
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jing-Ning Zhu | -- | 3781 | 2024-02-20 15:08:09 | | | |
| 2 | Sirius Huang | Meta information modification | 3781 | 2024-02-21 02:19:26 | | |
Video Upload Options
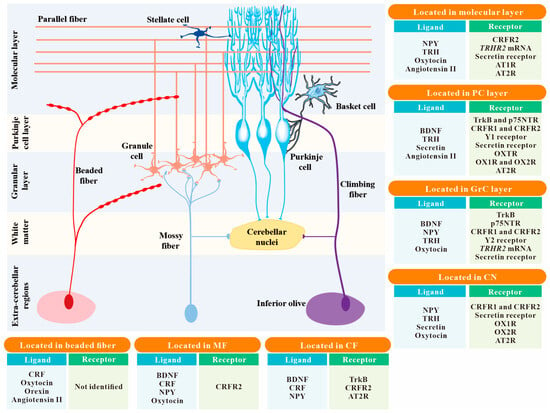
Although more than 30 different types of neuropeptides have been identified in various cell types and circuits of the cerebellum, their unique functions in the cerebellum remain poorly understood. Given the nature of their diffuse distribution, peptidergic systems are generally assumed to exert a modulatory effect on the cerebellum via adaptively tuning neuronal excitability, synaptic transmission, and synaptic plasticity within cerebellar circuits. Moreover, cerebellar neuropeptides have also been revealed to be involved in the neurogenetic and developmental regulation of the developing cerebellum, including survival, migration, differentiation, and maturation of the Purkinje cells and granule cells in the cerebellar cortex. On the other hand, cerebellar neuropeptides hold a critical position in the pathophysiology and pathogenesis of many cerebellar-related motor and psychiatric disorders, such as cerebellar ataxias and autism. A growing body of evidence has indicated neuropeptides as potential therapeutic targets to ameliorate these diseases effectively.
1. Introduction
| Order | Neuropeptide Name | Abbreviation | No. of Publications Available on PubMed in Last Two Decades (2004–2024) |
Potential Clinical Treatment |
|---|---|---|---|---|
| 1 | Brain-derived neurotrophic factor | BDNF | 403 | SCA6, SCA1 |
| 2 | Insulin-like growth factor 1 | IGF-1 | 110 | Metabolic diseases |
| 3 | Corticotropin-releasing factor (hormone) | CRF (CRH) | 68 | Ataxias |
| 4 | Angiotensin II | Ang II | 49 | ASD |
| 5 | Somatostatin | SS | 45 | |
| 6 | Cerebellin | CBLN | 41 | |
| 7 | Neuropeptide Y | NPY | 40 | SCA3 |
| 8 | Orexin | OX | 33 | Narcolepsy-cataplexy, anxiety |
| 9 | Thyrotropin-releasing hormone | TRH | 32 | Ataxias |
| 10 | Oxytocin | OXT | 29 | ASD |
| 11 | Calcitonin-gene-related peptide | CGRP | 28 | Migraine |
| 12 | Substance P | SP | 26 | |
| 13 | Secretin | SCT | 24 | ASD |
| 14 | Cholecystokinin | CCK | 22 | |
| 15 | Dynorphin | DYN | 17 |
2. BDNF
3. CRF
4. Angiotensin II
References
- Koziol, L.F.; Budding, D.; Andreasen, N.; D’Arrigo, S.; Bulgheroni, S.; Imamizu, H.; Ito, M.; Manto, M.; Marvel, C.; Parker, K.; et al. Consensus paper: The cerebellum’s role in movement and cognition. Cerebellum 2014, 13, 151–177.
- Hibi, M.; Shimizu, T. Development of the cerebellum and cerebellar neural circuits. Dev. Neurobiol. 2012, 72, 282–301.
- Hashimoto, M.; Hibi, M. Development and evolution of cerebellar neural circuits. Dev. Growth Differ. 2012, 54, 373–389.
- Marzban, H.; Del Bigio, M.R.; Alizadeh, J.; Ghavami, S.; Zachariah, R.M.; Rastegar, M. Cellular commitment in the developing cerebellum. Front. Cell. Neurosci. 2015, 8, 450.
- Kapfhammer, J.P. Cellular and molecular control of dendritic growth and development of cerebellar Purkinje cells. Prog. Histochem. Cytochem. 2004, 39, 131–182.
- Hanzel, M.; Rook, V.; Wingate, R.J.T. Mitotic granule cell precursors undergo highly dynamic morphological transitions throughout the external germinal layer of the chick cerebellum. Sci. Rep. 2019, 9, 15218.
- Galas, L.; Bénard, M.; Lebon, A.; Komuro, Y.; Schapman, D.; Vaudry, H.; Vaudry, D.; Komuro, H. Postnatal Migration of Cerebellar Interneurons. Brain Sci. 2017, 7, 62.
- Ito, M. Cerebellar circuitry as a neuronal machine. Prog. Neurobiol. 2006, 78, 272–303.
- Hashimoto, K.; Ichikawa, R.; Kitamura, K.; Watanabe, M.; Kano, M. Translocation of a “Winner” Climbing Fiber to the Purkinje Cell Dendrite and Subsequent Elimination of “Losers” from the Soma in Developing Cerebellum. Neuron 2009, 63, 106–118.
- Busch, S.E.; Hansel, C. Climbing fiber multi-innervation of mouse Purkinje dendrites with arborization common to human. Science 2023, 381, 420–427.
- Gao, Z.; van Beugen, B.J.; De Zeeuw, C.I. Distributed synergistic plasticity and cerebellar learning. Nat. Rev. Neurosci. 2012, 13, 619–635.
- Ito, M. Functional roles of neuropeptides in cerebellar circuits. Neuroscience 2009, 162, 666–672.
- Corbière, A.; Walet-Balieu, M.-L.; Chan, P.; Basille-Dugay, M.; Hardouin, J.; Vaudry, D. A Peptidomic Approach to Characterize Peptides Involved in Cerebellar Cortex Development Leads to the Identification of the Neurotrophic Effects of Nociceptin. Mol. Cell. Proteom. 2018, 17, 1737–1749.
- Bishop, G.A.; King, J.S. Neuropeptides in the Cerebellum; Springer International Publishing: Cham, Germany, 2023; pp. 231–236.
- Barde, Y.; Edgar, D.; Thoenen, H. Purification of a new neurotrophic factor from mammalian brain. EMBO J. 1982, 1, 549–553.
- Colucci-D’amato, L.; Speranza, L.; Volpicelli, F. Neurotrophic Factor BDNF, Physiological Functions and Therapeutic Potential in Depression, Neurodegeneration and Brain Cancer. Int. J. Mol. Sci. 2020, 21, 7777.
- Camuso, S.; La Rosa, P.; Fiorenza, M.T.; Canterini, S. Pleiotropic effects of BDNF on the cerebellum and hippocampus: Implications for neurodevelopmental disorders. Neurobiol. Dis. 2021, 163, 105606.
- Chen, A.I.; Zang, K.; Masliah, E.; Reichardt, L.F. Glutamatergic axon-derived BDNF controls GABAergic synaptic differentiation in the cerebellum. Sci. Rep. 2016, 6, 20201.
- Dieni, S.; Rees, S. Distribution of brain-derived neurotrophic factor and TrkB receptor proteins in the fetal and postnatal hippocampus and cerebellum of the guinea pig. J. Comp. Neurol. 2002, 454, 229–240.
- Rocamora, N.; García-Ladona, F.; Palacios, J.; Mengod, G. Differential expression of brain-derived neurotrophic factor, neurotrophin-3, and low-affinity nerve growth factor receptor during the postnatal development of the rat cerebellar system. Mol. Brain Res. 1993, 17, 1–8.
- Zanin, J.P.; Verpeut, J.L.; Li, Y.; Shiflett, M.W.; Wang, S.S.-H.; Santhakumar, V.; Friedman, W.J. The p75NTR Influences Cerebellar Circuit Development and Adult Behavior via Regulation of Cell Cycle Duration of Granule Cell Progenitors. J. Neurosci. 2019, 39, 9119–9129.
- Lindholm, D.; Dechant, G.; Heisenberg, C.; Thoenen, H. Brain-derived Neurotrophic Factor is a Survival Factor for Cultured Rat Cerebellar Granule Neurons and Protects them Against Glutamate-induced Neurotoxicity. Eur. J. Neurosci. 1993, 5, 1455–1464.
- Schwartz, P.M.; Borghesani, P.R.; Levy, R.L.; Pomeroy, S.L.; Segal, R.A. Abnormal cerebellar development and foliation in BDNF-/- mice reveals a role for neurotrophins in CNS patterning. Neuron 1997, 19, 269–281.
- Gao, W.Q.; Zheng, J.L.; Karihaloo, M. Neurotrophin-4/5 (NT-4/5) and brain-derived neurotrophic factor (BDNF) act at later stages of cerebellar granule cell differentiation. J. Neurosci. 1995, 15, 2656–2667.
- Rico, B.; Xu, B.; Reichardt, L.F. TrkB receptor signaling is required for establishment of GABAergic synapses in the cerebellum. Nat. Neurosci. 2002, 5, 225–233.
- Minichiello, L.; Klein, R. TrkB and TrkC neurotrophin receptors cooperate in promoting survival of hippocampal and cerebellar granule neurons. Minerva Anestesiol. 1996, 10, 2849–2858.
- Choo, M.; Miyazaki, T.; Yamazaki, M.; Kawamura, M.; Nakazawa, T.; Zhang, J.; Tanimura, A.; Uesaka, N.; Watanabe, M.; Sakimura, K.; et al. Retrograde BDNF to TrkB signaling promotes synapse elimination in the developing cerebellum. Nat. Commun. 2017, 8, 195.
- Kafitz, K.W.; Rose, C.R.; Thoenen, H.; Konnerth, A. Neurotrophin-evoked rapid excitation through TrkB receptors. Nature 1999, 401, 918–921.
- Huang, Y.; Ko, H.; Cheung, Z.H.; Yung, K.K.; Yao, T.; Wang, J.-J.; Morozov, A.; Ke, Y.; Ip, N.Y.; Yung, W.-H. Dual actions of brain-derived neurotrophic factor on GABAergic transmission in cerebellar Purkinje neurons. Exp. Neurol. 2012, 233, 791–798.
- Boxall, A.R. GABAergic mIPSCs in rat cerebellar Purkinje cells are modulated by TrkB and mGluR1-mediated stimulation of Src. J. Physiol. 2000, 524, 677–684.
- Arenas, Y.M.; Martínez-García, M.; Llansola, M.; Felipo, V. Enhanced BDNF and TrkB Activation Enhance GABA Neurotransmission in Cerebellum in Hyperammonemia. Int. J. Mol. Sci. 2022, 23, 11770.
- Huang, Y.; Wang, J.-J.; Yung, W.-H. Coupling Between GABA-A Receptor and Chloride Transporter Underlies Ionic Plasticity in Cerebellar Purkinje Neurons. Cerebellum 2013, 12, 328–330.
- Carter, A.R.; Chen, C.; Schwartz, P.M.; Segal, R.A. Brain-Derived Neurotrophic Factor Modulates Cerebellar Plasticity and Synaptic Ultrastructure. J. Neurosci. 2002, 22, 1316–1327.
- Takahashi, M.; Ishikawa, K.; Sato, N.; Obayashi, M.; Niimi, Y.; Ishiguro, T.; Yamada, M.; Toyoshima, Y.; Takahashi, H.; Kato, T.; et al. Reduced brain-derived neurotrophic factor (BDNF) mRNA expression and presence of BDNF-immunoreactive granules in the spinocerebellar ataxia type 6 (SCA6) cerebellum. Neuropathology 2012, 32, 595–603.
- Hourez, R.; Servais, L.; Orduz, D.; Gall, D.; Millard, I.; de Kerchove d’Exaerde, A.; Cheron, G.; Orr, H.T.; Pandolfo, M.; Schiffmann, S.N. Aminopyridines correct early dysfunction and delay neurodegeneration in a mouse model of spinocerebellar ataxia type 1. J. Neurosci. 2011, 31, 11795–11807.
- Jones, J.; Jaramillo-Merchán, J.; Bueno, C.; Pastor, D.; Viso-León, M.; Martínez, S. Mesenchymal stem cells rescue Purkinje cells and improve motor functions in a mouse model of cerebellar ataxia. Neurobiol. Dis. 2010, 40, 415–423.
- Firozan, B.; Goudarzi, I.; Salmani, M.E.; Lashkarbolouki, T.; Rezaei, A.; Abrari, K. Estradiol increases expression of the brain-derived neurotrophic factor after acute administration of ethanol in the neonatal rat cerebellum. Eur. J. Pharmacol. 2014, 732, 1–11.
- Sugiyama, A.; Kato, H.; Takakura, H.; Osawa, S.; Maeda, Y.; Izawa, T. Effects of physical activity and melatonin on brain-derived neurotrophic factor and cytokine expression in the cerebellum of high-fat diet-fed rats. Neuropsychopharmacol. Rep. 2020, 40, 291–296.
- Angelucci, F.; De Bartolo, P.; Gelfo, F.; Foti, F.; Cutuli, D.; Bossù, P.; Caltagirone, C.; Petrosini, L. Increased Concentrations of Nerve Growth Factor and Brain-Derived Neurotrophic Factor in the Rat Cerebellum After Exposure to Environmental Enrichment. Cerebellum 2009, 8, 499–506.
- Fernandes, M.S.d.S.; Santos, G.C.J.; Filgueira, T.O.; Gomes, D.A.; Barbosa, E.A.S.; dos Santos, T.M.; Câmara, N.O.S.; Castoldi, A.; Souto, F.O. Cytokines and Immune Cells Profile in Different Tissues of Rodents Induced by Environmental Enrichment: Systematic Review. Int. J. Mol. Sci. 2022, 23, 11986.
- Vale, W.; Spiess, J.; Rivier, C.; Rivier, J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science 1981, 213, 1394–1397.
- Bittencourt, J.C.; Sawchenko, P.E. Do Centrally Administered Neuropeptides Access Cognate Receptors?: An Analysis in the Central Corticotropin-Releasing Factor System. J. Neurosci. 2000, 20, 1142–1156.
- Wang, Y.; Chen, Z.-P.; Zhuang, Q.-X.; Zhang, X.-Y.; Li, H.-Z.; Wang, J.-J.; Zhu, J.-N. Role of Corticotropin-Releasing Factor in Cerebellar Motor Control and Ataxia. Curr. Biol. 2017, 27, 2661–2669.e5.
- Ezra-Nevo, G.; Volk, N.; Ramot, A.; Kuehne, C.; Tsoory, M.; Deussing, J.; Chen, A. Inferior olive CRF plays a role in motor performance under challenging conditions. Transl. Psychiatry 2018, 8, 107.
- Cummings, S.; Sharp, B.; Elde, R. Corticotropin-releasing factor in cerebellar afferent systems: A combined immunohistochemistry and retrograde transport study. J. Neurosci. 1988, 8, 543–554.
- Mizuno, Y.; Takahashi, K.; Totsune, K.; Ohneda, M.; Konno, H.; Murakami, O.; Satoh, F.; Sone, M.; Takase, S.; Itoyama, Y.; et al. Decrease in cerebellin and corticotropin-releasing hormone in the cerebellum of olivopontocerebellar atrophy and Shy-Drager syndrome. Brain Res. 1995, 686, 115–118.
- Libster, A.M.; Title, B.; Yarom, Y. Corticotropin-releasing factor increases Purkinje neuron excitability by modulating sodium, potassium, and Ih currents. J. Neurophysiol. 2015, 114, 3339–3350.
- Miyata, M.; Okada, D.; Hashimoto, K.; Kano, M.; Ito, M. Corticotropin-Releasing Factor Plays a Permissive Role in Cerebellar Long-Term Depression. Neuron 1999, 22, 763–775.
- Schmolesky, M.T.; De Ruiter, M.M.; De Zeeuw, C.I.; Hansel, C. The neuropeptide corticotropin-releasing factor regulates excitatory transmission and plasticity at the climbing fibre-Purkinje cell synapse. Eur. J. Neurosci. 2007, 25, 1460–1466.
- Cui, L.-N.; Zhang, X.-D.; Xu, Y.-H.; Li, B.-Y.; Qiu, D.-L.; Chu, C.-P. Activation CRF-R2 augments cerebellar climbing fiber-Purkinje cell synaptic transmission via presynaptic PKA pathway in mice. Neurosci. Lett. 2022, 777, 136584.
- Li, G.-G.; Piao, C.-J.; Wan, P.; Li, S.-Y.; Wei, Y.-X.; Zhao, G.-J.; Wu, W.-Y.; Hong, L.; Chu, C.-P.; Qiu, D.-L. Opposing actions of CRF-R1 and CB1 receptor on facial stimulation-induced MLI-PC plasticity in mouse cerebellar cortex. BMC Neurosci. 2022, 23, 39.
- Ezra-Nevo, G.; Prestori, F.; Locatelli, F.; Soda, T.; Brinke, M.M.T.; Engel, M.; Boele, H.-J.; Botta, L.; Leshkowitz, D.; Ramot, A.; et al. Cerebellar Learning Properties Are Modulated by the CRF Receptor. J. Neurosci. 2018, 38, 6751–6765.
- Wolf, O.T.; Minnebusch, D.; Daum, I. Stress impairs acquisition of delay eyeblink conditioning in men and women. Neurobiol. Learn. Mem. 2009, 91, 431–436.
- Wolf, O.T.; Bauser, D.S.; Daum, I. Eyeblink conditional discrimination learning in healthy young men is impaired after stress exposure. Psychophysiology 2011, 49, 164–171.
- Joëls, M.; Baram, T.Z. The neuro-symphony of stress. Nat. Rev. Neurosci. 2009, 10, 459–466.
- Wang, X.D.; Rammes, G.; Kraev, I.; Wolf, M.; Liebl, C.; Scharf, S.H.; Rice, C.J.; Wurst, W.; Holsboer, F.; Deussing, J.M.; et al. Forebrain CRF₁ modulates early-life stress-programmed cognitive deficits. J. Neurosci. 2011, 31, 13625–13634.
- Barmack, N.H.; Pettorossi, V.E. Adaptive Balance in Posterior Cerebellum. Front. Neurol. 2021, 12, 635259.
- Bishop, G.A.; King, J.S. Corticotropin Releasing Factor in the Embryonic Mouse Cerebellum. Exp. Neurol. 1999, 160, 489–499.
- Madtes, P.; King, J.S. The temporal and spatial development of CRF binding sites in the postnatal mouse cerebellum. Neurosci. Res. 1999, 34, 45–50.
- King, J.S.; Bishop, G.A. The distribution and cellular localization of CRF-R1 in the vermis of the postnatal mouse cerebellum. Exp. Neurol. 2002, 178, 175–185.
- Gounko, N.V.; Kalicharan, D.; Rybakin, V.; Gramsbergen, A.; Van Der Want, J.J.L. The dynamic developmental localization of the full-length corticotropin-releasing factor receptor type 2 in rat cerebellum. Eur. J. Neurosci. 2006, 23, 3217–3224.
- Tian, J.; Shan, X.; Bishop, G.; King, J. Presynaptic localization of a truncated isoform of the type 2 corticotropin releasing factor receptor in the cerebellum. Neuroscience 2006, 138, 691–702.
- Gounko, N.V.; Gramsbergen, A.; van der Want, J.J.L. Localization and functional roles of corticotropin-releasing factor receptor type 2 in the cerebellum. Cerebellum 2008, 7, 4–8.
- Gounko, N.V.; Swinny, J.D.; Kalicharan, D.; Jafari, S.; Corteen, N.; Seifi, M.; Bakels, R.; van der Want, J.J.L. Corticotropin-releasing factor and urocortin regulate spine and synapse formation: Structural basis for stress-induced neuronal remodeling and pathology. Mol. Psychiatry 2012, 18, 86–92.
- Swinny, J.D.; Metzger, F.; Ijkema-Paassen, J.; Gounko, N.V.; Gramsbergen, A.; Van Der Want, J.J.L. Corticotropin-releasing factor and urocortin differentially modulate rat Purkinje cell dendritic outgrowth and differentiation in vitro. Eur. J. Neurosci. 2004, 19, 1749–1758.
- Skeggs, L.T.; Marsh, W.H.; Kahn, J.R.; Shumway, N.P. The existence of two forms of hypertensin. J. Exp. Med. 1954, 99, 275–282.
- Phillips, M. Functions Of Angiotensin In The Central Nervous System. Annu. Rev. Physiol. 1987, 49, 413–435.
- Changaris, D.G.; Severs, W.B.; Keil, L.C. Localization of angiotensin in rat brain. J. Histochem. Cytochem. 1978, 26, 593–607.
- Erdmann, B.; Fuxe, K.; Ganten, D. Subcellular Localization of Angiotensin II Immunoreactivity in the Rat Cerebellar Cortex. Hypertension 1996, 28, 818–824.
- Elkahloun, A.G.; Saavedra, J.M. Candesartan Neuroprotection in Rat Primary Neurons Negatively Correlates with Aging and Senescence: A Transcriptomic Analysis. Mol. Neurobiol. 2019, 57, 1656–1673.
- Huang, Z.; Ohno, N.; Terada, N.; Saitoh, Y.; Chen, J.; Ohno, S. Immunohistochemical detection of angiotensin II receptors in mouse cerebellum and adrenal gland using “in vivo cryotechnique”. Histochem. Cell Biol. 2013, 140, 477–490.
- Reagan, L.P.; Flanagan-Cato, L.M.; Yee, D.K.; Ma, L.-Y.; Sakai, R.R.; Fluharty, S.J. Immunohistochemical mapping of angiotensin type 2 (AT2) receptors in rat brain. Brain Res. 1994, 662, 45–59.
- Jöhren, O.; Häuser, W.; Saavedra, J.M. Chemical lesion of the inferior olive reduces Sarcosine1–Angiotensin II binding to AT2 receptors in the cerebellar cortex of young rats. Brain Res. 1998, 793, 176–186.
- Côté, F.; Do, T.H.; Laflamme, L.; Gallo, J.-M.; Gallo-Payet, N. Activation of the AT2 Receptor of Angiotensin II Induces Neurite Outgrowth and Cell Migration in Microexplant Cultures of the Cerebellum. J. Biol. Chem. 1999, 274, 31686–31692.
- Tongroach, P.; Sanguanrungsirikul, S.; Tantisira, B.; Kunluan, P. Angiotensin II-induced depression of purkinje cell firing and possible modulatory action on GABA responses. Neurosci. Res. 1984, 1, 369–372.
- Haspula, D.; Clark, M.A. Contrasting Roles of Ang II and ACEA in the Regulation of IL10 and IL1β Gene Expression in Primary SHR Astroglial Cultures. Molecules 2021, 26, 3012.
- O’Connor, A.T.; Clark, M.A. Angiotensin II induces cyclooxygenase 2 expression in rat astrocytes via the angiotensin type 1 receptor. Neuropeptides 2019, 77, 101958.
- Vervoort, V.S.; Beachem, M.A.; Edwards, P.S.; Ladd, S.; Miller, K.E.; de Mollerat, X.; Clarkson, K.; DuPont, B.; Schwartz, C.E.; Stevenson, R.E.; et al. AGTR2 Mutations in X-Linked Mental Retardation. Science 2002, 296, 2401–2403.
- Butler, M.G.; Rafi, S.K.; Manzardo, A.M. High-Resolution Chromosome Ideogram Representation of Currently Recognized Genes for Autism Spectrum Disorders. Int. J. Mol. Sci. 2015, 16, 6464–6495.
- Bealer, S.L.; Crowley, W.R. Angiotensin II-induced release of oxytocin: Interaction with norepinephrine and role in lactation. Regul. Pept. 2002, 111, 41–46.
- Wang, S.S.; Kloth, A.D.; Badura, A. The cerebellum, sensitive periods, and autism. Neuron 2014, 83, 518–532.





