
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Adelina Vlad | -- | 3723 | 2024-02-17 10:32:59 | | | |
| 2 | Lindsay Dong | Meta information modification | 3723 | 2024-02-18 02:27:43 | | |
Video Upload Options
Skin diseases represent a global healthcare challenge due to their rising incidence and substantial socio-economic burden. While biological, immunological, and targeted therapies have brought a revolution in improving quality of life and survival rates for certain dermatological conditions, there remains a stringent demand for new remedies. Nature has long served as an inspiration for drug development. Recent studies have identified bitter taste receptors (TAS2Rs) in both skin cell lines and human skin. Additionally, bitter natural compounds have shown promising benefits in addressing skin aging, wound healing, inflammatory skin conditions, and even skin cancer. Thus, TAS2Rs may represent a promising target in all these processes.
1. Introduction
2. Bitter Taste Receptors (TAS2R)-Types and Signaling Pathways
3. Extraoral Bitter Taste Receptors and Their Biological Roles
4. Differential Expression of Bitter Taste Receptors in Human Skin
5. Functionality of Bitter Taste Receptors in Skin
5.1. Bitter Taste Receptors as Chemosensory Receptors
5.2. Bitter Taste Receptors as Regulators of Keratinocyte Differentiation and Skin Barrier Structural and Functional Integrity
5.3. Influence of Bitter Taste Receptors on Aging and Wound Healing
5.4. Bitter Taste Receptors as Regulators of Hair Follicle Growth
5.5. Bitter Taste Receptors as Modulators of Skin Immunity and Oral Microbiome
5.6. Bitter Taste Receptors as Regulators of Adipocyte Functions
5.7. Involvement of Bitter Taste Receptors in Skin and Oral Cancers
5.7.1. Skin Melanoma
5.7.2. Oral Squamous Cell Carcinoma
5.8. Other Potential Functions of Bitter Taste Receptors
6. Chemical and Orosensorial Complexity of Bitter Phytochemicals
7. Bitter Phytochemicals Active on Skin Inflammation, Skin Carcinogenesis, and Wound Healing
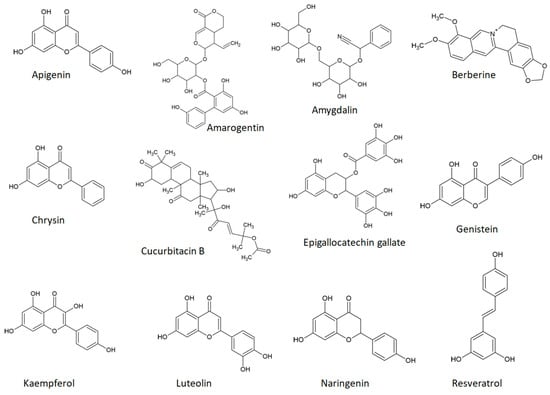
8. Phytochemical Bitter Taste Receptors agonists in Skin Aging, Inflammation, and Cancer: Insights into mammalian Target of rapamycin (mTOR) Signaling Pathways
9. Direct Involvement of Bitter Taste Receptors in the Bitter Phytochemicals’ Anti-Inflammatory and Anti-Cancer Effects
10. Conclusions
References
- McGrath, J.A.; Uitto, J. Structure and Function of the Skin. In Rook’s Textbook of Dermatology, 9th ed.; Griffiths, C.E.M., Barker, J., Bleiker, T.O., Chalmers, R., Creamer, D., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2016; pp. 1–52. ISBN 9781118441213.
- Flohr, C.; Hay, R. Putting the Burden of Skin Diseases on the Global Map. Br. J. Dermatol. 2021, 184, 189–190.
- Ujiie, H.; Rosmarin, D.; Schön, M.P.; Ständer, S.; Boch, K.; Metz, M.; Maurer, M.; Thaci, D.; Schmidt, E.; Cole, C.; et al. Unmet Medical Needs in Chronic, Non-Communicable Inflammatory Skin Diseases. Front. Med. 2022, 9, 875492.
- Carretero, M.; Guerrero-Aspizua, S.; Illera, N.; Galvez, V.; Navarro, M.; García-García, F.; Dopazo, J.; Jorcano, J.L.; Larcher, F.; del Rio, M. Differential Features between Chronic Skin Inflammatory Diseases Revealed in Skin-Humanized Psoriasis and Atopic Dermatitis Mouse Models. J. Investig. Dermatol. 2016, 136, 136–145.
- Snyder, A.M.; Brandenberger, A.U.; Taliercio, V.L.; Rich, B.E.; Webber, L.B.; Beshay, A.P.; Biber, J.E.; Hess, R.; Rhoads, J.L.W.; Secrest, A.M. Quality of Life Among Family of Patients with Atopic Dermatitis and Psoriasis. Int. J. Behav. Med. 2023, 30, 409–415.
- Narayanan, D.L.; Saladi, R.N.; Fox, J.L. Review: Ultraviolet Radiation and Skin Cancer. Int. J. Dermatol. 2010, 49, 978–986.
- Becerril, S.; Corchado-Cobos, R.; García-Sancha, N.; Revelles, L.; Revilla, D.; Ugalde, T.; Román-Curto, C.; Pérez-Losada, J.; Cañueto, J. Viruses and Skin Cancer. Int. J. Mol. Sci. 2021, 22, 5399.
- Rollan, M.P.; Cabrera, R.; Schwartz, R.A. Current Knowledge of Immunosuppression as a Risk Factor for Skin Cancer Development. Crit. Rev. Oncol. Hematol. 2022, 177, 103754.
- Facciolà, A.; Venanzi Rullo, E.; Ceccarelli, M.; D’Andrea, F.; Coco, M.; Micali, C.; Cacopardo, B.; Marino, A.; Cannavò, S.P.; Di Rosa, M.; et al. Malignant Melanoma in HIV: Epidemiology, Pathogenesis, and Management. Dermatol. Ther. 2020, 33, e13180.
- Nikolaou, V.; Stratigos, A.J.; Tsao, H. Hereditary Nonmelanoma Skin Cancer. Semin. Cutan. Med. Surg. 2012, 31, 204–210.
- Zocchi, L.; Lontano, A.; Merli, M.; Dika, E.; Nagore, E.; Quaglino, P.; Puig, S.; Ribero, S. Familial Melanoma and Susceptibility Genes: A Review of the Most Common Clinical and Dermoscopic Phenotypic Aspect, Associated Malignancies and Practical Tips for Management. J. Clin. Med. 2021, 10, 3760.
- Shreberk-Hassidim, R.; Ostrowski, S.M.; Fisher, D.E. The Complex Interplay between Nevi and Melanoma: Risk Factors and Precursors. Int. J. Mol. Sci. 2023, 24, 3541.
- Arafa, A.; Mostafa, A.; Navarini, A.A.; Dong, J.-Y. The Association between Smoking and Risk of Skin Cancer: A Meta-Analysis of Cohort Studies. Cancer Causes Control 2020, 31, 787–794.
- Matthews, N.H.; Fitch, K.; Li, W.-Q.; Morris, J.S.; Christiani, D.C.; Qureshi, A.A.; Cho, E. Exposure to Trace Elements and Risk of Skin Cancer: A Systematic Review of Epidemiologic Studies. Cancer Epidemiol. Biomark. Prev. 2019, 28, 3–21.
- Bennardo, L.; Passante, M.; Cameli, N.; Cristaudo, A.; Patruno, C.; Nisticò, S.P.; Silvestri, M. Skin Manifestations after Ionizing Radiation Exposure: A Systematic Review. Bioengineering 2021, 8, 153.
- Neagu, M.; Constantin, C.; Caruntu, C.; Dumitru, C.; Surcel, M.; Zurac, S. Inflammation: A Key Process in Skin Tumorigenesis (Review). Oncol. Lett. 2018, 17, 4068–4084.
- Ju, T.; Hernandez, L.; Mohsin, N.; Labib, A.; Frech, F.; Nouri, K. Evaluation of Risk in Chronic Cutaneous Inflammatory Conditions for Malignant Transformation. J. Eur. Acad. Dermatol. Venereol. 2023, 37, 231–242.
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249.
- Sackstein, R.; Schatton, T.; Barthel, S.R. T-Lymphocyte Homing: An Underappreciated yet Critical Hurdle for Successful Cancer Immunotherapy. Lab. Investig. 2017, 97, 669–697.
- Spałkowska, M.; Dyduch, G.; Broniatowska, E.; Damiani, G.; Wojas-Pelc, A. Molecular Proof of a Clinical Concept: Expression of Estrogen Alpha-, Beta-Receptors and G Protein-Coupled Estrogen Receptor 1 (GPER) in Histologically Assessed Common Nevi, Dysplastic Nevi and Melanomas. Medicina 2021, 57, 1228.
- Rees, J.L. The Genetics of Sun Sensitivity in Humans. Am. J. Hum. Genet. 2004, 75, 739–751.
- Yamaguchi, Y.; Passeron, T.; Watabe, H.; Yasumoto, K.; Rouzaud, F.; Hoashi, T.; Hearing, V.J. The Effects of Dickkopf 1 on Gene Expression and Wnt Signaling by Melanocytes: Mechanisms Underlying Its Suppression of Melanocyte Function and Proliferation. J. Investig. Dermatol. 2007, 127, 1217–1225.
- Labes, A. Marine Resources Offer New Compounds and Strategies for the Treatment of Skin and Soft Tissue Infections. Mar. Drugs 2023, 21, 387.
- Liu, J.-K. Natural Products in Cosmetics. Nat. Prod. Bioprospect. 2022, 12, 40.
- Nicholas-Haizelden, K.; Murphy, B.; Hoptroff, M.; Horsburgh, M.J. Bioprospecting the Skin Microbiome: Advances in Therapeutics and Personal Care Products. Microorganisms 2023, 11, 1899.
- Yoon, J.H.; Kim, M.-Y.; Cho, J.Y. Apigenin: A Therapeutic Agent for Treatment of Skin Inflammatory Diseases and Cancer. Int. J. Mol. Sci. 2023, 24, 1498.
- Peterle, L.; Sanfilippo, S.; Borgia, F.; Li Pomi, F.; Vadalà, R.; Costa, R.; Cicero, N.; Gangemi, S. The Role of Nutraceuticals and Functional Foods in Skin Cancer: Mechanisms and Therapeutic Potential. Foods 2023, 12, 2629.
- Dragoș, D.; Petran, M.; Gradinaru, T.-C.; Gilca, M. Phytochemicals and Inflammation: Is Bitter Better? Plants 2022, 11, 2991.
- Grădinaru, T.-C.; Gilca, M.; Vlad, A.; Dragoș, D. Relevance of Phytochemical Taste for Anti-Cancer Activity: A Statistical Inquiry. Int. J. Mol. Sci. 2023, 24, 16227.
- Reed, D.R.; Knaapila, A. Genetics of Taste and Smell. In Progress in Molecular Biology and Translational Science; Elsevier: Amsterdam, The Netherlands, 2010; Volume 94, pp. 213–240.
- Lindemann, B. Taste Reception. Physiol. Rev. 1996, 76, 718–766.
- Lang, T.; Di Pizio, A.; Risso, D.; Drayna, D.; Behrens, M. Activation Profile of TAS2R2, the 26th Human Bitter Taste Receptor. Mol. Nutr. Food Res. 2023, 67, e2200775.
- Sanematsu, K.; Yoshida, R.; Shigemura, N.; Ninomiya, Y. Structure, Function, and Signaling of Taste G-Protein-Coupled Receptors. Curr. Pharm. Biotechnol. 2014, 15, 951–961.
- Ahmad, R.; Dalziel, J.E. G Protein-Coupled Receptors in Taste Physiology and Pharmacology. Front. Pharmacol. 2020, 11, 587664.
- Talmon, M.; Pollastro, F.; Fresu, L.G. The Complex Journey of the Calcium Regulation Downstream of TAS2R Activation. Cells 2022, 11, 3638.
- Ho, H.K.-Y.; Bigliardi, P.L.; Stelmashenko, O.; Ramasamy, S.; Postlethwaite, M.; Bigliardi-Qi, M. Functionally Expressed Bitter Taste Receptor TAS2R14 in Human Epidermal Keratinocytes Serves as a Chemosensory Receptor. Exp. Dermatol. 2021, 30, 216–225.
- Meyerhof, W.; Batram, C.; Kuhn, C.; Brockhoff, A.; Chudoba, E.; Bufe, B.; Appendino, G.; Behrens, M. The Molecular Receptive Ranges of Human TAS2R Bitter Taste Receptors. Chem. Senses 2010, 35, 157–170.
- Ji, M.; Su, X.; Su, X.; Chen, Y.; Huang, W.; Zhang, J.; Gao, Z.; Li, C.; Lu, X. Identification of Novel Compounds for Human Bitter Taste Receptors. Chem. Biol. Drug Des. 2014, 84, 63–74.
- Di Pizio, A.; Niv, M.Y. Promiscuity and Selectivity of Bitter Molecules and Their Receptors. Bioorg. Med. Chem. 2015, 23, 4082–4091.
- Thalmann, S.; Behrens, M.; Meyerhof, W. Major Haplotypes of the Human Bitter Taste Receptor TAS2R41 Encode Functional Receptors for Chloramphenicol. Biochem. Biophys. Res. Commun. 2013, 435, 267–273.
- Lossow, K.; Hubner, S.; Roudnitzky, N.; Slack, J.P.; Pollastro, F.; Behrens, M.; Meyerhof, W. Comprehensive Analysis of Mouse Bitter Taste Receptors Reveals Different Molecular Receptive Ranges for Orthologous Receptors in Mice and Humans. J. Biol. Chem. 2016, 291, 15358–15377.
- Tuzim, K.; Korolczuk, A. An Update on Extra-Oral Bitter Taste Receptors. J. Transl. Med. 2021, 19, 440.
- Behrens, M.; Brockhoff, A.; Kuhn, C.; Bufe, B.; Winnig, M.; Meyerhof, W. The Human Taste Receptor HTAS2R14 Responds to a Variety of Different Bitter Compounds. Biochem. Biophys. Res. Commun. 2004, 319, 479–485.
- Brockhoff, A.; Behrens, M.; Massarotti, A.; Appending, G.; Meyerhof, W. Broad Tuning of the Human Bitter Taste Receptor HTAS2R46 to Various Sesquiterpene Lactones, Clerodane and Labdane Diterpenoids, Strychnine, and Denatonium. J. Agric. Food Chem. 2007, 55, 6236–6243.
- Yamazaki, T.; Sagisaka, M.; Ikeda, R.; Nakamura, T.; Matsuda, N.; Ishii, T.; Nakayama, T.; Watanabe, T. The Human Bitter Taste Receptor HTAS2R39 Is the Primary Receptor for the Bitterness of Theaflavins. Biosci. Biotechnol. Biochem. 2014, 78, 1753–1756.
- Foster, S.R.; Porrello, E.R.; Purdue, B.; Chan, H.-W.; Voigt, A.; Frenzel, S.; Hannan, R.D.; Moritz, K.M.; Simmons, D.G.; Molenaar, P.; et al. Expression, Regulation and Putative Nutrient-Sensing Function of Taste GPCRs in the Heart. PLoS ONE 2013, 8, e64579.
- Kertesz, Z.; Harrington, E.O.; Braza, J.; Guarino, B.D.; Chichger, H. Agonists for Bitter Taste Receptors T2R10 and T2R38 Attenuate LPS-Induced Permeability of the Pulmonary Endothelium in Vitro. Front. Physiol. 2022, 13, 794370.
- Kaji, I.; Karaki, S.; Fukami, Y.; Terasaki, M.; Kuwahara, A. Secretory Effects of a Luminal Bitter Tastant and Expressions of Bitter Taste Receptors, T2Rs, in the Human and Rat Large Intestine. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 296, G971–G981.
- Jeon, T.-I.; Seo, Y.-K.; Osborne, T.F. Gut Bitter Taste Receptor Signalling Induces ABCB1 through a Mechanism Involving CCK. Biochem. J. 2011, 438, 33–37.
- Liszt, K.I.; Wang, Q.; Farhadipour, M.; Segers, A.; Thijs, T.; Nys, L.; Deleus, E.; Van der Schueren, B.; Gerner, C.; Neuditschko, B.; et al. Human Intestinal Bitter Taste Receptors Regulate Innate Immune Responses and Metabolic Regulators in Obesity. J. Clin. Investig. 2022, 132, 1–16.
- Clark, A.A.; Dotson, C.D.; Elson, A.E.T.; Voigt, A.; Boehm, U.; Meyerhof, W.; Steinle, N.I.; Munger, S.D. TAS2R Bitter Taste Receptors Regulate Thyroid Function. FASEB J. 2015, 29, 164–172.
- Malki, A.; Fiedler, J.; Fricke, K.; Ballweg, I.; Pfaffl, M.W.; Krautwurst, D. Class I Odorant Receptors, TAS1R and TAS2R Taste Receptors, Are Markers for Subpopulations of Circulating Leukocytes. J. Leukoc. Biol. 2015, 97, 533–545.
- Tran, H.T.T.; Herz, C.; Ruf, P.; Stetter, R.; Lamy, E. Human T2R38 Bitter Taste Receptor Expression in Resting and Activated Lymphocytes. Front. Immunol. 2018, 9, 2949.
- Shaw, L.; Mansfield, C.; Colquitt, L.; Lin, C.; Ferreira, J.; Emmetsberger, J.; Reed, D.R. Personalized Expression of Bitter ‘Taste’ Receptors in Human Skin. PLoS ONE 2018, 13, e0205322.
- Reszka, E.; Nowakowska-Swirta, E.; Kupczyk, M.; Dudek, W.; Swierczynska-Machura, D.; Wittczak, T.; Rykała, J.; Przybek, M. Expression of Bitter Taste Receptors in the Human Skin In Vitro. J. Clin. Res. Bioeth. 2015, 6, 1000218.
- Wolfle, U.; Elsholz, F.A.; Kersten, A.; Haarhaus, B.; Muller, W.E.; Schempp, C.M. Expression and Functional Activity of the Bitter Taste Receptors TAS2R1 and TAS2R38 in Human Keratinocytes. Skin Pharmacol. Physiol. 2015, 28, 137–146.
- Talmon, M.; Massara, E.; Quaregna, M.; De Battisti, M.; Boccafoschi, F.; Lecchi, G.; Puppo, F.; Bettega Cajandab, M.A.; Salamone, S.; Bovio, E.; et al. Bitter Taste Receptor (TAS2R) 46 in Human Skeletal Muscle: Expression and Activity. Front. Pharmacol. 2023, 14, 1205651.
- Garcia-Esparcia, P.; Schluter, A.; Carmona, M.; Moreno, J.; Ansoleaga, B.; Torrejon-Escribano, B.; Gustincich, S.; Pujol, A.; Ferrer, I. Functional Genomics Reveals Dysregulation of Cortical Olfactory Receptors in Parkinson Disease: Novel Putative Chemoreceptors in the Human Brain. J. Neuropathol. Exp. Neurol. 2013, 72, 524–539.
- Wölfle, U.; Haarhaus, B.; Kersten, A.; Fiebich, B.; Hug, M.J.; Schempp, C.M. Salicin from Willow Bark Can Modulate Neurite Outgrowth in Human Neuroblastoma SH-SY5Y Cells. Phyther. Res. 2015, 29, 1494–1500.
- Ansoleaga, B.; Garcia-Esparcia, P.; Pinacho, R.; Haro, J.M.; Ramos, B.; Ferrer, I. Decrease in Olfactory and Taste Receptor Expression in the Dorsolateral Prefrontal Cortex in Chronic Schizophrenia. J. Psychiatr. Res. 2015, 60, 109–116.
- Ansoleaga, B.; Garcia-Esparcia, P.; Llorens, F.; Moreno, J.; Aso, E.; Ferrer, I. Dysregulation of Brain Olfactory and Taste Receptors in AD, PSP and CJD, and AD-Related Model. Neuroscience 2013, 248, 369–382.
- Zheng, M.; Simon, R.; Mirlacher, M.; Maurer, R.; Gasser, T.; Forster, T.; Diener, P.A.; Mihatsch, M.J.; Sauter, G.; Schraml, P. TRIO Amplification and Abundant MRNA Expression Is Associated with Invasive Tumor Growth and Rapid Tumor Cell Proliferation in Urinary Bladder Cancer. Am. J. Pathol. 2004, 165, 63–69.
- Wölfle, U.; Elsholz, F.; Kersten, A.; Haarhaus, B.; Schumacher, U.; Schempp, C. Expression and Functional Activity of the Human Bitter Taste Receptor TAS2R38 in Human Placental Tissues and JEG-3 Cells. Molecules 2016, 21, 306.
- Governini, L.; Semplici, B.; Pavone, V.; Crifasi, L.; Marrocco, C.; De Leo, V.; Arlt, E.; Gudermann, T.; Boekhoff, I.; Luddi, A.; et al. Expression of Taste Receptor 2 Subtypes in Human Testis and Sperm. J. Clin. Med. 2020, 9, 264.
- Semplici, B.; Luongo, F.P.; Passaponti, S.; Landi, C.; Governini, L.; Morgante, G.; De Leo, V.; Piomboni, P.; Luddi, A. Bitter Taste Receptors Expression in Human Granulosa and Cumulus Cells: New Perspectives in Female Fertility. Cells 2021, 10, 3127.
- Martin, L.T.P.; Nachtigal, M.W.; Selman, T.; Nguyen, E.; Salsman, J.; Dellaire, G.; Dupré, D.J. Bitter Taste Receptors Are Expressed in Human Epithelial Ovarian and Prostate Cancers Cells and Noscapine Stimulation Impacts Cell Survival. Mol. Cell. Biochem. 2019, 454, 203–214.
- Shah, A.S.; Ben-Shahar, Y.; Moninger, T.O.; Kline, J.N.; Welsh, M.J. Motile Cilia of Human Airway Epithelia Are Chemosensory. Science 2009, 325, 1131–1134.
- Grassin-Delyle, S.; Abrial, C.; Fayad-Kobeissi, S.; Brollo, M.; Faisy, C.; Alvarez, J.-C.; Naline, E.; Devillier, P. The Expression and Relaxant Effect of Bitter Taste Receptors in Human Bronchi. Respir. Res. 2013, 14, 134.
- Barham, H.P.; Cooper, S.E.; Anderson, C.B.; Tizzano, M.; Kingdom, T.T.; Finger, T.E.; Kinnamon, S.C.; Ramakrishnan, V.R. Solitary Chemosensory Cells and Bitter Taste Receptor Signaling in Human Sinonasal Mucosa. Int. Forum Allergy Rhinol. 2013, 3, 450–457.
- Lund, T.C.; Kobs, A.J.; Kramer, A.; Nyquist, M.; Kuroki, M.T.; Osborn, J.; Lidke, D.S.; Low-Nam, S.T.; Blazar, B.R.; Tolar, J. Bone Marrow Stromal and Vascular Smooth Muscle Cells Have Chemosensory Capacity via Bitter Taste Receptor Expression. PLoS ONE 2013, 8, e58945.
- Cheng, W.; Yao, M.; Liu, F. Bitter Taste Receptor as a Therapeutic Target in Orthopaedic Disorders. Drug Des. Devel. Ther. 2021, 15, 895–903.
- Wang, Y.; Zajac, A.L.; Lei, W.; Christensen, C.M.; Margolskee, R.F.; Bouysset, C.; Golebiowski, J.; Zhao, H.; Fiorucci, S.; Jiang, P. Metal Ions Activate the Human Taste Receptor TAS2R7. Chem. Senses 2019, 44, 339–347.
- Gaida, M.; Dapunt, U.; Hansch, G.M. Sensing Developing Biofilms: The Bitter Receptor T2R38 on Myeloid Cells. Pathog. Dis. 2016, 74, ftw004.
- Kang, W.; Wang, Y.; Li, J.; Xie, W.; Zhao, D.; Wu, L.; Wang, H.; Xie, S. TAS2R Supports Odontoblastic Differentiation of Human Dental Pulp Stem Cells in the Inflammatory Microenvironment. Stem Cell Res. Ther. 2022, 13, 374.
- Orsmark-Pietras, C.; James, A.; Konradsen, J.R.; Nordlund, B.; Söderhäll, C.; Pulkkinen, V.; Pedroletti, C.; Daham, K.; Kupczyk, M.; Dahlén, B.; et al. Transcriptome Analysis Reveals Upregulation of Bitter Taste Receptors in Severe Asthmatics. Eur. Respir. J. 2013, 42, 65–78.
- Singh, N.; Chakraborty, R.; Bhullar, R.P.; Chelikani, P. Differential Expression of Bitter Taste Receptors in Non-Cancerous Breast Epithelial and Breast Cancer Cells. Biochem. Biophys. Res. Commun. 2014, 446, 499–503.
- Carey, R.M.; Kim, T.; Cohen, N.A.; Lee, R.J.; Nead, K.T. Impact of Sweet, Umami, and Bitter Taste Receptor (TAS1R and TAS2R) Genomic and Expression Alterations in Solid Tumors on Survival. Sci. Rep. 2022, 12, 8937.
- Lee, R.J.; Cohen, N.A. Taste Receptors in Innate Immunity. Cell. Mol. Life Sci. 2015, 72, 217–236.
- Patel, N.N.; Workman, A.D.; Cohen, N.A. Role of Taste Receptors as Sentinels of Innate Immunity in the Upper Airway. J. Pathog. 2018, 2018, 9541987.
- Xi, R.; Zheng, X.; Tizzano, M. Role of Taste Receptors in Innate Immunity and Oral Health. J. Dent. Res. 2022, 101, 759–768.
- Tran, H.T.T.; Stetter, R.; Herz, C.; Spöttel, J.; Krell, M.; Hanschen, F.S.; Schreiner, M.; Rohn, S.; Behrens, M.; Lamy, E. Allyl Isothiocyanate: A TAS2R38 Receptor-Dependent Immune Modulator at the Interface Between Personalized Medicine and Nutrition. Front. Immunol. 2021, 12, 669005.
- Grassin-Delyle, S.; Salvator, H.; Mantov, N.; Abrial, C.; Brollo, M.; Faisy, C.; Naline, E.; Couderc, L.-J.; Devillier, P. Bitter Taste Receptors (TAS2Rs) in Human Lung Macrophages: Receptor Expression and Inhibitory Effects of TAS2R Agonists. Front. Physiol. 2019, 10, 1267.
- Zhou, Z.; Xi, R.; Liu, J.; Peng, X.; Zhao, L.; Zhou, X.; Li, J.; Zheng, X.; Xu, X. TAS2R16 Activation Suppresses LPS-Induced Cytokine Expression in Human Gingival Fibroblasts. Front. Immunol. 2021, 12, 726546.
- Liszt, K.I.; Ley, J.P.; Lieder, B.; Behrens, M.; Stöger, V.; Reiner, A.; Hochkogler, C.M.; Köck, E.; Marchiori, A.; Hans, J.; et al. Caffeine Induces Gastric Acid Secretion via Bitter Taste Signaling in Gastric Parietal Cells. Proc. Natl. Acad. Sci. USA 2017, 114, E6260–E6269.
- Grau-Bové, C.; Miguéns-Gómez, A.; González-Quilen, C.; Fernández-López, J.-A.; Remesar, X.; Torres-Fuentes, C.; Ávila-Román, J.; Rodríguez-Gallego, E.; Beltrán-Debón, R.; Blay, M.T.; et al. Modulation of Food Intake by Differential TAS2R Stimulation in Rat. Nutrients 2020, 12, 3784.
- Zagorchev, P.; Petkov, G.V.; Gagov, H.S. Bitter Taste Receptors as Regulators of Abdominal Muscles Contraction. Physiol. Res. 2019, 68, 991–995.
- Zhou, Y.-W.; Sun, J.; Wang, Y.; Chen, C.-P.; Tao, T.; Ma, M.; Chen, X.; Zhang, X.-N.; Yang, L.-Y.; Zhang, Z.-L.; et al. Tas2R Activation Relaxes Airway Smooth Muscle by Release of Gα t Targeting on AChR Signaling. Proc. Natl. Acad. Sci. USA 2022, 119, e2121513119.
- Costa, A.R.; Duarte, A.C.; Costa-Brito, A.R.; Gonçalves, I.; Santos, C.R.A. Bitter Taste Signaling in Cancer. Life Sci. 2023, 315, 121363.
- Zehentner, S.; Reiner, A.T.; Grimm, C.; Somoza, V. The Role of Bitter Taste Receptors in Cancer: A Systematic Review. Cancers 2021, 13, 5891.
- Seo, Y.; Kim, Y.-S.; Lee, K.E.; Park, T.H.; Kim, Y. Anti-Cancer Stemness and Anti-Invasive Activity of Bitter Taste Receptors, TAS2R8 and TAS2R10, in Human Neuroblastoma Cells. PLoS ONE 2017, 12, e0176851.
- Pham, H.; Hui, H.; Morvaridi, S.; Cai, J.; Zhang, S.; Tan, J.; Wu, V.; Levin, N.; Knudsen, B.; Goddard, W.A.; et al. A Bitter Pill for Type 2 Diabetes? The Activation of Bitter Taste Receptor TAS2R38 Can Stimulate GLP-1 Release from Enteroendocrine L-Cells. Biochem. Biophys. Res. Commun. 2016, 475, 295–300.
- Choi, J.-H. Variation in the TAS2R38 Bitterness Receptor Gene Was Associated with Food Consumption and Obesity Risk in Koreans. Nutrients 2019, 11, 1973.
- Sun, S.; Yang, Y.; Xiong, R.; Ni, Y.; Ma, X.; Hou, M.; Chen, L.; Xu, Z.; Chen, L.; Ji, M. Oral Berberine Ameliorates High-Fat Diet-Induced Obesity by Activating TAS2Rs in Tuft and Endocrine Cells in the Gut. Life Sci. 2022, 311, 121141.
- Lee, R.J.; Cohen, N.A. Role of the Bitter Taste Receptor T2R38 in Upper Respiratory Infection and Chronic Rhinosinusitis. Curr. Opin. Allergy Clin. Immunol. 2015, 15, 14–20.
- Luo, X.-C.; Chen, Z.-H.; Xue, J.-B.; Zhao, D.-X.; Lu, C.; Li, Y.-H.; Li, S.-M.; Du, Y.-W.; Liu, Q.; Wang, P.; et al. Infection by the Parasitic Helminth Trichinella Spiralis Activates a Tas2r-Mediated Signaling Pathway in Intestinal Tuft Cells. Proc. Natl. Acad. Sci. USA 2019, 116, 5564–5569.
- Wölfle, U.; Haarhaus, B.; Schempp, C.M. Amarogentin Displays Immunomodulatory Effects in Human Mast Cells and Keratinocytes. Mediat. Inflamm. 2015, 2015, 630128.
- Sakakibara, M.; Sumida, H.; Yanagida, K.; Miyasato, S.; Nakamura, M.; Sato, S. Bitter Taste Receptor T2R38 Is Expressed on Skin-Infiltrating Lymphocytes and Regulates Lymphocyte Migration. Sci. Rep. 2022, 12, 11790.
- Gherardini, J.; Rouille, T.; Ferholz, M.; Funk, W.; Rodríguez-Feliz, J.; Bauman, A.J.; Bíró, T.; Chéret, J.; Paus, R. 571 Human Hair Follicles Can “Taste”: Stimulation of the Bitter Taste Receptor TAS2R4 Inhibits Hair Growth Ex Vivo by Up-Regulating TGF-Β2. J. Investig. Dermatol. 2022, 142, S279.
- Chung, M.G.; Kim, Y.; Cha, Y.K.; Park, T.H.; Kim, Y. Bitter Taste Receptors Protect against Skin Aging by Inhibiting Cellular Senescence and Enhancing Wound Healing. Nutr. Res. Pract. 2022, 16, 1.
- Wölfle, U.; Haarhaus, B.; Seiwerth, J.; Cawelius, A.; Schwabe, K.; Quirin, K.-W.; Schempp, C. The Herbal Bitter Drug Gentiana Lutea Modulates Lipid Synthesis in Human Keratinocytes In Vitro and In Vivo. Int. J. Mol. Sci. 2017, 18, 1814.
- de Jesus, V.C.; Mittermuller, B.-A.; Hu, P.; Schroth, R.J.; Chelikani, P. Genetic Variants in Taste Genes Play a Role in Oral Microbial Composition and Severe Early Childhood Caries. iScience 2022, 25, 105489.
- Lumpkin, E.A.; Caterina, M.J. Mechanisms of Sensory Transduction in the Skin. Nature 2007, 445, 858–865.
- Keppel Hesselink, J.M.; Kopsky, D.J.; Bhaskar, A.K. Skin Matters! The Role of Keratinocytes in Nociception: A Rational Argument for the Development of Topical Analgesics. J. Pain Res. 2016, 10, 1–8.
- Umbayev, B.; Askarova, S.; Almabayeva, A.; Saliev, T.; Masoud, A.-R.; Bulanin, D. Galactose-Induced Skin Aging: The Role of Oxidative Stress. Oxidative Med. Cell. Longev. 2020, 2020, 7145656.
- Schwartzenfeld, D.M.; Karamikian, J. Chapter 18—Hair Transplantation. In Plastic Surgery Secrets Plus, 2nd ed.; Weinzweig, J., Ed.; Mosby: Philadelphia, PA, USA, 2010; pp. 123–127. ISBN 978-0-323-03470-8.
- Cancello, R.; Micheletto, G.; Meta, D.; Lavagno, R.; Bevilacqua, E.; Panizzo, V.; Invitti, C. Expanding the Role of Bitter Taste Receptor in Extra Oral Tissues: TAS2R38 Is Expressed in Human Adipocytes. Adipocyte 2020, 9, 7–15.
- Avau, B.; Bauters, D.; Steensels, S.; Vancleef, L.; Laermans, J.; Lesuisse, J.; Buyse, J.; Lijnen, H.R.; Tack, J.; Depoortere, I. The Gustatory Signaling Pathway and Bitter Taste Receptors Affect the Development of Obesity and Adipocyte Metabolism in Mice. PLoS ONE 2015, 10, e0145538.
- Shain, A.H.; Bastian, B.C. From Melanocytes to Melanomas. Nat. Rev. Cancer 2016, 16, 345–358.
- Uhlen, M.; Fagerberg, L.; Hallstrom, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, A.; Kampf, C.; Sjostedt, E.; Asplund, A.; et al. Proteomics. Tissue-Based Map of the Human Proteome. Science 2015, 347, 1260419.
- Kallini, J.R.; Hamed, N.; Khachemoune, A. Squamous Cell Carcinoma of the Skin: Epidemiology, Classification, Management, and Novel Trends. Int. J. Dermatol. 2015, 54, 130–140.
- Almangush, A.; Mäkitie, A.A.; Triantafyllou, A.; de Bree, R.; Strojan, P.; Rinaldo, A.; Hernandez-Prera, J.C.; Suárez, C.; Kowalski, L.P.; Ferlito, A.; et al. Staging and Grading of Oral Squamous Cell Carcinoma: An Update. Oral Oncol. 2020, 107, 104799.
- Carey, R.M.; McMahon, D.B.; Miller, Z.A.; Kim, T.; Rajasekaran, K.; Gopallawa, I.; Newman, J.G.; Basu, D.; Nead, K.T.; White, E.A.; et al. T2R Bitter Taste Receptors Regulate Apoptosis and May Be Associated with Survival in Head and Neck Squamous Cell Carcinoma. Mol. Oncol. 2022, 16, 1474–1492.
- Luongo, F.P.; Passaponti, S.; Haxhiu, A.; Raeispour, M.; Belmonte, G.; Governini, L.; Casarini, L.; Piomboni, P.; Luddi, A. Bitter Taste Receptors and Endocrine Disruptors: Cellular and Molecular Insights from an In Vitro Model of Human Granulosa Cells. Int. J. Mol. Sci. 2022, 23, 15540.
- Slominski, A.; Zbytek, B.; Nikolakis, G.; Manna, P.R.; Skobowiat, C.; Zmijewski, M.; Li, W.; Janjetovic, Z.; Postlethwaite, A.; Zouboulis, C.C.; et al. Steroidogenesis in the Skin: Implications for Local Immune Functions. J. Steroid Biochem. Mol. Biol. 2013, 137, 107–123.
- Gradinaru, T.-C.; Petran, M.; Dragos, D.; Gilca, M. PlantMolecularTasteDB: A Database of Taste Active Phytochemicals. Front. Pharmacol. 2022, 12, 751712.
- Dagan-Wiener, A.; Nissim, I.; Ben Abu, N.; Borgonovo, G.; Bassoli, A.; Niv, M.Y. Bitter or Not? BitterPredict, a Tool for Predicting Taste from Chemical Structure. Sci. Rep. 2017, 7, 12074.
- Soares, S.; Silva, M.S.; García-Estevez, I.; Groβmann, P.; Brás, N.; Brandão, E.; Mateus, N.; de Freitas, V.; Behrens, M.; Meyerhof, W. Human Bitter Taste Receptors Are Activated by Different Classes of Polyphenols. J. Agric. Food Chem. 2018, 66, 8814–8823.
- Roland, W.S.U.; Van Buren, L.; Gruppen, H.; Driesse, M.; Gouka, R.J.; Smit, G.; Vincken, J.P. Bitter Taste Receptor Activation by Flavonoids and Isoflavonoids: Modeled Structural Requirements for Activation of HTAS2R14 and HTAS2R39. J. Agric. Food Chem. 2013, 61, 10454–10466.
- Sakurai, T.; Misaka, T.; Ueno, Y.; Ishiguro, M.; Matsuo, S.; Ishimaru, Y.; Asakura, T.; Abe, K. The Human Bitter Taste Receptor, HTAS2R16, Discriminates Slight Differences in the Configuration of Disaccharides. Biochem. Biophys. Res. Commun. 2010, 402, 595–601.
- Chialva, F.; Dada, G. Bitterness in Alcoholic Beverages. In Bitternes in Foods and Beverages. Developments in Food Science; Rouseff, R.L., Ed.; Elsevier: Amsterdam, The Netherlands, 1990; Volume 25, pp. 103–122.
- Steinhardt, R.G.; Calvin, A.D.; Dodd, E.A. Taste-Structure Correlation with α-D-Mannose and β-D-Mannose. Science 1962, 135, 367–368.
- Hellfritsch, C.; Brockhoff, A.; Stähler, F.; Meyerhof, W.; Hofmann, T. Human Psychometric and Taste Receptor Responses to Steviol Glycosides. J. Agric. Food Chem. 2012, 60, 6782–6793.
- Dragos, D.; Gilca, M. Taste of Phytocompounds: A Better Predictor for Ethnopharmacological Activities of Medicinal Plants Than The Phytochemical Class? J. Ethnopharmacol. 2018, 220, 129–146.
- Trinh, X.-T.; Long, N.-V.; Van Anh, L.T.; Nga, P.T.; Giang, N.N.; Chien, P.N.; Nam, S.-Y.; Heo, C.-Y. A Comprehensive Review of Natural Compounds for Wound Healing: Targeting Bioactivity Perspective. Int. J. Mol. Sci. 2022, 23, 9573.
- Ali, F.; Rahul; Naz, F.; Jyoti, S.; Siddique, Y.H. Health Functionality of Apigenin: A Review. Int. J. Food Prop. 2017, 20, 1197–1238.
- Shankar, E.; Goel, A.; Gupta, K.; Gupta, S. Plant Flavone Apigenin: An Emerging Anticancer Agent. Curr. Pharmacol. Rep. 2017, 3, 423–446.
- Hostetler, G.L.; Ralston, R.A.; Schwartz, S.J. Flavones: Food Sources, Bioavailability, Metabolism, and Bioactivity. Adv. Nutr. 2017, 8, 423–435.
- Behrens, M.; Meyerhof, W. The Vertebrate Gustatory System. In Flavour; Guichard, E., Salles, C., Morzel, M., Le Bon, A.M., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2016; pp. 57–78. ISBN 9781118929384.
- Kashyap, P.; Shikha, D.; Thakur, M.; Aneja, A. Functionality of Apigenin as a Potent Antioxidant with Emphasis on Bioavailability, Metabolism, Action Mechanism and in Vitro and in Vivo Studies: A Review. J. Food Biochem. 2022, 46, e13950.
- Ginwala, R.; Bhavsar, R.; Chigbu, D.I.; Jain, P.; Khan, Z.K. Potential Role of Flavonoids in Treating Chronic Inflammatory Diseases with a Special Focus on the Anti-Inflammatory Activity of Apigenin. Antioxidants 2019, 8, 35.
- Zhang, X.; Wang, G.; Gurley, E.C.; Zhou, H. Flavonoid Apigenin Inhibits Lipopolysaccharide-Induced Inflammatory Response through Multiple Mechanisms in Macrophages. PLoS ONE 2014, 9, e107072.
- Lee, J.-H.; Zhou, H.Y.; Cho, S.Y.; Kim, Y.S.; Lee, Y.S.; Jeong, C.S. Anti-Inflammatory Mechanisms of Apigenin: Inhibition of Cyclooxygenase-2 Expression, Adhesion of Monocytes to Human Umbilical Vein Endothelial Cells, and Expression of Cellular Adhesion Molecules. Arch. Pharm. Res. 2007, 30, 1318–1327.
- Imran, M.; Aslam Gondal, T.; Atif, M.; Shahbaz, M.; Batool Qaisarani, T.; Hanif Mughal, M.; Salehi, B.; Martorell, M.; Sharifi-Rad, J. Apigenin as an Anticancer Agent. Phyther. Res. 2020, 34, 1812–1828.
- Rahmani, A.H.; Alsahli, M.A.; Almatroudi, A.; Almogbel, M.A.; Khan, A.A.; Anwar, S.; Almatroodi, S.A. The Potential Role of Apigenin in Cancer Prevention and Treatment. Molecules 2022, 27, 6051.
- Nayaka, H.B.; Londonkar, R.L.; Umesh, M.K.; Tukappa, A. Antibacterial Attributes of Apigenin, Isolated from Portulaca oleracea L. Int. J. Bacteriol. 2014, 2014, 175851.
- Xu, X.; Miao, J.; Shao, Q.; Gao, Y.; Hong, L. Apigenin Suppresses Influenza A Virus-induced RIG-I Activation and Viral Replication. J. Med. Virol. 2020, 92, 3057–3066.
- Khandelwal, N.; Chander, Y.; Kumar, R.; Riyesh, T.; Dedar, R.K.; Kumar, M.; Gulati, B.R.; Sharma, S.; Tripathi, B.N.; Barua, S.; et al. Antiviral Activity of Apigenin against Buffalopox: Novel Mechanistic Insights and Drug-Resistance Considerations. Antivir. Res. 2020, 181, 104870.
- Thomas, S.D.; Jha, N.K.; Jha, S.K.; Sadek, B.; Ojha, S. Pharmacological and Molecular Insight on the Cardioprotective Role of Apigenin. Nutrients 2023, 15, 385.
- Qiao, Y.; Zhang, Z.; Zhai, Y.; Yan, X.; Zhou, W.; Liu, H.; Guan, L.; Peng, L. Apigenin Alleviates Obesity-Associated Metabolic Syndrome by Regulating the Composition of the Gut Microbiome. Front. Microbiol. 2021, 12, 805827.
- Meng, X.; Zheng, S.; Yin, Z.; Wang, X.; Yang, D.; Zou, T.; Li, H.; Chen, Y.; Liao, C.; Xie, Z.; et al. Apigenin Ameliorates Imiquimod-Induced Psoriasis in C57BL/6J Mice by Inactivating STAT3 and NF-ΚB. Food Sci. Hum. Wellness 2024, 13, 211–224.
- Fiorito, S.; Epifano, F.; Marchetti, L.; Palumbo, L.; Mascioli, F.; Bastianini, M.; Cardellini, F.; Spogli, R.; Genovese, S. An Improved Method for the Isolation of Amarogentin, the Bitter Principle of Yellow Gentian Roots. Food Chem. 2021, 364, 130383.
- Zhang, L.; Ulriksen, E.S.; Hoel, H.; Sandvik, L.; Malterud, K.E.; Inngjerdingen, K.T.; Inngjerdingen, M.; Wangensteen, H. Phytochemical Characterization and Anti-Inflammatory Activity of a Water Extract of Gentiana Purpurea Roots. J. Ethnopharmacol. 2023, 301, 115818.
- Kumar, V.; Sood, H.; Chauhan, R.S. Detection of Intermediates through High-Resolution Mass Spectrometry for Constructing Biosynthetic Pathways for Major Chemical Constituents in a Medicinally Important Herb, Swertia Chirayita. Nat. Prod. Res. 2015, 29, 1449–1455.
- Perez, J.J. Amygdalin Analogs for the Treatment of Psoriasis. Future Med. Chem. 2013, 5, 799–808.
- Bufe, B.; Hofmann, T.; Krautwurst, D.; Raguse, J.-D.; Meyerhof, W. The Human TAS2R16 Receptor Mediates Bitter Taste in Response to β-Glucopyranosides. Nat. Genet. 2002, 32, 397–401.
- Hui, K.; Huihua, Q.; Baoping, Q.; Wenhao, Z.; Yan, Z.; Xueqian, W.; Qingguo, W. Correlation between the Transdermal Characteristics of Pseudoephedrine and Amygdalin in Majiepingchuan in Vitro. J. Tradit. Chin. Med. 2016, 36, 238–242.
- Haidar, H.; Lauricella, M.; Jurjus, R.; Daouk, H.; Leone, A.; Capello, F.; Eid, A.; Jurjus, A. Amygdalin Improves Burn Wound Healing in Diabetic Rats. FASEB J. 2019, 33, 662.17.
- Gago-López, N.; Lagunas Arnal, C.; Perez, J.J.; Wagner, E.F.; Gago-López, N.; Lagunas Arnal, C.; Perez, J.J.; Wagner, E.F. Topical Application of an Amygdalin Analogue Reduces Inflammation and Keratinocyte Proliferation in a Psoriasis Mouse Model. Exp. Dermatol. 2021, 30, 1662–1674.
- Yu, Y.; Hao, G.; Zhang, Q.; Hua, W.; Wang, M.; Zhou, W.; Zong, S.; Huang, M.; Wen, X. Berberine Induces GLP-1 Secretion through Activation of Bitter Taste Receptor Pathways. Biochem. Pharmacol. 2015, 97, 173–177.
- Behrens, M.; Gu, M.; Fan, S.; Huang, C.; Meyerhof, W. Bitter Substances from Plants Used in Traditional Chinese Medicine Exert Biased Activation of Human Bitter Taste Receptors. Chem. Biol. Drug Des. 2018, 91, 422–433.
- Andoh, T.; Yoshihisa, Y.; Rehman, M.U.; Tabuchi, Y.; Shimizu, T. Berberine Induces Anti-Atopic Dermatitis Effects through the Downregulation of Cutaneous EIF3F and MALT1 in NC/Nga Mice with Atopy-like Dermatitis. Biochem. Pharmacol. 2021, 185, 114439.
- Li, D.X.; Zhang, J.; Zhang, Y.; Zhao, P.W.; Yang, L.M. Inhibitory Effect of Berberine on Human Skin Squamous Cell Carcinoma A431 Cells. Genet. Mol. Res. 2015, 14, 10553–10568.
- Kou, Y.; Li, L.; Li, H.; Tan, Y.; Li, B.; Wang, K.; Du, B. Berberine Suppressed Epithelial Mesenchymal Transition through Cross-Talk Regulation of PI3K/AKT and RARα/RARβ in Melanoma Cells. Biochem. Biophys. Res. Commun. 2016, 479, 290–296.
- Liu, J.-F.; Lai, K.; Peng, S.-F.; Maraming, P.; Huang, Y.-P.; Huang, A.-C.; Chueh, F.-S.; Huang, W.-W.; Chung, J.-G. Berberine Inhibits Human Melanoma A375.S2 Cell Migration and Invasion via Affecting the FAK, UPA, and NF-ΚB Signaling Pathways and Inhibits PLX4032 Resistant A375.S2 Cell Migration In Vitro. Molecules 2018, 23, 2019.
- Ammam, A.; Zemour, H.; Kaid, M.; Villemin, D.; Soufan, W.; Belhouadjeb, F.A. Assessment of the Anti-Inflammatory and Analgesic Effects of Opuntia Ficus Indica L. Cladodes Extract. Libyan J. Med. 2023, 18, 2275417.
- Goławska, S.; Łukasik, I.; Chojnacki, A.A.; Chrzanowski, G. Flavonoids and Phenolic Acids Content in Cultivation and Wild Collection of European Cranberry Bush Viburnum opulus L. Molecules 2023, 28, 2285.
- Chan, C.W.; Deadman, B.J.; Manley-Harris, M.; Wilkins, A.L.; Alber, D.G.; Harry, E. Analysis of the Flavonoid Component of Bioactive New Zealand Mānuka (Leptospermum Scoparium) Honey and the Isolation, Characterisation and Synthesis of an Unusual Pyrrole. Food Chem. 2013, 141, 1772–1781.
- Woźniak, M.; Mrówczyńska, L.; Kwaśniewska-Sip, P.; Waśkiewicz, A.; Nowak, P.; Ratajczak, I. Effect of the Solvent on Propolis Phenolic Profile and Its Antifungal, Antioxidant, and In Vitro Cytoprotective Activity in Human Erythrocytes Under Oxidative Stress. Molecules 2020, 25, 4266.
- Li, H.-J.; Wu, N.-L.; Pu, C.-M.; Hsiao, C.-Y.; Chang, D.-C.; Hung, C.-F. Chrysin Alleviates Imiquimod-Induced Psoriasis-like Skin Inflammation and Reduces the Release of CCL20 and Antimicrobial Peptides. Sci. Rep. 2020, 10, 2932.
- Li, Z.J.; Shin, J.-M.; Choi, D.-K.; Lim, S.K.; Yoon, T.-J.; Lee, Y.H.; Sohn, K.-C.; Im, M.; Lee, Y.; Seo, Y.-J.; et al. Inhibitory Effect of Cucurbitacin B on Imiquimod-Induced Skin Inflammation. Biochem. Biophys. Res. Commun. 2015, 459, 673–678.
- Yamazaki, T.; Narukawa, M.; Mochizuki, M.; Misaka, T.; Watanabe, T. Activation of the HTAS2R14 Human Bitter-Taste Receptor by (−)-Epigallocatechin Gallate and (−)-Epicatechin Gallate. Biosci. Biotechnol. Biochem. 2013, 77, 1981–1983.
- Khan, N.; Afaq, F.; Saleem, M.; Ahmad, N.; Mukhtar, H. Targeting Multiple Signaling Pathways by Green Tea Polyphenol (−)-Epigallocatechin-3-Gallate. Cancer Res. 2006, 66, 2500–2505.
- Zhang, S.; Liu, X.; Mei, L.; Wang, H.; Fang, F. Epigallocatechin-3-Gallate (EGCG) Inhibits Imiquimod-Induced Psoriasis-like Inflammation of BALB/c Mice. BMC Complement. Altern. Med. 2016, 16, 334.
- Fukutake, M.; Takahashi, M.; Ishida, K.; Kawamura, H.; Sugimura, T.; Wakabayashi, K. Quantification of Genistein and Genistin in Soybeans and Soybean Products. Food Chem. Toxicol. 1996, 34, 457–461.
- Record, I.R.; Broadbent, J.L.; King, R.A.; Dreosti, I.E.; Head, R.J.; Tonkin, A.L. Genistein Inhibits Growth of B16 Melanoma Cellsin Vivo Andin Vitro and Promotes Differentiationin Vitro. Int. J. Cancer 1997, 72, 860–864.
- Yan, C.; Han, R. Genistein Suppresses Adhesion-Induced Protein Tyrosine Phosphorylation and Invasion of B16-BL6 Melanoma Cells. Cancer Lett. 1998, 129, 117–124.
- Yan, C.-H.; Chen, X.-G.; Li, V.; Han, R. Effects of Genistein, A Soybean-Derived Isoflavone, on Proliferation and Differentiation of B16-BL6 Mouse Melanoma Cells. J. Asian Nat. Prod. Res. 1999, 1, 285–299.
- Wang, H.-Z.; Zhang, Y.; Xie, L.-P.; Yu, X.-Y.; Zhang, R.-Q. Effects of Genistein and Daidzein on the Cell Growth, Cell Cycle, and Differentiation of Human and Murine Melanoma Cells1 1The Abbreviations Used Are: BSA, Bovine Serum Albumin; FBS, Fetal Bovine Serum; ECM, Extracellular Matrix; DMSO, Dimethyl Sulfoxide. J. Nutr. Biochem. 2002, 13, 421–426.
- Farina, H.G.; Pomies, M.; Alonso, D.F.; Gomez, D.E. Antitumor and Antiangiogenic Activity of Soy Isoflavone Genistein in Mouse Models of Melanoma and Breast Cancer. Oncol. Rep. 2006, 16, 885–891.
- Wang, H.; Zhu, Y.; Li, C.; Xie, L.; Chen, G.; Nie, Y.; Zhang, R. Effects of Genistein on Cell Cycle and Apoptosis of Two Murine Melanoma Cell Lines. Tsinghua Sci. Technol. 2007, 12, 372–380.
- Cui, S.; Wang, J.; Wu, Q.; Qian, J.; Yang, C.; Bo, P. Genistein Inhibits the Growth and Regulates the Migration and Invasion Abilities of Melanoma Cells via the FAK/Paxillin and MAPK Pathways. Oncotarget 2017, 8, 21674–21691.
- Dabeek, W.M.; Marra, M.V. Dietary Quercetin and Kaempferol: Bioavailability and Potential Cardiovascular-Related Bioactivity in Humans. Nutrients 2019, 11, 2288.
- Liu, C.; Liu, H.; Lu, C.; Deng, J.; Yan, Y.; Chen, H.; Wang, Y.; Liang, C.-L.; Wei, J.; Han, L.; et al. Kaempferol Attenuates Imiquimod-Induced Psoriatic Skin Inflammation in a Mouse Model. Clin. Exp. Immunol. 2019, 198, 403–415.
- Wang, J.; Li, G.; Zhang, H.; Xie, Y. Protective Effect of Luteolin on Atopic Dermatitis Murine Model via IgE Mediated Immune Response. Isr. J. Plant Sci. 2021, 68, 99–106.
- Zhou, W.; Hu, M.; Zang, X.; Liu, Q.; Du, J.; Hu, J.; Zhang, L.; Du, Z.; Xiang, Z. Luteolin Attenuates Imiquimod–Induced Psoriasis-like Skin Lesions in BALB/c Mice via Suppression of Inflammation Response. Biomed. Pharmacother. 2020, 131, 110696.
- Dagan-Wiener, A.; Di Pizio, A.; Nissim, I.; Bahia, M.S.; Dubovski, N.; Margulis, E.; Niv, M.Y. BitterDB: Taste Ligands and Receptors Database in 2019. Nucleic Acids Res. 2019, 47, D1179–D1185.
- Fierro, F.; Peri, L.; Hübner, H.; Tabor-Schkade, A.; Waterloo, L.; Löber, S.; Pfeiffer, T.; Weikert, D.; Dingjan, T.; Margulis, E.; et al. Inhibiting a Promiscuous GPCR: Iterative Discovery of Bitter Taste Receptor Ligands. Cell. Mol. Life Sci. 2023, 80, 114.
- Kim, T.-H.; Kim, G.-D.; Ahn, H.-J.; Cho, J.-J.; Park, Y.S.; Park, C.-S. The Inhibitory Effect of Naringenin on Atopic Dermatitis Induced by DNFB in NC/Nga Mice. Life Sci. 2013, 93, 516–524.
- Choi, J.; Lee, D.-H.; Jang, H.; Park, S.-Y.; Seol, J.-W. Naringenin Exerts Anticancer Effects by Inducing Tumor Cell Death and Inhibiting Angiogenesis in Malignant Melanoma. Int. J. Med. Sci. 2020, 17, 3049–3057.
- Kumar, R.; Bhan Tiku, A. Naringenin Suppresses Chemically Induced Skin Cancer in Two-Stage Skin Carcinogenesis Mouse Model. Nutr. Cancer 2020, 72, 976–983.
- Burns, J.; Yokota, T.; Ashihara, H.; Lean, M.E.J.; Crozier, A. Plant Foods and Herbal Sources of Resveratrol. J. Agric. Food Chem. 2002, 50, 3337–3340.
- Kjær, T.N.; Thorsen, K.; Jessen, N.; Stenderup, K.; Pedersen, S.B. Resveratrol Ameliorates Imiquimod-Induced Psoriasis-Like Skin Inflammation in Mice. PLoS ONE 2015, 10, e0126599.
- Shen, Y.; Xu, J. Resveratrol Exerts Therapeutic Effects on Mice With Atopic Dermatitis. Wounds A Compend. Clin. Res. Pract. 2019, 31, 279–284.
- Karuppagounder, V.; Arumugam, S.; Thandavarayan, R.A.; Pitchaimani, V.; Sreedhar, R.; Afrin, R.; Harima, M.; Suzuki, H.; Nomoto, M.; Miyashita, S.; et al. Resveratrol Attenuates HMGB1 Signaling and Inflammation in House Dust Mite-Induced Atopic Dermatitis in Mice. Int. Immunopharmacol. 2014, 23, 617–623.
- Assaf, H.; Adly, M.A.; Hussein, M.R. Aging and Intrinsic Aging: Pathogenesis and Manifestations. In Textbook of Aging Skin; Farage, M.A., Miller, K.W., Maibach, H.I., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 129–138. ISBN 978-3-540-89656-2.
- Stallone, G.; Infante, B.; Prisciandaro, C.; Grandaliano, G. MTOR and Aging: An Old Fashioned Dress. Int. J. Mol. Sci. 2019, 20, 2774.
- Karagianni, F.; Pavlidis, A.; Malakou, L.S.; Piperi, C.; Papadavid, E. Predominant Role of MTOR Signaling in Skin Diseases with Therapeutic Potential. Int. J. Mol. Sci. 2022, 23, 1693.
- Chamcheu, J.C.; Esnault, S.; Adhami, V.M.; Noll, A.L.; Banang-Mbeumi, S.; Roy, T.; Singh, S.S.; Huang, S.; Kousoulas, K.G.; Mukhtar, H. Fisetin, a 3,7,3′,4′-Tetrahydroxyflavone Inhibits the PI3K/Akt/MTOR and MAPK Pathways and Ameliorates Psoriasis Pathology in 2D and 3D Organotypic Human Inflammatory Skin Models. Cells 2019, 8, 1089.
- Gong, C.; Xia, H. Resveratrol Suppresses Melanoma Growth by Promoting Autophagy through Inhibiting the PI3K/AKT/MTOR Signaling Pathway. Exp. Ther. Med. 2019, 19, 1878–1886.
- Ekoff, M.; Choi, J.-H.; James, A.; Dahlén, B.; Nilsson, G.; Dahlén, S.-E. Bitter Taste Receptor (TAS2R) Agonists Inhibit IgE-Dependent Mast Cell Activation. J. Allergy Clin. Immunol. 2014, 134, 475–478.





