Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Anthony Burke | -- | 5037 | 2024-02-27 11:30:49 | | | |
| 2 | Camila Xu | Meta information modification | 5037 | 2024-02-28 03:25:55 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Carreiro, E.P.; Federsel, H.; Hermann, G.J.; Burke, A.J. Stereoselective Catalytic Synthesis of Bioactive Compounds in NADESs. Encyclopedia. Available online: https://encyclopedia.pub/entry/55534 (accessed on 05 May 2026).
Carreiro EP, Federsel H, Hermann GJ, Burke AJ. Stereoselective Catalytic Synthesis of Bioactive Compounds in NADESs. Encyclopedia. Available at: https://encyclopedia.pub/entry/55534. Accessed May 05, 2026.
Carreiro, Elisabete P., Hans-Jürgen Federsel, Gesine J. Hermann, Anthony J. Burke. "Stereoselective Catalytic Synthesis of Bioactive Compounds in NADESs" Encyclopedia, https://encyclopedia.pub/entry/55534 (accessed May 05, 2026).
Carreiro, E.P., Federsel, H., Hermann, G.J., & Burke, A.J. (2024, February 27). Stereoselective Catalytic Synthesis of Bioactive Compounds in NADESs. In Encyclopedia. https://encyclopedia.pub/entry/55534
Carreiro, Elisabete P., et al. "Stereoselective Catalytic Synthesis of Bioactive Compounds in NADESs." Encyclopedia. Web. 27 February, 2024.
Copy Citation
Deep eutectic solvents (DESs) are a mixture of two or more components, and at a particular composition, they become liquids at room temperature. When the compounds that constitute the DESs are primary metabolites namely, amino acids, organic acids, sugars, or choline derivatives, the DESs are called natural deep eutectic solvents (NADESs).
natural deep eutectic solvents (NADESs)
asymmetric catalysis
organocatalysis
biocatalysis
metal-based catalysis
1. Introduction
The vast majority of drugs launched on the market over the recent 2-to-3 decades exists as single enantiomers (or are achiral); this stems from the fact that for chiral molecules one enantiomer brings the therapeutic benefit whilst the other has no effect, or can have deleterious effects, as is the case for thalidomide (commercialized as Contergan®) administered in the 1960s as a racemate to pregnant women, where the (R)-enantiomer is a sedative and the (S)-enantiomer a teratogen in a number of countries [1]. (It must be pointed out that, in the special case of thalidomide, just administering the (R)-isomer to patients would not suffice from a safety point of view, as the molecule is sterically unstable under physiological conditions, leading to bio-inversion and formation of the undesired (S)-enantiomer.) The current market for single enantiomer drugs is more than EUR 300 billion. Many stereoselective methods are available to make single-enantiomer chiral drugs, but the most promising by far is via asymmetric catalysis. Many important and even block-buster drugs have been obtained using this methodology [2][3]. Asymmetric catalytic synthesis can be achieved through three principally distinct methods: metal-based catalysis, organocatalysis, and biocatalysis. Although catalysis, in general, is a green methodology, the overarching considerations in all of these approaches are the (1) cost of the catalyst, (2) catalytic activity throughout the reaction, and (3) recycling of the catalyst. There are many solutions; the catalyst can be immobilized—and traditionally, this was achieved via its attachment to an appropriate solid support [4]—and then used either under batch conditions or in continuous flow. Under these conditions, the reactions are heterogeneous and entail certain disadvantages (support type, pore size, stability of catalyst, immobilization method, existence of linker, and interactions between the solid support and the components); this can be overcome by using liquids to immobilize the catalysts, the most common being ionic liquids [5], or biphasic fluorous liquids [6], but the most recent is the Deep Eutectic Solvent (DES) category [7]. DESs have emerged as very useful liquids for many applications, but their most striking contribution is in the field of catalysis, particularly biocatalysis [7] and organocatalysis [8]. What is a DES? A DES is a liquid that is obtained when a hydrogen-bond acceptor (HBA), like a quaternary ammonium salt, is mixed with a hydrogen bond donor (HBD) [9]. Their melting points are lower than those of their components, and they readily mix with water but gratifyingly are immiscible with most organic solvents. DESs are recyclable, reusable, stable, non-flammable, biodegradable, and non-toxic mixtures. They have properties similar to ionic liquids, but unlike ionic liquids, they are non-toxic and are not biodegradable. One other significant advantage is their solubility properties, as well as their ability to stabilize transition states, particularly for organocatalyzed reactions. Some informative reviews on the subject have been published by the Alicante/Perugia groups [8], del Amo [9], and Domínguez de Maria [10].
2. Deep Eutectic Solvent (DES) Characteristics
The excellent physicochemical properties of DESs allow for the possibility of being used as greener solvents and an alternative to those commonly used in chemical synthesis. As shown above, DESs are built from hydrogen-bond donor and hydrogen-bond acceptor components and can be easily adapted for specific applications. The most widely used hydrogen bond donor components include urea, thiourea, phenol, glucose, citric acid, malonic acid, sorbitol, and glycerol. The most commonly used hydrogen bond acceptors (HBA) are choline chloride, betaine, glycine, and proline [9]. DESs have been applied in numerous chemical operations, including polymerizations, biotransformations, metal-catalyzed reactions, sample preparations, and catalytic techniques, as well as in the design of new pharmaceutical formulations [11]. Traditionally, the HBA was a halide salt, like choline chloride (ChCl), but recently, other types of DESs—the so-called Natural DESs or NADESs, which are obtained from organic acids, amino acids, and sugars, have been used more often [11]. One excellent example is the NADES developed from the HBA: N,N,N-trimethylglycine (betaine) [12]. The solvent category with the most similarity to NADES is the room-temperature ionic liquid (RTIL) category, which has enjoyed much success and application in stereoselective transformations over the last several decades, over time; however, the observations and evidence accumulated indicated that members of the RTIL family were tainted by the lack of certain green features and technical applicability, a high perceived cost, and poor biodegradability and biocompatibility, pointing in the direction of unsatisfactory sustainability and, thus, making the case for substitution by NADES.
3. Asymmetric Organocatalysis in DESs
The area of organocatalysis is perhaps the newest member to the field of asymmetric catalysis. Although various types of organocatalytic transformations have been used over the last 100 years, including the landmark Hajos–Parrish–Wiechert–Eder–Sauer intramolecular Aldol reaction used as an inspiration in the 1970s to make drug molecules. It, however, only became an important catalytic tool two decades ago with the developments of the 2022 Nobelists in chemistry, Benjamin List and David MacMillan [13]. More recently, asymmetric organocatalysis has been hooked up with DESs. The 2021 review by the Alicante/Perugia group provides a number of interesting examples of the application of asymmetric organocatalysis in NADESs for accessing some key target molecules (Figure 1) [8]. Most of the reactions that have been studied to date fall into the very limited categories of Aldol condensation [14], Michael addition [15], and α-aminations [16], as well as a report on an integrated biocatalytic/organocatalytic sequence that provides chiral 1,3-diols with yields of up to 70% and 99% ee. In fact, prior to 2016, there were no reports on the application of NADES to support organocatalysis [10], and the expectations were very high.
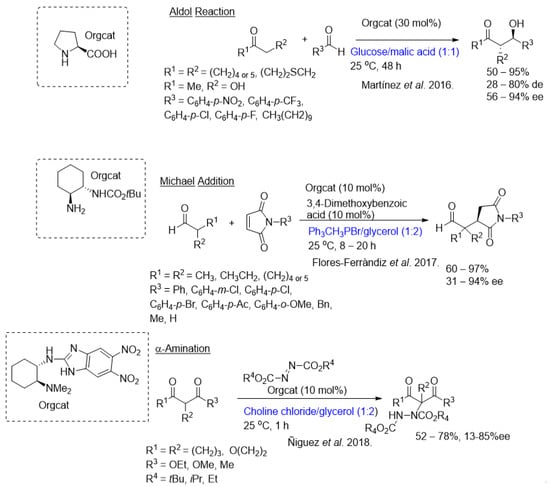
More recently, researchers' group successfully immobilized cinchona-squaramides to size-controlled porous glass beads and effectively used them in Michael additions of acetylacetone and methyl 2-oxocyclopentane-1-carboxylate to trans-β-nitrostyrene [17]. the application of this catalyst in the same reactions will be showed, but using NADES systems [18]. The results were highly enlightening, affording the Michael adducts in very high yields and stereoselectivities (Scheme 1). Yields of up to 99% and enantioselectivities of up to 99% ee could be achieved after 24 h at 40 °C (the reaction had to be run at this temperature to maintain the NADES in the liquid form and as viscous as possible). Three types of NADESs were investigated: betaine/sorbitol/water, betaine/xylitol/water, and betaine/mannitol/water. It was with the former that the best results were achieved; this was presumed to be due to the structural characteristics of the sorbitol. In the interest of recyclability, several reaction cycles were conducted with the same NADES and organocatalyst; in the case of methyl 2-oxocyclopentane-1-carboxylate, up to 10 cycles could be successfully performed without any significant drop in yield and stereoselectivities.
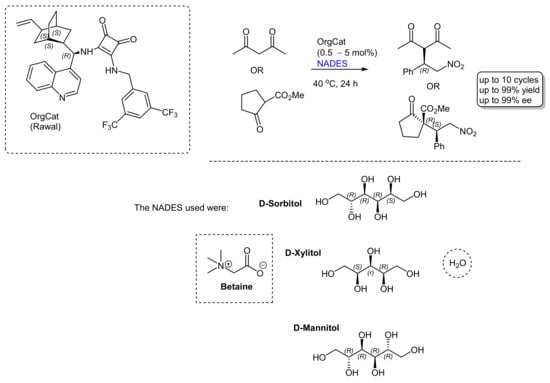
One other question is the role of the NADES solvent in organocatalyzed reactions. A recent experience in the Michael reaction is that the solvent or the components of the solvent compete with the organocatalyst. For example, the Michael reaction with methyl 2-oxocyclopentane-1-carboxylate occurred in betaine/sorbitol/water with a yield of 99% and a diastereoselectivity of 58% ee without any catalyst present. Some good support for this conjecture came from the density functional theory (DFT) calculations, which showed that there was a lowering of the reaction transition state by the sorbitol component. This was assumed to be due to key H-bonding between the sorbitol and the reaction transition state complex. Even stronger support came from the observation that one of the reactions in the betaine/sorbitol/water was highly enantioselective (for acetylacetone) with 75% ee. This phenomenon is not uncommon, and in fact both base and acid catalyzed reactions have been successfully executed in NADES (reactions like Perkin, Knoevenagel, and Aldol condensations) [10]. It appears, in fact, that NADESs are not “non-innocent” solvents, manifesting subtle interactions with the reaction intermediates, possibly also stabilizing the transition states via H-bond interactions. This discussion naturally leads to the role of chiral NADES as a medium for conducting asymmetric organocatalysis. A good example was the use of various L-proline-derived NADESs (as the HBA component) in asymmetric Michael reactions that involved butan-2-one and nitrostyrene [19]. Amongst the NADESs that were tested, the best performers consisted of diethyleneglycol/proline (3/1) (92% yield, syn/anti 85/15, ee 26/rac) and glycolic acid/proline (3/1) (15% yield, syn/anti 60/40, ee 86/95). This exciting aspect was also reviewed in 2022 by Tang and Yao [20], who alerted to the paucity of examples currently known and mentioned that there were decreases in the enantioselectivity of the reaction due to NADES recycling.
A more recent development has been with eutectogels and their use in asymmetric organocatalysis. In 2021, the group of Ramón and D’Anna reported on the application of these materials for asymmetric Aldol reactions [21]. These materials have been categorized as “catalytic soft materials” and are a combination of Low-Molecular-Weight Gelators (LMWGs) and NADES. They were designed to act as specific organized vessels to perform organocatalytic reactions. In this work, a variety of canonical amino acids, like proline, cysteine, asparagine, etc. (all of L-configuration), were used for a variety of Aldol reactions between ketones and aldehydes—employing initially the reagents acetone and p-nitrobenzaldehyde as the benchmark (Scheme 2). To determine the best LMWG and NADES combination, using the reaction of acetone with p-nitrobenzaldehyde indicated that a gel derived from using L-proline (5 wt%) in the NADES, ChCl/Urea (1:2) at 20 °C for 24 h, afforded the aldol product in an 87% yield and 68% ee (Scheme 2). The scope of the reaction was evaluated, and in some cases, enantioselectivities of up to 95% could be achieved; however, although in those cases where diastereomers were formed, the diastereoselectivities were only moderate. Michael additions were also conducted, using isobutyraldehyde or maleimide with trans-β-nitrostyrene, a base additive or an acid additive and 5 mol% proline, but unfortunately the yields were very poor.
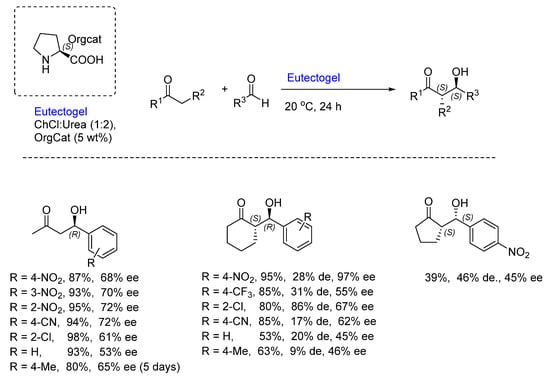
Scheme 2. The application of an L-proline-based eutectogel in the Aldol reaction.
Overall, this is a very promising, enabling technology that has much potential to offer for the synthesis of biologically active compounds but is still in its infancy.
4. Biocatalysis in DESs
The long-lasting paradigm that enzymes could actually function in a strictly aqueous environment was finally debunked in the mid-1980s by Klibanov and co-workers when they showed that the reactions could be run in organic solvents [22][23]. Since then, it has been shown by numerous examples how efficient biocatalytic reactions can be performed in “unconventional” systems such as “classical” organic solvents, room-temperature ionic liquids (RTIL), supercritical fluids, and of course, NADESs [24]. The latter category, along with RTILs, has brought several advantages for conducting enzyme-driven transformations, having enjoyed enormous attention as being broadly useful in organic chemistry. The ground-making work on the use of NADES in biocatalysis was performed by Kazlauskas’s group in 2008; they demonstrated that it was entirely feasible to run biocatalysis in these nonconventional systems, as was showcased by different biotransformations relying on hydrolases (CALB type) as catalytic species in a variety of choline chloride-based NADESs (Scheme 3) [25]. It was found that the catalytic activity was 20-fold better in the NADES system than in the original aqueous system.
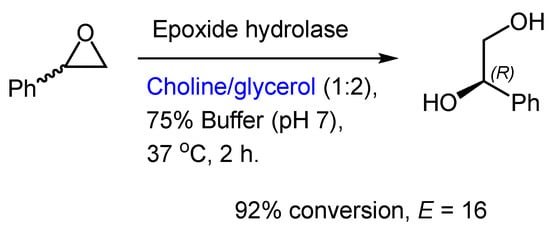
Scheme 3. Pioneering work from Kazlauskas’s group on the deracemization of styrene oxide [25].
This pioneering work showed the far superior performance in the NADES chosen to conduct this ring-opening hydrolysis, with 92% conversion in comparison to <5% in a “standard” aqueous buffer. It seems that the (R)-enantiomer was the favored isomer, and the enantioselectivity of the kinetic resolution was moderate (E = 16).
They have also been used successfully in trans-esterification reactions. Several articles disclose the use of glucose and several acyl donors for enzymatic transesterifications in DESs. For example, in 2015, Pöhnlein et al. reported the transesterification of glucose with vinyl hexanoate, using immobilized CALB (Novozym 435) in different DESs [26]. Ester products were obtained in the reactions that used both ChCl/urea and ChCl/glucose DESs. The beauty of this approach was that, in one case, the NADES behaved as the solvent and the substrate. In 2020, the same two hydrophilic DESs were reported for the transesterification of glucose with vinyl decanoate [27].
The same group also explored, for the first time, the use of a hydrophobic DES (menthol/decanoic acid) as a suitable reaction medium for sugar ester production [28]. The yields obtained with these NADESs were significantly higher than with hydrophilic NADESs. Different reaction parameters were investigated to optimize the synthesis further. Twenty milligrams/mL iCALB and 0.5 M glucose resulted in the highest initial reaction velocity for the esterification reaction, and the highest initial reaction velocity was observed with 1.5 M glucose in the transesterification reaction. The enzyme was reusable for at least five cycles, without significant loss of activity.
On the downside, a recent study from Ubialli’s group addressed the problems involved in the synthesis of sugar fatty acid esters from disaccharides, like lactose, using choline chloride-based NADES [29]. They encountered difficulties in conducting this transformation using standard Novozym lipases (Scheme 4). An important point was raised by these authors, where they correctly recognized that the NADES does not behave as a simple solvent, but in fact, its individual components interact with the reaction system’s constituents. (“Overall, the conclusion is that DESs are not innocent solvents because they interact with the substrates and the catalysts and influence the reaction outcome or directly hamper it”.) They also liked the choice of DES to the choice of catalyst for a reaction, particularly a chiral catalyst, where there is no such thing as a ‘one-size-fits-all’ solution. “Rather than being a simple ‘one-size-fits-all’ solution, incorporating a DES in a reaction needs a profound study for each particular case, starting from the genuine motivation to use them (solubility, sustainability, etc.), considering the enzyme and substrate compatibility, and eventually the ease of a downstream unit”.
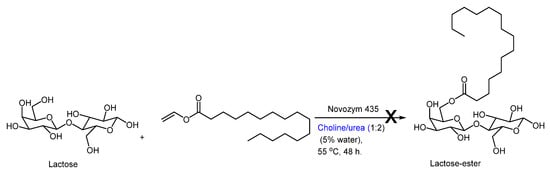
Scheme 4. Failed transesterification of lactose with vinyl palmitate (acyl donor) by Ubialli and co-workers [29].
The enzyme porcine pancreas lipase (PPL) was used in the enzymatic catalysis of the aldol reaction condensation of p-nitrobenzaldehyde and acetone in the same NADES ChCl/glycerol, as described above (Scheme 5) [30].
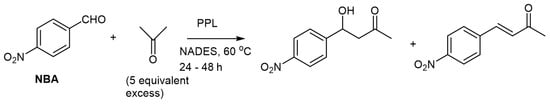
Scheme 5. PPL catalyzed aldol reaction in NADES [30].
It was noted that the product composition could be directly correlated with the choice of DES, as the DESs differ in their hydrophobicity. The initial reaction velocity of the aldol addition was higher in the hydrophilic NADES, ChCl:Gly (1:1.5). Unfortunately, this DES was limited by the solubility for the p-nitrobenzaldehyde (4-NBA) substrate. To overcome this problem, a NADES consisting of tetrabutylammonium chloride/4-NBA (2.2:1.5) was prepared and evaluated. The 4-NBA containing DES exhibited the highest solubility for 4-NBA. However, the fastest reaction with a final yield of 1296.5 mM, or 71% yield and 82% conversion after 32 h, was achieved when no NADES was used (solvent-less reaction conditions) and improving the productivity up to 40.5 mM h−1 compared to reactions performed in NADES. An enone side-product was obtained in most cases, but particularly for the more hydrophilic NADES, like ChCl/glycerol. BSA-catalyzed aldol additions were also investigated but did not exhibit specificity either for an aldol or olefin formation. Unfortunately, the authors did not discuss the stereochemical aspects of the reaction, such as the configuration of the major enantiomer or the direction of enantioselectivity.
The Henry reaction has also been explored in NADES. The Henry reaction is simple, but it is useful from a pharmaceutical point of view due to the presence of a vicinal benzylic chiral alcohol and a nitro group when aldehydes are used and nitro–amino compounds when imines are used (aza-Henry variant). The useful lipase-catalyzed (Aspergillus niger [lipase AS “Amano”]) C-C bond forming Henry reaction between nitromethane and carbonyl acceptors (4-NBA and other aromatic aldehydes) has been demonstrated to proceed in an excellent manner in DESs, such as ChCl/glycerol (1:2). By the addition of up to 30 vol% of H2O, it was found that the lipase activity could be improved, and under optimized conditions, a best yield of >92% was achieved (Scheme 6) [31].
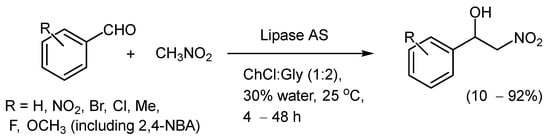
Scheme 6. Lipase-catalyzed Henry reactions, as reported by Zhang and co-workers [31].
Interestingly, when the aza-Henry reaction was conducted between benzaldehyde N-tosyl imine and nitromethane in ChCl:Gly (1:2), the product was obtained in 17% yield after 40 h, proving that the NADES could catalyze the reaction. This is a similar observation to one of ours for a Michael addition reaction [18]. The reactions with the Lipase present gave slightly better yields (up to 39%). Unfortunately, the reactions were not enantioselective, but the authors expressed a desire to use site-directed mutagenesis and alterations in the reaction medium to make the outcomes enantioselective.
It was demonstrated that the “classical” benzoin condensation can operate at high efficiency under DES conditions (Scheme 7). As shown by Maugeri and Domínguez de María, benzaldehyde gives (R)-benzoin in a benzaldehyde lyase (BAL)-catalyzed enantioselective C-C bond-forming reaction in NADES/buffer mixtures in the presence of thiamine diphosphate (ThDP) and Mg2+ ions. The enzyme at hand is a thiamine diphosphate-dependent lyase (ThDP-lyase). The NADES/buffer mixtures were selected according to their composition and proportion. Three different NADES mixtures were studied, ChCl/glycerol, ChCl/urea, and ChCl/xylitol, and the best was ChCl/glycerol (1:2). Due to the high viscosity of NADESs coupled with the stability/activity of the enzyme, a certain quantity of buffer is required in the reaction; the best ratio found was 60:40 v/v. The reaction proceeds with 95% conversion and offers the product in an outstanding enantiopurity of >99% (R-isomer) [32].

Scheme 7. BAL-catalyzed enantioselective C-C bond formations in DES/buffer mixtures [32].
The preparation of chiral bioactive compounds, in many instances, requires enantiopure precursors, consequently implying that stereoselective reactions that can be catalyzed by biocatalysts in unconventional solvents, such as NADES solvents. As already mentioned, these solvents are very viscous and sometimes compromise the activity of the biocatalyst and, consequently, the productivity and enantioselectivity of the reaction. However, NADESs are increasingly being used in biocatalytic reactions, even though in smaller quantities than the buffer co-solvents. Further examples of the great potential of NADESs for biocatalysis are shown in Scheme 8. All of these examples are bioreductions of acetophenone derivatives, which have provided key enantiopure intermediates to important bioactive compounds, such as AD101 (SCH-350581) [33][34], Ticagrelor [35] and Aprepitant [36][37]. Xia et al. reported the preparation of (R)-1-[4-(trifluoromethyl)phenyl]ethanol through the bioreduction of 4-(trifluoromethyl)acetophenone biocatalyzed by recombinant E. coli LXCAR-S154Y and Geotrichum ZJPH1810, using 3% NADES/DES as co-solvents in a buffer (Scheme 8). The Betaine (B)/Lysine (Lys) (NADES) was chosen from the four DESs and 13 NADESs examined in this reaction, providing the desired product in up to 92.4% yield and up to 99% ee (preparative scale) at a substrate concentration of 400 mM [34]. He et al. described a stereoselective bioreduction of 2-chloro-1-(3,4-difluorophenyl)ethanone to (S)-2-chloro-1-(3,4-difluorophenyl)ethanol catalyzed by recombinant Escherichia coli cells in reaction media containing 1% choline acetate (ChAc)/Lys with a buffer (Scheme 8). This ChAc/Lys mixture was selected from several NADESs due to enhanced coenzyme regeneration and improved cell membrane permeability during bioreduction, which explains the high conversion. The result was excellent, and the enzymatic reduction of the keto-molecule precursor made it possible to obtain the (S)-chlorohydrin with a yield of 87% and enantioselectivity of 99% ee with 1 M of substrate. Furthermore, the substrate loading in the established ChAc/Lys buffer mixture was increased 3.3-fold compared to the aqueous medium [35]. Another example reported by Zhu et al. was the enantioselective synthesis of (S)-1-[3,5-bis(trifluoromethyl)phenyl]ethanol in 86% yield and 99% ee through the bioreduction of the corresponding carbonyl group promoted by whole cells of Candida tropicalis 104 in 1% w/v NADES (mixture of ChCl/Trehalose (Tre) (1:1)) in a buffer (Scheme 8). The use of NADES as a reaction medium has been shown to be advantageous, increasing yield, cell viability, and cell membrane permeability to the substrate and, at the same time, protecting the cell [37].
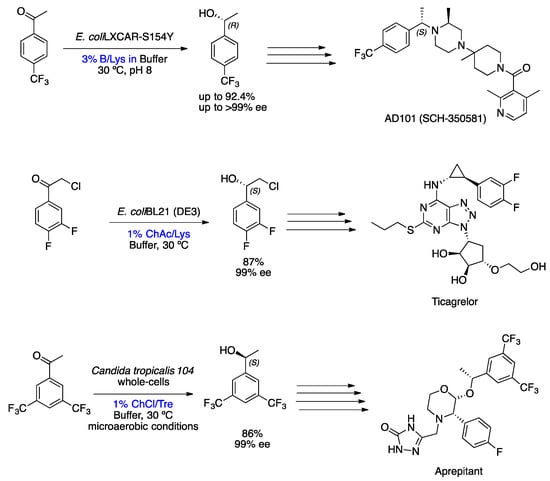
5. Metal-Based Asymmetric Catalysis (MBAC) in DES
Metal-based asymmetric catalysis is very important for the preparation of bioactive chiral compounds in the pharmaceutical industry, but the toxicity of heavy metals and the volatile organic solvents jeopardizes its sustainability [38]. The use of DESs in asymmetric metal-based catalysis is the way to make these reactions more sustainable since both the solvent and the metal catalyst can be reused several times. So far, DESs have shown their versatility in coupling reactions based on transition metals (Suzuki, Hiyama, Heck, and Sonogashira) and aminocarbonylations or Pd-catalyzed C-S bond formation reactions, among others. In most of these reactions, the DESs performed well, in that they gave very good-to-excellent yields, and it was possible to reuse the solvents and the catalysts several times, while maintaining the activity [38][39]. Furthermore, these reactions are part of the synthetic pathway for various bioactive compounds [40][41]. Despite its extraordinary performance in metal-based catalysis reactions, its application in asymmetric catalytic synthesis based on metals is rare. So far, only a few examples have been reported in the literature, and the metals have been used in stoichiometric quantities rather than catalytic. Peñaranda-Gómez et al. reported a sustainable and diastereoselective method for the preparation of tetrahydroquinoline derivatives from star anise oil, using a mixture of choline chloride (ChCl)/zinc chloride as the DES (Scheme 9) [42]. This diastereoselective synthesis is a multicomponent inverse electron-demand aza-Diels−Alder reaction, carried out with anise oil (the main component is trans-anethole), anilines, and aromatic aldehydes in ChCl/ZnCl2 mixtures (3:2) at 110 °C for 3 h, providing a total of 22 tetrahydroquinoline derivatives in low-to-excellent yields with high diastereoselectivity [42]. The diastereoisomers formed have the cis-(2e,4e) configuration, which was determined by NMR analysis. In addition, DESs can be recycled for three cycles and still maintain their activity. The DESs in this reaction act as a solvent and catalyst and are crucial to the reaction. It should be emphasized that tetrahydroquinoline derivatives are value-added compounds in the field of medicinal chemistry. Differential scanning calorimetry (DSC) determined the best temperature and composition of DESs for this multicomponent reaction.
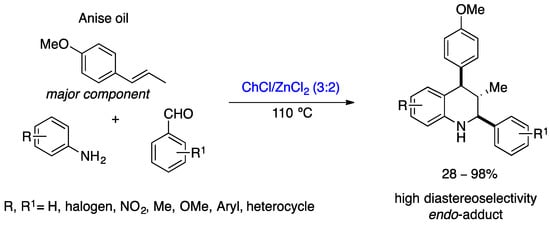
Scheme 9. Diastereoselective synthesis of tetrahydroquinoline derivatives in DES described by Peñaranda-Gómez et al. [42].
Recently, Sanabria-Sánchez et al. developed a multicomponent, diastereoselective, one-pot Povarov reaction in DESs for the preparation of a wide variety of dihydroisoindolo[2,1-a]quinolin-11-ones derivatives. Similar to the methodology described by Peñaranda-Gómez et al. [42], the DESs act as both the solvent and catalyst. Various DESs consisting of Lewis or Brønsted acids were studied, such as ChCl/ZnCl2, Urea/ZnCl2, ChCl/MgCl2, ChCl/SnCl2, ChCl/APTS, ChCl/CF3SO3H, and ZnCl2/ethylene glycol and reaction conditions to choose the best system, ChCl/ZnCl2 (1:2) at 110 °C for 4.5 h (Scheme 10) [43]. Twenty dihydroisoindolo[2,1-a]quinoline-11-one derivatives were prepared with yields of up to 87% and high diastereoselectivities by reacting a wide variety of anilines, alkenes (trans-anethole, methyl eugenol, and indene), and 2-formylbenzoic acid in ChCl/ZnCl2 (1:2) at 110 °C for 4.5 h. 1H NMR analysis of the compounds confirmed the production of a single diastereoisomer, the 6,6a-trans isomer. In addition, the DES mixture could be reused for six cycles without loss of activity.
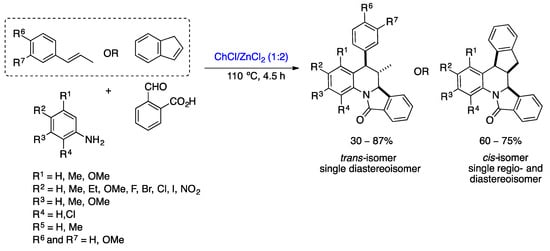
Scheme 10. Synthesis of dihydroisoindolo[2,1-a]quinolin-11-one derivatives via the Povarov reaction in DESs [43].
The development of more sustainable methods for the preparation of homoallylic alcohols is of great interest to the pharmaceutical industry. González-Gallardo et al. reported the allylation of various carbonyl compounds via indium organometallic and allyl chloride derivatives in DESs. Several DESs were studied for this reaction, such as choline chloride (ChCl)/ethylene glycol (1:2), acetylcholine chloride (AcChCl)/acetamide (1:2), ChCl/glycerol (1:2), ChCl/urea (1:2), MePPh3Br/glycerol (1:2), ChCl/L-malic acid (1:1), and ChCl/D-galactose (5:2). To the reactions carried out with the DESs ChCl/ethylene glycol (1:2) and AcChCl/acetamide (1:2), ammonium salts (NH4Cl or NH4OAc) were added to stabilize the species of In, furnishing the desired product with excellent yields. Once both of the DESs gave excellent yields, the AcChCl/acetamide mixture was selected as a solvent due to its lower toxicity [44]. Twenty homoallyl alcohol derivatives were prepared in moderate-to-excellent yields through the Barbier allylation of a wide range of carbonyl derivatives and chloroallyl substrates mediated by indium (stabilized by NH4Cl) in DESs at 25 °C for 12 h. However, only two of them showed diastereoselectivity, those prepared from γ-substituted allyl chloride derivatives (crotyl chloride and cinnamyl chloride). In both cases, the anti-adduct was the sole or the major diastereoisomer formed according to Scheme 11. The recyclability of DESs and NH4Cl was studied over six cycles, but performance was only maintained over four cycles. In any case, it can be emphasized that DESs perform well in organometallic reactions, making these reactions more sustainable due to their lower toxicity and good recyclability.
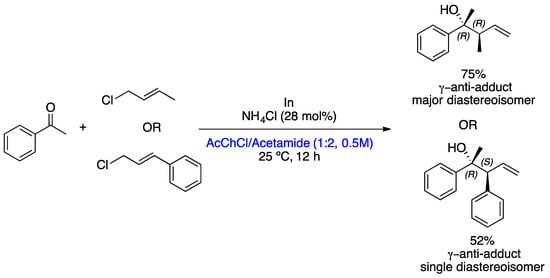
Scheme 11. Barbier allylation of acetophenone mediated by In in DESs [44].
Another promising approach is the chemoenzymatic cascade, which combines transition metal catalysis and biocatalysis for the asymmetric synthesis of bioactive molecules [45]. Basically, metal/enzymatic cascades and sequential transformations consist primarily of the role of metal species (Pd, Ru, Au, Ir, and Fe) in catalyzing various organic reactions (C-C couplings, isomerizations, and hydrations) and their combination with stereospecific enzymes, including alcohol dehydrogenases, alcohol oxidases, aldolases, amine dehydrogenases, and amino acid dehydrogenases, among others. The use of DESs in metal/enzymatic cascades and sequential transformations is rare, but the few results that have been reported are very promising for the pharmaceutical industry.
The first successful example of the integration of metal and biological catalysis in DESs/buffer mixtures was reported in 2018 [46]. This chemoenzymatic cascade consists of the ruthenium-catalyzed isomerization reaction of allylic alcohols to an α,β-saturated ketone, followed by enantioselective enzymatic reduction promoted by ketoreductases (KREDs), providing enantiopure alcohols (Scheme 12). This chemoenzymatic cascade was carried out using two different approaches, a sequential reaction (one-pot in two steps) and a simultaneous (concurrent) reaction (one-pot in one step). The choice of solvent mixture, based on DESs and buffers, was made carefully, making it possible for both reactions to take place without inhibiting the metal catalyst or the enzyme. The best mixture of DESs was ChCl/glycerol (1:2) or ChCl/sorbitol (1:1) for enantioselective bioreduction; however, for the chemoenzymatic reactions, the first mixture of DESs was chosen due to the high viscosity of the sorbitol-based DES, even at 40 °C. In the case of the sequential approach (one-pot in two stages), the first stage involves the Ru-catalyzed isomerization of racemic allylic alcohols in DESs buffer, i-PrOH at 50 °C, after consumption of the starting material; for the second stage, the temperature was reduced to 30 °C and KRED and its cofactor (NADP+), obtaining the desired chiral alcohols in good-to-excellent yields and high enantioselectivities. With regard to the second approach, the simultaneous reaction (one pot in one step), both catalysts were added at the start of the reaction; for this reason, the enzyme showed poor stability in the aqueous buffer, resulting in incomplete reactions, providing the desired product mixed with unreacted ketone, and in some cases, allyl alcohol was also obtained, indicating slow isomerization. However, five chiral saturated alcohols were prepared in low-to-excellent yields and with excellent enantioselectivities via the chemoenzymatic reaction of allylic alcohols catalyzed by KRED and Ru complex (10% mol) at 40 °C in ChCl/Gly (1:2)/buffer (4:1) (Scheme 12).
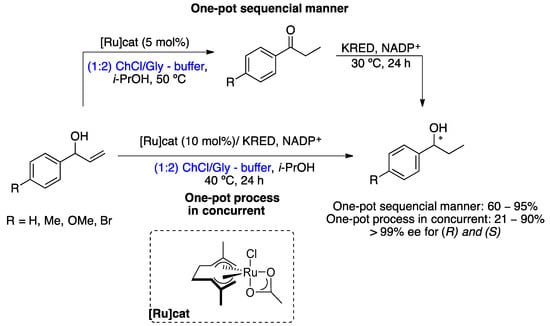
Scheme 12. Chemoenzymatic cascade for the preparation of enantiopure saturated alcohols from racemic allylic alcohols in DES/buffer mixtures [46].
In 2018, Paris et al. described the first application of DESs as a solvent in Suzuki coupling, followed by bioreduction for the preparation of enantiopure biaryl alcohols in very good yields (up to 86%) and excellent ee (>99%) (Scheme 13) [47]. The two-step method consists of the palladium-catalyzed Suzuki coupling of 4′-haloacetophenones with arylboronic acids at 70–100 °C in a choline chloride/glycerol (Gly) buffer mixture, followed by the enzymatic reduction of the ketone formed catalyzed by alcohol dehydrogenases at 30 °C. Separately, both the palladium-catalyzed coupling reactions and the bioreductions were previously carried out in mixtures of DES and DES/buffer. Subsequently, the reaction conditions were optimized with regard to the choice of components and the proportion of DES mixture: the proportion of DES and buffer and also the substrate concentration. Using these ChCl/Gly (1:2)/buffer (4:1) conditions at 100 °C for the Suzuki coupling step for a 200 mM substrate, it was possible to prepare 10 biaryl ketone derivatives in high yields. In addition, the reaction conditions for enzymatic reductions were optimized, requiring dilution to 75 mM with a buffer containing i-PrOH, ketoreductases (KREDs), and cofactor (NADP+) to establish an efficient bioreduction of the transiently formed ketone. Using these conditions, it was possible to reduce a wide variety of substrates. Furthermore, using the stereocomplementary ADH enzymes of Lactobacillus kefir and Rhodococcus ruber, it is possible to obtain enantiopure (R)- and (S)-biaryl alcohols.
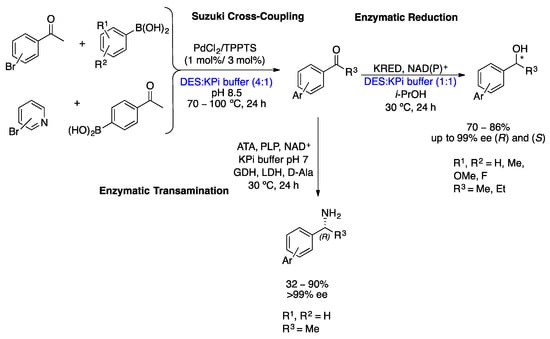
Later, the same groups of Gröger and González-Sabín reported the preparation of enantiopure biaryl amines through a chemoenzymatic cascade in DESs/buffer mixture, which consists of a palladium-catalyzed cross-coupling reaction followed by enantioselective enzymatic amination (Scheme 13) [48]. As described above, the first step was the optimization of the reaction conditions by choosing the most suitable solvent for both reactions to take place in excellent yields. The best DES (co-solvent) for this chemoenzymatic cascade was (1:2) ChCl/Gly. In addition, biocatalysis using amine transaminases (ATAs) in DESs has not been reposted before. The reactions were carried out according to the schematic representation (Scheme 13), the Pd-catalyzed step was carried out at 200 mM substrate loading in a DES/buffer mixture (4:1), and the biocatalysis step was carried out at 25 mM before dilution to a buffer containing 10% w/w DES due to enzyme stability problems. Five (R)-biaryl amines were prepared using this methodology in low to very good yields with excellent enantioselectivities (>99% ee) [48].
The preparation of chiral compounds through asymmetric catalytic syntheses carried out in DESs has not been widely explored so far, perhaps because, in general, chiral metal complexes (consisting of a chiral ligand coordinated to a metal) are probably very sensitive to the aqueous medium and end up decomposing; another aspect is that most DESs contain other components that coordinate with the metal, giving rise to racemic products. An alternative is to resort to the chemoenzymatic cascade or the use of organocatalysts for the preparation of chiral compounds, since both approaches have shown very promising results using DESs as reaction media.
References
- Gal, J. Chiral Drugs from a Historical Point of View in Chirality in Drug Research; Francotte, E., Lindner, W., Eds.; Wiley-VCH: Weinheim, Germany, 2006; pp. 3–26.
- Federsel, H.-J. Stereoselective Synthesis of Drugs–An Industrial Perspective in Chirality in Drug Research; Francotte, E., Lindner, W., Eds.; Wiley-VCH: Weinheim, Germany, 2006; pp. 29–65.
- Burke, A.J.; Marques, C.S.; Turner, N.J.; Hermann, G.J. Active Pharmaceutical Ingredients in Synthesis–Catalytic Processes in Research and Development; Wiley-VCH: Weinheim, Germany, 2018.
- Blaser, H.-U.; Pugìn, B.; Studer, M. Enantioselective Heterogenous Catalysis: Academic and Industrial Challenges in Chiral Catalyst Immobilization and Recycling; De Vos, D.E., Vankelecom, I.F.J., Jacobs, P.A., Eds.; Wiley-VCH: Weinheim, Germany, 2000; pp. 1–42.
- Wasserscheid, P.; Welton, T. (Eds.) Ionic Liquids in Synthesis; Wiley-VCH: Weinheim, Germany, 2003.
- Horváth, I.T.; Rábai, J. Facil catalyst separation without water: Fluorous biphase hydroformylation of olefins. Science 1994, 266, 72–75.
- Abbott, A.P.; Capper, G.; Davies, D.L.; Munro, H.L.; Rasheed, R.K.; Tambyrajah, V. Preparation of novel, moisture-stable, Lewis-acidic ionic liquids containing quaternary ammonium salts with functional side chains. Chem. Commun. 2001, 19, 2010–2011.
- Alonso, D.A.; Burlingham, S.-J.; Chinchilla, R.; Guillena, G.; Ramón, D.J.; Tiecco, M. Asymmetric Organocatalysis in Deep Eutectic Solvents. Eur. J. Org. Chem. 2021, 29, 4065–4071.
- Fanjul-Mosteirín, N.; Amo, V. Organocatalytic transformations in deep eutectic solvents: Green methodologies made greener. Tetrahedron 2021, 84, 131967.
- Guajardo, N.; Müller, C.R.; Schrebler, R.; Carlesi, C.; Domínguez de Maria, P. Deep eutectic solventes for organocatalysis, biotransformations, and multistep organocatalyst/enzyme combinations. ChemCatChem 2016, 8, 1020–1027.
- Hansen, B.B.; Spittle, S.; Chen, B.; Poe, D.; Zhang, Y.; Klein, J.M.; Horton, A.; Adhikari, L.; Zelovich, T.; Doherty, B.W.; et al. Deep Eutectic Solvents: A Review of Fundamentals and Applications. Chem. Rev. 2020, 3, 1232–1285.
- Paiva, A.; Craveiro, R.; Aroso, I.; Martins, M.; Reis, R.L.; Duarte, A.R.C. Natural Deep Eutectic Solvents−Solvents for the 21st Century. ACS Sustain. Chem. Eng. 2014, 5, 1063–1071.
- Burke, A.J. Update on Asymmetric Organocatalytic Approaches for Accessing Drugs. Expert Opin. Drug Discov. 2023, 18, 37–46.
- Martínez, R.; Berbegal, L.; Guillena, G.; Ramón, D.J. Bio-renewable enantioselective aldol reaction in natural deep eutectic solvents. Green Chem. 2016, 18, 1724–1730.
- Flores-Ferràndiz, J.; Chinchilla, R. Organocatalytic enantioselective conjugate addition of aldehydes to maleimides in deep eutectic solvents. Tetrahedron Asymmetry 2017, 28, 302–306.
- Ñíguez, D.R.; Khazaeli, P.; Alonso, D.A.; Guillen, G. Deep Eutectic Mixtures as Reaction Media for the Enantioselective Organocatalyzed α-Amination of 1,3-Dicarbonyl Compounds. Catalysts 2018, 8, 217.
- Amorim, A.C.; Fonseca, D.P.; Carreiro, E.P.; Hermann, G.J.; Federsel, H.-J.; Burke, A.J. Immobilization of Functionalized epi-Cinchonine Organocatalysts on Controlled Porous Glass-Beads: Applications in Batch and Continuous Flow. Synlett 2022, 33, 1756–1762.
- Amorim, A.C.; Fonseca, D.P.; Carreiro, E.P.; Ramalho, J.P.; Duarte, A.R.; Hermann, G.J.; Federsel, H.J.; Burke, A.J. Sustainable OrganoCatalyzed Enantioselective Catalytic Michael Additions in Betaine derived Deep Eutectic Solvents. SynOpen 2023, 7, 374–380.
- Tiecco, M.; Alonso, D.A.; Ñíguez, D.R.; Ciancaleoni, G.; Guillen, G.; Ramón, D.J.; Bonillo, A.A.; Germani, R. L-Proline-based deep eutectic solvents based on their structural features. J. Mol. Liq. 2020, 313, 113573.
- Tang, J.; Yao, S. Discovery of Deep Eutectic Solvents in Organic Synthesis: An insight into their current applications and prospect. Curr. Org. Chem. 2022, 26, 2230–2234.
- Saavedra, B.; Meli, A.; Rizzo, C.; Ramón, D.J.; D’Anna, F. Natural eutectogels: Sustainable catalytic systems for C-C bond formation reactions. Green Chem. 2021, 23, 6555–6565.
- Zaks, A.; Klibanov, A.M. Enzyme-catalyzed processes in organic solvents. Proc. Natl. Acad. Sci. USA 1985, 82, 3192–3196.
- Klibanov, A.M. Improving enzymes by using them in organic solvents. Nature 2001, 409, 241–246.
- Pätzold, M.; Siebenhaller, S.; Kara, S.; Liese, A.; Syldatk, C.; Holtmann, D. Deep Eutectic Solvents as Efficient Solvents in Biocatalysis. Trends Biotechnol. 2019, 37, 943–959.
- Gorke, J.T.; Srienc, F.; Kazlauskas, R.J. Hydrolase-catalyzed biotransformations in deep eutectic solvents. Chem. Commun. 2008, 10, 1235–1237.
- Pöhnlein, M.; Ulrich, J.; Kirschhöfer, F.; Nusser, M.; Muhle-Goll, C.; Kannengiesser, B.; Brenner-Weiß, G.; Luy, B.; Liese, A.; Syldatk, C.; et al. Lipase-catalyzed synthesis of glucose-6-O-hexanoate in deep eutectic solvents. Eur. J. Lipid Sci. Technol. 2015, 117, 161–166.
- Hollenbach, R.; Bindereif, B.; Van der Schaaf, U.S.; Ochsenreither, K.; Syldatk, C. Optimization of glycolipid synthesis in hydrophilic deep eutectic solvents. Front. Bioeng. Biotechnol. 2020, 8, 382.
- Hollenbach, R.; Ochsenreither, K.; Syldatk, C. Enzymatic synthesis of glucose monodecanoate in a hydrophobic deep eutectic solvent. Int. J. Mol. Sci. 2020, 21, 4342.
- Semproli, R.; Chanquia, S.N.; Bittner, J.P.; Müller, S.; Domínguez de María, P.; Kara, S.; Ubiali, D. Deep Eutectic Solvents for the Enzymatic Synthesis of Sugar Esters: A Generalizable Strategy? ACS Sustain. Chem. Eng. 2023, 11, 5926–5936.
- Milker, S.; Pätzold, M.; Bloh, J.Z.; Holtmann, D. Comparison of deep eutectic solvents and solvent-free reaction conditions for aldol production. Mol. Catal. 2019, 466, 70–74.
- Tian, X.; Zhang, S.; Zheng, L. Enzyme-Catalyzed Henry Reaction in Choline Chloride-Based Deep Eutectic Solvents. J. Microbiol. Biotechnol. 2016, 26, 80–88.
- Maugeri, Z.; Domínguez de María, P. Benzaldehyde lyase (BAL)-catalyzed enantioselective C-C bond formation in deep-eutectic-solvents–buffer mixtures. J. Mol. Catal. B Enzym. 2017, 107, 120–123.
- Tsamis, F.; Gavrilov, S.; Kajumo, F.; Seibert, C.; Kuhmann, S.; Ketas, T.; Trkola, A.; Palani, A.; Clader, J.W.; Tagat, J.R.; et al. Analysis of the mechanism by which the small-Molecule CCR5 antagonists SCH-351125 and SCH-350581 inhibit human immunodeficiency virus type 1 entry. J. Virol. 2003, 77, 5201–5208.
- Xia, N.; Xiong, L.; Bi, S.; Qian, F.; Wang, P. Development of biocompatible DES/NADES as co-solvents for efficient biosynthesis of chiral alcohols. Bioprocess Biosyst. Eng. 2020, 43, 1987–1997.
- He, Y.; Huang, Q.; Wang, P. Design and evaluation of novel bio-based deep eutectic solvents for highly efficient bioproduction of chiral aryl alcohol. J. Chem. Technol. Biotechnol. 2020, 95, 1980.
- Zhang, Z.H.; Yang, Y.P.; Lu, P.; Li, X.Q.; Chang, J.H.; Zheng, R.S.; Zhou, L.; Chen, S.S.; Chen, X.P.; Ren, B.Y.; et al. Fosaprepitant versus aprepitant in the prevention of chemotherapy-induced nausea and vomiting in patients receiving cisplatin-based chemotherapy: A multicenter, randomized, double-blind, double-simulated, positive-controlled phase III trial. Ann. Transl. Med. 2020, 8, 234.
- Zhu, Z.; Bi, S.; Ye, N.; Wang, P. High-Efficient Production of (S)-1-ethanol via Whole-Cell Catalyst in Deep-Eutectic Solvent-Containing Micro-Aerobic Medium System. Molecules 2020, 25, 1855.
- Farina, V.; Reeves, J.T.; Senanayake, C.H.; Song, J.J. Asymmetric Synthesis of Active Pharmaceutical Ingredients. Chem. Rev. 2006, 106, 2734–2793.
- Marset, X.; Guillena, G. Deep Eutectic Solvents as à-la-Carte Medium for Transition-Metal-Catalyzed Organic Processes. Molecules 2022, 27, 8445.
- Peng, L.; Hu, Z.; Lu, Q.; Tang, Z.; Jiao, Y.; Xu, X. DESs: Green solvents for transition metal catalyzed organic reactions. Chin. Chem. Lett. 2019, 30, 2151–2156.
- Cicco, L.; Dilauro, G.; Perna, F.M.; Vitale, P.; Capriati, V. Advances in deep eutectic solvents and water: Applications in metal- and biocatalyzed processes, in the synthesis of APIs, and other biologically active compounds. Org. Biomol. Chem. 2021, 19, 2558–2577.
- Peñaranda-Gómez, A.; Rodríguez Bejarano, O.; Kouznetsov, V.V.; Ochoa-Puentes, C. One-Pot Diastereoselective Synthesis of Tetrahydroquinolines from Star Anise Oil in a Choline Chloride/Zinc Chloride Eutectic Mixture. ACS Sustain. Chem. Eng. 2019, 7, 18630–18639.
- Sanabria-Sánchez, C.M.; Kouznetsov, V.V.; Ochoa-Puentes, C. Diastereoselective multicomponent synthesis of dihydroisoindolo quinolin-11-ones mediated by eutectic solvents. RSC Adv. 2023, 13, 26189–26195.
- González-Gallardo, N.; Saavedra, B.; Guillena, G.; Ramón, D.J. Indium-mediated allylation of carbonyl compounds in deep eutectic solvents. Appl. Organomet. Chem. 2021, 35, e6418.
- González-Granda, S.; Escot, L.; Lavandera, I.; Gotor-Fernández, V. Chemoenzymatic Cascades Combining Biocatalysis and Transition Metal Catalysis for Asymmetric Synthesis. Angew. Chem. Int. Ed. 2023, 62, e202217713.
- Cicco, L.; Ríos-Lombardía, N.; Rodríguez-Álvarez, M.J.; Moris, F.; Perna, F.M.; Capriati, V.; García-Álvarez, J.; González-Sabín, J. Programming Cascade Reactions Interfacing Biocatalysis with Transition-Metal Catalysis in Deep Eutectic Solvents as Biorenewable Reaction Media. Green Chem. 2018, 20, 3468–3475.
- Paris, J.; Ríos-Lombardía, N.; Morís, F.; Gröger, H.; González-Sabín, J. Novel insights into the combination of metal- and biocatalysis: Cascade one-pot synthesis of enantiomerically pure biaryl alcohols in deep eutectic solvents. ChemCatChem 2018, 10, 4417–4423.
- Paris, J.; Telzerow, A.; Ríos-Lombardía, N.; Steiner, K.; Schwab, H.; Morís, F.; Gröger, H.; González-Sabín, J. Enantioselective One-Pot Synthesis of Biaryl-Substituted Amines by Combining Palladium and Enzyme Catalysis in Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2019, 7, 5486–5493.
More
Information
Subjects:
Chemistry, Organic
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
513
Revisions:
2 times
(View History)
Update Date:
28 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





