HNSCC studies have associated CASP8 mutations with radioresistance and poor survival outcomes [130], as is the case for OSCC [131]. In this manner, knockdown of CASP8 enhances the radiosensitizing effects of certain compounds through the induction of necroptosis. Additionally, as seen in other cell death mechanisms, the investigation of necroptosis-related genes that may be relevant in prognosis prediction has also been explored. Bioinformatic analyses and in vitro experiments identified six genes (hypoxanthine phosphoribosyltransferase 1-HPRT1, PGAM family member 5, mitochondrial serine/threonine protein phosphatase-PGAM5, BH3 interacting domain death agonist-BID, survival of motor neuron 1, telomeric-SMN1, FADD, and KIAA1191) contributing to OSCC development, metastasis, and immune modulation. These genes may play a role in the regulation of cell death pathways, including necroptosis [132]. Moreover, a study in HNSCC emphasized the prevalence of necroptosis and its association with poor overall survival and progression-free survival. Approximately half of the necrosis in HNSCC was attributed to necroptosis, indicating its significance as an independent risk factor for adverse clinical outcomes [133].
In summary, necroptosis induction is a promising alternative in the treatment of oral cancers, and the expression of necroptosis-related genes depicts prognostic potential for predicting OSCC outcomes. However, one of the main challenges in inducing necroptosis as a cancer treatment strategy remains in establishing specificity in a manner by which little toxicity is archived [124].
References
- Safa, A.R. Resistance to Cell Death and Its Modulation in Cancer Stem Cells. Crit. Rev. Oncog. 2016, 21, 203.
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Agostinis, P.; Alnemri, E.S.; Altucci, L.; Amelio, I.; Andrews, D.W.; et al. Molecular mechanisms of cell death: Recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018, 25, 486–541.
- Yan, G.; Elbadawi, M.; Efferth, T. Multiple cell death modalities and their key features (Review). World Acad. Sci. J. 2020, 2, 39–48.
- Peng, F.; Liao, M.; Qin, R.; Zhu, S.; Peng, C.; Fu, L.; Chen, Y.; Han, B. Regulated cell death (RCD) in cancer: Key pathways and targeted therapies. Signal Transduct. Target. Ther. 2022, 7, 286.
- Tong, X.; Tang, R.; Xiao, M.; Xu, J.; Wang, W.; Zhang, B.; Liu, J.; Yu, X.; Shi, S. Targeting cell death pathways for cancer therapy: Recent developments in necroptosis, pyroptosis, ferroptosis, and cuproptosis research. J. Hematol. Oncol. 2022, 15, 174.
- Wang, X.; Hua, P.; He, C.; Chen, M. Non-apoptotic cell death-based cancer therapy: Molecular mechanism, pharmacological modulators, and nanomedicine. Acta Pharm. Sin. B 2022, 12, 3567–3593.
- Xi, Y.; Gao, L.; Li, S.; Sun, K.; Chen, P.; Cai, Z.; Ren, W.; Zhi, K. The role of novel programmed cell death in head and neck squamous cell carcinoma: From mechanisms to potential therapies. Front. Pharmacol. 2023, 14, 1228985.
- Bedoui, S.; Herold, M.J.; Strasser, A. Emerging connectivity of programmed cell death pathways and its physiological implications. Nat. Rev. Mol. Cell Biol. 2020, 21, 678–695.
- Li, J.; He, D.; Li, S.; Xiao, J.; Zhu, Z. Ferroptosis: The emerging player in remodeling triple-negative breast cancer. Front. Immunol. 2023, 14, 1284057.
- Jia, Y.-J.; Zhang, Y.; Ma, X.-B.; Wang, Y.; Tian, Y.-Q.; He, P.-X.; Xu, Y.-C. Ferroptosis: Opportunities and Challenges in Cancer. J. Explor. Res. Pharmacol. 2023, 8, 243–253.
- Frank, D.; Vince, J.E. Pyroptosis versus necroptosis: Similarities, differences, and crosstalk. Cell Death Differ. 2019, 26, 99–114.
- Bertheloot, D.; Latz, E.; Franklin, B.S. Necroptosis, pyroptosis and apoptosis: An intricate game of cell death. Cell. Mol. Immunol. 2021, 18, 1106–1121.
- Kayagaki, N.; Webster, J.D.; Newton, K. Control of Cell Death in Health and Disease. Annu. Rev. Pathol. Mech. Dis. 2023, 19, 157–180.
- Gulia, S.; Chandra, P.; Das, A. The Prognosis of Cancer Depends on the Interplay of Autophagy, Apoptosis, and Anoikis within the Tumor Microenvironment. Cell Biochem. Biophys. 2023, 81, 621–658.
- Li, J.; Cao, F.; Yin, H.; Huang, Z.; Lin, Z.; Mao, N.; Sun, B.; Wang, G. Ferroptosis: Past, present and future. Cell Death Dis. 2020, 11, 88.
- Stockwell, B.R.; Angeli, J.P.F.; Bayir, H.; Bush, A.I.; Conrad, M.; Dixon, S.J.; Fulda, S.; Gascón, S.; Hatzios, S.K.; Kagan, V.E.; et al. Ferroptosis: A Regulated Cell Death Nexus Linking Metabolism, Redox Biology, and Disease. Cell 2017, 171, 273–285.
- Zhang, C.; Liu, X.; Jin, S.; Chen, Y.; Guo, R. Ferroptosis in cancer therapy: A novel approach to reversing drug resistance. Mol. Cancer 2022, 21, 47.
- Hou, W.; Xie, Y.; Song, X.; Sun, X.; Lotze, M.T.; Zeh, H.J., 3rd; Kang, R.; Tang, D. Autophagy promotes ferroptosis by degradation of ferritin. Autophagy 2016, 12, 1425–1428.
- Chen, Y.; Fan, Z.; Hu, S.; Lu, C.; Xiang, Y.; Liao, S. Ferroptosis: A New Strategy for Cancer Therapy. Front. Oncol. 2022, 12, 830561.
- Tang, D.; Chen, X.; Kang, R.; Kroemer, G. Ferroptosis: Molecular mechanisms and health implications. Cell Res. 2021, 31, 107–125.
- Cao, J.Y.; Dixon, S.J. Mechanisms of ferroptosis. Cell. Mol. Life Sci. 2016, 73, 2195–2209.
- Ying, J.F.; Lu, Z.B.; Fu, L.Q.; Tong, Y.; Wang, Z.; Li, W.F.; Mou, X.Z. The role of iron homeostasis and iron-mediated ROS in cancer. Am. J. Cancer Res. 2021, 11, 1895.
- Seibt, T.M.; Proneth, B.; Conrad, M. Role of GPX4 in ferroptosis and its pharmacological implication. Free. Radic. Biol. Med. 2018, 133, 144–152.
- Dodson, M.; Castro-Portuguez, R.; Zhang, D.D. NRF2 plays a critical role in mitigating lipid peroxidation and ferroptosis. Redox Biol. 2019, 23, 101107.
- Jiang, L.; Kon, N.; Li, T.; Wang, S.-J.; Su, T.; Hibshoosh, H.; Baer, R.; Gu, W. Ferroptosis as a p53-mediated activity during tumour suppression. Nature 2015, 520, 57–62.
- Hassannia, B.; Vandenabeele, P.; Berghe, T.V. Targeting Ferroptosis to Iron Out Cancer. Cancer Cell 2019, 35, 830–849.
- Hsieh, C.-H.; Hsieh, H.-C.; Shih, F.-H.; Wang, P.-W.; Yang, L.-X.; Shieh, D.-B.; Wang, Y.-C. An innovative NRF2 nano-modulator induces lung cancer ferroptosis and elicits an immunostimulatory tumor microenvironment. Theranostics 2021, 11, 7072–7091.
- Ma, Q. Role of Nrf2 in Oxidative Stress and Toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426.
- Sun, X.; Ou, Z.; Chen, R.; Niu, X.; Chen, D.; Kang, R.; Tang, D. Activation of the p62-Keap1-NRF2 pathway protects against ferroptosis in hepatocellular carcinoma cells. Hepatology 2016, 63, 173–184.
- Zheng, S.; Mo, J.; Zhang, J.; Chen, Y. HIF-1α inhibits ferroptosis and promotes malignant progression in non-small cell lung cancer by activating the Hippo-YAP signalling pathway. Oncol. Lett. 2023, 25, 1–9.
- Jun, J.C.; Rathore, A.; Younas, H.; Gilkes, D.; Polotsky, V.Y. Hypoxia-Inducible Factors and Cancer. Curr. Sleep Med. Rep. 2017, 3, 1–10.
- Wu, J.; Minikes, A.M.; Gao, M.; Bian, H.; Li, Y.; Stockwell, B.R.; Chen, Z.-N.; Jiang, X. Intercellular interaction dictates cancer cell ferroptosis via NF2–YAP signalling. Nature 2019, 572, 402–406.
- Wang, W.; Green, M.; Choi, J.E.; Gijón, M.; Kennedy, P.D.; Johnson, J.K.; Liao, P.; Lang, X.; Kryczek, I.; Sell, A.; et al. CD8+ T cells regulate tumour ferroptosis during cancer immunotherapy. Nature 2019, 569, 270–274.
- Sun, S.; Shen, J.; Jiang, J.; Wang, F.; Min, J. Targeting ferroptosis opens new avenues for the development of novel therapeutics. Signal Transduct. Target. Ther. 2023, 8, 372.
- Lei, G.; Mao, C.; Yan, Y.; Zhuang, L.; Gan, B. Ferroptosis, radiotherapy, and combination therapeutic strategies. Protein Cell 2021, 12, 836–857.
- Lei, G.; Zhuang, L.; Gan, B. Targeting ferroptosis as a vulnerability in cancer. Nat. Rev. Cancer 2022, 22, 381–396.
- Luo, L.; Wang, H.; Tian, W.; Zeng, J.; Huang, Y.; Luo, H. Targeting ferroptosis for cancer therapy: Iron metabolism and anticancer immunity. Am. J. Cancer Res. 2021, 11, 5508–5525.
- Cosialls, E.; El Hage, R.; Dos Santos, L.; Gong, C.; Mehrpour, M.; Hamaï, A. Ferroptosis: Cancer Stem Cells Rely on Iron until “to Die for” It. Cells 2021, 10, 2981.
- Taylor, W.R.; Fedorka, S.R.; Gad, I.; Shah, R.; Alqahtani, H.D.; Koranne, R.; Kuganesan, N.; Dlamini, S.; Rogers, T.; Al-Hamashi, A.; et al. Small-Molecule Ferroptotic Agents with Potential to Selectively Target Cancer Stem Cells. Sci. Rep. 2019, 9, 5926.
- Wang, Y.; Yu, L.; Ding, J.; Chen, Y. Iron Metabolism in Cancer. Int. J. Mol. Sci. 2019, 20, 95.
- Wang, H.; Zhang, Z.; Ruan, S.; Yan, Q.; Chen, Y.; Cui, J.; Wang, X.; Huang, S.; Hou, B. Regulation of iron metabolism and ferroptosis in cancer stem cells. Front. Oncol. 2023, 13, 1251561.
- Gnanapradeepan, K.; Basu, S.; Barnoud, T.; Budina-Kolomets, A.; Kung, C.-P.; Murphy, M.E. The p53 Tumor Suppressor in the Control of Metabolism and Ferroptosis. Front. Endocrinol. 2018, 9, 124.
- Liu, Y.; Gu, W. p53 in ferroptosis regulation: The new weapon for the old guardian. Cell Death Differ. 2022, 29, 895–910.
- Poltorack, C.D.; Dixon, S.J. Understanding the role of cysteine in ferroptosis: Progress & paradoxes. FEBS J. 2021, 289, 374–385.
- Liu, M.-Z.; Kong, N.; Zhang, G.-Y.; Xu, Q.; Xu, Y.; Ke, P.; Liu, C. The critical role of ferritinophagy in human disease. Front. Pharmacol. 2022, 13, 933732.
- Chung, C.; Lin, C.; Chen, C.; Hsueh, C.; Chang, Y.; Wang, C.; Chu, P.; Tai, S.; Yang, M. Ferroptosis Signature Shapes the Immune Profiles to Enhance the Response to Immune Checkpoint Inhibitors in Head and Neck Cancer. Adv. Sci. 2023, 10, 2204514.
- Zhu, T.; Shi, L.; Yu, C.; Dong, Y.; Qiu, F.; Shen, L.; Qian, Q.; Zhou, G.; Zhu, X. Ferroptosis Promotes Photodynamic Therapy: Supramolecular Photosensitizer-Inducer Nanodrug for Enhanced Cancer Treatment. Theranostics 2019, 9, 3293–3307.
- Sun, K.; Ren, W.; Li, S.; Zheng, J.; Huang, Y.; Zhi, K.; Gao, L. MiR-34c-3p upregulates erastin-induced ferroptosis to inhibit proliferation in oral squamous cell carcinomas by targeting SLC7A11. Pathol. Res. Pract. 2022, 231, 153778.
- Wang, L.; Wang, C.; Li, X.; Tao, Z.; Zhu, W.; Su, Y.; Choi, W.S. Melatonin and erastin emerge synergistic anti-tumor effects on oral squamous cell carcinoma by inducing apoptosis, ferroptosis, and inhibiting autophagy through promoting ROS. Cell. Mol. Biol. Lett. 2023, 28, 36.
- Han, L.; Li, L.; Wu, G. Induction of ferroptosis by carnosic acid-mediated inactivation of Nrf2/HO-1 potentiates cisplatin responsiveness in OSCC cells. Mol. Cell. Probes 2022, 64, 101821.
- Wang, Z.-Q.; Li, Y.-Q.; Wang, D.-Y.; Shen, Y.-Q. Natural product piperlongumine inhibits proliferation of oral squamous carcinoma cells by inducing ferroptosis and inhibiting intracellular antioxidant capacity. Transl. Cancer Res. 2022, 12, 2911–2922.
- Zhao, Y.; Zhu, S. Nrf2/HO-1 Alleviates Disulfiram/Copper-Induced Ferroptosis in Oral Squamous Cell Carcinoma. Biochem. Genet. 2023. online ahead of print.
- Yang, J.; Cao, X.-H.; Luan, K.-F.; Huang, Y.-D. Circular RNA FNDC3B Protects Oral Squamous Cell Carcinoma Cells From Ferroptosis and Contributes to the Malignant Progression by Regulating miR-520d-5p/SLC7A11 Axis. Front. Oncol. 2021, 11, 672724.
- Xu, C.; Shen, Y.; Shi, Y.; Zhang, M.; Zhou, L. Eukaryotic translation initiation factor 3 subunit B promotes head and neck cancer via CEBPB translation. Cancer Cell Int. 2022, 22, 161.
- Yu, Y.; MohamedAl-Sharani, H.; Zhang, B. EZH2-mediated SLC7A11 upregulation via miR-125b-5p represses ferroptosis of TSCC. Oral Dis. 2021, 29, 880–891.
- Li, J.; Xiao, W.; Wei, W.; Wu, M.; Xiong, K.; Lyu, J.; Li, Y. HSPA5, as a ferroptosis regulator, may serve as a potential therapeutic for head and neck squamous cell carcinoma. Mol. Immunol. 2023, 158, 79–90.
- Zhou, Q.; Wang, X.; Zhang, Y.; Wang, L.; Chen, Z. Inhibition of AEBP1 predisposes cisplatin-resistant oral cancer cells to ferroptosis. BMC Oral Health 2022, 22, 478.
- Lee, J.; You, J.H.; Shin, D.; Roh, J.-L. Inhibition of Glutaredoxin 5 predisposes Cisplatin-resistant Head and Neck Cancer Cells to Ferroptosis. Theranostics 2020, 10, 7775–7786.
- Tomita, K.; Nagasawa, T.; Kuwahara, Y.; Torii, S.; Igarashi, K.; Roudkenar, M.H.; Roushandeh, A.M.; Kurimasa, A.; Sato, T. MiR-7-5p Is Involved in Ferroptosis Signaling and Radioresistance Thru the Generation of ROS in Radioresistant HeLa and SAS Cell Lines. Int. J. Mol. Sci. 2021, 22, 8300.
- You, J.H.; Lee, J.; Roh, J.-L. Mitochondrial pyruvate carrier 1 regulates ferroptosis in drug-tolerant persister head and neck cancer cells via epithelial-mesenchymal transition. Cancer Lett. 2021, 507, 40–54.
- Wu, X.; Wu, Q.; Wang, Y.; Liu, Y.; Li, Z.; Liu, Q.; Huang, Z.; Li, M.; Zhang, B.; Zhan, Q. Aqueous-soluble components of sporoderm-removed Ganoderma lucidum spore powder promote ferroptosis in oral squamous cell carcinoma. Chin. J. Cancer Res. 2023, 35, 176–190.
- Li, J.; Jia, Y.-C.; Ding, Y.-X.; Bai, J.; Cao, F.; Li, F. The crosstalk between ferroptosis and mitochondrial dynamic regulatory networks. Int. J. Biol. Sci. 2023, 19, 2756–2771.
- Wang, Z.; Han, J.; Guo, Z.; Wu, H.; Liu, Y.; Wang, W.; Zhang, C.; Liu, J. Ginseng-based carbon dots inhibit the growth of squamous cancer cells by increasing ferroptosis. Front. Oncol. 2023, 13, 1097692.
- Huang, K.-J.; Wei, Y.-H.; Chiu, Y.-C.; Wu, S.-R.; Shieh, D.-B. Assessment of zero-valent iron-based nanotherapeutics for ferroptosis induction and resensitization strategy in cancer cells. Biomater. Sci. 2019, 7, 1311–1322.
- Bartolacci, C.; Andreani, C.; El-Gammal, Y.; Scaglioni, P.P. Lipid Metabolism Regulates Oxidative Stress and Ferroptosis in RAS-Driven Cancers: A Perspective on Cancer Progression and Therapy. Front. Mol. Biosci. 2021, 8, 706650.
- Jehl, A.; Conrad, O.; Burgy, M.; Foppolo, S.; Vauchelles, R.; Ronzani, C.; Etienne-Selloum, N.; Chenard, M.-P.; Danic, A.; Dourlhes, T.; et al. Blocking EREG/GPX4 Sensitizes Head and Neck Cancer to Cetuximab through Ferroptosis Induction. Cells 2023, 12, 733.
- Hirata, Y.; Cai, R.; Volchuk, A.; Steinberg, B.E.; Saito, Y.; Matsuzawa, A.; Grinstein, S.; Freeman, S.A. Lipid peroxidation increases membrane tension, Piezo1 gating, and cation permeability to execute ferroptosis. Curr. Biol. 2023, 33, 1282–1294.
- Yang, J.; Mo, J.; Dai, J.; Ye, C.; Cen, W.; Zheng, X.; Jiang, L.; Ye, L. Cetuximab promotes RSL3-induced ferroptosis by suppressing the Nrf2/HO-1 signalling pathway in KRAS mutant colorectal cancer. Cell Death Dis. 2021, 12, 1079.
- Zhang, C.; Wang, C.; Yang, Z.; Bai, Y.; Shukuya, T.; Poh, M.-E.; Ekman, S.; Li, J.; Xu, Y.; Deng, S. Identification of GPX4 as a therapeutic target for lung adenocarcinoma after EGFR-TKI resistance. Transl. Lung Cancer Res. 2022, 11, 786–801.
- Liu, J.; An, W.; Zhao, Q.; Liu, Z.; Jiang, Y.; Li, H.; Wang, D. Hyperbaric oxygen enhances X-ray induced ferroptosis in oral squamous cell carcinoma cells. Oral Dis. 2022. online ahead of print.
- Stępień, K.; Ostrowski, R.P.; Matyja, E. Hyperbaric oxygen as an adjunctive therapy in treatment of malignancies, including brain tumours. Med. Oncol. 2016, 33, 101.
- Zhang, P.; Cui, Y.; Wang, J.; Cheng, J.; Zhu, L.; Liu, C.; Yue, S.; Pang, R.; Guan, J.; Xie, B.; et al. Dual-stimuli responsive smart nanoprobe for precise diagnosis and synergistic multi-modalities therapy of superficial squamous cell carcinoma. J. Nanobiotechnology 2023, 21, 4.
- Sato, K.; Shi, L.; Ito, F.; Ohara, Y.; Motooka, Y.; Tanaka, H.; Mizuno, M.; Hori, M.; Hirayama, T.; Hibi, H.; et al. Non-thermal plasma specifically kills oral squamous cell carcinoma cells in a catalytic Fe(II)-dependent manner. J. Clin. Biochem. Nutr. 2019, 65, 8–15.
- Lu, T.; Zhang, Z.; Pan, X.; Zhang, J.; Wang, X.; Wang, M.; Li, H.; Yan, M.; Chen, W. Caveolin-1 promotes cancer progression via inhibiting ferroptosis in head and neck squamous cell carcinoma. J. Oral Pathol. Med. 2022, 51, 52–62.
- Wang, S.; Wu, Z.-Z.; Zhu, S.-W.; Wan, S.-C.; Zhang, M.-J.; Zhang, B.-X.; Yang, Q.-C.; Xiao, Y.; Li, H.; Mao, L.; et al. CTLA-4 blockade induces tumor pyroptosis via CD8+ T cells in head and neck squamous cell carcinoma. Mol. Ther. 2023, 31, 2154–2168.
- Zhou, J.-Y.; Wang, W.-J.; Zhang, C.-Y.; Ling, Y.-Y.; Hong, X.-J.; Su, Q.; Li, W.-G.; Mao, Z.-W.; Cheng, B.; Tan, C.-P.; et al. Ru(II)-modified TiO2 nanoparticles for hypoxia-adaptive photo-immunotherapy of oral squamous cell carcinoma. Biomaterials 2022, 289, 121757.
- Zhu, S.-W.; Ye, M.; Ma, X.; Wu, Z.-Z.; Wan, S.-C.; Yang, S.-C.; Li, H.; Xu, Z.; Sun, Z.-J. pH-responsive nanoprodrugs combining a Src inhibitor and chemotherapy to potentiate antitumor immunity via pyroptosis in head and neck cancer. Acta Biomater. 2022, 154, 497–509.
- Zhu, W.; Zhang, J.; Wang, M.; Zhai, R.; Xu, Y.; Wang, J.; Wang, M.; Zhang, H.; Liu, L. Development of a prognostic pyroptosis-related gene signature for head and neck squamous cell carcinoma patient. Cancer Cell Int. 2022, 22, 62.
- Zi, M.; Xingyu, C.; Yang, C.; Xiaodong, S.; Shixian, L.; Shicheng, W. Improved antitumor immunity of chemotherapy in OSCC treatment by Gasdermin-E mediated pyroptosis. Apoptosis 2022, 28, 348–361.
- Liu, X.; Zhan, W.; Gao, G.; Jiang, Q.; Zhang, X.; Zhang, H.; Sun, X.; Han, W.; Wu, F.-G.; Liang, G. Apoptosis-Amplified Assembly of Porphyrin Nanofiber Enhances Photodynamic Therapy of Oral Tumor. J. Am. Chem. Soc. 2023, 145, 7918–7930.
- Nan, Z.; Dou, Y.; Chen, A.; Wang, K.; Sun, J.; Meng, Z.; Neckenig, M.; Ai, D.; Liu, S.; Dong, Z.; et al. Identification and validation of a prognostic signature of autophagy, apoptosis and pyroptosis-related genes for head and neck squamous cell carcinoma: To imply therapeutic choices of HPV negative patients. Front. Immunol. 2023, 13, 1100417.
- Yan, L.; Sun, Y.; Guo, J.; Jia, R. PD-L1 Exon 3 Is a Hidden Switch of Its Expression and Function in Oral Cancer Cells. Int. J. Mol. Sci. 2023, 24, 8193.
- Yu, P.; Zhang, X.; Liu, N.; Tang, L.; Peng, C.; Chen, X. Pyroptosis: Mechanisms and diseases. Signal Transduct. Target. Ther. 2021, 6, 128.
- Fang, Y.; Tian, S.; Pan, Y.; Li, W.; Wang, Q.; Tang, Y.; Yu, T.; Wu, X.; Shi, Y.; Ma, P.; et al. Pyroptosis: A new frontier in cancer. Biomed. Pharmacother. 2020, 121, 109595.
- Kelley, N.; Jeltema, D.; Duan, Y.; He, Y. The NLRP3 Inflammasome: An Overview of Mechanisms of Activation and Regulation. Int. J. Mol. Sci. 2019, 20, 3328.
- Jiang, M.; Qi, L.; Li, L.; Li, Y. The caspase-3/GSDME signal pathway as a switch between apoptosis and pyroptosis in cancer. Cell Death Discov. 2020, 6, 112.
- Bhat, A.A.; Thapa, R.; Afzal, O.; Agrawal, N.; Almalki, W.H.; Kazmi, I.; Alzarea, S.I.; Altamimi, A.S.A.; Prasher, P.; Singh, S.K.; et al. The pyroptotic role of Caspase-3/GSDME signalling pathway among various cancer: A Review. Int. J. Biol. Macromol. 2023, 242, 124832.
- Zhang, Z.; Li, X.; Wang, Y.; Wei, Y.; Wei, X. Involvement of inflammasomes in tumor microenvironment and tumor therapies. J. Hematol. Oncol. 2023, 16, 24.
- Vyas, D.; Laput, G.; Vyas, A. Chemotherapy-enhanced inflammation may lead to the failure of therapy and metastasis. OncoTargets Ther. 2014, 7, 1015–1023.
- Moossavi, M.; Parsamanesh, N.; Bahrami, A.; Atkin, S.L.; Sahebkar, A. Role of the NLRP3 inflammasome in cancer. Mol. Cancer 2018, 17, 158.
- Sharma, B.R.; Kanneganti, T.-D. NLRP3 inflammasome in cancer and metabolic diseases. Nat. Immunol. 2021, 22, 550–559.
- Liu, X.; Yin, L.; Shen, S.; Hou, Y. Inflammation and cancer: Paradoxical roles in tumorigenesis and implications in immunotherapies. Genes Dis. 2023, 10, 151–164.
- Lu, L.; Zhang, Y.; Tan, X.; Merkher, Y.; Leonov, S.; Zhu, L.; Deng, Y.; Zhang, H.; Zhu, D.; Tan, Y.; et al. Emerging mechanisms of pyroptosis and its therapeutic strategy in cancer. Cell Death Discov. 2022, 8, 338.
- Wang, X.; Liu, K.; Gong, H.; Li, D.; Chu, W.; Zhao, D.; Wang, X.; Xu, D. Death by histone deacetylase inhibitor quisinostat in tongue squamous cell carcinoma via apoptosis, pyroptosis, and ferroptosis. Toxicol. Appl. Pharmacol. 2020, 410, 115363.
- Bhuyan, S.; Pal, B.; Pathak, L.; Saikia, P.J.; Mitra, S.; Gayan, S.; Mokhtari, R.B.; Li, H.; Ramana, C.V.; Baishya, D.; et al. Targeting hypoxia-induced tumor stemness by activating pathogen-induced stem cell niche defense. Front. Immunol. 2022, 13, 933329.
- Luo, Q.; Li, X.; Gan, G.; Yang, M.; Chen, X.; Chen, F. PPT1 Reduction Contributes to Erianin-Induced Growth Inhibition in Oral Squamous Carcinoma Cells. Front. Cell Dev. Biol. 2021, 9, 764263.
- Yang, M.; Luo, Q.; Chen, X.; Chen, F. Bitter melon derived extracellular vesicles enhance the therapeutic effects and reduce the drug resistance of 5-fluorouracil on oral squamous cell carcinoma. J. Nanobiotechnol. 2021, 19, 259.
- Yao, Y.; Shen, X.; Zhou, M.; Tang, B. Periodontal Pathogens Promote Oral Squamous Cell Carcinoma by Regulating ATR and NLRP3 Inflammasome. Front. Oncol. 2021, 11, 722797.
- Lu, X.; Guo, T.; Zhang, X. Pyroptosis in Cancer: Friend or Foe? Cancers 2021, 13, 3620.
- Feng, X.; Luo, Q.; Zhang, H.; Wang, H.; Chen, W.; Meng, G.; Chen, F. The role of NLRP3 inflammasome in 5-fluorouracil resistance of oral squamous cell carcinoma. J. Exp. Clin. Cancer Res. 2017, 36, 81.
- Feng, X.; Luo, Q.; Wang, H.; Zhang, H.; Chen, F. MicroRNA-22 suppresses cell proliferation, migration and invasion in oral squamous cell carcinoma by targeting NLRP3. J. Cell. Physiol. 2018, 233, 6705–6713.
- Huang, Z.; Zhang, Q.; Wang, Y.; Chen, R.; Wang, Y.; Huang, Z.; Zhou, G.; Li, H.; Rui, X.; Jin, T.; et al. Inhibition of caspase-3-mediated GSDME-derived pyroptosis aids in noncancerous tissue protection of squamous cell carcinoma patients during cisplatin-based chemotherapy. Am. J. Cancer Res. 2020, 10, 4287.
- Yue, E.; Tuguzbaeva, G.; Chen, X.; Qin, Y.; Li, A.; Sun, X.; Dong, C.; Liu, Y.; Yu, Y.; Zahra, S.M.; et al. Anthocyanin is involved in the activation of pyroptosis in oral squamous cell carcinoma. Phytomedicine 2019, 56, 286–294.
- Xin, Y.; Zhang, J.; Jiang, Q.; Qiu, J. Construction of prognostic signature of patients with oral squamous cell carcinoma based on pyroptosis-related long non-coding RNAs. Front. Surg. 2022, 9, 935765.
- Wei, X.; Xie, F.; Zhou, X.; Wu, Y.; Yan, H.; Liu, T.; Huang, J.; Wang, F.; Zhou, F.; Zhang, L. Role of pyroptosis in inflammation and cancer. Cell. Mol. Immunol. 2022, 19, 971–992.
- Zeng, D.; Wang, X.; Zhang, S.; Zheng, A.; Huang, Q.; Cao, L. Pyroptosis-related gene-based prognostic signature for predicting the overall survival of oral squamous cell carcinoma patients. Front. Surg. 2022, 9, 903271.
- Dhuriya, Y.K.; Sharma, D. Necroptosis: A regulated inflammatory mode of cell death. J. Neuroinflammation 2018, 15, 199.
- Nicolè, L.; Sanavia, T.; Cappellesso, R.; Maffeis, V.; Akiba, J.; Kawahara, A.; Naito, Y.; Radu, C.M.; Simioni, P.; Serafin, D.; et al. Necroptosis-driving genes RIPK1, RIPK3 and MLKL-p are associated with intratumoral CD3+ and CD8+ T cell density and predict prognosis in hepatocellular carcinoma. J. Immunother. Cancer 2022, 10, e004031.
- Grootjans, S.; Vanden Berghe, T.; Vandenabeele, P. Initiation and execution mechanisms of necroptosis: An overview. Cell Death Differ. 2017, 24, 1184–1195.
- Pinci, F.; Gaidt, M.M.; Jung, C.; Nagl, D.; Kuut, G.; Hornung, V. Tumor necrosis factor is a necroptosis-associated alarmin. Front. Immunol. 2022, 13, 1074440.
- Seo, J.; Nam, Y.W.; Kim, S.; Oh, D.-B.; Song, J. Necroptosis molecular mechanisms: Recent findings regarding novel necroptosis regulators. Exp. Mol. Med. 2021, 53, 1007–1017.
- Deragon, M.A.; McCaig, W.D.; Truong, P.V.; Metz, K.R.; Carron, K.A.; Hughes, K.J.; Knapp, A.R.; Dougherty, M.J.; LaRocca, T.J. Mitochondrial Trafficking of MLKL, Bak/Bax, and Drp1 Is Mediated by RIP1 and ROS which Leads to Decreased Mitochondrial Membrane Integrity during the Hyperglycemic Shift to Necroptosis. Int. J. Mol. Sci. 2023, 24, 8609.
- Juan, C.A.; de la Lastra, J.M.P.; Plou, F.J.; Pérez-Lebeña, E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. Int. J. Mol. Sci. 2021, 22, 4642.
- Choi, M.E.; Price, D.R.; Ryter, S.W.; Choi, A.M.K. Necroptosis: A crucial pathogenic mediator of human disease. J. Clin. Investig. 2019, 4, 128834.
- Galluzzi, L.; Kepp, O.; Chan, F.K.-M.; Kroemer, G. Necroptosis: Mechanisms and Relevance to Disease. Annu. Rev. Pathol. Mech. Dis. 2017, 12, 103–130.
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as anticancer mechanism: Function and dysfunction of its modulators and targeted therapeutic strategies. Aging 2016, 8, 603–619.
- Gong, Y.; Fan, Z.; Luo, G.; Yang, C.; Huang, Q.; Fan, K.; Cheng, H.; Jin, K.; Ni, Q.; Yu, X.; et al. The role of necroptosis in cancer biology and therapy. Mol. Cancer 2019, 18, 100.
- Sprooten, J.; De Wijngaert, P.; Vanmeerbeek, I.; Martin, S.; Vangheluwe, P.; Schlenner, S.; Krysko, D.V.; Parys, J.B.; Bultynck, G.; Vandenabeele, P.; et al. Necroptosis in Immuno-Oncology and Cancer Immunotherapy. Cells 2020, 9, 1823.
- Yan, J.; Wan, P.; Choksi, S.; Liu, Z.-G. Necroptosis and tumor progression. Trends Cancer 2022, 8, 21–27.
- Khamseh, M.E.; Sheikhi, A.; Shahsavari, Z.; Ghorbani, M.; Akbari, H.; Imani, M.; Panahi, M.; Alimohammadi, A.; Ameri, M.; Nazem, S.; et al. Evaluation of the expression of necroptosis pathway mediators and its association with tumor characteristics in functional and non-functional pituitary adenomas. BMC Endocr. Disord. 2022, 22, 1.
- Zhang, T.; Wang, Y.; Inuzuka, H.; Wei, W. Necroptosis pathways in tumorigenesis. Semin. Cancer Biol. 2022, 86, 32–40.
- Wu, Y.; Dong, G.; Sheng, C. Targeting necroptosis in anticancer therapy: Mechanisms and modulators. Acta Pharm. Sin. B 2020, 10, 1601–1618.
- Murphy, J.M.; Rodriguez, Y.A.R.; Jeong, K.; Ahn, E.-Y.E.; Lim, S.-T.S. Targeting focal adhesion kinase in cancer cells and the tumor microenvironment. Exp. Mol. Med. 2020, 52, 877–886.
- Su, Z.; Yang, Z.; Xie, L.; DeWitt, J.P.; Chen, Y. Cancer therapy in the necroptosis era. Cell Death Differ. 2016, 23, 748–756.
- Sulkshane, P.; Teni, T. BH3 mimetic Obatoclax (GX15-070) mediates mitochondrial stress predominantly via MCL-1 inhibition and induces autophagy-dependent necroptosis in human oral cancer cells. Oncotarget 2016, 8, 60060–60079.
- Basit, F.; Cristofanon, S.; Fulda, S. Obatoclax (GX15-070) triggers necroptosis by promoting the assembly of the necrosome on autophagosomal membranes. Cell Death Differ. 2013, 20, 1161–1173.
- Huang, Y.-C.; Yuan, T.-M.; Liu, B.-H.; Liu, K.-L.; Wung, C.-H.; Chuang, S.-M. Capsaicin Potentiates Anticancer Drug Efficacy Through Autophagy-Mediated Ribophorin II Downregulation and Necroptosis in Oral Squamous Cell Carcinoma Cells. Front. Pharmacol. 2021, 12, 676813.
- Yuan, T.-M.; Liang, R.-Y.; Chueh, P.J.; Chuang, S.-M. Role of ribophorin II in the response to anticancer drugs in gastric cancer cell lines. Oncol. Lett. 2015, 9, 1861–1868.
- Gupta, P.; Singh, A.; Verma, A.K.; Kant, S.; Pandey, A.K.; Mishra, A.; Khare, P.; Prakash, V. Nanoencapsulation of Docetaxel Induces Concurrent Apoptosis and Necroptosis in Human Oral Cancer Cells (SCC-9) via TNF-α/RIP1/RIP3 Pathway. Indian J. Clin. Biochem. 2022, 38, 351–360.
- Uzunparmak, B.; Gao, M.; Lindemann, A.; Erikson, K.; Wang, L.; Lin, E.; Frank, S.J.; Gleber-Netto, F.O.; Zhao, M.; Skinner, H.D.; et al. Caspase-8 loss radiosensitizes head and neck squamous cell carcinoma to SMAC mimetic–induced necroptosis. J. Clin. Investig. 2020, 5, 139837.
- Bhosale, P.G.; Kennedy, R.A.; Watt, F.M. Caspase activation in tumour-infiltrating lymphocytes is associated with lymph node metastasis in oral squamous cell carcinoma. J. Pathol. 2023, 261, 43–54.
- Huang, K.; Gu, X.; Xu, H.; Li, H.; Shi, M.; Wei, D.; Wang, S.; Li, Y.; Liu, B.; Li, Y. Prognostic Value of Necroptosis-Related Genes Signature in Oral Squamous Cell Carcinoma. Cancers 2023, 15, 4539.
- Li, J.; Huang, S.; Zeng, L.; Li, K.; Yang, L.; Gao, S.; Guan, C.; Zhang, S.; Lao, X.; Liao, G.; et al. Necroptosis in head and neck squamous cell carcinoma: Characterization of clinicopathological relevance and in vitro cell model. Cell Death Dis. 2020, 11, 391.
More
Information
Subjects:
Cell Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
478
Revisions:
2 times
(View History)
Update Date:
29 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





