
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Valmary Michelle Van Breda | -- | 2302 | 2024-02-26 13:27:52 | | | |
| 2 | Mona Zou | Meta information modification | 2302 | 2024-02-27 08:32:06 | | | | |
| 3 | Mona Zou | Meta information modification | 2302 | 2024-02-27 08:34:47 | | | | |
| 4 | Mona Zou | -15 word(s) | 2287 | 2024-02-29 08:59:08 | | |
Video Upload Options
Low-temperature treatments preceding alcoholic fermentation are becoming increasingly popular and have been used in winemaking as a tool to improve wine colour, aroma, and quality. Additionally, the pre-fermentative treatment of grapes with cryogenic agents protects the grape juice (must) from oxidation by reducing the diffusion of atmospheric oxygen into the liquid phase during the winemaking process. Resultant wines were reported to have enhanced varietal aromas, increased complexity, and higher thiol levels. Indications are that increased contact time between skin and juice improves the extraction of the compounds and/or precursors. Recently, there has been considerable interest in the production of wines with enhanced varietal aromas and improved quality by applying innovative winemaking technologies.
1. Effect of Cryogenic Technologies on Physicochemical and Aroma Compounds of Grape Must and Wine
1.1. Physicochemical Parameters
1.2. Effect of Cryogenic Technologies on Grape Aroma Compounds
| Aroma Development Stage | Compound | Origin |
|---|---|---|
| Varietal | Precursors (free or bound) | Grape berry (skin and pulp) |
| Pre-fermentative | C6 compounds | Enzymatic/catalytic reactions due to processing (crushing of berries) |
| Fermentation | Ethyl esters, fusel alcohols, fatty acids, thiols | Microorganism metabolism (yeast and bacterial) |
| Post-fermentation | Oxidation of volatile aroma compounds; increase in fatty acids, esters, aldehydes, ketones, and polyphenols | Wine aging (bottle, barrel, storage, aging on lees) |
1.3. Effect of Cryogenic Technologies on Varietal Thiols
| Cultivar | Compound & Chemical Structure |
Aroma Description |
Aroma Perception in Wine (ng L–1) |
Range in Wine (ng L–1) |
Range in SA 1 Wine (ng L–1) |
|---|---|---|---|---|---|
| Sauvignon blanc | 4–methyl–4–sulfanylpentan–2–one (4MSP)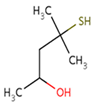 |
Boxwood, blackcurrant | 0.8 | 0–88 | 0–21.9 |
| Chenin blanc | 0–23 | n.d. * | |||
| Sauvignon blanc | 3–sulfanylhexyl acetate (3SHA) 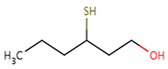 |
Passionfruit, tropical, boxwood | 4 | 0–106 | 23–151 |
| Chenin blanc | 0–100 | 5–253 | |||
| Sauvignon blanc | 3–sulfanylhexan–1–ol (3SH) 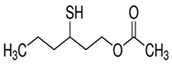 |
Grapefruit, tropical, passionfruit | 60 | 350–5664 | 178–904 |
| Chenin blanc | 10–1368 | 99–1124 | |||
| Sauvignon blanc | benzyl mercaptan (BM)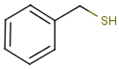 |
Smoke, toasty, struck flint | 0.3 | 0.6–5.5 | n.d. * |
| Chenin Blanc | 30–40 | n.d. * | |||
| Sauvignon blanc | 2–furfurylthiol (FFT)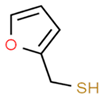 |
Roasted Coffee | 0.4 | 1–36 | n.d. * |
| Chenin blanc | 14 | n.d. * |
1.4. Effect of Cryogenic Technologies on Methoxypyrazines
| Compound & Chemical Structure | Aroma Description | Aroma Perception in Water (ng L–1) |
Aroma Perception in Wine (ng L–1) |
Range in Wine (ng L–1) |
|---|---|---|---|---|
2–methoxy–3–isobutylpyrazine (ibMP)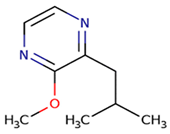 |
vegetative, green pepper | 1–2 | 2–163 | 2–30 |
2–methoxy–3–isopropylpyrazine (ipMP)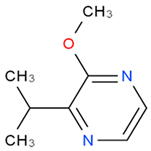 |
earthy, mushroom, cooked, or canned asparagus, green beans | 1–2 | 2–16 | <10 |
2–methoxy–3–sec–butylpyrazine (sbMP)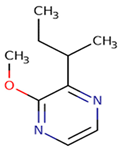 |
green (peas, bell pepper, galbanum), ivy leaves, bell pepper | 1–2 | 2–16 | <10 |
2. Effect of Cryogenic Technologies on Sensory Properties of Wine
References
- Zhang, S.; Petersen, M.A.; Liu, J.; Toldam-Andersen, T.B. Influence of pre-fermentation treatments on wine volatile and sensory profile of the new disease tolerant cultivar Solaris. Molecules 2015, 20, 21609–21625.
- Aleixandre-Tudo, J.L.; du Toit, W. Cold maceration application in red wine production and its effects on phenolic compounds: A review. LWT Food Sci. Technol. 2018, 95, 200–208.
- Santesteban, L.G.; Miranda, C.; Royo, J.B. Influence of the freezing method on the changes that occur in grape samples after frozen storage. J. Sci. Food Agric. 2013, 93, 3010–3015.
- Olarte Mantilla, S.M.; Collins, C.; Iland, P.G.; Kidman, C.M.; Jordans, C.; Bastian, S.E.P. Comparison of sensory attributes of fresh and frozen wine grape berries using Berry Sensory Assessment. Aust. J. Grape Wine Res. 2013, 19, 349–357.
- Naviglio, D.; Formato, A.; Scaglione, G.; Montesano, D.; Pellegrino, A.; Villecco, F.; Gallo, M. Study of the grape cryo–maceration process at different temperatures. Foods 2018, 7, 107.
- Bestulić, E.; Rossi, S.; Plavša, T.; Horvat, I.; Lukić, I.; Bubola, M.; Peršurić, A.S.I.; Jeromel, A.; Radeka, S. Comparison of different maceration and non-maceration treatments for enhancement of phenolic composition, colour intensity, and taste attributes of Malvazija istarska (Vitis vinifera L.) white wines. J. Food Compos. Anal. 2022, 109, 104472.
- Naranjo, A.; Martínez-Lapuente, L.; Ayestarán, B.; Guadalupe, Z.; Pérez, I.; Canals, C.; Adell, E. Aromatic and sensory characterization of Maturana Blanca wines made with different technologies. Beverages 2021, 7, 10.
- Robinson, A.L.; Boss, P.K.; Solomon, P.S.; Trengove, R.D.; Heymann, H.; Ebeler, S.E. Origins of grape and wine aroma. Part 1. Chemical components and viticultural impacts. Am. J. Enol. Vitic. 2014, 65, 1–24.
- Coetzee, C.; du Toit, W.J. A comprehensive review on Sauvignon blanc aroma with a focus on certain positive volatile thiols. Food Res. Int. 2012, 45, 287–298.
- Pedrosa-López, M.D.C.; Aragón-García, F.; Ruíz-Rodríguez, A.; Piñeiro, Z.; Durán-Guerrero, E.; Palma, M. Effects from the freezing of either whole or crushed grapes on the volatile compounds contents in Muscat wines. Foods 2022, 11, 1782.
- Carillo, M.; Formato, A.; Fabiani, A.; Scaglione, G.; Pucillo, G.P. An inertizing and cooling process for grapes cryomaceration. Electron. J. Biotechnol. 2011, 14, 2–14.
- JagatiÄ, A.M.; Prusina, T.; IviÄ, S. Influence of cold maceration treatment on aromatic and sensory properties of Vugava wine (Vitis vinifera L.). J. Microbiol. Biotechnol. Food Sci. 2020, 10, 49–53.
- Cataldo, E.; Salvi, L.; Paoli, F.; Fucile, M.; Mattii, G.B. Effect of agronomic techniques on aroma composition of white grapevines: A Review. Agronomy 2021, 11, 2027.
- Chen, K.; Li, J. A glance into the aroma of white wine. In White Wine Technology; Morata, A., Ed.; Academic Press: Cambridge, MA, USA, 2022; pp. 313–326.
- October, F.M. Effect of Yeasts and Oenological Parameters on Acetaldehyde Production during Alcoholic Fermentation of South African Grape Musts. Master’s Thesis, Stellenbosch University, Stellenbosch, South Africa, 2020.
- Pinu, F.R.; Edwards, P.J.; Jouanneau, S.; Kilmartin, P.A.; Gardner, R.C. Villas-Boas, S.G. Sauvignon blanc metabolomics: Grape juice metabolites affecting the development of varietal thiols and other aroma compounds in wines. Metabolomics 2014, 10, 556–573.
- Ruiz, J.; Kiene, F.; Belda, I.; Fracassetti, D.; Marquina, D.; Navascués, E.; Calderón, F.; Benito, A.; Rauhut, D.; Santos, A.; et al. Effects on varietal aromas during wine making: A review of the impact of varietal aromas on the flavor of wine. Appl. Microbiol. Biotechnol. 2019, 103, 7425–7450.
- Moreno-Pérez, A.; Vila-López, R.; Fernández-Fernández, J.I.; Martínez-Cutillas, A.; Gil-Muñoz, R. Influence of cold pre-fermentation treatments on the major volatile compounds of three wine varieties. Food Chem. 2013, 139, 770–776.
- Aleixandre-Tudo, J.L.; Weightman, C.; Panzeri, V.; Nieuwoudt, H.H.; Du Toit, W.J. Effect of skin contact before and during alcoholic fermentation on the chemical and sensory profile of South African Chenin blanc white wines. S. Afr. J. Enol Vitic. 2015, 36, 366–377.
- Olejar, K.J.; Fedrizzi, B.; Kilmartin, P.A. Influence of harvesting technique and maceration process on aroma and phenolic attributes of Sauvignon blanc wine. Food Chem. 2015, 183, 181–189.
- Ferreira, V.; Lopez, R. The actual and potential aroma of winemaking grapes. Biomolecules 2019, 9, 818.
- Ouellet, É.; Pedneault, K. Impact of frozen storage on the free volatile compound profile of grape berries. Am. J. Enol. Vitic. 2016, 67, 239–244.
- Bonnaffoux, H.; Delpech, S.; Rémond, E.; Schneider, R.; Roland, A.; Cavelier, F. Revisiting the evaluation strategy of varietal thiol biogenesis. Food Chem. 2018, 268, 126–133.
- Wilson, C.L. Chemical Evaluation and Sensory Relevance of Thiols in South African Chenin Blanc Wines. Ph.D. Thesis, Stellenbosch University, Stellenbosch, South Africa, 2017. Available online: https://hdl.handle.net/10019.1/101250 (accessed on 15 March 2021).
- Alti-Palacios, L.; Martínez, J.; Teixeira, J.A.; Câmara, J.S.; Perestrelo, R. Influence of cold pre-fermentation maceration on the volatilomic pattern and aroma of white wines. Foods 2023, 12, 1135.
- Ruiz-Rodríguez, A.; Durán-Guerrero, E.; Natera, R.; Palma, M.; Barroso, C.G. Influence of two different cryoextraction procedures on the quality of wine produced from muscat grapes. Foods 2020, 9, 1529.
- Hart, R.S.; Ndimba, B.K.; Jolly, N.P. Characterisation of thiol-releasing and lower volatile acidity-forming intra-genus hybrid yeast strains for Sauvignon blanc wine. S. Afr. J. Enol. Vitic. 2017, 38, 144–155.
- Chen, L.; Capone, D.L.; Nicholson, E.L.; Jeffery, D.W. Investigation of intraregional variation, grape amino acids, and pre-fermentation freezing on varietal thiols and their precursors for Vitis vinifera Sauvignon blanc. Food Chem. 2019, 295, 637–645.
- Benkwitz, F.; Nicolau, L.; Lund, C.; Beresford, M.; Wohlers, M.; Kilmartin, P.A. Evaluation of key odorants in Sauvignon blanc wines using three different methodologies. J. Agric. Food Chem. 2012, 60, 6293–6302.
- Coetzee, C.; Schulze, A.; Mokwena, L.; Du Toit, W.J.; Buica, A. Investigation of thiol levels in young commercial South African Sauvignon blanc and Chenin blanc wines using propiolate derivatization and GC-MS/MS. S. Afr. J. Enol. Vitic. 2018, 39, 180–184.
- Wilson, C.; Brand, J.; du Toit, W.; Buica, A. Matrix effects influencing the perception of 3-mercaptohexan-1-ol (3MH) and 3-mercaptohexyl acetate (3MHA) in different Chenin blanc wines by Projective Mapping (PM) with Ultra Flash profiling (UFP) intensity ratings. Food Res. Int. 2019, 121, 633–640.
- Hart, R.S.; Jolly, N.P.; Ndimba, B.K. Characterisation of hybrid yeasts for the production of varietal Sauvignon blanc wine-A review. J. Microbiol. Methods 2019, 165, 105699.
- Hart, R.S.; Jolly, N.P.; Mohamed, G.; Booyse, M.; Ndimba, B.K. Characterisation of Saccharomyces cerevisiae hybrids selected for low volatile acidity formation and the production of aromatic Sauvignon blanc wine. Afr. J. Biotechnol. 2016, 15, 2068–2081.
- Visan, L.; Tamba-Berehoiu, R.M.; Popa, C.N.; Danaila–Guidea, S.M.; Culea, R. Aromatic compounds in wines. Sci. Papers 2018, 18, 423–430.
- Dimopoulou, M.; Troianou, V.; Toumpeki, C.; Gosselin, Y.; Dorignac, É.; Kotseridis, Y. Effect of strains from different Saccharomyces species used in different inoculation schemes on chemical composition and sensory characteristics of Sauvignon blanc wine. OENO One 2020, 54, 745–759.
- Capone, D.L.; Jeffery, D.W. Effects of transporting and processing Sauvignon blanc grapes on 3-mercaptohexan-1-ol precursor concentrations. J. Agric. Food Chem. 2011, 59, 4659–4667.
- Capone, D.L.; Ristic, R.; Pardon, K.H.; Jeffery, D.W. Simple quantitative determination of potent thiols at ultratrace levels in wine by derivatization and high-performance liquid chromatography-tandem mass spectrometry (HPLC-MS/MS) analysis. Anal. Chem. 2015, 87, 1226–1231.
- Jeffery, D.W. Spotlight on varietal thiols and precursors in grapes and wines. Aust. J. Chem. 2016, 69, 1323–1330.
- Casassa, L.F.; Sari, S.E. Sensory and chemical effects of two alternatives of pre-fermentative cold soak in Malbec wines during winemaking and bottle ageing. Int. J. Food Sci. Technol. 2015, 50, 1044–1055.
- Suklje, K.; Gobler, N.; Coetzee, Z.; Lisjak, K.; Deloire, A. Methoxypyrazines and Greenness in Wines: Myth or Reality? A Few Perspectives. Wineland. 2013. Available online: https://www.wineland.co.za/methoxypyrazines-greenness-wines-myth-reality-perspectives/ (accessed on 2 December 2021).
- Sidhu, D.; Lund, J.; Kotseridis, Y.; Saucier, C. Methoxypyrazine analysis and influence of viticultural and enological procedures on their levels in grapes, musts, and wines. Crit. Rev. Food Sci. Nutr. 2015, 55, 485–502.
- Lei, Y.; Xie, S.; Guan, X.; Song, C.; Zhang, Z.; Meng, J. Methoxypyrazines biosynthesis and metabolism in grape: A review. Food Chem. 2018, 245, 1141–1147.
- Aleixandre-Tudo, J.L.; Alvarez, I.; Lizama, V.; Nieuwoudt, H.; Garcia, M.J.; Aleixandre, J.L.; Du Toit, W.J. Modelling phenolic and volatile composition to characterize the effects of pre-fermentative cold soaking in Tempranillo wines. LWT Food Sci. Technol. 2016, 66, 193–200.
- Baron, M.; Prusova, B.; Tomaskova, L.; Kumsta, M.; Sochor, J. Terpene content of wine from the aromatic grape variety ‘Irsai Oliver’ (Vitis vinifera L.) depends on maceration time. Open Life Sci. 2017, 12, 42–50.
- Vilanova, M.; Escudero, A.; Graña, M.; Cacho, J. Volatile composition and sensory properties of Northwest Spain white wines. Food Res. Int. 2013, 54, 562–568.
- Robinson, A.L.; Boss, P.K.; Solomon, P.S.; Trengove, R.D.; Heymann, H.; Ebeler, S.E. Origins of grape and wine aroma. Part 2. Chemical and sensory analysis. Am. J. Enol. Vitic. 2014, 65, 25–42.
- Karabagias, I.K.; Sykalia, D.; Mannu, A.; Badeka, A.V. Physico–chemical parameters complemented with aroma compounds fired up the varietal discrimination of wine using statistics. Eur. Food Res. Technol. 2020, 246, 2233–2248.
- Baiano, A.; Terracone, C.; Longobardi, F.; Ventrella, A.; Agostiano, A.; Del Nobile, M.A. Effects of different vinification technologies on physical and chemical characteristics of Sauvignon blanc wines. Food Chem. 2012, 135, 2694–2701.
- Malićanin, M.; Danilović, B.; Stamenković Stojanović, S.; Cvetković, D.; Lazić, M.; Karabegović, I.; Savić, D. Pre-Fermentative Cold Maceration and Native Non-Saccharomyces Yeasts as a Tool to Enhance Aroma and Sensory Attributes of Chardonnay Wine. Horticulturae 2022, 8, 212.
- Benucci, I.; Cerreti, M.; Liburdi, K.; Nardi, T.; Vagnoli, P.; Ortiz-Julien, A.; Esti, M. Pre-fermentative cold maceration in presence of non-Saccharomyces strains: Evolution of chromatic characteristics of Sangiovese red wine elaborated by sequential inoculation. Food Res. Int. 2018, 107, 257–266.





