
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Giuseppe Brisinda | -- | 3218 | 2024-02-27 07:10:21 | | | |
| 2 | Lindsay Dong | Meta information modification | 3218 | 2024-02-28 03:40:18 | | |
Video Upload Options
Enteric fistulas are a common problem in gastrointestinal tract surgery and remain associated with significant mortality rates, due to complications such as sepsis, malnutrition, and electrolyte imbalance. The increasingly widespread use of open abdomen techniques for the initial treatment of abdominal sepsis and trauma has led to the observation of so-called entero-atmospheric fistulas. Because of their clinical complexity, the proper management of enteric fistula requires a multidisciplinary team. The main goal of the treatment is the closure of enteric fistula, but also mortality reduction and improvement of patients’ quality of life are fundamental. Successful management of patients with enteric fistula requires the establishment of controlled drainage, management of sepsis, prevention of fluid and electrolyte depletion, protection of the skin, and provision of adequate nutrition.
1. Introduction
2. Etiology of ECF
3. EAF and OA
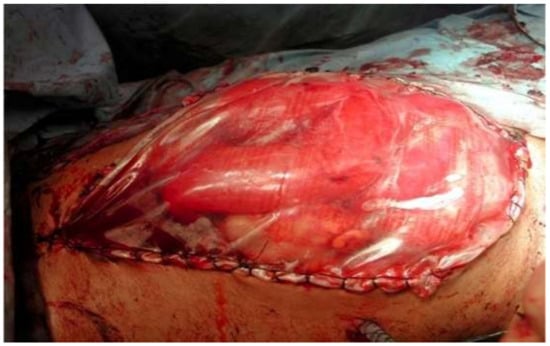
4. Classification and Evolution
5. Physiopathology
6. Outcomes
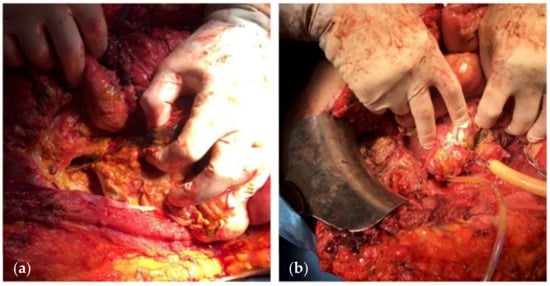
7. Treatment
- (a)
-
Recognition and management of sepsis: organ dysfunction or progressive organ failure should be promptly managed;
- (b)
-
Source control is crucial for the resolution of sepsis;
- (c)
-
Antibiotic regimen based on culture results;
- (d)
-
Reducing fistula output: nil per os, a nasogastric tube as well as an attempt to reduce secretions of the gastrointestinal tract by administering proton pump inhibitors and to reduce enteric and pancreatic secretions using somatostatin or octreotide [79];
- (e)
-
Nutritional support: In the presence of OA, the patient is in a hypercatabolic state. Hypercatabolism is further worsened by the presence of an EAF. The main parameters on which nutritional support should be based are the following: (a) increased caloric requirements, usually calculated by 30–35 kcal/kg/day; (b) increased protein depriving, calculated by adding 1.5 g protein/kg/day and 2 g protein losses for each liter of fluid collected from the raw surface of the OA; and (c) deficiencies of vitamins and trace elements. Adequate nutritional support based on the patient’s nutritional status, a positive nitrogen balance, adequate trace minerals, and vitamin replacement, along with glycemic control, may allow the surgeon to proceed to surgical treatment of the fistula. Additionally, several known parameters, such as weight, prealbumin, albumin, and transferrin, are correlated with postoperative mortality and morbidity and spontaneous fistula closure rates [65][80][81].
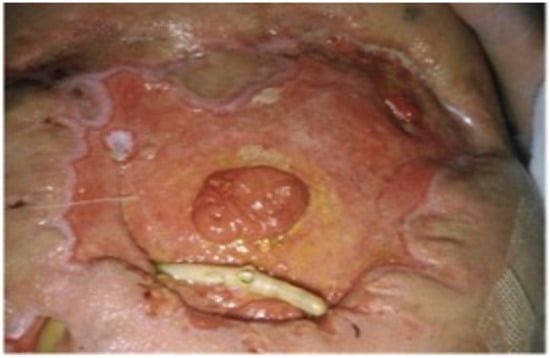
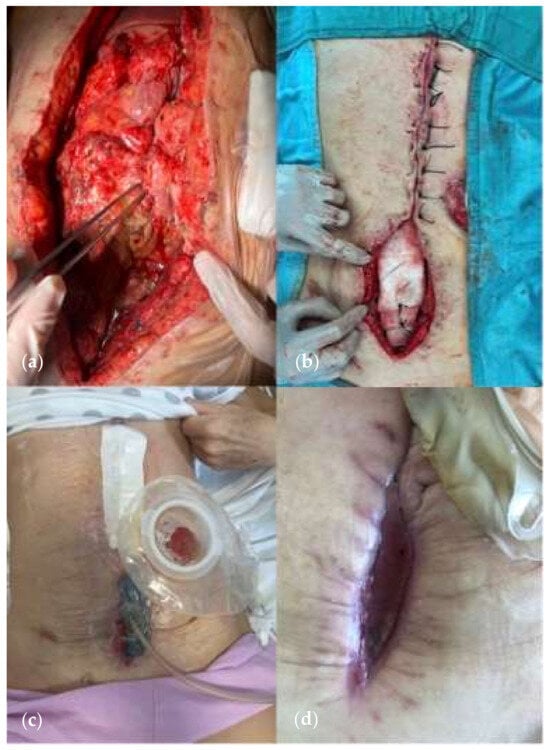
8. NPWT Assisted Closure
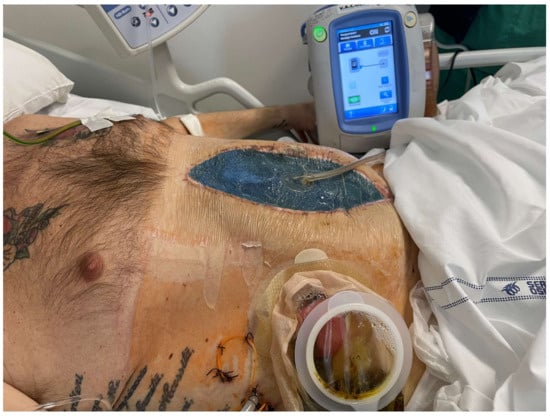
9. Conclusions
References
- Pepe, G.; Magalini, S.; Callari, C.; Persiani, R.; Lodoli, C.; Gui, D. Vacuum Assisted Closure (VAC) therapyTM as a swiss knife multi-tool for enteric fistula closure: Tips and tricks: A pilot study. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 2527–2532.
- Di Saverio, S.; Tarasconi, A.; Inaba, K.; Navsaria, P.; Coccolini, F.; Costa Navarro, D.; Mandrioli, M.; Vassiliu, P.; Jovine, E.; Catena, F.; et al. Open abdomen with concomitant enteroatmospheric fistula: Attempt to rationalize the approach to a surgical nightmare and proposal of a clinical algorithm. J. Am. Coll. Surg. 2015, 220, e23–e33.
- Campos, A.C.; Andrade, D.F.; Campos, G.M.; Matias, J.E.; Coelho, J.C. A multivariate model to determine prognostic factors in gastrointestinal fistulas. J. Am. Coll. Surg. 1999, 188, 483–490.
- Galie, K.L.; Whitlow, C.B. Postoperative enterocutaneous fistula: When to reoperate and how to succeed. Clin. Colon. Rectal Surg. 2006, 19, 237–246.
- Fischer, P.E.; Fabian, T.C.; Magnotti, L.J.; Schroeppel, T.J.; Bee, T.K.; Maish, G.O., 3rd; Savage, S.A.; Laing, A.E.; Barker, A.B.; Croce, M.A. A ten-year review of enterocutaneous fistulas after laparotomy for trauma. J. Trauma. 2009, 67, 924–928.
- Bruhin, A.; Ferreira, F.; Chariker, M.; Smith, J.; Runkel, N. Systematic review and evidence based recommendations for the use of negative pressure wound therapy in the open abdomen. Int. J. Surg. 2014, 12, 1105–1114.
- Brisinda, G.; Chiarello, M.M.; Crocco, A.; Adams, N.J.; Fransvea, P.; Vanella, S. Postoperative mortality and morbidity after D2 lymphadenectomy for gastric cancer: A retrospective cohort study. World J. Gastroenterol. 2022, 28, 381–398.
- Navsaria, P.; Nicol, A.; Hudson, D.; Cockwill, J.; Smith, J. Negative pressure wound therapy management of the “open abdomen” following trauma: A prospective study and systematic review. World J. Emerg. Surg. 2013, 8, 4.
- Fitzgerald, C.A.; Broecker, J.; Park, C.; Dumas, R.P. Primary Repair Versus Resection for AAST Grade I and II Colon Injuries: Does the Type of Repair Matter? J. Surg. Res. 2023, 295, 370–375.
- Vengail, S.; Chandrakar, D.; Naik, A.K.; Nayak, A.K.; Mahajan, A.; Dutta, P. Assessment of Risk Factors for Enteric Fistula and Intra-Abdominal Sepsis in Patients with Open Abdomen in Trauma: An Original Research. J. Pharm. Bioallied Sci. 2023, 15, S273–S276.
- Fico, V.; Altieri, G.; Di Grezia, M.; Bianchi, V.; Chiarello, M.M.; Pepe, G.; Tropeano, G.; Brisinda, G. Surgical complications of oncological treatments: A narrative review. World J. Gastrointest. Surg. 2023, 15, 1056–1067.
- Roberts, D.J.; Leppaniemi, A.; Tolonen, M.; Mentula, P.; Bjorck, M.; Kirkpatrick, A.W.; Sugrue, M.; Pereira, B.M.; Petersson, U.; Coccolini, F.; et al. The open abdomen in trauma, acute care, and vascular and endovascular surgery: Comprehensive, expert, narrative review. BJS Open 2023, 7, zrad084.
- Dubose, J.J.; Lundy, J.B. Enterocutaneous fistulas in the setting of trauma and critical illness. Clin. Colon. Rectal Surg. 2010, 23, 182–189.
- Whelan, J.F., Jr.; Ivatury, R.R. Enterocutaneous fistulas: An overview. Eur. J. Trauma. Emerg. Surg. 2011, 37, 251–258.
- Latifi, R.; Joseph, B.; Kulvatunyou, N.; Wynne, J.L.; O’Keeffe, T.; Tang, A.; Friese, R.; Rhee, P.M. Enterocutaneous fistulas and a hostile abdomen: Reoperative surgical approaches. World J. Surg. 2012, 36, 516–523.
- Polk, T.M.; Schwab, C.W. Metabolic and nutritional support of the enterocutaneous fistula patient: A three-phase approach. World J. Surg. 2012, 36, 524–533.
- Bradley, M.J.; Dubose, J.J.; Scalea, T.M.; Holcomb, J.B.; Shrestha, B.; Okoye, O.; Inaba, K.; Bee, T.K.; Fabian, T.C.; Whelan, J.F.; et al. Independent predictors of enteric fistula and abdominal sepsis after damage control laparotomy: Results from the prospective AAST Open Abdomen registry. JAMA Surg. 2013, 148, 947–954.
- Wainstein, D.E.; Sisco, P.; Deforel, M.L.; Irigoyen, M.; Devoto, J.; Zarate, J.M. Systematic and Specific Treatment of Patients with Enteroatmospheric Fistulas: From Initial Conservative Treatment to Definitive Surgery. Surg. Technol. Int. 2016, 28, 73–81.
- Gribovskaja-Rupp, I.; Melton, G.B. Enterocutaneous Fistula: Proven Strategies and Updates. Clin. Colon. Rectal Surg. 2016, 29, 130–137.
- Haffejee, A.A. Surgical management of high output enterocutaneous fistulae: A 24-year experience. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 309–316.
- Alvarez, C.; McFadden, D.W.; Reber, H.A. Complicated enterocutaneous fistulas: Failure of octreotide to improve healing. World J. Surg. 2000, 24, 533–537; discussion 538.
- Erdmann, D.; Drye, C.; Heller, L.; Wong, M.S.; Levin, S.L. Abdominal wall defect and enterocutaneous fistula treatment with the Vacuum-Assisted Closure (V.A.C.) system. Plast. Reconstr. Surg. 2001, 108, 2066–2068.
- Gunn, L.A.; Follmar, K.E.; Wong, M.S.; Lettieri, S.C.; Levin, L.S.; Erdmann, D. Management of enterocutaneous fistulas using negative-pressure dressings. Ann. Plast. Surg. 2006, 57, 621–625.
- Davis, K.G.; Johnson, E.K. Controversies in the care of the enterocutaneous fistula. Surg. Clin. N. Am. 2013, 93, 231–250.
- Sartelli, M.; Abu-Zidan, F.M.; Ansaloni, L.; Bala, M.; Beltran, M.A.; Biffl, W.L.; Catena, F.; Chiara, O.; Coccolini, F.; Coimbra, R.; et al. The role of the open abdomen procedure in managing severe abdominal sepsis: WSES position paper. World J. Emerg. Surg. 2015, 10, 35.
- Coccolini, F.; Biffl, W.; Catena, F.; Ceresoli, M.; Chiara, O.; Cimbanassi, S.; Fattori, L.; Leppaniemi, A.; Manfredi, R.; Montori, G.; et al. The open abdomen, indications, management and definitive closure. World J. Emerg. Surg. 2015, 10, 32.
- Chiara, O.; Cimbanassi, S.; Biffl, W.; Leppaniemi, A.; Henry, S.; Scalea, T.M.; Catena, F.; Ansaloni, L.; Chieregato, A.; de Blasio, E.; et al. International consensus conference on open abdomen in trauma. J. Trauma. Acute Care Surg. 2016, 80, 173–183.
- Pereira, B.; Duchesne, J.; Concon-Filho, A.; Leppaniemi, A. Entero-atmospheric fistula migration: A new management alternative for complex septic open abdomen. Anaesthesiol. Intensive Ther. 2020, 52, 56–62.
- Di Saverio, S.; Tarasconi, A.; Walczak, D.A.; Cirocchi, R.; Mandrioli, M.; Birindelli, A.; Tugnoli, G. Classification, prevention and management of entero-atmospheric fistula: A state-of-the-art review. Langenbecks Arch. Surg. 2016, 401, 1–13.
- Wainstein, D.E.; Calvi, R.J.; Rezzonico, F.; Deforel, M.L.; Perrone, N.; Sisco, P. Management of enteroatmospheric fistula: A ten-year experience following fifteen years of learning. Surgery 2023, 173, 1079–1085.
- Chiarello, M.M.; Fransvea, P.; Cariati, M.; Adams, N.J.; Bianchi, V.; Brisinda, G. Anastomotic leakage in colorectal cancer surgery. Surg. Oncol. 2022, 40, 101708.
- Brisinda, G.; Chiarello, M.M.; Pepe, G.; Cariati, M.; Fico, V.; Mirco, P.; Bianchi, V. Anastomotic leakage in rectal cancer surgery: Retrospective analysis of risk factors. World J. Clin. Cases 2022, 10, 13321–13336.
- Andreyev, H.J.; Davidson, S.E.; Gillespie, C.; Allum, W.H.; Swarbrick, E. Practice guidance on the management of acute and chronic gastrointestinal problems arising as a result of treatment for cancer. Gut 2012, 61, 179–192.
- Birch, J.C.; Khatri, G.; Watumull, L.M.; Arriaga, Y.E.; Leyendecker, J.R. Unintended Consequences of Systemic and Ablative Oncologic Therapy in the Abdomen and Pelvis. Radiographics 2018, 38, 1158–1179.
- Andreyev, H.J.; Vlavianos, P.; Blake, P.; Dearnaley, D.; Norman, A.R.; Tait, D. Gastrointestinal symptoms after pelvic radiotherapy: Role for the gastroenterologist? Int. J. Radiat. Oncol. Biol. Phys. 2005, 62, 1464–1471.
- Andreyev, J. Gastrointestinal complications of pelvic radiotherapy: Are they of any importance? Gut 2005, 54, 1051–1054.
- Zheng, Y.; Gao, W.; Spratt, D.E.; Sun, Y.; Xing, L. Management of gastrointestinal perforation related to radiation. Int. J. Clin. Oncol. 2020, 25, 1010–1015.
- Reginelli, A.; Sangiovanni, A.; Vacca, G.; Belfiore, M.P.; Pignatiello, M.; Viscardi, G.; Clemente, A.; Urraro, F.; Cappabianca, S. Chemotherapy-induced bowel ischemia: Diagnostic imaging overview. Abdom. Radiol. 2022, 47, 1556–1564.
- Katabathina, V.S.; Restrepo, C.S.; Betancourt Cuellar, S.L.; Riascos, R.F.; Menias, C.O. Imaging of oncologic emergencies: What every radiologist should know. Radiographics 2013, 33, 1533–1553.
- Sussman, J.J. Surgical emergencies in the cancer patient. In Surgery: Basic Science and Clinical Evidence; Norton, J.A., Ed.; Springer-Verlag: New York, NY, USA, 2007; p. 2117.
- Heimann, D.M.; Schwartzentruber, D.J. Gastrointestinal perforations associated with interleukin-2 administration. J. Immunother. 2004, 27, 254–258.
- Cronin, C.G.; O’Connor, M.; Lohan, D.G.; Keane, M.; Roche, C.; Bruzzi, J.F.; Murphy, J.M. Imaging of the gastrointestinal complications of systemic chemotherapy. Clin. Radiol. 2009, 64, 724–733.
- Liaw, C.C.; Huang, J.S.; Wang, H.M.; Wang, C.H. Spontaneous gastroduodenal perforation in patients with cancer receiving chemotherapy and steroids. Report of four cases combining 5-fluorouracil infusion and cisplatin with antiemetics dexamethasone. Cancer 1993, 72, 1382–1385.
- Rose, P.G.; Piver, M.S. Intestinal perforation secondary to paclitaxel. Gynecol. Oncol. 1995, 57, 270–272.
- Hapani, S.; Chu, D.; Wu, S. Risk of gastrointestinal perforation in patients with cancer treated with bevacizumab: A meta-analysis. Lancet Oncol. 2009, 10, 559–568.
- Garant, A.; Des Groseilliers, S.; Martin, L.; Ferland, E.; Vuong, T. Late anastomotic dehiscence during bevacizumab therapy for patients with colorectal cancer. Clin. Oncol. 2011, 23, 497–498.
- Kabbinavar, F.F.; Flynn, P.J.; Kozloff, M.; Ashby, M.A.; Sing, A.; Barr, C.E.; Grothey, A. Gastrointestinal perforation associated with bevacizumab use in metastatic colorectal cancer: Results from a large treatment observational cohort study. Eur. J. Cancer 2012, 48, 1126–1132.
- Badgwell, B.D.; Camp, E.R.; Feig, B.; Wolff, R.A.; Eng, C.; Ellis, L.M.; Cormier, J.N. Management of bevacizumab-associated bowel perforation: A case series and review of the literature. Ann. Oncol. 2008, 19, 577–582.
- Bonifazi, M.; Rossi, M.; Moja, L.; Scigliano, V.D.; Franchi, M.; La Vecchia, C.; Zocchetti, C.; Negri, E. Bevacizumab in clinical practice: Prescribing appropriateness relative to national indications and safety. Oncologist 2012, 17, 117–124.
- Yetisir, F.; Sarer, A.E. Operative Management of Enteroatmospheric Fistula in Bjorck 4 Open Abdomen Patients by the Help of Laparoscopic Lateral Approach. Indian. J. Surg. 2017, 79, 173–176.
- Serrano Concha, K.; Morales Mayorga, H.; Acosta Farina, D.; Mendoza Saldarreaga, L.; Polit Guerrero, V.; Oliveros Rivero, J.; Acosta Bowen, D. Negative pressure device used in pediatric patients with Hostile abdomen. Case series. Cir. Pediatr. 2024, 37, 37–41.
- Theodorou, A.; Jedig, A.; Manekeller, S.; Willms, A.; Pantelis, D.; Matthaei, H.; Schafer, N.; Kalff, J.C.; von Websky, M.W. Long Term Outcome After Open Abdomen Treatment: Function and Quality of Life. Front. Surg. 2021, 8, 590245.
- Bleier, J.I.; Hedrick, T. Metabolic support of the enterocutaneous fistula patient. Clin. Colon. Rectal Surg. 2010, 23, 142–148.
- Barie, P.S. Serious intra-abdominal infections. Curr. Opin. Crit. Care 2001, 7, 263–267.
- Waibel, B.H.; Rotondo, M.F. Damage control in trauma and abdominal sepsis. Crit. Care Med. 2010, 38, S421–S430.
- Bjorck, M.; Kirkpatrick, A.W.; Cheatham, M.; Kaplan, M.; Leppaniemi, A.; De Waele, J.J. Amended Classification of the Open Abdomen. Scand. J. Surg. 2016, 105, 5–10.
- Mahoney, E.J.; Bugaev, N.; Appelbaum, R.; Goldenberg-Sandau, A.; Baltazar, G.A.; Posluszny, J.; Dultz, L.; Kartiko, S.; Kasotakis, G.; Como, J.; et al. Management of the open abdomen: A systematic review with meta-analysis and practice management guideline from the Eastern Association for the Surgery of Trauma. J. Trauma. Acute Care Surg. 2022, 93, e110–e118.
- Coccolini, F.; Roberts, D.; Ansaloni, L.; Ivatury, R.; Gamberini, E.; Kluger, Y.; Moore, E.E.; Coimbra, R.; Kirkpatrick, A.W.; Pereira, B.M.; et al. The open abdomen in trauma and non-trauma patients: WSES guidelines. World J. Emerg. Surg. 2018, 13, 7.
- Kirkpatrick, A.W.; Roberts, D.J.; Jaeschke, R.; De Waele, J.J.; De Keulenaer, B.L.; Duchesne, J.; Bjorck, M.; Leppaniemi, A.; Ejike, J.C.; Sugrue, M.; et al. Methodological background and strategy for the 2012-2013 updated consensus definitions and clinical practice guidelines from the abdominal compartment society. Anaesthesiol. Intensive Ther. 2015, 47, s63–s77.
- Sermoneta, D.; Di Mugno, M.; Spada, P.L.; Lodoli, C.; Carvelli, M.E.; Magalini, S.C.; Cavicchioni, C.; Bocci, M.G.; Martorelli, F.; Brizi, M.G.; et al. Intra-abdominal vacuum-assisted closure (VAC) after necrosectomy for acute necrotising pancreatitis: Preliminary experience. Int. Wound J. 2010, 7, 525–530.
- Brooke, J.; El-Ghaname, A.; Napier, K.; Sommerey, L. Nurses Specialized in Wound, Ostomy and Continence Canada (NSWOCC) Management of Enterocutaneous Fistula and Enteroatmospheric Fistula: Development of Best Practice Recommendations. J. Wound Ostomy Cont. Nurs. 2019, 46, 346–347.
- Coccolini, F.; Ceresoli, M.; Kluger, Y.; Kirkpatrick, A.; Montori, G.; Salvetti, F.; Fugazzola, P.; Tomasoni, M.; Sartelli, M.; Ansaloni, L.; et al. Open abdomen and entero-atmospheric fistulae: An interim analysis from the International Register of Open Abdomen (IROA). Injury 2019, 50, 160–166.
- Williams, L.J.; Zolfaghari, S.; Boushey, R.P. Complications of enterocutaneous fistulas and their management. Clin. Colon. Rectal Surg. 2010, 23, 209–220.
- Dudrick, S.J.; Panait, L. Metabolic consequences of patients with gastrointestinal fistulas. Eur. J. Trauma. Emerg. Surg. 2011, 37, 215–225.
- Pironi, L.; Arends, J.; Baxter, J.; Bozzetti, F.; Pelaez, R.B.; Cuerda, C.; Forbes, A.; Gabe, S.; Gillanders, L.; Holst, M.; et al. ESPEN endorsed recommendations. Definition and classification of intestinal failure in adults. Clin. Nutr. 2015, 34, 171–180.
- Klek, S.; Forbes, A.; Gabe, S.; Holst, M.; Wanten, G.; Irtun, O.; Damink, S.O.; Panisic-Sekeljic, M.; Pelaez, R.B.; Pironi, L.; et al. Management of acute intestinal failure: A position paper from the European Society for Clinical Nutrition and Metabolism (ESPEN) Special Interest Group. Clin. Nutr. 2016, 35, 1209–1218.
- Levy, E.; Frileux, P.; Cugnenc, P.H.; Honiger, J.; Ollivier, J.M.; Parc, R. High-output external fistulae of the small bowel: Management with continuous enteral nutrition. Br. J. Surg. 1989, 76, 676–679.
- Hollington, P.; Mawdsley, J.; Lim, W.; Gabe, S.M.; Forbes, A.; Windsor, A.J. An 11-year experience of enterocutaneous fistula. Br. J. Surg. 2004, 91, 1646–1651.
- Quinn, M.; Falconer, S.; McKee, R.F. Management of Enterocutaneous Fistula: Outcomes in 276 Patients. World J. Surg. 2017, 41, 2502–2511.
- Visschers, R.G.; Olde Damink, S.W.; Winkens, B.; Soeters, P.B.; van Gemert, W.G. Treatment strategies in 135 consecutive patients with enterocutaneous fistulas. World J. Surg. 2008, 32, 445–453.
- Boolaky, K.N.; Tariq, A.H.; Hardcastle, T.C. Open abdomen in the trauma ICU patient: Who? when? why? and what are the outcome results? Eur. J. Trauma. Emerg. Surg. 2022, 48, 953–961.
- Marinis, A.; Gkiokas, G.; Argyra, E.; Fragulidis, G.; Polymeneas, G.; Voros, D. “Enteroatmospheric fistulae”—Gastrointestinal openings in the open abdomen: A review and recent proposal of a surgical technique. Scand. J. Surg. 2013, 102, 61–68.
- Becker, H.P.; Willms, A.; Schwab, R. Small bowel fistulas and the open abdomen. Scand. J. Surg. 2007, 96, 263–271.
- Kaushal, M.; Carlson, G.L. Management of enterocutaneous fistulas. Clin. Colon. Rectal Surg. 2004, 17, 79–88.
- Samad, S.; Anele, C.; Akhtar, M.; Doughan, S. Implementing a Pro-forma for Multidisciplinary Management of an Enterocutaneous Fistula: A Case Study. Ostomy Wound Manag. 2015, 61, 46–52.
- Martinez, J.L.; Luque-de-Leon, E.; Mier, J.; Blanco-Benavides, R.; Robledo, F. Systematic management of postoperative enterocutaneous fistulas: Factors related to outcomes. World J. Surg. 2008, 32, 436–443; discussion 444.
- Tang, Q.Q.; Hong, Z.W.; Ren, H.J.; Wu, L.; Wang, G.F.; Gu, G.S.; Chen, J.; Zheng, T.; Wu, X.W.; Ren, J.A.; et al. Nutritional Management of Patients With Enterocutaneous Fistulas: Practice and Progression. Front. Nutr. 2020, 7, 564379.
- Urbanek, L.; Veverkova, L.; Zak, J.; Reska, M. Nutrition in open abdomen. Rozhl. Chir. 2021, 100, 83–87.
- Assenza, M.; Rossi, D.; De Gruttola, I.; Ballanti, C. Enterocutaneous fistula treatment: Case report and review of the literature. G. Chir. 2018, 39, 143–151.
- Meguid, M.M.; Muscaritoli, M. Current uses of total parenteral nutrition. Am. Fam. Physician 1993, 47, 383–394.
- Lloyd, D.A.; Gabe, S.M.; Windsor, A.C. Nutrition and management of enterocutaneous fistula. Br. J. Surg. 2006, 93, 1045–1055.
- Connolly, P.T.; Teubner, A.; Lees, N.P.; Anderson, I.D.; Scott, N.A.; Carlson, G.L. Outcome of reconstructive surgery for intestinal fistula in the open abdomen. Ann. Surg. 2008, 247, 440–444.
- Rahbour, G.; Siddiqui, M.R.; Ullah, M.R.; Gabe, S.M.; Warusavitarne, J.; Vaizey, C.J. A meta-analysis of outcomes following use of somatostatin and its analogues for the management of enterocutaneous fistulas. Ann. Surg. 2012, 256, 946–954.
- Lynch, A.C.; Delaney, C.P.; Senagore, A.J.; Connor, J.T.; Remzi, F.H.; Fazio, V.W. Clinical outcome and factors predictive of recurrence after enterocutaneous fistula surgery. Ann. Surg. 2004, 240, 825–831.
- Rahbour, G.; Gabe, S.M.; Ullah, M.R.; Thomas, G.P.; Al-Hassi, H.O.; Yassin, N.A.; Tozer, P.J.; Warusavitarne, J.; Vaizey, C.J. Seven-year experience of enterocutaneous fistula with univariate and multivariate analysis of factors associated with healing: Development of a validated scoring system. Colorectal Dis. 2013, 15, 1162–1170.
- Runstrom, B.; Hallbook, O.; Nystrom, P.O.; Sjodahl, R.; Olaison, G. Outcome of 132 consecutive reconstructive operations for intestinal fistula—Staged operation without primary anastomosis improved outcome in retrospective analysis. Scand. J. Surg. 2013, 102, 152–157.
- Rabel, T.; Bonnot, P.E.; Hadeedi, O.; Kepenekian, V.; Bernard, L.; Friggeri, A.; Glehen, O.; Passot, G. Negative-Pressure Wound Therapy for Open Abdomen in Surgical Reintervention after Curative Surgery of Peritoneal Malignancy Increases the Risk of Recurrence. Adv. Skin. Wound Care 2023, 36, 1–5.
- Piccoli, M.; Agresta, F.; Attina, G.M.; Amabile, D.; Marchi, D.; “Complex Abdominal Wall Study” Italian Collaborative Group. “Complex abdominal wall” management: Evidence-based guidelines of the Italian Consensus Conference. Updates Surg. 2019, 71, 255–272.
- Gefen, R.; Garoufalia, Z.; Zhou, P.; Watson, K.; Emile, S.H.; Wexner, S.D. Treatment of enterocutaneous fistula: A systematic review and meta-analysis. Tech. Coloproctol. 2022, 26, 863–874.
- Suter, K.J.L.; Fairweather, L.; Al-Habbal, Y.; Houli, N.; Jacobs, R.; Bui, H.T. How to isolate a high output enteroatmospheric fistula in the open abdomen with negative pressure therapy: An institution’s step by step guide to the VAC donut. ANZ J. Surg. 2023, 93, 682–686.
- Sun, R.; Xu, X.; Luo, S.; Zhao, R.; Tian, W.; Huang, M.; Yao, Z. An alternative negative pressure treatment for enteroatmospheric fistula resulting from small intestinal leakage caused by incision dehiscence. Heliyon 2023, 9, e22045.
- Goverman, J.; Yelon, J.A.; Platz, J.J.; Singson, R.C.; Turcinovic, M. The “Fistula VAC”, a technique for management of enterocutaneous fistulae arising within the open abdomen: Report of 5 cases. J. Trauma. 2006, 60, 428–431; discussion 431.
- Wang, G.; Ren, J.; Liu, S.; Wu, X.; Gu, G.; Li, J. “Fistula patch”: Making the treatment of enteroatmospheric fistulae in the open abdomen easier. J. Trauma. Acute Care Surg. 2013, 74, 1175–1177.
- Miranda, L.E.; Miranda, A.C. Enteroatmospheric fistula management by endoscopic gastrostomy PEG tube. Int. Wound J. 2017, 14, 915–917.
- Morykwas, M.J.; Argenta, L.C.; Shelton-Brown, E.I.; McGuirt, W. Vacuum-assisted closure: A new method for wound control and treatment: Animal studies and basic foundation. Ann. Plast. Surg. 1997, 38, 553–562.
- Trevelyan, S.L.; Carlson, G.L. Is TNP in the open abdomen safe and effective? J. Wound Care 2009, 18, 24–25.
- Bee, T.K.; Croce, M.A.; Magnotti, L.J.; Zarzaur, B.L.; Maish, G.O., 3rd; Minard, G.; Schroeppel, T.J.; Fabian, T.C. Temporary abdominal closure techniques: A prospective randomized trial comparing polyglactin 910 mesh and vacuum-assisted closure. J. Trauma. 2008, 65, 337–342; discussion 342–334.
- Fischer, J.E. A cautionary note: The use of vacuum-assisted closure systems in the treatment of gastrointestinal cutaneous fistula may be associated with higher mortality from subsequent fistula development. Am. J. Surg. 2008, 196, 1–2.
- Demetriades, D. Total management of the open abdomen. Int. Wound J. 2012, 9 (Suppl. S1), 17–24.
- Caro, A.; Olona, C.; Jimenez, A.; Vadillo, J.; Feliu, F.; Vicente, V. Treatment of the open abdomen with topical negative pressure therapy: A retrospective study of 46 cases. Int. Wound J. 2011, 8, 274–279.





