Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Hideki Kawanishi | -- | 1556 | 2024-02-23 03:26:12 | | | |
| 2 | Peter Tang | Meta information modification | 1556 | 2024-02-23 09:03:34 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Kawanishi, H. Middle Molecular Uremic Toxin and Blood Purification Therapy. Encyclopedia. Available online: https://encyclopedia.pub/entry/55372 (accessed on 21 May 2026).
Kawanishi H. Middle Molecular Uremic Toxin and Blood Purification Therapy. Encyclopedia. Available at: https://encyclopedia.pub/entry/55372. Accessed May 21, 2026.
Kawanishi, Hideki. "Middle Molecular Uremic Toxin and Blood Purification Therapy" Encyclopedia, https://encyclopedia.pub/entry/55372 (accessed May 21, 2026).
Kawanishi, H. (2024, February 23). Middle Molecular Uremic Toxin and Blood Purification Therapy. In Encyclopedia. https://encyclopedia.pub/entry/55372
Kawanishi, Hideki. "Middle Molecular Uremic Toxin and Blood Purification Therapy." Encyclopedia. Web. 23 February, 2024.
Copy Citation
The purpose of blood purification therapy is to remove uremic toxins, and middle molecules (MMs) are a specific target. An MM is defined as a solute that passes through the glomerulus with a molecular weight in the range of 0.5–58 kDa, and new classifications of “small-middle 0.5–15 kDa,” “medium-middle 15–25 kDa,” and “large-middle 25–58 kDa” were proposed.
blood purification
middle molecules
hemodiafiltration
MCO-membrane
α1-microglobulin
1. Introduction
The objective of blood purification in the treatment of patients with kidney failure or end-stage renal disease is to eliminate uremic toxins from the blood. Chronic dialysis therapy, the primary treatment for kidney failure, was pioneered in the 1960s by the Scribner Group in Seattle [1]. Uremic toxins are waste products that accumulate in the body when the kidneys can no longer effectively filter and excrete them. Various techniques, including hemodialysis (HD) and the removal of uremic toxins, have been developed to achieve this goal. Online hemodiafiltration (HDF), an advanced blood purification technique, was introduced by Henderson et al. [2]. However, the clinical application of online HDF has been limited in the U.S. [3] and has predominantly advanced in Europe [4][5]. Recently, there has been a growing number of patients in Japan and other Asian countries opting for this treatment [5].
The definition of uremic toxins has been a subject of debate since the 1970s [6]. In 2003, the EUROPEAN UREMIC TOXIN WORK GROUP (EUTOX), led by Vanholder et al., provided a comprehensive definition and classification [7]. According to them, solutes were categorized as free water-soluble low molecular weight solutes (small solutes), middle molecules, and protein-bound solutes. Additionally, they assessed the level of evidence for these uremic toxins [8]. Dialysis therapy, based on membrane separation technology, targets small to middle molecules. However, middle molecules (MMs), with molecular weights ranging from 0.5 to 58 kD, are widely distributed, and a diverse array of target substances has caused some confusion, including in terminology. A new classification was proposed in 2021 to clarify middle molecules [9].
2. Middle Molecules and the Blood Purification Setting (Figure 1)
The removal efficiency of HDF is primarily determined by the volume of replacement fluid, although removal characteristics vary depending on the dilution method employed. Post-dilution HDF, when combined with a protein-non-leakage hemodiafilter, demonstrates efficacy in the removal of small-middle molecules such as βMG. In contrast, for the targeting of medium to larger-middle molecules (e.g., κ-free light chain (FLC) 22 kDa, λ-FLCα1 44 kDa, α1-microglobulin (αMG), 33 kDa), a protein-leakage type hemodiafilter is preferred. However, the drawback of excessive albumin leakage in post-dilution HDF arises due to the challenges in effectively separating albumin from larger-middle substances. In such instances, pre-diluted HDF, capable of discriminating between albumin and larger-middle molecules, is the preferred choice.
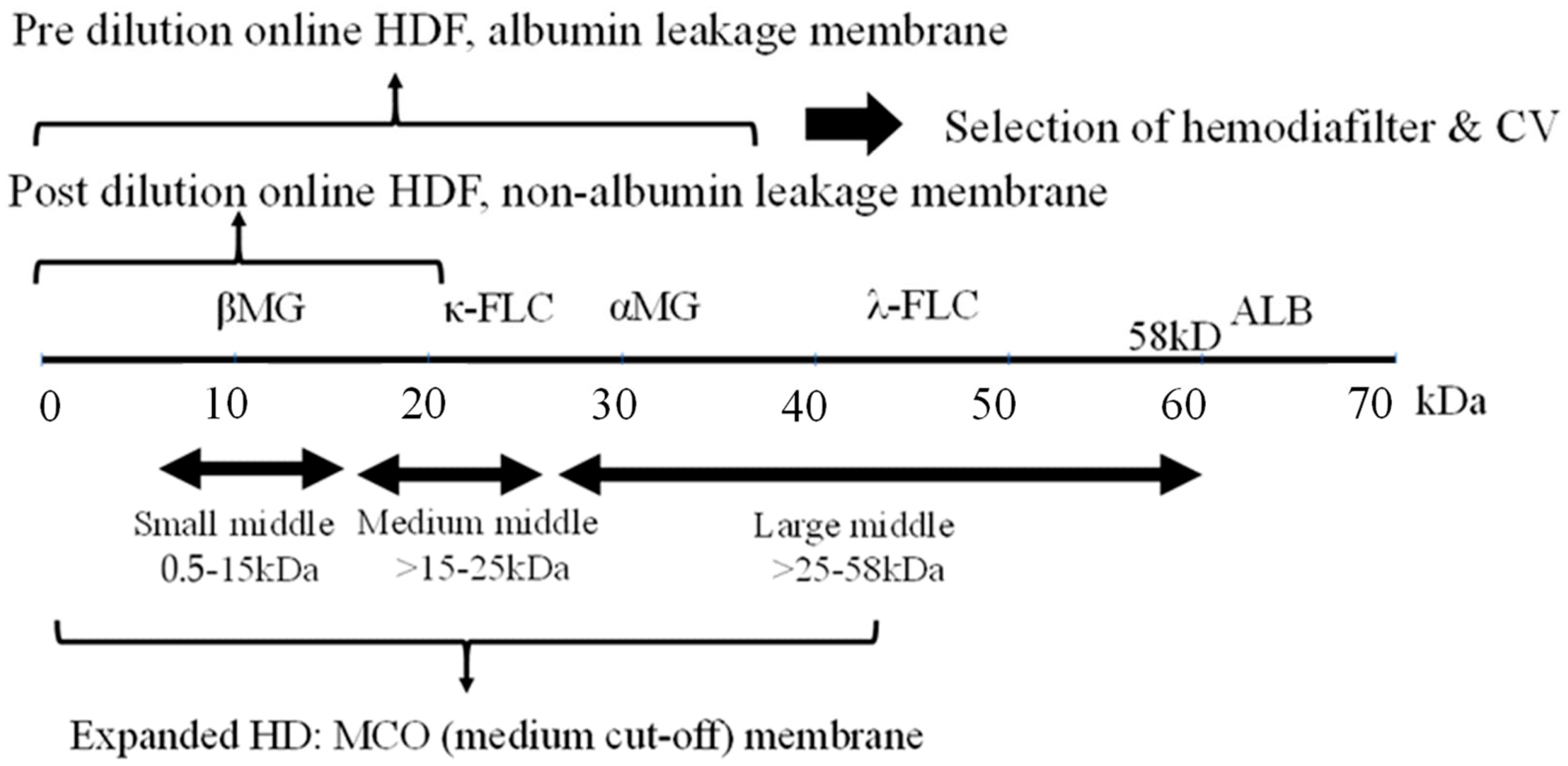
Figure 1. Selection of blood purification modality on middle molecules. Definition of middle molecules (MM)s: MWs 0.5 to <58 KD. CV: convection volume, βMG: β2 microglobulin, FLC: free light chain, ALB: albumin.
As a new classification of MM, large pore membrane dialyzers have been used for the removal by HD in the medium to large-middle region [10]. Initially, a high cutoff (HCO) membrane (average pore size 10 nm) was used to remove FLC (κ- 22 kDa, λ- 44 kDa), the causative agent of renal damage due to multiple myeloma, but albumin loss was excessive and it was difficult to use in common HD.
Subsequently, medium cutoff (MCO) membranes (average pore size around 5 nm) were developed and are widely used. Terminology of expanded HD is also used in some cases to indicate that MM removal is more efficient than conventional HD. These MCO membrane dialyzers have about the same performance as protein-leakage filters (dialyzers) in Japan, which means that the super high-flux-albumin leaking dialyzer therapy in Japan is now accepted in Europe, where protein leakage has tended to be denied. Comparisons between this MCO membrane HD and post-dilution HDF have also been made [11], but at present only the name MCO precedes it and no clear criteria have been established. The new definition of MMs requires standards based on a set range of substances to be removed.
3. Impact of Highly Efficient Blood Purification on Patient Survival
3.1. Impact on HDF for Survival
The patient survival outcomes associated with HDF have been subject to extensive review [5]. The most recent investigations have focused on the survival effects of high CV achieved through post-dilution online HDF in European settings. Randomized controlled trials (RCTs), including the Dutch CONTRAST trial [12], comparing post-dilution HDF versus low flux HD, and the Turkish study [13], ESHOL study [14], and French study [15], which all compare post-dilution HDF versus high-flux HD, have collectively reported that augmenting CV with HDF (with CV values of 22 L [12], 17.2 L [13], 20.7 L [14], and 22.9–23.9 L [15] in each respective study) demonstrates a proportionate enhancement in patient survival.
However, it is essential to note that these RCTs did not explicitly establish CV as a designated treatment objective and did not definitively account for the potential presence of confounding factors. In cases where a higher CV might have been attained, such as in individuals characterized as “healthy” with fewer comorbidities, the presence of native vascular access, and higher blood flow, the risk of mortality was observed to be lower. It is worth acknowledging that these factors were not systematically controlled for in the study design.
Furthermore, the relatively low mean age of the study participants (53 years) and the absence of comprehensive information regarding the selection process and participating centers raised questions about the generalizability of the findings to the broader dialysis population. Consequently, it remained uncertain whether the procedure was universally applicable to all dialysis patients [16]. The theoretical significance of increased solute removal as a determinant of improved life expectancy also warranted further clarification, as no specific upper limit for CV had been proposed.
Conversely, an observational study conducted using Euro-DOPPS 4–5 failed to demonstrate a discernible advantage of HDF [17]. Likewise, an RCT in Australia, comparing post-dilution HDF with a CV of 24.7 L to high-flux HD, did not reveal any substantial survival benefit or improvements in neurological symptoms [18]. It is plausible that the prolonged dialysis duration (5 h) and the favorable survival rates (1-year survival exceeding 90%) in this particular study may have contributed to the absence of differential outcomes.
To address the limitations observed in prior HDF studies, two extensive RCTs were conducted in Europe [16]: the CONVINCE study (Comparison of high-dose HDF with high-flux HD) and the H4RT (High-volume HDF versus High-flux HD Registry Trial). These trials encompassed not only assessments of life expectancy but also explored outcomes related to hospitalization, quality of life, and cost-effectiveness.
In the primary analysis of CONVINCE conducted in 2023, a notable 33% reduction in all-cause mortality over a span of 3 years was reported in the high-dose HDF group (CV > 23 L) comprising 683 patients in comparison to the high-flux HD group with 677 patients. Subsequent sub-analyses highlighted specific benefits, particularly among patients without cardiovascular disease (CVD) or diabetes [19].
Nevertheless, it is essential to acknowledge that this RCT coincided with the COVID-19 pandemic, which could potentially have impacted hospitalization rates and other variables. Ongoing investigations and further analyses are slated for the future to gain a more comprehensive understanding of these findings [20].
3.2. Efficacy of Expanded HD
A meta-analysis examining the efficacy of expanded HD revealed superior clearance of medium to large-middle molecules, such as κ/λ FLC, in comparison to high-flux HD and post-dilution HDF. Additionally, it was associated with reduced albumin loss when contrasted with HDF [21]. However, an equivalent meta-analysis focusing on HDF did not demonstrate a survival advantage for expanded HD [22]. This underscores the continued scarcity of long-term survival evidence for expanded HD, as supported by the limited number of studies or randomized controlled trials available to date. Consequently, additional research is warranted to address this knowledge gap and provide further insights into its potential benefits.
4. The Functional Classification of Dialyzer
The functional classification of dialyzers in Japan was initially established by the JSDT. In 1996, it was categorized into Type I and Type II based on in vitro βMG clearance. Subsequently, in 2006, the classification was expanded to encompass Types I-IV. In 2013, further refinement was achieved with the adoption of Type Ia, Ib, IIa, and IIb classifications, which integrated in vitro albumin sieving coefficient (SC) along with βMG clearance.
An analysis of the JRDR utilizing the 2006 classification framework revealed a significant 35% reduction in 3-year mortality among patients using Type V dialyzers when compared to Type IV dialyzers as the reference group. This result emphasizes the effectiveness of employing the so-called super high-flux membranes [23].
As a consequence of the development of MCO membranes and the increasing adoption of Japanese protein-leak dialyzers, predominantly in Europe since approximately 2015, the Storr classification was introduced. This classification incorporated albumin permeability alongside βMG [24]. This framework aligns with the principles outlined in the 2013 Japanese classification.
In line with this evolution, the extracorporeal blood purification meeting in November 2022 Rome introduced a classification system. This classification categorizes patients based on treatment modality (rather than membrane permeability) into low-flux HD, high-flux HD, and expanded HD, with albumin permeability serving as an additional indicator [25].
Japan has taken a leading role in the development of functional classifications for dialyzers, with Japanese dialyzers exhibiting superior performance in comparison to those from other regions [23][26]. It is of paramount importance to determine whether this enhanced performance translates into improved clinical outcomes. A prospective observational cohort study titled “Japanese study of the effects of α1-microglobulin reduction rates on survival; JAMREDS” [27] has recently been initiated. This study aims to investigate the prognosis and occurrence of cardiovascular events in dialysis patients, with the goal of elucidating the theory behind the regeneration of α1-microglobulin’s antioxidant function [28].
References
- Scribner, B.H.; Caner, J.E.; Buri, R.; Quinton, W. The technique of continous hemodialysis. Trans. Am. Soc. Artif. Intern. Organs 1960, 6, 88–103.
- Henderson, L.W.; Sanfelippo, M.L.; Beans, E. “On line” preparation of sterile pyrogen-free electrolyte solution. Trans. Am. Soc. Artif. Intern. Organs 1978, 24, 465–467.
- Ward, R.A.; Vienken, J.; Silverstein, D.M.; Ash, S.; Canaud, B. Regulatory Considerations for Hemodiafiltration in the United States. Clin. J. Am. Soc. Nephrol. 2018, 13, 1444–1449.
- Rindi, P.; Pilone, N.; Riccò, V.; Cioni, L. Clinical experience with a new hemodiafiltration (HDF) system. ASAIO Trans. 1988, 34, 765–768.
- Canaud, B.; Köhler, K.; Sichart, J.M.; Möller, S. Global prevalent use, trends and practices in haemodiafiltration. Nephrol. Dial. Transplant. 2020, 35, 398–407.
- Bergström, J.; Fürst, P. Uremic toxins. Kidney Int. Suppl. 1978, S9–S12.
- Vanholder, R.; De Smet, R.; Glorieux, G.; Argilés, A.; Baurmeister, U.; Brunet, P.; Clark, W.; Cohen, G.; De Deyn, P.P.; Deppisch, R.; et al. Review on uremic toxins: Classification, concentration, and interindividual variability. Kidney Int. 2003, 63, 1934–1943.
- Vanholder, R.; Pletinck, A.; Schepers, E.; Glorieux, G. Biochemical and Clinical Impact of Organic Uremic Retention Solutes: A Comprehensive Update. Toxins 2018, 10, 33.
- Rosner, M.H.; Reis, T.; Husain-Syed, F.; Vanholder, R.; Hutchison, C.; Stenvinkel, P.; Blankestijn, P.J.; Cozzolino, M.; Juillard, L.; Kashani, K.; et al. Classification of uremic toxins and their role in kidney failure. Clin. J. Am. Soc. Nephrol. 2021, 16, 1918–1928.
- Ronco, C.; Marchionna, N.; Brendolan, A.; Neri, M.; Lorenzin, A.; Martínez Rueda, A.J. Expanded haemodialysis: From operational mechanism to clinical results. Nephrol. Dial. Transplant. 2018, 33 (Suppl. 3), iii41–iii47.
- Maduell, F.; Rodas, L.; Broseta, J.J.; Gomez, M.; Xipell, M.; Guillen, E.; Montagud-Marrahi, E.; Arias-Guillén, M.; Fontseré, N.; Vera, M.; et al. Medium cut-off dialyzer versus eight hemodiafiltration dialyzers: Comparison using a global removal score. Blood Purif. 2019, 48, 167–174.
- Grooteman, M.P.; van den Dorpel, M.A.; Bots, M.L.; Penne, E.L.; van der Weerd, N.C.; Mazairac, A.H.; den Hoedt, C.H.; van der Tweel, I.; Lévesque, R.; Nubé, M.J.; et al. Effect of Online Hemodiafiltration on All-Cause Mortality and Cardiovascular Outcomes. J. Am. Soc. Nephrol. 2012, 23, 1087–1096.
- Ok, E.; Asci, G.; Toz, H.; Ok, E.S.; Kircelli, F.; Yilmaz, M.; Hur, E.; Demirci, M.S.; Demirci, C.; Duman, S.; et al. Mortality and cardiovascular events in online haemodiafiltration (OL-HDF) compared with high-flux dialysis: Results from the Turkish OL-HDF Study. Nephrol. Dial. Transplant. 2013, 28, 192–202.
- Maduell, F.; Moreso, F.; Pons, M.; Ramos, R.; Mora-Macià, J.; Carreras, J.; Soler, J.; Torres, F.; Campistol, J.M.; Martinez-Castelao, A. High-Efficiency Postdilution Online Hemodiafiltration Reduces All-Cause Mortality in Hemodialysis Patients. J. Am. Soc. Nephrol. 2013, 24, 487–497.
- Morena, M.; Jaussent, A.; Chalabi, L.; Leray-Moragues, H.; Chenine, L.; Debure, A.; Thibaudin, D.; Azzouz, L.; Patrier, L.; Maurice, F.; et al. Treatment tolerance and patient-reported outcomes favor online hemodiafiltration compared to high-flux hemodialysis in the elderly. Kidney Int. 2017, 91, 1495–1509.
- Vernooij, R.W.M.; Bots, M.L.; Strippoli, G.F.M.; Canaud, B.; Cromm, K.; Woodward, M.; Blankestijn, P.J. CONVINCE scientific committee. CONVINCE in the context of existing evidence on haemodiafiltration. Nephrol. Dial. Transplant. 2022, 37, 1006–1013.
- Locatelli, F.; Karaboyas, A.; Pisoni, R.L.; Robinson, B.M.; Fort, J.; Vanholder, R.; Rayner, H.C.; Kleophas, W.; Jacobson, S.H.; Combe, C.; et al. Mortality risk in patients on hemodiafiltration versus hemodialysis: A ‘real-world’ comparison from the DOPPS. Nephrol. Dial. Transplant. 2018, 33, 683–689.
- Kang, A.; Arnold, R.; Gallagher, M.; Snelling, P.; Green, J.; Fernando, M.; Kiernan, M.C.; Hand, S.; Grimley, K.; Burman, J.; et al. Effect of Hemodiafiltration on the Progression of Neuropathy with Kidney Failure: A Randomized Controlled Trial. Clin. J. Am. Soc. Nephrol. 2021, 16, 1365–1375.
- Blankestijn, P.J.; Vernooij, R.W.M.; Hockham, C.; Strippoli, G.F.M.; Canaud, B.; Hegbrant, J.; Barth, C.; Covic, A.; Cromm, K.; Cucui, A.; et al. Effect of Hemodiafiltration or Hemodialysis on Mortality in Kidney Failure. N. Engl. J. Med. 2023, 389, 700–709.
- Shroff, R.; Basile, C.; van der Sande, F.; Mitra, S. Haemodiafiltration for all: Are we CONVINCE? Nephrol. Dial. Transplant. 2023, 38, 2663–2665.
- Zhao, Y.; Gan, L.; Niu, Q.; Ni, M.; Zuo, L. Efficacy and safety of expanded hemodialysis in hemodialysis patients: A meta-analysis and systematic review. Ren. Fail. 2022, 44, 541–550.
- Mitchell, C.R.; Hornig, C.; Canaud, B. Systematic review to compare the outcomes associated with the modalities of expanded hemodialysis (HDx) versus high-flux hemodialysis and/or hemodiafiltration (HDF) in patients with end-stage kidney disease (ESKD). Semin. Dial. 2023, 36, 86–106.
- Abe, M.; Masakane, I.; Wada, A.; Nakai, S.; Nitta, K.; Nakamoto, H. Super high-flux membrane dialyzers improve mortality in patients on hemodialysis: A 3-year nationwide cohort study. Clin. Kidney J. 2021, 15, 473–483.
- Storr, M.; Ward, R.A. Membrane innovation: Closer to native kidneys. Nephrol. Dial. Transplant. 2018, 33, iii22–iii27.
- Meijers, B.; Vega, A.; Juillard, L.; Kawanishi, H.; Kirsch, A.H.; Maduell, F.; Massy, Z.A.; Mitra, S.; Vanholder, R.; Ronco, C.; et al. Extracorporeal Techniques in End-Stage Kidney Disease. Blood Purif. 2023, 2, 929–944.
- Saito, A. Definition of high-performance membranes—From the clinical point of view. Contrib. Nephrol. 2011, 173, 1–10.
- Naganuma, T.; Takemoto, Y.; Kamada, N.; Kawanishi, H. Hemodiafiltration in Japan: Current status and future directions. Ren. Replace. Ther. 2023, 9, 23.
- Kawanishi, H. Development of online hemodiafiltration in Japan. Ren. Replace. Ther. 2021, 7, 51.
More
Information
Subjects:
Urology & Nephrology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
952
Revisions:
2 times
(View History)
Update Date:
23 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





