Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Masaru Tanaka | -- | 1707 | 2024-02-21 12:08:11 | | | |
| 2 | Wendy Huang | Meta information modification | 1707 | 2024-02-22 06:11:32 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Jászberényi, M.; Thurzó, B.; Bagosi, Z.; Vécsei, L.; Tanaka, M. Role of Orexins in Regulation of Stress Response. Encyclopedia. Available online: https://encyclopedia.pub/entry/55292 (accessed on 26 May 2026).
Jászberényi M, Thurzó B, Bagosi Z, Vécsei L, Tanaka M. Role of Orexins in Regulation of Stress Response. Encyclopedia. Available at: https://encyclopedia.pub/entry/55292. Accessed May 26, 2026.
Jászberényi, Miklós, Balázs Thurzó, Zsolt Bagosi, László Vécsei, Masaru Tanaka. "Role of Orexins in Regulation of Stress Response" Encyclopedia, https://encyclopedia.pub/entry/55292 (accessed May 26, 2026).
Jászberényi, M., Thurzó, B., Bagosi, Z., Vécsei, L., & Tanaka, M. (2024, February 21). Role of Orexins in Regulation of Stress Response. In Encyclopedia. https://encyclopedia.pub/entry/55292
Jászberényi, Miklós, et al. "Role of Orexins in Regulation of Stress Response." Encyclopedia. Web. 21 February, 2024.
Copy Citation
The orexin/hypocretin neuropeptide family has emerged as a focal point of neuroscientific research following the discovery that this family plays a crucial role in a variety of physiological and behavioral processes. These neuropeptides serve as powerful neuromodulators, intricately shaping autonomic, endocrine, and behavioral responses across species. Notably, they serve as master regulators of vigilance and stress responses; however, their roles in food intake, metabolism, and thermoregulation appear complementary and warrant further investigation.
orexins
neuropeptides
stress response
hypocretin
neuroendocrine
HPA axis
1. The Hypocretin/Orexin Peptide and Receptor Family
The hypocretin/orexin system represents an extremely complex neuropeptide network in the CNS [1][2]. The seminal papers [3][4][5] that dealt with the discovery of these ligands and their receptors also demonstrated the hyperphagic [3] and neuroexcitatory activity [4] of orexins and the unique distribution pattern of the system. The orexin/hypocretin system, similarly to melanin-concentrating hormone (MCH)-positive neurons [6], has a well-circumscribed expression in the hypothalamus (Figure 1) [4][7]. Its cell bodies are restricted to the lateral, dorsal, dorsomedial, and perifornical areas, and the whole population does not exceed 50,000–80,000 cells in the hypothalamus. However, its axon terminals reach distant regions, and its receptors are scattered throughout the whole CNS [5].
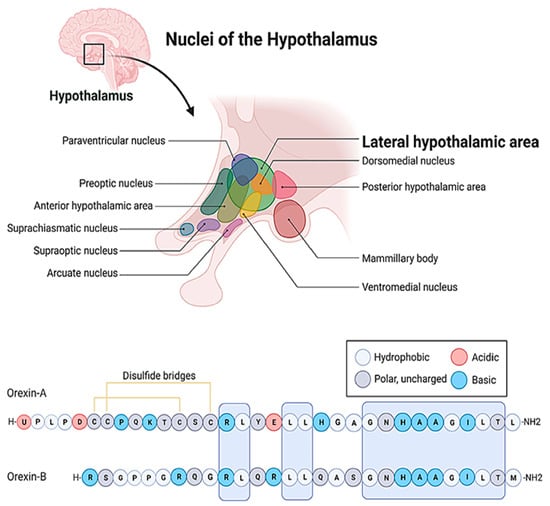
Figure 1. The localization of the lateral hypothalamic area and the amino acid sequences of the orexin/hypocretin family peptides. The letters stand for the one letter code of amino acids. A: Alanine, C: Cysteine, D: Aspartic acid, E: Glutamic acid, G: Glycine, H: Histidine, I: Isoleucine, K: Lysine, L: Leucine, M: Methionine, N: Asparagine, P: Proline, Q: Glutamine, R: Arginine, S: Serine, T: Threonine, U: Pyroglutamic acid, Y: Tyrosine.
At the cellular level, so far, two ligands (orexin-A, orexin-B) and two receptors (OX1R and OX2R) of the system have been described (Figure 1) [3][4]. The peptides biochemically belong to the incretin family, but they bear weak structural resemblance only to a few members of the group [3][4]. Even orexin-A and orexin-B differ by 50% of their primary structure. Both peptides are cleft from pre-pro-orexin (PPO) and are amidated C-terminally, but orexin-A is larger, comprising 33 amino acids, while orexin-B consists of only 28 residues [8]. Orexin-A is also less prone to proteolytic degradation because it comprises an N-terminal pyroglutamate residue and two disulfide bonds. Additionally, orexin-A is more hydrophobic, and therefore it can bypass more efficiently the blood–brain barrier (BBB) [9]. These orexins also exhibit significantly different receptor affinities [3][4][10], which is definitely attributed to the fact that the orexin receptors (OXRs) share only 64% amino acid identity [8][11]. The two receptor subtypes create diversity within the cellular signaling pathways [8][11][12][13][14]. Both OX1R and OX2R activity is mediated by Gq11, which, in turn, leads to the activation of phospholipase C (PLC), phospholipase A (PLA), and phospholipase D, ultimately resulting in an increase in cytosolic Ca2+ and the activation of protein kinase C (PKC). In addition, OX1R can elevate the intracellular Ca2+ level by activating non-selective cation channels (NSCCs) [11]. On the other hand, OX2R can also inhibit adenyl cyclase (AC) and protein kinase A (PKA) through the G-protein-coupled pathway. The potential dimerization of the OXRs and the structural overlap between OXRs and some other GPCRs lend further diversity to the signal transduction of the system [11]. For example, certain neuropeptide receptors, such as the type-2 neuropeptide-Y (NPY) receptor, the thyrotropin-releasing hormone (TRH) receptor, the cholecystokinin (CCK) type-A receptor, and the NK2 neurokinin receptor, show some similarities (26%, 25%, 23%, and 20% identity, respectively) to the orexin receptors [3]. The highest structural similarity is exhibited by the neuropeptide FF (NPFF) receptor of the RF-amide peptide family, which is 37% identical to OX1R and 35% identical to OX2R, respectively [15][16].
Neither the distribution of the immunoreactivity of the two orexins [17][18] nor the expression of OX1R [10][19][20][21] and OX2R [10][20][21][22] completely overlaps. This, together with the aforementioned distinct features of the pharmacokinetics of orexin peptides and the differences in the signal transduction of OX1R and OX2R [12][13][14], must be responsible for some divergence in the physiologic and pathophysiologic actions of orexin-A and orexin-B [18].
2. The Role of Orexins in the Regulation of the Stress Response
The reaction of our neuroendocrine regulation to adverse challenges is provided by the interaction between the sympathoadrenal (SA) system and the HPA axis [23]. Although they represent two distinct pathways, the line between them is frequently blurred, even in scientific literature. Perhaps this is due to their interwoven functions, as they complement each other’s activity while trying to maintain the homeostatic balance of challenged individuals. However, the SA response described by Cannon [24] is carried out according to the cooperation of the autonomic nervous system and the adrenal medulla, while the stress response, discovered by Selye [25], relies on the reaction of the HPA system, one of the central neuroendocrine axes later described by Schally, Guillemin [26], and Vale [27]. Unfortunately, by now, the terminology has been oversimplified, and stress response (though it has several stages) is frequently used as an umbrella term for both responses. Only in meticulous descriptions are these two neuroendocrine reactions clearly separated. To avoid confusion, for the HPA response, the most suitable term is the synonym (general adaptation syndrome: GAS) coined later by Selye [28]. Nonetheless, the distinction between the two pathways is of crucial importance because it helps clarify many contradictions in the literature. Some conflicting responses to certain stress paradigms could be easily resolved by clear discrimination between the two potential targets of adverse stimuli, that is, the SA system and the HPA axis.
It is well known that many neuropeptides modulate the activity of the HPA axis. For instance, NPY, neurotensin (NT), ghrelin, apelin, and endomorphins activate [29][30][31][32][33][34][35] while oxytocin and natriuretic peptides inhibit the system [36][37][38][39]. The output of the HPA axis is quite uniform: it begins with the pituitary translation and cleavage of pro-opiomelanocortin (POMC), yielding adrenocorticotropic hormone (ACTH), which, upon secretion, stimulates glucocorticoid release from the adrenal cortex [25][28]. However, in sharp contrast with the output, the input of the HPA axis is extremely diverse and involves a multitude of neuropeptides in signal transduction [33]. Therefore, it is not surprising that the modality (systemic or neurogenic) and schedule (acute, repeated, or chronic) of the stressors strongly influence the extent of the HPA response [40]. Systemic challenges (e.g. osmotic, immune, etc.) perturb the homeostatic balance of the organism, which is directly projected to the brainstem, while neurogenic paradigms (fear, pain) are processed by the cerebral centers [23]. The responses to these two types of challenges are signaled in a dichotomized manner in the brain. The corticotrope-releasing hormone (CRH)-positive neurons of the PVN are responsible for the acute and processed stimuli, while parvocellular arginine vasopressin (AVP) cells in the PVN and the SON maintain responsiveness to chronic, repeated, and homeostatic challenges [41]. It is also worth noting that neuropeptide modulation perfectly complements the built-in brakes of the GAS: the stepwise ultrashort, short, and long loop feedback mechanisms provided by CRH, ACTH, and the glucocorticoids, as well as the potent anti-inflammatory action of the glucocorticoids [23]. These mechanisms are called stress coping or stress resilience, and they harness the severe inflammatory response (SIRS), which otherwise could consume the organism [23][42][43].
As far as the effect of orexins on the HPA axis and the SA system is concerned, the two responses work hand in hand. Namely, in both responses, orexins play a predominantly stimulatory role [44][45]. However, according to the data from the literature, they are stimulated separately. It seems that the SA system is uniformly activated by orexin-A, which stimulates the OX1Rs expressed in the neurons of the nucleus of the solitary tract (NST), the LC, and the sympathetic neurons [44][46][47][48][49][50][51]. Ultimately, it is not a far-fetched idea to state that the perifornical, dorsal, dorsomedial, and lateral hypothalamic foci of orexin-positive neurons can be identified with those in the caudal hypothalamic region, which were demonstrated to be essential for an intact “fight or flight” and “sham rage” response by Philip Bard and Walter Hess [52][53].
However, as for the HPA axis, the picture is more complex. Soon after the discovery of the dense orexinergic innervation of the hypothalamic centers (PVN and SON) of the GAS, the scientific rivalry surrounding this highly coveted topic begot several important papers, which established that orexin neurons can activate the HPA axis predominantly at the hypothalamic level [47][54][55]. The main targets of the orexin neurons are the OX2Rs [56] expressed in the CRH-positive perikarya of the PVN [5][55]. Nonetheless, later publications showed that the connection between the orexin- and CRH-positive neuron population is bidirectional since abundant CRH-positive fibers land in the orexinergic perikarya of the hypothalamus [57][58][59]. Apparently, orexin-evoked HPA activation also involves the release of noradrenaline and NPY [60][61][62][63], which can significantly diversify its processing [42][43].
As far as the input of the HPA axis is concerned, the activity of orexins appears to be stressor- and schedule-specific [44][45]. In an acute setting, the challenges processed with heightened arousal (aversive odors, novelty, and contextual fear) give rise to more conspicuous activation of the orexin neurons (verified according to c-fos expression) than systemic challenges (e.g., cold exposure) or long-lasting procedures (e.g., restraint and immobilization) [44][45][64][65]. Nevertheless, while acute stress mostly activates the orexin neurons, experiments with chronic or repeated stressors returned mixed results [44][45], the findings of which may reflect an adaptation to unavoidable and permanent challenges. Further studies have revealed that the involvement of the orexins in the GAS depends on not only the modalities and schedule of the applied stressor but also the species and gender of the investigated subjects. Females and strains with better stress resilience phenotypes release less orexin in response to adverse stimuli [44][45].
Regarding the output of the HPA axis, orexins have been proven to stimulate the HPA axis not only at the hypothalamic but also at the pituitary and adrenal levels [44][45]. This finding is of crucial importance as peripheral activation stabilizes the HPA response to prolonged stimuli. It nurtures sufficient basal activity but also prevents an exaggerated hypothalamic response by maintaining negative feedback through the release of ACTH and glucocorticoids. Apparently, the orexin/hypocretin system also plays a crucial role in the cooperation and seamless integration of the GAS and the “fight or flight” response. Even the earliest publications which dealt with the orexin system demonstrated the dense innervation of the BNST, a limbic center, which harmonizes the activity of the SA system and the GAS [5]. Therefore, it is not unrealistic at all to conceive of orexins as the coordinators of the stress response to challenges with heightened arousal [44][45].
References
- Xia, L.; Liu, H.Y.; Wang, B.Y.; Lin, H.N.; Wang, M.C.; Ren, J.X. A review of physiological functions of orexin: From instinctive responses to subjective cognition. Medicine 2023, 102, e34206.
- Soya, S.; Sakurai, T. Evolution of Orexin Neuropeptide System: Structure and Function. Front. Neurosci. 2020, 14, 691.
- Sakurai, T.; Amemiya, A.; Ishii, M.; Matsuzaki, I.; Chemelli, R.M.; Tanaka, H.; Williams, S.C.; Richardson, J.A.; Kozlowski, G.P.; Wilson, S.; et al. Orexins and orexin receptors: A family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 1998, 92, 573–585.
- de Lecea, L.; Kilduff, T.S.; Peyron, C.; Gao, X.; Foye, P.E.; Danielson, P.E.; Fukuhara, C.; Battenberg, E.L.; Gautvik, V.T.; Bartlett, F.S., 2nd; et al. The hypocretins: Hypothalamus-specific peptides with neuroexcitatory activity. Proc. Natl. Acad. Sci. USA 1998, 95, 322–327.
- Peyron, C.; Tighe, D.K.; van den Pol, A.N.; de Lecea, L.; Heller, H.C.; Sutcliffe, J.G.; Kilduff, T.S. Neurons containing hypocretin (orexin) project to multiple neuronal systems. J. Neurosci. 1998, 18, 9996–10015.
- Bittencourt, J.C.; Presse, F.; Arias, C.; Peto, C.; Vaughan, J.; Nahon, J.L.; Vale, W.; Sawchenko, P.E. The melanin-concentrating hormone system of the rat brain: An immuno- and hybridization histochemical characterization. J. Comp. Neurol. 1992, 319, 218–245.
- Lopez-Lopez, C.; Dietrich, M.O.; Metzger, F.; Loetscher, H.; Torres-Aleman, I. Disturbed cross talk between insulin-like growth factor I and AMP-activated protein kinase as a possible cause of vascular dysfunction in the amyloid precursor protein/presenilin 2 mouse model of Alzheimer’s disease. J. Neurosci. 2007, 27, 824–831.
- Couvineau, A.; Nicole, P.; Gratio, V.; Voisin, T. The Orexin receptors: Structural and anti-tumoral properties. Front. Endocrinol. 2022, 13, 931970.
- Kastin, A.J.; Akerstrom, V. Orexin A but not orexin B rapidly enters brain from blood by simple diffusion. J. Pharmacol. Exp. Ther. 1999, 289, 219–223.
- Sutcliffe, J.G.; de Lecea, L. The hypocretins: Excitatory neuromodulatory peptides for multiple homeostatic systems, including sleep and feeding. J. Neurosci. Res. 2000, 62, 161–168.
- Wang, C.; Wang, Q.; Ji, B.; Pan, Y.; Xu, C.; Cheng, B.; Bai, B.; Chen, J. The Orexin/Receptor System: Molecular Mechanism and Therapeutic Potential for Neurological Diseases. Front. Mol. Neurosci. 2018, 11, 220.
- Kukkonen, J.P. G-protein-dependency of orexin/hypocretin receptor signalling in recombinant Chinese hamster ovary cells. Biochem. Biophys. Res. Commun. 2016, 476, 379–385.
- Kukkonen, J.P. OX2 orexin/hypocretin receptor signal transduction in recombinant Chinese hamster ovary cells. Cell. Signal. 2016, 28, 51–60.
- Kukkonen, J.P. Orexin/Hypocretin Signaling. Curr. Top. Behav. Neurosci. 2017, 33, 17–50.
- Bonini, J.A.; Jones, K.A.; Adham, N.; Forray, C.; Artymyshyn, R.; Durkin, M.M.; Smith, K.E.; Tamm, J.A.; Boteju, L.W.; Lakhlani, P.P.; et al. Identification and characterization of two G protein-coupled receptors for neuropeptide FF. J. Biol. Chem. 2000, 275, 39324–39331.
- Laemmle, B.; Schindler, M.; Beilmann, M.; Hamilton, B.S.; Doods, H.N.; Wieland, H.A. Characterization of the NPGP receptor and identification of a novel short mRNA isoform in human hypothalamus. Regul. Pept. 2003, 111, 21–29.
- Cutler, D.J.; Morris, R.; Sheridhar, V.; Wattam, T.A.; Holmes, S.; Patel, S.; Arch, J.R.; Wilson, S.; Buckingham, R.E.; Evans, M.L.; et al. Differential distribution of orexin-A and orexin-B immunoreactivity in the rat brain and spinal cord. Peptides 1999, 20, 1455–1470.
- Smart, D.; Jerman, J. The physiology and pharmacology of the orexins. Pharmacol. Ther. 2002, 94, 51–61.
- Hervieu, G.J.; Cluderay, J.E.; Harrison, D.C.; Roberts, J.C.; Leslie, R.A. Gene expression and protein distribution of the orexin-1 receptor in the rat brain and spinal cord. Neuroscience 2001, 103, 777–797.
- Trivedi, P.; Yu, H.; MacNeil, D.J.; Van der Ploeg, L.H.; Guan, X.M. Distribution of orexin receptor mRNA in the rat brain. FEBS Lett. 1998, 438, 71–75.
- Lu, X.Y.; Bagnol, D.; Burke, S.; Akil, H.; Watson, S.J. Differential distribution and regulation of OX1 and OX2 orexin/hypocretin receptor messenger RNA in the brain upon fasting. Horm. Behav. 2000, 37, 335–344.
- Mitsukawa, K.; Kimura, H. Orexin 2 receptor (OX2R) protein distribution measured by autoradiography using radiolabeled OX2R-selective antagonist EMPA in rodent brain and peripheral tissues. Sci. Rep. 2022, 12, 8473.
- Melmed, S.; Auchus, R.J.; Goldfine, A.B.; Koenig, R.J.; Rosen, C.J. Williams Textbook of Endocrinology, 14th ed.; Elsevier: Philadelphia, PA, USA, 2020.
- Cannon, W.B. The emergency function of the adrenal medulla in pain and the major emotions. Am. J. Physiol.-Leg. Content 1914, 33, 356–372.
- Selye, H. A Syndrome produced by Diverse Nocuous Agents. Nature 1936, 138, 32.
- Wade, N. Guillemin and schally: A race spurred by rivalry. Science 1978, 200, 510–513.
- Vale, W.; Spiess, J.; Rivier, C.; Rivier, J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science 1981, 213, 1394–1397.
- Selye, H. The general adaptation syndrome and the diseases of adaptation. J. Allergy 1946, 17, 231.
- Jaszberenyi, M.; Bujdoso, E.; Telegdy, G. Behavioral, neuroendocrine and thermoregulatory actions of apelin-13. Neuroscience 2004, 129, 811–816.
- Jaszberenyi, M.; Bujdoso, E.; Bagosi, Z.; Telegdy, G. Mediation of the behavioral, endocrine and thermoregulatory actions of ghrelin. Horm. Behav. 2006, 50, 266–273.
- Jaszberenyi, M.; Bagosi, Z.; Thurzo, B.; Foldesi, I.; Telegdy, G. Endocrine and behavioral effects of neuromedin S. Horm. Behav. 2007, 52, 631–639.
- Bujdoso, E.; Jaszberenyi, M.; Tomboly, C.; Toth, G.; Telegdy, G. Behavioral and neuroendocrine actions of endomorphin-2. Peptides 2001, 22, 1459–1463.
- Aguilera, G. Regulation of the hypothalamic-pituitary-adrenal axis by neuropeptides. Horm. Mol. Biol. Clin. Investig. 2011, 7, 327–336.
- Bujdoso, E.; Jaszberenyi, M.; Tomboly, C.; Toth, G.; Telegdy, G. Effects of endomorphin-1 on open-field behavior and on the hypothalamic-pituitary-adrenal system. Endocrine 2001, 14, 221–224.
- Rostene, W.H.; Alexander, M.J. Neurotensin and neuroendocrine regulation. Front. Neuroendocrinol. 1997, 18, 115–173.
- Perras, B.; Schultes, B.; Behn, B.; Dodt, C.; Born, J.; Fehm, H.L. Intranasal atrial natriuretic peptide acts as central nervous inhibitor of the hypothalamo-pituitary-adrenal stress system in humans. J. Clin. Endocrinol. Metab. 2004, 89, 4642–4648.
- Kuppusamy, T.; Ramaswamy, P.; Perumal, M.; Silambanan, S.; Prabu Kumar, A. A short note on oxytocin and stress attenuation. Bioinformation 2021, 17, 921–923.
- Jaszberenyi, M.; Bujdoso, E.; Telegdy, G. Effects of C-type natriuretic peptide on pituitary-adrenal activation in rats. Neuroreport 1998, 9, 2601–2603.
- Jaszberenyi, M.; Bujdoso, E.; Telegdy, G. Effects of brain natriuretic peptide on pituitary-adrenal activation in rats. Life Sci. 2000, 66, 1655–1661.
- Kronenberg, H.; Williams, R.H. Williams Textbook of Endocrinology, 11th ed.; Saunders/Elsevier: Philadelphia, PA, USA, 2008.
- Carrasco, G.A.; Van de Kar, L.D. Neuroendocrine pharmacology of stress. Eur. J. Pharmacol. 2003, 463, 235–272.
- Pan, W.; Kastin, A.J. Urocortin and the brain. Prog. Neurobiol. 2008, 84, 148–156.
- Henckens, M.J.; Deussing, J.M.; Chen, A. Region-specific roles of the corticotropin-releasing factor-urocortin system in stress. Nat. Rev. Neurosci. 2016, 17, 636–651.
- Grafe, L.A.; Bhatnagar, S. Orexins and stress. Front. Neuroendocrinol. 2018, 51, 132–145.
- Sargin, D. The role of the orexin system in stress response. Neuropharmacology 2019, 154, 68–78.
- Saper, C.B.; Scammell, T.E.; Lu, J. Hypothalamic regulation of sleep and circadian rhythms. Nature 2005, 437, 1257–1263.
- Hagan, J.J.; Leslie, R.A.; Patel, S.; Evans, M.L.; Wattam, T.A.; Holmes, S.; Benham, C.D.; Taylor, S.G.; Routledge, C.; Hemmati, P.; et al. Orexin A activates locus coeruleus cell firing and increases arousal in the rat. Proc. Natl. Acad. Sci. USA 1999, 96, 10911–10916.
- Zheng, H.; Patterson, L.M.; Berthoud, H.R. Orexin-A projections to the caudal medulla and orexin-induced c-Fos expression, food intake, and autonomic function. J. Comp. Neurol. 2005, 485, 127–142.
- de Oliveira, C.V.; Rosas-Arellano, M.P.; Solano-Flores, L.P.; Ciriello, J. Cardiovascular effects of hypocretin-1 in nucleus of the solitary tract. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H1369–H1377.
- Yamashita, A.; Moriya, S.; Nishi, R.; Kaminosono, J.; Yamanaka, A.; Kuwaki, T. Aversive emotion rapidly activates orexin neurons and increases heart rate in freely moving mice. Mol. Brain 2021, 14, 104.
- Kuwaki, T. Orexin (hypocretin) participates in central autonomic regulation during fight-or-flight response. Peptides 2021, 139, 170530.
- Purves, D. Neuroscience, 6th ed.; Oxford University Press: New York, NY, USA, 2018.
- Johnson, P.L.; Molosh, A.; Fitz, S.D.; Truitt, W.A.; Shekhar, A. Orexin, stress, and anxiety/panic states. Prog. Brain Res. 2012, 198, 133–161.
- Jaszberenyi, M.; Bujdoso, E.; Pataki, I.; Telegdy, G. Effects of orexins on the hypothalamic-pituitary-adrenal system. J. Neuroendocrinol. 2000, 12, 1174–1178.
- Samson, W.K.; Taylor, M.M.; Follwell, M.; Ferguson, A.V. Orexin actions in hypothalamic paraventricular nucleus: Physiological consequences and cellular correlates. Regul. Pept. 2002, 104, 97–103.
- Yun, S.; Wennerholm, M.; Shelton, J.E.; Bonaventure, P.; Letavic, M.A.; Shireman, B.T.; Lovenberg, T.W.; Dugovic, C. Selective Inhibition of Orexin-2 Receptors Prevents Stress-Induced ACTH Release in Mice. Front. Behav. Neurosci. 2017, 11, 83.
- Winsky-Sommerer, R.; Yamanaka, A.; Diano, S.; Borok, E.; Roberts, A.J.; Sakurai, T.; Kilduff, T.S.; Horvath, T.L.; de Lecea, L. Interaction between the corticotropin-releasing factor system and hypocretins (orexins): A novel circuit mediating stress response. J. Neurosci. 2004, 24, 11439–11448.
- Sakamoto, F.; Yamada, S.; Ueta, Y. Centrally administered orexin-A activates corticotropin-releasing factor-containing neurons in the hypothalamic paraventricular nucleus and central amygdaloid nucleus of rats: Possible involvement of central orexins on stress-activated central CRF neurons. Regul. Pept. 2004, 118, 183–191.
- Blasiak, A.; Gundlach, A.L.; Hess, G.; Lewandowski, M.H. Interactions of Circadian Rhythmicity, Stress and Orexigenic Neuropeptide Systems: Implications for Food Intake Control. Front. Neurosci. 2017, 11, 127.
- Jaszberenyi, M.; Bujdoso, E.; Telegdy, G. The role of neuropeptide Y in orexin-induced hypothalamic-pituitary-adrenal activation. J. Neuroendocrinol. 2001, 13, 438–441.
- Hirota, K.; Kushikata, T.; Kudo, M.; Kudo, T.; Lambert, D.G.; Matsuki, A. Orexin A and B evoke noradrenaline release from rat cerebrocortical slices. Br. J. Pharmacol. 2001, 134, 1461–1466.
- Brunton, P.J.; Bales, J.; Russell, J.A. Neuroendocrine stress but not feeding responses to centrally administered neuropeptide Y are suppressed in pregnant rats. Endocrinology 2006, 147, 3737–3745.
- Russell, S.H.; Small, C.J.; Dakin, C.L.; Abbott, C.R.; Morgan, D.G.; Ghatei, M.A.; Bloom, S.R. The central effects of orexin-A in the hypothalamic-pituitary-adrenal axis in vivo and in vitro in male rats. J. Neuroendocrinol. 2001, 13, 561–566.
- Vanderhaven, M.W.; Cornish, J.L.; Staples, L.G. The orexin-1 receptor antagonist SB-334867 decreases anxiety-like behavior and c-Fos expression in the hypothalamus of rats exposed to cat odor. Behav. Brain Res. 2015, 278, 563–568.
- Furlong, T.M.; Vianna, D.M.; Liu, L.; Carrive, P. Hypocretin/orexin contributes to the expression of some but not all forms of stress and arousal. Eur. J. Neurosci. 2009, 30, 1603–1614.
More
Information
Subjects:
Medicine, Research & Experimental
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
756
Revisions:
2 times
(View History)
Update Date:
22 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





