Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Monica Florin-Christensen | -- | 1828 | 2024-02-26 14:02:57 | | | |
| 2 | Camila Xu | -30 word(s) | 1798 | 2024-02-27 03:13:33 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Wieser, S.N.; Giuliano, S.M.; Reategui Ordoñez, J.; Barriga Marcapura, X.; Olivera, L.V.M.; Chavez Fumagalli, M.A.; Schnittger, L.; Florin-Christensen, M. Sarcocystis Infecting Old World and South American camelids. Encyclopedia. Available online: https://encyclopedia.pub/entry/55467 (accessed on 08 June 2026).
Wieser SN, Giuliano SM, Reategui Ordoñez J, Barriga Marcapura X, Olivera LVM, Chavez Fumagalli MA, et al. Sarcocystis Infecting Old World and South American camelids. Encyclopedia. Available at: https://encyclopedia.pub/entry/55467. Accessed June 08, 2026.
Wieser, Sarah N., Susana M. Giuliano, Juan Reategui Ordoñez, Ximena Barriga Marcapura, Luis V. M. Olivera, Miguel Angel Chavez Fumagalli, Leonhard Schnittger, Mónica Florin-Christensen. "Sarcocystis Infecting Old World and South American camelids" Encyclopedia, https://encyclopedia.pub/entry/55467 (accessed June 08, 2026).
Wieser, S.N., Giuliano, S.M., Reategui Ordoñez, J., Barriga Marcapura, X., Olivera, L.V.M., Chavez Fumagalli, M.A., Schnittger, L., & Florin-Christensen, M. (2024, February 26). Sarcocystis Infecting Old World and South American camelids. In Encyclopedia. https://encyclopedia.pub/entry/55467
Wieser, Sarah N., et al. "Sarcocystis Infecting Old World and South American camelids." Encyclopedia. Web. 26 February, 2024.
Copy Citation
Sarcocystis spp. are coccidian protozoans belonging to the Apicomplexa phylum. As with other members of this phylum, they are obligate intracellular parasites with complex cellular machinery for the invasion of host cells. Sarcocystis spp. display dixenous life cycles, involving a predator and a prey as definitive and intermediate hosts, respectively. Specifically, these parasites develop sarcocysts in the tissues of their intermediate hosts, ranging in size from microscopic to visible to the naked eye, depending on the species. When definitive hosts consume sarcocysts, infective forms are produced in the digestive system and discharged into the environment via feces.
South American camelids
Old World camels
Sarcocystis
sarcocysts
1. Introduction
Sarcocystosis is a parasitic infection caused by different species of protozoans belonging to the Sarcocystis genus. With over 200 described species, different Sarcocystis spp. infect mammals, including humans, as well as birds and reptiles. Their life cycles involve a predator and a prey that serve as definitive and intermediate hosts, respectively. Typical of the intermediate host phase of Sarcocystis is the formation of sarcocysts—wall-surrounded capsules in which the parasites divide asexually—which may be micro or macroscopic, depending on the species [1].
Sarcocystis complex life cycles alternate between invading, dividing, and sexual stages. After a predator ingests the meat of a prey containing sarcocysts, bradyzoites—the infective stage borne in these structures—invade the goblet cells of the predator’s intestine. Micro- and macrogametes are formed, and gamete fusion leads to the formation of an oocyst which, after being excreted with the feces into the environment, sporulates to form four sporozoites. Intermediate hosts ingest oocysts when grazing or drinking contaminated pasture or water, and enzymes in their intestine lead to the liberation of sporozoites. Individual sporozoites can also be found in stools due to occasional breakage of the thin oocyst wall, and can contaminate water and pastures, which leads to direct ingestion of sporozoites by the intermediate host. Once in the intestinal lumen, sporozoites invade the endothelial cells of mesenteric lymph node arteries, where they reproduce asexually by schizogony, forming schizonts with lobed nuclei that have the appearance of a rosette. Merozoites eventually bud off and are released into the bloodstream, disseminating the infection in the intermediate host. They can be found free or inside mononuclear cells, where they undergo binary division by endodyogeny. When merozoites invade the endothelial cells of downstream arterioles, capillaries, and veins, a new cycle of schizogony is initiated. In turn, invasion of a myocyte or a nervous cell by a merozoite leads to the formation of sarcocysts, the final stage in the intermediate host. The parasite remains inside a parasitophorous vacuole (PV), and the parasitophorous vacuolar membrane (PMV) together with parasite secretions form a wall that provides a safe microenvironment for parasite multiplication. Depending on the Sarcocystis species, final-destination cells can be myocytes of skeletal or cardiac muscles, or neural cells. In the sarcocysts, merozoites transform into metrocytes, which rapidly divide by endodyogeny and eventually turn into infective bradyzoites [1][2][3] (Figure 1).
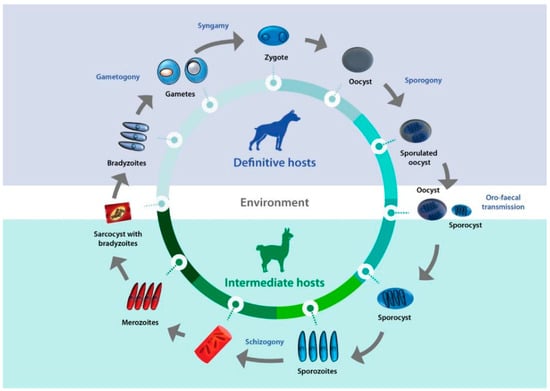
Figure 1. Life cycle of Sarcocystis. A typical life cycle of Sarcocystis species is shown, exemplified with a llama and a dog as intermediate and definitive hosts, respectively.
Most Sarcocystis spp. that infect livestock have a worldwide distribution and, in general, occur with high prevalence in both industrialized and developing countries [3]. The economic burden of Sarcocystis infections of livestock is related to abortions, low meat and/or milk yield, poor body growth, and outbreaks of clinical sarcocystosis that can be fatal. Additionally, carcasses destined for human consumption can be condemned by sanitary authorities when abundant macroscopic Sarcocystis spp. sarcocysts or Sarcocystis spp.-associated lesions due to eosinophilic myositis are encountered. Eosinophilic myositis is an inflammatory condition of striated muscles that leads to necrosis of the affected areas which has been described in cattle and some other mammals [1][4][5]. For most Sarcocystis species, especially those infecting wild animals, their effect on host fitness is unknown.
Camelids are Artiodactyla mammals grouped in the Camelidae family, the only extant family of the Tylopoda suborder (from the Greek: “feet with cushions”). They regurgitate and rechew food but are not ruminants and are differentiated from the latter by several anatomical features, including their stomach with three compartments, lack of horns, and the presence of real canine teeth and plantar cushions, as well as other physiological and behavioral features. Importantly, camelids differ from ruminants in their susceptibility to microbial and parasitic diseases [6].
Old World camelids (OWCs) belong to the genus Camelus, which comprises three extant species: C. dromedarius (one-humped camel or dromedary), C. bactrianus (two-humped or Bactrian camel), both of which have been domesticated, and the wild and highly endangered C. ferus (two humps). The population size of OWCs has been estimated to be at least 35.5 million heads, of which 95% are dromedaries [7]. South American camelids (SACs) comprise four species: the wild Lama guanicoe (guanaco) and Vicugna vicugna (vicuña), and the domesticated Lama glama (llama) and Vicugna pacos (alpaca), with an overall estimated population of 10 million heads [8]. Notably, llamas and alpacas have been introduced to farms in some European countries, South Africa, and Australia and, thus, their geographic distribution and numbers of heads are considerably larger than those mentioned here [7][8].
Both OWCs and SACs are adapted to harsh environmental conditions, including extreme temperatures, intense solar radiation, water scarcity, and poor pastures. Under such rough settings, most other livestock species are either unable to thrive or show a significant decline in production. Camelids thus constitute an attractive livestock choice in a scenario of climate change, shortage of water, and reduction in agricultural areas [9].
Camelids have a long history of association with humans. Human groups who lived as gatherers and hunters must have found in camels a good source of food and hides, and through their domestication some 3 to 7 thousand years ago (kya), they became important suppliers of vital goods to ancient civilizations in the Old and New Worlds [10][11]. Currently, camelids continue to be an important asset to a considerable number of human populations, and among other benefits, their meat is a valuable source of animal protein and an attractive product for the gourmet cuisine [12][13][14].
Both OWCs and SACs act as intermediate hosts for some Sarcocystis spp., sometimes with a relevant negative impact on local economies [15][16][17][18].
2. Sarcocystis Infecting OWCs and SACs
Four Sarcocystis spp. have so far been described to infect camelids: S. cameli and S. ippeni for OWCs, and S. aucheniae and S. masoni for SACs [3][17][19].
The first observation of Sarcocystis infections in OWCs was carried out in Egypt by Mason (1910), who reported macroscopic sarcocysts in the muscles of camels and used the name S. cameli to refer to the etiological agent. Later, several additional case reports of Sarcocystis species infecting OWCs appeared in the literature, which were designated as S. ippeni, S. camelicanis, S. camelocanis, and S. miescheri, depending on their sarcocyst wall or oocyst features. A taxonomic revision of OWCs sarcocystosis led to the acceptance of S. cameli and S. ippeni as valid species, while S. camelicanis, S. camelocanis, and S. miescheri were considered synonymous with S. cameli. Importantly, the vast majority of Sarcocystis reports are from dromedary camels [16][19][20].
In the case of SAC, the first description of a macroscopic sarcocyst in a llama took place in 1913, and the corresponding parasite was named S. aucheniae [21]. Later, the names S. tilopodi and S. guanicoecanis were used for parasites forming sarcocysts in guanaco [22][23]. A Sarcocystis species forming microscopic cysts in SACs was initially named S. lamacanis [24][25]. Electron microscopy and molecular studies established S. aucheniae as the only species forming macroscopic cysts in llamas, alpacas, and guanacos, while the species forming microscopic cysts was redescribed as S. masoni in honor of Dr Eugene Mason. Thus, the names S. tilopodi, S. guanicoecanis, and S. lamacanis are currently considered invalid [3][17][26].
In different Sarcocystis spp., sarcocysts vary in shape (globular, filamentous, fusiform), size, and other characteristics, such as the presence or absence of internal partitions and variations in their wall ultrastructure [2][3]. In the case of Sarcocystis spp. that infect camelids, both S. aucheniae and S. cameli generate macroscopic sarcocysts that are visible to the naked eye (oval, 5–20 mm × 2 mm, and fusiform, 1.5–5 mm × 0.2–0.4 mm, respectively). Additionally, microscopic cysts of S. cameli (700 × 100 µm) are commonly found in camel tissues. In turn, only microcysts were described for S. masoni (fusiform, 800 × 95 µm) and S. ippeni (globular, 100–120 × 50–100 µm) [3][17][19] (Table 1).
Table 1. Characteristics of sarcocysts produced by camelid-infecting Sarcocystis sp.
| Intermediate Host | Species | Sarcocyst | Ref. | ||||
|---|---|---|---|---|---|---|---|
| Shape, Size (Length × Width) |
Cyst Wall | Location | |||||
| Type | Thickness (µm) |
Villar Protrusions (vp) Size and Aspect | |||||
| OWC | S. cameli | Fusiform, microscopic (700 × 100 µm) and macroscopic (1.5–5.0 × 0.2–0.4 mm) |
9j | <2 | 3.0 × 0.5 µm finger-like vp |
cardiac and skeletal muscle | [3][19][20][27][28] |
| S. ippeni | Globular, microscopic (100–120 × 50–100 µm) |
32 | 2.3–3.0 | 1.0 × 0.25–1.2 µm thorn-like vp |
skeletal muscle | [3][19] | |
| SAC | S. aucheniae | Oval, macroscopic (0.5–2.0 × 0.2 cm) |
21 | 6–10 | 3–4.5 × 2.5–3.5 µm branched vp, cauliflower-like wall |
skeletal muscle | [1][17][26][29][30] |
| S. masoni | Fusiform, microscopic (800 × 95 µm) |
9j | 2.5–3.5 | 2–2.8 × 0.5–0.7 µm conical to cylindrical vp |
cardiac and skeletal muscle | 17 | |
In all Sarcocystis spp., the sarcocyst wall essentially consists of the PVM covering a granular, electron-dense layer from which septa can arise. When present, septa cross the cyst, separating its cavity into compartments, where metrocytes and bradyzoites are found. The number of parasites contained in a sarcocyst varies with the species and the stage of maturation: young cysts as small as 5 μm in diameter might contain only two parasites, while a mature macroscopic cyst can contain 20 million, as has been observed for S. aucheniae [3][26].
The sarcocyst wall can remain relatively simple in some species and, in others, form projections (villar protrusions) of different sizes and shapes that bulge outwardly and can contain microfilaments, microtubules, electron-dense bodies, minute granules, and small vesicles [1][3]. At least 82 ultrastructural types of cyst wall have been described for different Sarcocystis spp. [3]. Notably, the cyst walls of both S. cameli and S. masoni have a common “9j” conformation, characterized by the presence of upright finger-like villar protrusions with knob-like structures arising from the PVM, in which microtubules can be observed [3][17]. S. aucheniae presents a ‘type 21’ sarcocyst wall ultrastructure, with highly branched cauliflower-like villar protrusions, similar to that of S. gigantea [1][3][17]. Finally, S. ippeni has a characteristic ‘type 32’ sarcocyst wall structure. This type of ultrastructure is characterized by thorn-like villar protrusions with microtubules radiating into the granular layer and has not been previously described in any other Sarcocystis species [3] (Table 1).
Figure 2 exemplifies the different aspects of sarcocysts produced by S. aucheniae in the skeletal muscles of llamas and alpacas.
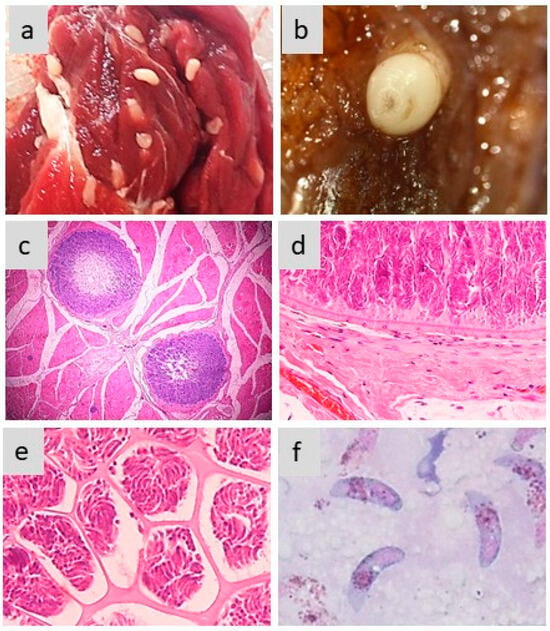
Figure 2. Morphology of sarcocysts and bradyzoites in S. aucheniae. (a,b) Macroscopic sarcocysts in llama (a) and alpaca (b) skeletal muscle; (c) hematoxylin eosin-dyed cross-section of alpaca skeletal muscle with two macroscopic sarcocysts, in which zoites are located to the periphery and the center is empty (100×); (d,e) details of hematoxylin eosin-dyed section of a macroscopic sarcocyst showing the morphology of the cell wall (d) and compartments with thousands of banana-shaped bradyzoites (e) (400×); (f) bradyzoites observed in a cyst stained with Giemsa (1000×). The photographs were obtained by S.N.W. and L.V.M.O.
References
- Decker Franco, C.; Schnittger, L.; Florin-Christensen, M. Sarcocystis. In Parasitic Protozoa of Farm Animals and Pets; Springer International Publishing: Cham, Switzerland, 2018; pp. 103–124. ISBN 9783319701325.
- Fayer, R. Sarcocystis spp. in human infections. Clin. Microbiol. Rev. 2004, 17, 894–902.
- Dubey, J.P.; Calero-Bernal, R.; Rosenthal, B.M.; Speer, C.A.; Fayer, R. Sarcocystosis of Animals and Humans, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2016; ISBN 9781498710152.
- Dubey, J.P.; Rosenthal, B.M. Bovine Sarcocystosis: Sarcocystis species, diagnosis, prevalence, economic and public health considerations, and association of Sarcocystis species with eosinophilic myositis in cattle. Int. J. Parasitol. 2023, 53, 463–475.
- Poulsen, C.S.; Stensvold, C.R. Current status of epidemiology and diagnosis of human sarcocystosis. J. Clin. Microbiol. 2014, 52, 3524–3530.
- Fowler, M.E. Camelids are not ruminants. Zoo Wild Anim. Med. 2008, 375–385.
- FAOSTAT. Available online: http://www.fao.org/faostat/en/#home (accessed on 13 December 2023).
- Midagari Camélidos Sudamericanos. Available online: https://www.midagri.gob.pe/portal/datero/40-sector-agrario/situacion-de-las-actividades-de-crianza-y-producci/298-camelidos-sudamericanos?start=1 (accessed on 13 December 2023).
- Zarrin, M.; Riveros, J.L.; Ahmadpour, A.; De Almeida, A.M.; Konuspayeva, G.; Vargas-Bello-Pérez, E.; Faye, B.; Hernández-Castellano, L.E. Camelids: New players in the international animal production context; Springer: Cham, Switzerland, 2020; Volume 52, pp. 903–913.
- Fowler, M.E. Husbandry and diseases of camelids. Rev. Sci. Tech. 1996, 15, 155–169.
- Yacobaccio, H.D. The domestication of South American camelids: A Review. Anim. Front. 2021, 11, 43–51.
- Mamani-Linares, L.W.; Gallo, C.B. Meat quality, proximate composition and muscle fatty acid profile of young llamas (Lama glama) supplemented with hay or concentrate during the dry season. Meat Sci. 2014, 96, 394–399.
- Mamani-Linares, L.W.; Gallo, C.B. Meat quality attributes of the longissimus lumborum muscle of the Kh’ara genotype of llama (Lama glama) reared extensively in Northern Chile. Meat Sci. 2013, 94, 89–94.
- Kadim, I.T.; Al-Amri, I.S.; Alkindi, A.Y.; Haq, Q.M.I. Nutritional values and health benefits of dromedary camel meat. Anim. Front. 2022, 12, 61–70.
- Saeed, M.A.; Rashid, M.H.; Vaughan, J.; Jabbar, A. Sarcocystosis in South American camelids: The state of play revisited. Parasites Vectors 2018, 11, 146.
- Saeed, M.A.; Vaughan, J.L.; Jabbar, A. An update on sarcocystosis in one-humped camels (Camelus dromedarius). Parasitology 2018, 145, 1367–1377.
- Moré, G.; Regensburger, C.; Gos, M.L.; Pardini, L.; Verma, S.K.; Ctibor, J.; Serrano-Martínez, M.E.; Dubey, J.P.; Venturini, M.C. Sarcocystis masoni, n. sp. (Apicomplexa: Sarcocystidae), and redescription of Sarcocystis aucheniae from llama (Lama glama), guanaco (Lama guanicoe) and alpaca (Vicugna pacos). Parasitology 2016, 143, 617–626.
- Giuliano, S.M.; Reategui Ordonez, J.; Barriga Marcopuda, X.; Florin-Christensen, M. Situación actual de la calidad de carne de camélidos sudamericanos (llama y alpaca) en Argentina y Perú y su relación con infestación con Sarcocystis aucheniae. In Proceedings of the XXIII Reunión Nacional de la Asociación Boliviana de Producción Animal—ABOPA, Oruro, Bolivia, 26–28 October 2023; Available online: https://www.cifumss.agro.bo/abopa/index.html (accessed on 22 February 2023).
- Dubey, J.P.; Hilali, M.; Van Wilpe, E.; Calero-Bernal, R.; Verma, S.K.; Abbas, I.E. A review of sarcocystosis in camels and redescription of Sarcocystis cameli and Sarcocystis ippeni sarcocysts from the one-humped camel (Camelus dromedarius). Parasitology 2015, 142, 1481–1492.
- Dubey, J.P.; A’aji, N.N.; Mowery, J.D.; Verma, S.K.; Calero-Bernal, R. Identification of macroscopic sarcocysts of Sarcocystis cameli from one-humped camel (Camelus dromedarius) in Iraq. J. Parasitol. 2017, 103, 168–169.
- Brumpt, E. Precis de Parasitologie, 2nd ed.; Masson et Cie: Paris, France, 1913.
- Quiroga, D.; Lombadero, O.; Zorrila, R. Sarcocystis tilpodi n.sp. en guanacos (Lama guanicoe) de la Repúplica Argentina. Gaceta Veterinaria 1969, 31, 67–70.
- Gorman, T.R.; Alcaíno, H.A.; Muñuz, H.; Cunazza, C. Sarcocystis sp. in guanaco (Lama guanicoe) and effect of temperature on its viability. Vet. Parasitol. 1984, 15, 95–101.
- Leguía, G. Enfermedades Parasitarias y Atlas Parasitológico de Camélidos Sudamericanos; Primera, Ed.; De Mar: Lima, Perú, 1999.
- Leguía, G. The epidemiology and economic impact of llama parasites. Parasitol. Today 1991, 7, 54–56.
- Carletti, T.; Martin, M.; Romero, S.; Morrison, D.A.; Marcoppido, G.; Florin-Christensen, M.; Schnittger, L. Molecular identification of Sarcocystis aucheniae as the macrocyst-forming parasite of llamas. Vet. Parasitol. 2013, 198, 396–400.
- Metwally, D.M.; Al-Otaibi, T.T.; Al-Turaiki, I.M.; El-Khadragy, M.F.; Alajmi, R.A. Identification of Sarcocystis spp. in one-humped camels (Camelus dromedarius) from Riyadh and Dammam, Saudi Arabia, via histological and phylogenetic approaches. Animals 2020, 10, 1108.
- Motamedi, G.R.; Dalimi, A.; Nouri, A.; Aghaeipour, K. Ultrastructural and molecular characterization of Sarcocystis isolated from camel (Camelus dromedarius) in Iran. Parasitol. Res. 2011, 108, 949–954.
- Regensburger, C.; Gos, M.L.; Ctibor, J.; Moré, G. Morphological and molecular characteristics of Sarcocystis aucheniae isolated from meat of guanaco (Lama guanicoe). J. Food Qual. Hazards Cont. 2015, 2, 118–121.
- Schnieder, T.; Kaup, F.J.; Drommer, W.; Thiel, W.; Rommel, M. Fine structure and development of Sarcocystis aucheniae in llamas. Z. Parasitenkd. 1984, 70, 451–458.
More
Information
Subjects:
Parasitology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
650
Revisions:
2 times
(View History)
Update Date:
27 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





