Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Nirmal Mazumder | -- | 3421 | 2024-02-19 04:39:03 | | | |
| 2 | Catherine Yang | Meta information modification | 3421 | 2024-02-19 06:13:03 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Govindaraju, I.; Sana, M.; Chakraborty, I.; Rahman, M.H.; Biswas, R.; Mazumder, N. Dietary Acrylamide. Encyclopedia. Available online: https://encyclopedia.pub/entry/55131 (accessed on 02 April 2026).
Govindaraju I, Sana M, Chakraborty I, Rahman MH, Biswas R, Mazumder N. Dietary Acrylamide. Encyclopedia. Available at: https://encyclopedia.pub/entry/55131. Accessed April 02, 2026.
Govindaraju, Indira, Maidin Sana, Ishita Chakraborty, Md. Hafizur Rahman, Rajib Biswas, Nirmal Mazumder. "Dietary Acrylamide" Encyclopedia, https://encyclopedia.pub/entry/55131 (accessed April 02, 2026).
Govindaraju, I., Sana, M., Chakraborty, I., Rahman, M.H., Biswas, R., & Mazumder, N. (2024, February 19). Dietary Acrylamide. In Encyclopedia. https://encyclopedia.pub/entry/55131
Govindaraju, Indira, et al. "Dietary Acrylamide." Encyclopedia. Web. 19 February, 2024.
Copy Citation
In today’s fast-paced world, people increasingly rely on a variety of processed foods due to their busy lifestyles. The enhanced flavors, vibrant colors, and ease of accessibility at reasonable prices have made ready-to-eat foods the easiest and simplest choice to satiate hunger, especially those that undergo thermal processing. However, these foods often contain an unsaturated amide called ‘Acrylamide’, known by its chemical name 2-propenamide, which is a contaminant formed when a carbohydrate- or protein-rich food product is thermally processed at more than 120 °C through methods like frying, baking, or roasting.
dietary acrylamide
Maillard’s reaction
acrylamide toxicity
starch
1. Introduction
For centuries, people have employed thermal processing to prepare food before consumption. The heating process holds several significant benefits for food products, including enhancing nutritional quality, preventing microbial contamination, improving flavor, and extending the shelf life of foods. While thermal processing offers various advantages, it also gives rise to unwanted heat-induced toxins as a by-product, known as “thermally processed contaminants”. Acrylamide is one such chemical that has garnered considerable curiosity in recent years [1].
Acrylamide, known by its chemical name 2-propenamide (C3H5NO), is an odorless, white, water-soluble, highly reactive, and crystalline solid with a molecular weight of 71.08 gmol−1 [2]. It has become a significant concern as it is found in most thermally processed carbohydrate-rich foods. It was categorized as a potential human carcinogen, neurotoxicant, and genotoxicant by the International Agency for Research Cancer (IARC) in 1994. The Swedish National Food Administration (SNFA) was the first to report the presence of acrylamide in foods in 2002 when they were subjected to higher temperatures [3]. Subsequent studies from various countries, including the United Kingdom, the United States of America, Norway, Germany, and Switzerland, confirmed the presence of acrylamide in baked and fried dishes cooked at high temperatures [4]. Potato chips, crisps, coffee, pastry, and sweet biscuits were identified by the Joint Expert Committee on Food Additives (JECFA) as significant sources of acrylamide in daily diets worldwide, particularly in carbohydrate-rich foods cooked at temperatures higher than 120 °C [1].
2. Detection of Acrylamide in Food
As acrylamide is found in food products and poses a significant threat to human health, determining its content has become an intriguing challenge. Many experimental studies have been conducted to assess the dietary acrylamide content in food products. There are several detection methods for the determination of acrylamide content in food products, such as electronic tongue and electronic nose, Surface Enhanced Raman Spectroscopy (SERS), Gas Chromatography–Mass Spectroscopy (GC–MS), Liquid Chromatography –tandem Mass Spectroscopy (LC–MS/MS), hemoglobin nanoparticles (HbNPs), and fluorescent biosensors.
2.1. Electronic Tongue and Nose
An electronic tongue, or E-tongue, is a potentiometric sensor device capable of detecting the acrylamide content in food through electrostatic interactions [5]. It comprises polymeric lipid sensor membranes that interact with the acrylamide’s amine group, generating potentiometric signals (Figure 1). It was examined in a study using the liquid state of acrylamide solutions in a homemade two-cylindrical sensor array with lipid polymeric membranes. The samples were prepared by homogenizing the fine-grained olive to aqueous paste using deionized water, and the brine solutions were also diluted using deionized water to obtain the same proportions as olive. E-tongue membranes showed an increased response with a rising acrylamide content in olive oil and brine solutions, as reported by Martin-Vertedor et al. [6]. In contrast, an electronic nose, or E-nose, is a sensor device with four gas sensor chips and metal oxide sensors and a data collection block that evaluates organic compounds responsible for the odor of cooked food. The microdetector collects values detected by the sensors, determining the food’s acrylamide content. Thermal treatment can change the aromatic and phenolic profiles of olive oil, thereby altering the acrylamide content of the food. A study established a linear relationship between the aromatic status of the olive oil and the acrylamide content using E-nose, where the samples were prepared by crushing the olive, homogenizing by using Mili-Q water, and centrifugating at 4 °C [7]. These electronic devices surpass conventional methods as they are cost-effective, rapid, user-friendly, efficient, reliable, and accurate for detecting acrylamide content in an aqueous state; see Figure 1.
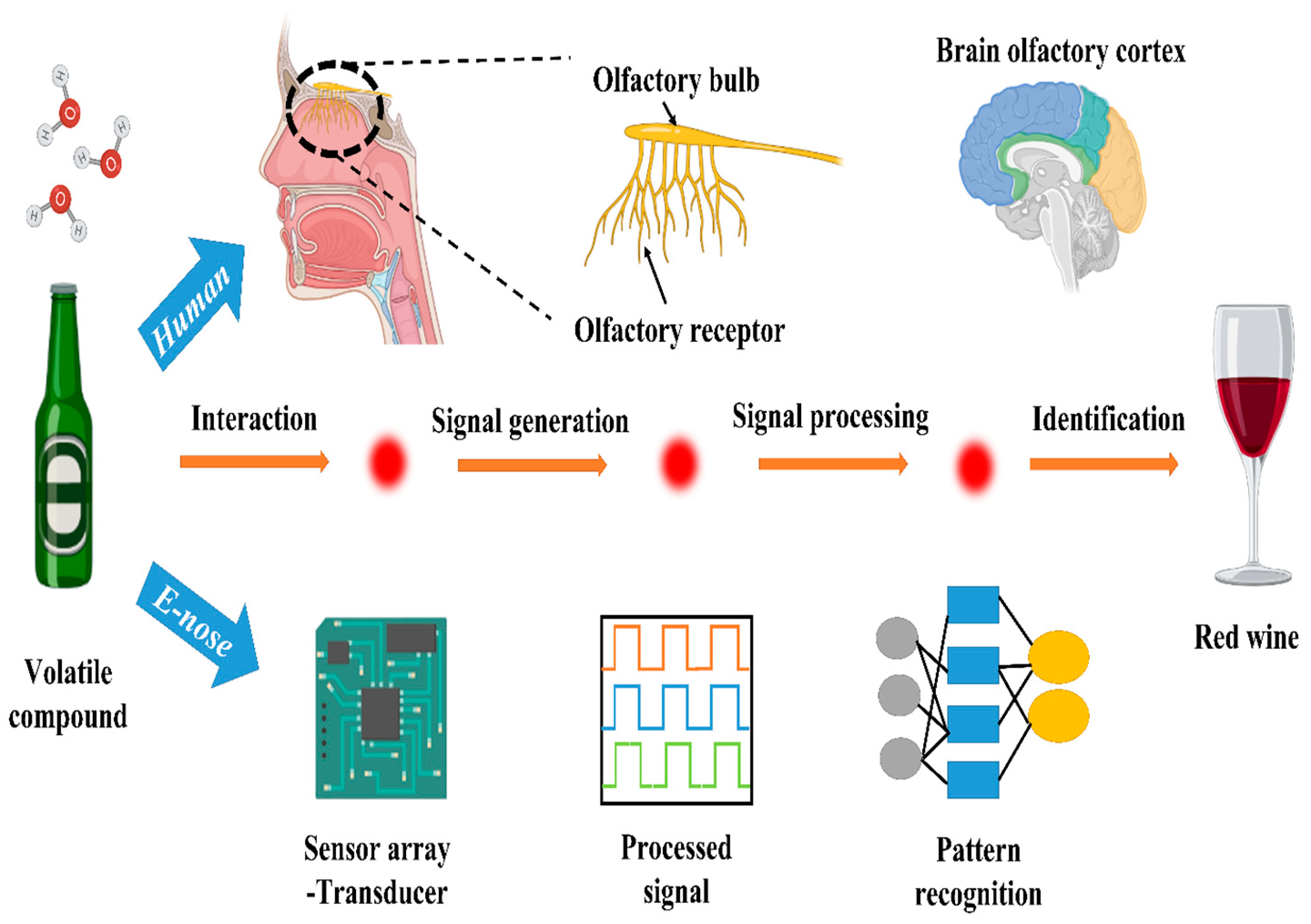
Figure 1. Schematic representation showing the working mechanism of E-nose for the detection of food materials.
2.2. Liquid Chromatography–Tandem Mass Spectrometry (LC–MS/MS)
Liquid Chromatography–Mass Spectrometry (LC–MS/MS) is one of the most precise and accurate methods for detecting the acrylamide content in food. Liquid Chromatography is an analytical technique used to determine the water-soluble compounds that are non-volatile. As roasted coffee contains toxic components like acrylamide, several experiments were conducted to determine the specific method for the detection of acrylamide content in coffee. In a study report, the acrylamide content of the coffee was determined by using LC-MS/MS with different brands of Turkish coffee. The highest acrylamide content was observed in instant coffee, ranging from 16.5 to 79.5 ng mL−1 [8]. Whereas Mesías et al. [9] detected the acrylamide content in the breakfast cereals, including fruits and nuts, using the same above-mentioned method. The acrylamide content was highest in wheat products, ranging from 197 to 639 µg/kg when baked for 35 min at 200 °C. The authors of [10] analyzed and observed the fact that processed food contained a higher amount of acrylamide content and have quoted that this is the reason for the increase in the acrylamide content in the food. The acrylamide content of the medicine homologous food Atractylodis Macrocephalae Rhizoma was detected by the above method using graphite carbon nanotubes as an extraction sorbet under processing temperatures ranging from 80 to 210 °C over 5 min to 2 h. The acrylamide concentration was highest with a concentration of 9826 µg/kg when processed at 150 °C for 60 min, as reported by Zhu et al. [11].
2.3. Gas Chromatography–Mass Spectrometry (GC–MS)
Gas Chromatography–Mass Spectrometry (GC–MS) is another method for detecting volatile acrylamide in food products. [12]. An experimental study used GC-MS coupled with microextraction and derivatization to determine the acrylamide content in cereal food products. The limit of detection was observed to be 0.6 ng g−1, and the acrylamide content of the samples, such as wafers, was determined to be more than 100 ng g−1. Baking temperature and time also influenced the acrylamide level of food products when determined using the above method, as reported by Nematollahi et al. [13]. Moreover, in another study, roasted seeds and nuts were examined by dispersive liquid–liquid microextraction followed by GC-MS, and acrylamide levels ranged between 33.36 and 250.90 µg kg−1 due to variations in the roasting temperature and time as observed by Nematollahi et al. [14].
2.4. Hemoglobin Nanoparticles (HbNPs)
Proteins such as hemoglobin are redox-active and are used as biosensors in detecting acrylamide content in food products as they have a configuration similar to that of glycidamide. These can induce the formation of hemoglobin adducts, causing harmful health effects. Therefore, HbNPs were prepared and used to detect the acrylamide content in foods such as bread, nuts, potato crisps, biscuits, and also snacks like Kurkure by optimizing the pH, time, and acrylamide concentration and were evaluated by various parameters such as the limit of detection (LOD) showing 0.1 nmol L−1, linearity, and recovery percentage as reported in a study by Yadav et al. [15]. In another study, hemoglobin was fixed with the carbon paste electrode to determine the acrylamide content, especially in French fries, which showed the limit of detection of 0.06 nmoL L−1 [16]. However, the hemoglobin carbon ionic liquid paste electrode showed a low limit of detection of 5 × 10−3 nmol L−1 M, as reported in a study by Li et al. [17].
2.5. Fluorescence Biosensor
Due to the high sensitivity and selectivity properties of the fluorescent, it has been of great significance in the field of the food industry in determining the acrylamide content. Fluorescence biosensors, utilizing gold nanoparticles, double-stranded DNA, and carbon quantum dots (CQDs), demonstrate high sensitivity in detecting acrylamide content in food products. This method produces a difference in the fluorescence intensity based on the absence or presence of acrylamide compounds in the food. In the presence of acrylamide, adducts and single-stranded DNA are formed. Therefore, the complementary strand gets absorbed on the surface of the gold nanoparticle, thereby quenching, as reported in a study by Asnaashari et al. [18]. Moreover, fluorescent biosensors based on carbon quantum dots (CQDs) and single-stranded DNA, in the presence of acrylamide, these single-stranded DNA were bound to acrylamide through hydrogen bonding. The presence of acrylamide content in the food showed a higher fluorescence signal than without acrylamide, as reported in other studies [19].
2.6. Surface-Enhanced Raman Spectroscopy (SERS)
Since the 1970s, the intensity of the Raman signal has attained great significance in various fields. Acrylamide content in the fried food was detected using SERS, synthesizing re-oxidized graphene oxide (rGO)/Au nanoparticles. This synthesized rGO/Au composite was mixed with the food sample and observed using a Raman spectrometer at the peak of △v = 1478 cm−1 [20]. However, using SiO2/Ag nanocomposite immersed in filter paper to detect the acrylamide content using the same above method in food products such as cookies, bread, and chips showed no or weak detection signaling with the limit of detection of 0.02 nmol L−1, as reported by Wu et al. [21]. Biodegradable gold-based SERS detected the presence of acrylamide content in the food by showing the peak at 1447 cm−1 in an aqueous solution.
Several detection methods as described above showed a great detection range, among which E-tongue and E-nose are quick and cost-effective, user-friendly, and accurate with an LOD of 2.5 ng g−1 compared to other conventional methods, whereas HbNPs are much more specific and sensitive in detecting the acrylamide content in food due to their catalytical activity with an LOD of 0.06 nmol L−1 as shown in Table 1.
Table 1. Comparison data showing LOD and acrylamide detection range of various detection method.
| Detection Method | Limit of Detection (LOD) | Range of Detection | Sample Model | References |
|---|---|---|---|---|
| E-tongue and nose | 2.5 × 10−3 μg kg−1 | 2.5 × 10−3–20×10−3 μg kg−1 | Olive and brine solution | [6] |
| Fluorescence biosensor | 0.5 × 10−6 nmol L−1 | 0.05 M–10−7 nmol L−1 | Potato fries | [18] |
| 2.41 × 10−2 nmol L−1 | 1 × 10−1–5 × 103 nmol L−1 | Bread crust | [19] | |
| HbNPs | 0.1 nmol L−1 | 0.05–100 nmol L−1 | Bread, nuts, potato crips, biscuits | [15] |
| 0.06 nmol L−1 | 10–171 nmol L−1 | French fries | [16] | |
| SERS | 2 µg kg−1 | 5–100 µg kg−1 | Fried food | [20] |
| 0.02 nmol L−1 | 0.1–5 × 104 nmol L−1 | Cookies, chips, and bread | [21] | |
| LC-MS/MS | 3 ng mL−1 | 16.8–72.8 ng mL−1 | Coffee | [8] |
| 6 µg kg−1 | 197–639 µg kg−1 | Breakfast cereals | [9] | |
| 2 μg kg−1 | 95.8–9826 μg kg−1 | Medicine homologous foods (Atractylodis Macrocephalae Rhizoma) | [11] | |
| GC-MS | 0.6 µg kg−1 | 1–500 µg kg−1 | Bread, biscuits, wafers, cakes, cookies, and crackers | [13] |
| 0.6 µg kg−1 | 33.36–250.90 µg kg−1 | Roasted nuts and seeds | [14] |
3. Mitigation Strategies of Dietary Acrylamide
Several studies have been conducted to reduce the amount of acrylamide content in carbohydrate-rich foods at the industrial and household levels. The Food and Drug Administration (FDA) and European Regulation (EU) reported mitigation strategies to reduce the acrylamide content in food. It was a challenge to the food industry to reduce the acrylamide content of the food without changing the texture, taste, or appearance of the food product. There are several mitigation strategies to overcome the effect of acrylamide content in food, such as air and vacuum-frying, blanching, additives, pH and water content, fermentation, hydrocolloid coating, and cooking under a controlled atmosphere.
3.1. Air and Vacuum-Frying
The air-frying strategy has proven effective in reducing the acrylamide content of food. Vacuum-frying is another method of frying to avoid acrylamide formation conducted at a lower temperature and minimum pressure, which is below 6.65 kPa. It consists of a vacuum chamber, a pump to provide low pressure, and a refrigerator condenser to collect the rising steam [22]. Vacuum-frying of the potato crisps reduced the acrylamide content in the food even among the potato containing a higher level of reducing sugar molecules, not exceeding 250 µg kg−1 and moisture content <2.5%, as vacuum-frying operates under lower temperatures, limiting unwanted oxidation processes [23]. Vacuum-frying also significantly reduces the acrylamide content of the food. It removes water content by maintaining a lower temperature under pressure, thereby reducing the concentration of Maillard reaction precursors. Moreover, in other reports, the vacuum-frying method reduced the acrylamide content of the food products, ranging from 72 to 98%, with a moisture content of 4.83% when compared with the conventional baking method at the same moisture level [24].
3.2. Blanching
Blanching, a process where food products are soaked in hot water for some time before cooking, has proven effective in reducing the acrylamide content in food. This process enhances texture, maintains uniformity [25], and prevents the enzymatic browning of food by eliminating the soluble sugar molecules [22]. The blanching process leaches out the glucose and asparagine contents. It inactivates the enzymes, which reduces the precursors required for the Maillard reaction, thereby reducing the acrylamide content in the food. In an experimental study, the blanching process decreased the acrylamide content in the food till the end of the gastric stage of digestion, which can be correlated to the decrease in Schiff base as the free asparagine and reducing sugar contents are leached out [26]. Blanching temperature and time also play a vital role in reducing the acrylamide content. Blanching at a higher temperature, about 70 °C, for a shorter period (10–15 min), was much more efficient as it lowered the acrylamide content [27].
3.3. Addition of Additives
The addition of substances like amino acids, antioxidants, enzymes, salts, and vitamins can reduce the acrylamide content in food [1]. The addition of amino acids such as lysine, glycine, and alanine reduced the acrylamide content in the food as it has a nucleophilic component that binds covalently, thus eliminating acrylamide [28]. Glycine and lysine can have a positive effect by competing with asparagine for the carbonyl group of the sugar moiety and/or forming adducts with acrylamide once it has formed. The SH group of cysteine (or other thiols) can benefit in two ways: forming an adduct with acrylamide and undergoing heat-induced H2S elimination to generate dehydroalanine [CH2=CH(NH2)COOH]. As with acrylamide, the NH2 group of asparagine can then engage in addition reactions with the dehydroalanine’s double bond in a competitive manner. In theory, serine can also be converted to dehydroalanine by removing H2O. The addition of antioxidants also reduces the acrylamide content in the food, influencing the Maillard reaction. Antioxidants extracted from bamboo leaves have been shown to decrease the acrylamide content, as they block the oxidation process of the Maillard reaction to a certain extent [29]. Enzymes such as asparaginase will significantly reduce the acrylamide content in the food as they hydrolyze the asparagine to aspartic acid without altering the taste of the food product, thereby reducing the concentration of the precursor necessary for the Maillard reaction [30]. As reported in a study, the salt solution can lower the acrylamide content in the food, such as NaCl and CaCl2, where the cations inhibit Schiff base formation by interacting with the free asparagine present in the food [31]. Acrylamide generation in the carbohydrate-rich food was reduced after adding vitamins due to their antioxidative properties. Vitamin C (ascorbic acid) and B1 (thiamine) reduced the acrylamide content in the food by almost 60%, and Vitamin B2 (riboflavin) and B5 (pantothenic acid) decreased approximately 30% of the acrylamide content when examined using the amino acid/sugar chemical model system [32]. Acrylamide content was decreased in food when the reducing sugars were replaced with the nonreducing sucrose powder; however, acrylamide content is reduced at very high temperatures due to the rate of degradation exceeding the rate of formation [33].
3.4. pH and Water Content
pH plays a vital role in controlling the acrylamide formation in the food product, as the Maillard reaction is mainly influenced by pH. Lowering the pH by adding acids like citric acid can enormously reduce acrylamide formation. Under acidic conditions, the non-protonated amine is converted to protonated amine, blocking the nucleophilic attack on the Schiff base and thereby reducing the possibility of the formation of acrylamide via the Maillard reactions [34]. The acrylamide formation decreased in the buffered solution of sodium acetate when unsaturated lipids were added to examine the effect of formation under acidic conditions [35]. The water content of the food product also impacts the formation of dietary acrylamide; the water activity is less than 0.4 of the acrylamide content in the food [1].
3.5. Fermentation
The fermentation process of the food product can limit the acrylamide content by altering the time taken for the fermentation. Some studies showed reduced acrylamide content in the food by 39% and 26% when processed under lactic acid fermentation using Streptococcus lutetiensis and Lactobacillus plantarum, respectively; it was also tested using in vitro digestion processes in the gastric stage, where it removed 30% of the acrylamide content, and in the intestine stage, it eliminated about 40%, as observed in a study by Albedwawi [36]. Moreover, another study reported that Pediococcus acidilactici lactic acid bacteria strain reduced the acrylamide content to 5.64 µg kg−1 in bread when inoculated and fermented for about 16 h. Lactic acid bacteria fermentation influenced the taste properties and increased the softness of the bread, as reported by Nachi et al. [37]. Since most people consume roasted coffee, the acrylamide content in the roasted coffee was controlled by using the yeast fermentation process, which reduced it by 70% when mixed with Saccharomyces cerevisiae, also known as baker’s yeast, and some sugar in a tightly closed container and fermented for 48 h at 30 °C, as reported by Akıllıoglu et al. [38]. Prolonged fermentation of carbohydrate-rich food products resulted in a decrease in acrylamide content, attributed to the increased utilization of asparagine over an extended duration.
3.6. Hydrocolloid-Based Coating
Hydrocolloids are long-chain polymers that are hydrophilic in nature, comprising amino and carbonyl groups with a higher molecular weight. Hydrocolloid coating reduces the acrylamide content of food products and is one of the natural and essential mitigation strategies for controlling the acrylamide content of food products [39]. Its ability to bind to lipids, oxygen, and carbon dioxide prevents excess oil absorption and decreases acrylamide formation during the frying process [22]. Hydrocolloid solutions showed positive results in reducing acrylamide formation, which increased the water retention capacity and hindered the Maillard reaction. It also increased the Margin of Exposure (MOE) of the coated French fries, which controlled the carcinogenic toxicity induced by dietary acrylamide [40]. In another study, the rate of acrylamide content decreased when it was coated with aqueous antioxidant plant extracts of Zataria multiflora and Allium hirtifolium, as they contain aldehyde groups in their structures, which limits the free asparagine. Along with the above method, the hydrocolloid coating using alginate and pectin also reduced the acrylamide content in the food as they lower the rate of heat transfer and core temperature of the food products, thereby preventing water evaporation and thus controlling the moisture content of the food, as reported by Zokaei et al. [41]. However, another study has also reported that a lower concentration of hydrocolloid coating reduced the level of acrylamide content in the fish nuggets in the presence of chitosan and gum Arabic, as they contain amino acids and proteins that would influence the formation of acrylamide in the fish crust [42].
3.7. Inhibitory and Inert Baking Atmosphere
Using an inert and inhibitory baking atmosphere can reduce acrylamide formation. An anaerobic baking atmosphere using inert gases such as nitrogen and carbon dioxide decreased the acrylamide content by 50%, whereas inhibitory gases like sulfur dioxide (SO2) decreased the acrylamide content by 99%. It was observed that the reason for completely blocking the acrylamide formation in the bread when baked under an SO2 atmosphere is due to the sulfur atom’s nucleophilic structure, which binds more quickly to the carbonyl group of the reducing sugar than the amino acids, thereby hindering the Maillard reaction. Other than that, eliminating the oxygen from the baking atmosphere can also control the rate of the Maillard reaction. These atmospheric conditions also alter the sensory properties of the bread, where the sensorial properties of the sulfur dioxide atmosphere baked bread got worse compared to the inert gas atmosphere even though the rate of reduction of acrylamide content was higher, as reported by Gülcan et al. [43].
The formation of acrylamide content in the food can be controlled by utilizing various mitigation strategies, among which air and vacuum-frying are two of the most effective strategies with a reduction percentage of 72–98%, and the least effective is hydrocolloid coating, with a reduction percentage of 48%, as discussed above and shown in Table 2. Blanching or hydrocolloid coating followed by cooking at a controlled temperature and maintaining the acidic pH reduced the formation of acrylamide content in food. Moreover, utilizing additives and yeast fermentation is a very promising technology for controlling acrylamide formation by reducing the free asparagine molecules present in food products. Therefore, using mitigation strategies is one of the best ways to avoid the toxicity caused by the intake of acrylamide-rich food.
Table 2. Comparison data showing the reduction percentage of acrylamide using various mitigation strategies.
| Mitigation Strategies |
Reduction Percentage (%) | Sample Model | Reference |
|---|---|---|---|
| Air- and vacuum-frying | 72–98% | Potato chips | [24] |
| Blanching | 65% and 96% | French fries and potato crisps | [27] |
| Additives | 30–60% | Amino acid/sugar chemical model | [32] |
| Fermentation | 70% | Roasted coffee | [38] |
| Hydrocolloid coating | 48% | French fries | [40] |
| Inhibitory and inert baking atmosphere | 50–99% | Bread | [43] |
References
- Rifai, L.; Saleh, F.A. A Review on Acrylamide in Food: Occurrence, Toxicity, and Mitigation Strategies. Int. J. Toxicol. 2020, 39, 93–102.
- Semla, M.; Goc, Z.; Martiniaková, M.; Omelka, R.; Formicki, G. Acrylamide: A Common Food Toxin Related to Physiological Functions and Health. Physiol. Res. 2017, 66, 205–217.
- Ferrer-Aguirre, A.; Romero-González, R.; Vidal, J.L.M.; Frenich, A.G. Simple and Fast Determination of Acrylamide and Metabolites in Potato Chips and Grilled Asparagus by Liquid Chromatography Coupled to Mass Spectrometry. Food Anal. Methods 2016, 9, 1237–1245.
- Timmermann, C.; Mølck, S.; Kadawathagedara, M.; Bjerregaard, A.; Törnqvist, M.; Brantsæter, A.; Pedersen, M. A Review of Dietary Intake of Acrylamide in Humans. Toxics 2021, 9, 155.
- Das, J.; Mishra, H.N. Recent Advances in Sensors for Detecting Food Pathogens, Contaminants, and Toxins: A Review. Eur. Food Res. Technol. 2022, 248, 1125–1148.
- Martín-Vertedor, D.; Rodrigues, N.; Marx, Í.M.G.; Dias, L.G.; Veloso, A.C.A.; Pereira, J.A.; Peres, A.M. Assessing Acrylamide Content in Sterilized Californian-Style Black Table Olives Using HPLC-MS-QQQ and a Potentiometric Electronic Tongue. LWT 2020, 129, 109605.
- Ghalebi, M.; Hamidi, S.; Nemati, M. High-Performance Liquid Chromatography Determination of Acrylamide after Its Extraction from Potato Chips. Pharm. Sci. 2019, 25, 338–344.
- Başaran, B.; Aydın, F.; Kaban, G. The Determination of Acrylamide Content in Brewed Coffee Samples Marketed in Turkey. Food Addit. Contam. Part A 2020, 37, 280–287.
- Mesías, M.; Sáez-Escudero, L.; Morales, F.J.; Delgado-Andrade, C. Reassessment of Acrylamide Content in Breakfast Cereals. Evolution of the Spanish Market from 2006 to 2018. Food Control 2019, 105, 94–101.
- Hai, Y.D.; Tran-Lam, T.-T.; Nguyen, T.Q.; Vu, N.D.; Ma, K.H.; Le, G.T. Acrylamide in Daily Food in the Metropolitan Area of Hanoi, Vietnam. Food Addit. Contam. Part B 2019, 12, 159–166.
- Zhu, B.; Xu, X.; Ye, X.; Zhou, F.; Qian, C.; Chen, J.; Zhang, T.; Ding, Z. Determination and Risk Assessment of Acrylamide in Thermally Processed Atractylodis Macrocephalae Rhizoma. Food Chem. 2021, 352, 129438.
- Schouten, M.A.; Tappi, S.; Angeloni, S.; Cortese, M.; Caprioli, G.; Vittori, S.; Romani, S. Acrylamide Formation and Antioxidant Activity in Coffee during Roasting—A Systematic Study. Food Chem. 2021, 343, 128514.
- Nematollahi, A.; Kamankesh, M.; Hosseini, H.; Ghasemi, J.; Hosseini-Esfahani, F.; Mohammadi, A. Investigation and Determination of Acrylamide in the Main Group of Cereal Products Using Advanced Microextraction Method Coupled with Gas Chromatography-Mass Spectrometry. J. Cereal Sci. 2019, 87, 157–164.
- Nematollahi, A.; Kamankesh, M.; Hosseini, H.; Hadian, Z.; Ghasemi, J.; Mohammadi, A. Investigation and Determination of Acrylamide in 24 Types of Roasted Nuts and Seeds Using Microextraction Method Coupled with Gas Chromatography–Mass Spectrometry: Central Composite Design. J. Food Meas. Charact. 2020, 14, 1249–1260.
- Yadav, N.; Chhillar, A.K.; Pundir, C.S. Preparation, Characterization and Application of Haemoglobin Nanoparticles for Detection of Acrylamide in Processed Foods. Int. J. Biol. Macromol. 2018, 107, 1000–1013.
- Navarro, K.M.; Silva, J.C.; Ossick, M.V.; Nogueira, A.B.; Etchegaray, A.; Mendes, R.K. Low-Cost Electrochemical Determination of Acrylamide in Processed Food Using a Hemoglobin-Iron Magnetic Nanoparticle-Chitosan Modified Carbon Paste Electrode. Anal. Lett. 2021, 54, 1180–1192.
- Li, N.; Liu, X.; Zhu, J.; Zhou, B.; Jing, J.; Wang, A.; Xu, R.; Wen, Z.; Shi, X.; Guo, S. Simple and Sensitive Detection of Acrylamide Based on Hemoglobin Immobilization in Carbon Ionic Liquid Paste Electrode. Food Control 2020, 109, 106764.
- Asnaashari, M.; Esmaeilzadeh Kenari, R.; Farahmandfar, R.; Taghdisi, S.M.; Abnous, K. Fluorescence Quenching Biosensor for Acrylamide Detection in Food Products Based on Double-Stranded DNA and Gold Nanoparticles. Sens. Actuators B Chem. 2018, 265, 339–345.
- Wei, Q.; Zhang, P.; Liu, T.; Pu, H.; Sun, D.-W. A Fluorescence Biosensor Based on Single-Stranded DNA and Carbon Quantum Dots for Acrylamide Detection. Food Chem. 2021, 356, 129668.
- Cheng, J.; Zhang, S.; Wang, S.; Wang, P.; Su, X.-O.; Xie, J. Rapid and Sensitive Detection of Acrylamide in Fried Food Using Dispersive Solid-Phase Extraction Combined with Surface-Enhanced Raman Spectroscopy. Food Chem. 2019, 276, 157–163.
- Wu, L.; Zhang, W.; Liu, C.; Foda, M.F.; Zhu, Y. Strawberry-like SiO2/Ag Nanocomposites Immersed Filter Paper as SERS Substrate for Acrylamide Detection. Food Chem. 2020, 328, 127106.
- Zhang, X.; Zhang, M.; Adhikari, B. Recent Developments in Frying Technologies Applied to Fresh Foods. Trends Food Sci. Technol. 2020, 98, 68–81.
- Belkova, B.; Hradecky, J.; Hurkova, K.; Forstova, V.; Vaclavik, L.; Hajslova, J. Impact of Vacuum Frying on Quality of Potato Crisps and Frying Oil. Food Chem. 2018, 241, 51–59.
- Akkurt, K.; Mogol, B.A.; Gökmen, V. Mitigation of Acrylamide in Baked Potato Chips by Vacuum Baking and Combined Conventional and Vacuum Baking Processes. LWT 2021, 144, 111211.
- Maan, A.A.; Anjum, M.A.; Khan, M.K.I.; Nazir, A.; Saeed, F.; Afzaal, M.; Aadil, R.M. Acrylamide Formation and Different Mitigation Strategies during Food Processing—A Review. Food Rev. Int. 2022, 38, 70–87.
- Sansano, M.; Heredia, A.; Peinado, I.; Andrés, A. Dietary Acrylamide: What Happens during Digestion. Food Chem. 2017, 237, 58–64.
- Ofosu, I.W.; Ankar-Brewoo, G.M.; Lutterodt, H.E.; Benefo, E.O.; Menyah, C.A. Estimated Daily Intake and Risk of Prevailing Acrylamide Content of Alkalized Roasted Cocoa Beans. Sci. Afr. 2019, 6, e00176.
- Hu, H.; Liu, X.; Jiang, L.; Zhang, Q.; Zhang, H. The Relationship between Acrylamide and Various Components during Coffee Roasting and Effect of Amino Acids on Acrylamide Formation. J. Food Process. Preserv. 2021, 45, e15421.
- Sharif, R.; Shahar, S.; Rajab, N.F.; Fenech, M. Dietary Pattern, Genomic Stability and Relative Cancer Risk in Asian Food Landscape. Nutr. Cancer 2022, 74, 1171–1187.
- Jia, R.; Wan, X.; Geng, X.; Xue, D.; Xie, Z.; Chen, C. Microbial L-Asparaginase for Application in Acrylamide Mitigation from Food: Current Research Status and Future Perspectives. Microorganisms 2021, 9, 1659.
- Liyanage, D.W.K.; Yevtushenko, D.P.; Konschuh, M.; Bizimungu, B.; Lu, Z.-X. Processing Strategies to Decrease Acrylamide Formation, Reducing Sugars and Free Asparagine Content in Potato Chips from Three Commercial Cultivars. Food Control 2021, 119, 107452.
- Wang, X.; Xu, L. Influence Factors on the Formation of Acrylamide in the Amino Acid/Sugar Chemical Model System. J. Food Nutr. Res. 2014, 2, 344–348.
- Aarabi, F.; Seyedain Ardebili, M. The Effect of Sugar Type and Baking Condition on Formation of Acrylamide in Industrial Rotary Moulded Biscuit. J. Food Meas. Charact. 2020, 14, 2230–2239.
- Shakeri, F.; Shakeri, S.; Ghasemi, S.; Troise, A.D.; Fiore, A. Effects of Formulation and Baking Process on Acrylamide Formation in Kolompeh, a Traditional Cookie in Iran. J. Chem. 2019, 2019, 1425098.
- Wang, Y.; Hu, H.; McClements, D.J.; Nie, S.; Shen, M.; Li, C.; Huang, Y.; Zhong, Y.; Chen, J.; Zeng, M.; et al. PH and Lipid Unsaturation Impact the Formation of Acrylamide and 5-Hydroxymethylfurfural in Model System at Frying Temperature. Food Res. Int. 2019, 123, 403–413.
- Albedwawi, A.; Al Sakkaf, R.; Yusuf, A.; Osaili, T.; Al-Nabulsi, A.; Liu, S.-Q.; Palmisano, G.; Ayyash, M. Acrylamide Elimination by Lactic Acid Bacteria: Screening, Optimization, In Vitro Digestion, and Mechanism. Microorganisms 2022, 10, 557.
- Nachi, I.; Fhoula, I.; Smida, I.; Ben Taher, I.; Chouaibi, M.; Jaunbergs, J.; Bartkevics, V.; Hassouna, M. Assessment of Lactic Acid Bacteria Application for the Reduction of Acrylamide Formation in Bread. LWT 2018, 92, 435–441.
- Akıllıoglu, H.G.; Gökmen, V. Mitigation of Acrylamide and Hydroxymethyl Furfural in Instant Coffee by Yeast Fermentation. Food Res. Int. 2014, 61, 252–256.
- Zhang, N.; Zhou, Q.; Fan, D.; Xiao, J.; Zhao, Y.; Cheng, K.-W.; Wang, M. Novel Roles of Hydrocolloids in Foods: Inhibition of Toxic Maillard Reaction Products Formation and Attenuation of Their Harmful Effects. Trends Food Sci. Technol. 2021, 111, 706–715.
- Aiswarya, R.; Baskar, G. Enzymatic Mitigation of Acrylamide in Fried Potato Chips Using Asparaginase from Aspergillus Terreus. Int. J. Food Sci. Technol. 2018, 53, 491–498.
- Zokaei, M.; Kamankesh, M.; Abedi, A.-S.; Moosavi, M.H.; Mohammadi, A.; Rezvani, M.; Shojaee-Aliabadi, S.; Khaneghah, A.M. Reduction in Acrylamide Formation in Potato Crisps: Application of Extract and Hydrocolloid-Based Coatings. J. Food Prot. 2020, 83, 754–761.
- Jiang, Y.; Qin, R.; Jia, C.; Rong, J.; Hu, Y.; Liu, R. Hydrocolloid Effects on Nε-Carboxymethyllysine and Acrylamide of Deep-Fried Fish Nuggets. Food Biosci. 2021, 39, 100797.
- Gülcan, Ü.; Candal Uslu, C.; Mutlu, C.; Arslan-Tontul, S.; Erbaş, M. Impact of Inert and Inhibitor Baking Atmosphere on HMF and Acrylamide Formation in Bread. Food Chem. 2020, 332, 127434.
More
Information
Subjects:
Food Science & Technology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
583
Revisions:
2 times
(View History)
Update Date:
19 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





