Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yong Chool Boo | -- | 1955 | 2024-01-31 09:02:11 | | | |
| 2 | Wendy Huang | Meta information modification | 1955 | 2024-01-31 11:15:09 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Boo, Y.C. Plant-Derived Compounds in Preventing and Treating Keloid Scars. Encyclopedia. Available online: https://encyclopedia.pub/entry/54565 (accessed on 27 March 2026).
Boo YC. Plant-Derived Compounds in Preventing and Treating Keloid Scars. Encyclopedia. Available at: https://encyclopedia.pub/entry/54565. Accessed March 27, 2026.
Boo, Yong Chool. "Plant-Derived Compounds in Preventing and Treating Keloid Scars" Encyclopedia, https://encyclopedia.pub/entry/54565 (accessed March 27, 2026).
Boo, Y.C. (2024, January 31). Plant-Derived Compounds in Preventing and Treating Keloid Scars. In Encyclopedia. https://encyclopedia.pub/entry/54565
Boo, Yong Chool. "Plant-Derived Compounds in Preventing and Treating Keloid Scars." Encyclopedia. Web. 31 January, 2024.
Copy Citation
Keloid is a disease in which fibroblasts abnormally proliferate and synthesize excessive amounts of extracellular matrix, including collagen and fibronectin, during the healing process of skin wounds, causing larger scars that exceed the boundaries of the original wound. Various phenolic compounds, terpenoids, alkaloids, and other plant-derived compounds could modulate different cell signaling pathways associated with the pathogenesis of keloids. For now, many studies are limited to in vitro experiments; additional research and development are needed to proceed to clinical trials.
keloid
plant-derived compounds
phenolic compounds
terpenoids
alkaloids
1. Introduction
Keloid is a type of scarring that occurs due to abnormally high cell proliferation and the excessive accumulation of extracellular matrix (ECM) during the healing process of a skin injury [1][2]. Keloids are similar to hypertrophic scars in that they involve the proliferation of dermal fibroblasts and accumulation of ECM, but keloids differ in that they grow beyond the boundaries of the initial injury [3]. In hypertrophic scars, collagen or other ECM components show a wavy or spiral pattern arranged in a specific direction, but, in keloids, ECM does not show a consistent or regular pattern [4]. Additionally, hypertrophic scars gradually become smaller and lessen over time, but keloids are different in that they grow larger or persist [5][6]. Therefore, keloids are sometimes classified as benign fibroproliferative skin tumors [7].
Keloids and hypertrophic scars are both aesthetically disfiguring and functionally defective and can cause pruritus (itchiness), pressure, or pain depending on their shape, size, and location [5][8]. In treating keloids, surgical excision, cryotherapy, radiation, laser treatment, photodynamic therapy, pressure therapy, silicone gel sheeting, and pharmacotherapy are currently used alone or in combinations [9][10][11][12][13]. In pharmacotherapy, steroids, retinoids, interferons, imiquimod, etc. are administered by intralesional injection or topical application [9][10][11][12][13]. However, the outcomes are usually unsatisfactory, and further technical development is needed.
Studies have been conducted extensively to prevent and treat keloids by using various natural products in parallel with the application of various surgical, physical, and pharmacological therapies [9][12]. The following sections present experimental results from studies on the biological activities of plant-derived compounds in KFs at the cellular and molecular level. The compounds are described by classifying them into phenolic compounds, terpenoids, alkaloids, and others. The chemical structure of each compound is shown in Figure 1. The proposed therapeutic targets of plant-derived compounds are summarized in Table 1.
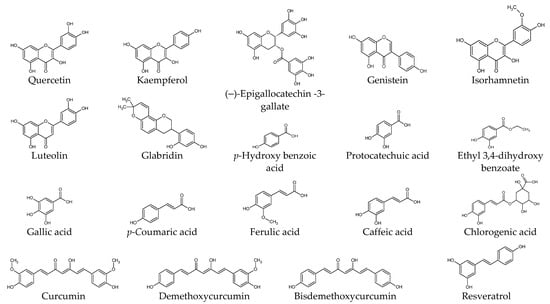
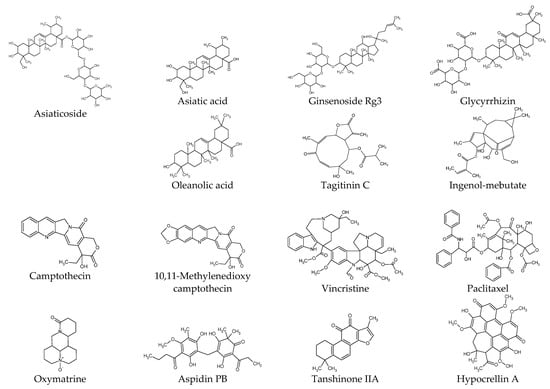
Figure 1. Chemical structures of natural products.
Table 1. Proposed therapeutic targets of plant-derived extracts and compounds. The targets modulated by each compound are indicated by check mark (√).
| Compounds | Proliferation/ Viability |
Migration/ Invasion |
Apoptosis | ECM Production |
TGF-β Level |
TGFβR Level |
SMAD Pathway |
AKT Pathway |
ERK Pathway |
Additional Targets |
Literature |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Quercetin | √ | √ | √ | √ | √ | √ | √ | IGF1R | [14][15][16][17][18] | ||
| Kaempferol | √ | [18] | |||||||||
| (–)-Epigallocatechin -3-gallate |
√ | √ | √ | √ | STAT3 | [19][20][21] | |||||
| Genistein | CTGF | [22] | |||||||||
| Luteolin | √ | √ | FRAT1 | [23] | |||||||
| Glabridin | √ | √ | √ | √ | √ | [24] | |||||
| Isorhamnetin | √ | √ | √ | √ | S1PR1 | [25] | |||||
| Protocatechuic acid | √ | √ | [18] | ||||||||
| Gallic acid | √ | √ | [18] | ||||||||
| p-Coumaric acid | √ | [18] | |||||||||
| Ferulic acid | √ | [18] | |||||||||
| Chlorogenic acid | √ | √ | [18] | ||||||||
| Curcumin | √ | √ | √ | √ | [18][26] | ||||||
| Demethoxycurcumin | √ | √ | √ | [26] | |||||||
| Bisdemethoxycurcumin | √ | √ | √ | [26] | |||||||
| Resveratrol | √ | √ | √ | √ | √ | HSP47, α-SMA | [27] | ||||
| Asiaticoside | √ | √ | √ | √ | √ | √ | p38, GDF-9 | [28] | |||
| Asiatic acid | √ | √ | PAI-1, PPARγ | [29][30] | |||||||
| Ginsenoside Rg3 | √ | √ | √ | √ | √ | Angiogenesis | [31] | ||||
| Tagitinin C | √ | √ | [32] | ||||||||
| Ingenol-mebutate | √ | √ | miR-34a | [33] | |||||||
| Glycyrrhizin | √ | √ | √ | √ | √ | √ | √ | NF-κB, HMGB1, Autophagy | [34] | ||
| Oleanolic acid | √ | √ | √ | MMP1 | [35] | ||||||
| Camptothecin | √ | [36] | |||||||||
| 10,11-Methylenedioxy camptothecin |
√ | √ | √ | PAI-1 | [37] | ||||||
| Oxymatrine | √ | √ | [38] | ||||||||
| Vincristine | √ | [39] | |||||||||
| Paclitaxel | √ | √ | √ | √ | √ | [40] | |||||
| Aspidin PB | √ | √ | √ | CTGF | [41] | ||||||
| Tanshinone IIA | √ | √ | Survivin | [42] | |||||||
| Selenium- polysaccharide |
√ | √ | PARP | [43] | |||||||
| Photodynamic therapy with Hypocrellin A | √ | √ | √ | √ | √ | √ | √ | Autophagy, α-SMA |
[44] |
2. Phenolic Compounds
Quercetin attenuated KF proliferation (IC50, 25 μg mL−1) and lowered the expression levels of TGF-β1, TGFβR1/2, collagen 1/3, and fibronectin [14][15][16][17]. It lowered the expression levels of SMAD2/3/4 and reduced the phosphorylation of SMAD2/3, and the formation of the SMAD2/3/4 complex [17]. It lowered the expression levels of the insulin-like growth factor 1 receptor (IGF-1R) β subunit, insulin receptor substrate (IRS) 1, PI3K p85 subunit, c-Raf, and reduced the phosphorylation of c-Raf, MEK1/2, ERK1/2, ETS like protein (ELK) 1, and AKT1 in KFs [15].
(–)-Epigallocatechin-3-gallate (EGCG) attenuated the proliferation, migration, and collagen production of KFs and NFs, and reduced the phosphorylation of STAT3, but not that of SMAD2/3, in KFs [19]. Green tea extract and EGCG lowered the expression level of collagen 1 and reduced the phosphorylation of AKT, eukaryotic translation initiation factor 4E-binding protein (4E-BP), and p70S6K in KFs stimulated by human leukemic mast cell line HMC-1 [21].
CTGF protein levels were higher in KFs compared to NFs, and genistein reduced the CTGF protein levels in KFs [22]. Genistein, at different concentrations (37 or 370 μM), had variable effects on the mRNA expression levels of subunit proteins of AP-1, such as c-Jun, c-Fos, and FosB, in skin keratinocytes, NFs, and KFs [45].
Luteolin decreased the KF viability and the expression levels of cyclin D1, BCL-2, and FRAT1, and increased cell apoptosis, p21, and BAX expression [23]. The pro-apoptotic effects of luteolin were abolished by overexpressed FRAT1, a GSKβ3 inhibitor causing β-catenin activation in the Wnt signaling pathway, and siRNA-mediated FRAT1 depletion increased cell apoptosis [23].
Glabridin, a component of Glycyrrhiza glabra, reduced KF proliferation and collagen production and induced apoptosis by inhibiting the PI3K/AKT and TGF-β1/SMAD signaling pathways in vitro [24].
Isorhamnetin inhibited the proliferation, migration, invasion, and fibrogenesis of KFs [25]. It lowered the expression level of S1PR1 and reduced the phosphorylation of PI3K and AKT [25]. S1PR1 upregulation abolished the inhibitory effects of isorhamnetin on KF proliferation, migration, invasion, and fibrogenesis.
KF proliferation was inhibited by curcumin (2.5 and 5 μg mL−1), gallic acid (5 and 10 μg mL−1), quercetin (10 and 20 μg mL−1), kaempferol (20 μg mL−1), protocatechuic acid (100 and 200 μg mL−1), p-coumaric acid (400 μg mL−1), ferulic acid (400 μg mL−1), and chlorogenic acid (400 μg mL−1) [18]. p-Hydroxy benzoic acid had no effect, and caffeic acid was very toxic [18]. These effects were attributed to cell cycle arrest rather than apoptosis [18]. The cell proliferation was resumed after the removal of each phytochemical and relatively slow recovery was seen with quercetin, chlorogenic acid, or curcumin [18]. Quercetin, gallic acid, protocatechuic acid, and chlorogenic acid more effectively inhibited the collagen lattice contraction by NFs and hypertrophic scar-derived fibroblasts (HSFs) than other compounds [18]. The collagen lattice contraction resumed when each compound was removed, and the recovery was slowest with quercetin [18].
Curcuminoids (25–100 nM), consisting of curcumin, demethoxycurcumin, and bisdemethoxycurcumin, lowered the cellular levels of total soluble collagens, pro-collagen 1, fibronectin, and TGF-β1, and reduced the phosphorylation of SMAD2 in KFs stimulated with bleomycin [26]. Curcumin was the major form of curcuminoids that entered and accumulated inside cells [26].
Resveratrol attenuated cell proliferation, induced apoptosis, and lowered the expression levels of TGF-β1, collagen 1, α-SMA, and heat shock protein (HSP) 47, which is involved in collagen folding and remodeling [46][47], in KFs but not in NFs [27]. It also attenuated cell proliferation, induced apoptosis, and reduced the collagen synthesis of KFs under hypoxia by downregulating hypoxia-inducible factor (HIF)-1α.
3. Terpenoids
Asiaticoside, a component of Centella asiatica, attenuated KF proliferation and lowered the expression levels of collagen 1/3 and TGFβR1/2 [28]. Asiaticoside did not affect the expression levels or the phosphorylation of SMAD2/3/4 but increased the expression level of SMAD7, which acts as an intracellular antagonist of the TGF-β signaling pathway [28]. Asiaticoside attenuated KF proliferation, invasion, and the phosphorylation of ERK1/2, p38 MAPK, and SMAD2/3 (linker region) stimulated by GDF-9 [29].
Asiatic acid from Centella asiatica suppressed the TGF-β1-induced expression of collagen 1 and plasminogen activator inhibitor-1 (PAI-1) and the phosphorylation of SMAD2/3, while increasing SMAD7 expression [30]. These effects of asiatic acid on KFs were abrogated by PPAR-γ antagonist GW9662 or peroxisome proliferator-activated receptor gamma (PPAR γ) siRNA [30].
Ginsenoside Rg3 (50 or 100 µg mL−1) attenuated the proliferation, migration, invasion, angiogenesis, and collagen synthesis of KFs and inhibited the TGF-β/SMAD and ERK-mediated signaling pathways [31].
Tagitinin C reduced KF viability after 72 h (IC50, 0.122 μg mL−1), as potently as mitomycin C (IC50, 0.120 μg mL−1) [32]. Tagitinin C at IC50 decreased keloid collagen deposition to 53.1% of the control level, whereas mitomycin C IC50 decreased it to 60.4% [32]. Tagitinin C and mitomycin C were less toxic to NFs (IC50; 35.05 μg mL−1 and 16.21 μg mL−1, respectively) [32]. The selective cytotoxicity index of tagitinin C and mitomycin C on KFs versus NFs was calculated to be 287 and 135, respectively [32].
Treatment of KFs with ingenol-mebutate induced morphological alterations and DNA fragmentation, which were associated with reduced cell growth and increased apoptosis [33]. It induced the expression of miR-34a in a p53-dependent manner and upregulated proapoptotic genes, such as caspase-10, while downregulating antiapoptotic genes, such as BCL-2 [33].
Glycyrrhizin, a component of Glycyrrhiza glabra, lowered the expression level of HMGB1 in KFs and attenuated cell proliferation and autophagy while increasing apoptosis [34]. Glycyrrhizin inhibited the expressions of ERK1/2, AKT, and NF-κB induced by HMGB1 [34]. Glycyrrhizin lowered the expression levels of TGF-β1, SMAD2/3, ERK1/2, collagen 1/3, fibronectin, and elastin in KFs [34].
Oleanolic acid attenuated the proliferation of KFs [35]. It lowered the expression levels of intra- and extracellular fibronectin, procollagen 1, and α-SMA while increasing MMP1 [35]. It inhibited the phosphorylation of SMAD2 and SMAD3 and attenuated the increases in fibronectin, procollagen 1, and α-SMA and the decrease in MMP1 in KFs stimulated with TGF-β1.
4. Alkaloids
Camptothecin, originally isolated from Camptotheca acuminata, is a topoisomerase inhibitor that has been used in cancer therapy [48]. Camptothecin lowered the expression levels of collagen 1/3 in KFs without causing cellular toxicity [36]. Its effects on the collagen 3 level were relatively smaller, and consequently, the ratios of collagen 1 to collagen 3 were decreased by the camptothecin treatment [48].
10,11-Methylenedioxycamptothecin loaded in hyaluronic acid nanoemulsions were delivered percutaneously to the keloid lesion area in a mouse model [37]. Its internalization by KFs and delivery to the nucleus resulted in decreased cell proliferation [37]. It increased the expression levels of TGF-β1, SMAD3, and SMAD7, and downregulated PAI-1 in KFs, implicating an overall suppression of the TGF-β-mediated signaling pathway [37].
Oxymatrine, an alkaloid compound extracted from Sophora japonica, lowered the expression levels of collagen and SMAD3 in KFs in vitro without affecting the expression levels of TGF-β1, TGFβR1/2, SMAD4, and SMAD7 [38]. Oxymatrine inhibited the phosphorylation and nuclear translocation of SMAD3 induced by TGF-β1 [38]. Thus oxymatrine could attenuate collagen synthesis by inhibiting the TGF-β/SMAD signaling pathway.
Vincristine is one of the vinca alkaloids originally separated from Catharanthus roseus, and is used as an anticancer drug [49]. Vincristine inhibited cell proliferation by inducing cell cycle arrest in the G2/M phase and promoting apoptosis in SH-SY5Y human neuroblastoma cells [50]. Vincristine showed cytotoxicity to the primary KFs and NFs, with higher potency to the latter [39]. The resistance of KFs could be largely abrogated by verapamil (a calcium channel blocker) [39].
The treatment of KFs with paclitaxel or LY294002 (a PI3K inhibitor) lowered their expression levels of TNF-α, IL-6, TGF-β1, α-SMA, and collagen 1 [40]. Paclitaxel also blocked the AKT/GSK3β signaling pathway in KFs and keloid tissues [40]. Paclitaxel-cholesterol-loaded liposomes inhibited KF proliferation, migration, and invasion, and promoted apoptosis and cell cycle arrest in the G2/M phases more effectively than paclitaxel itself in vitro [40]. Paclitaxel-cholesterol-loaded liposomes had better performance in inhibiting keloid growth compared to paclitaxel in the keloid-bearing BALB/c nude mouse model [40].
5. Other Compounds
Aspidin PB inhibited the expression of collagen 1, CTGF, and α-SMA in KFs stimulated by TGF-β1 [41]. It inhibited both the SMAD2/3-mediated signaling pathway and the PI3K/AKT-mediated signaling pathway stimulated by TGF-β1 [41].
Tanshinone IIA attenuated the proliferation of KFs, whereas it did not affect the proliferation of NFs [42]. It increased the percentages of KF cells in the G0/G1 phases and the cells undergoing early apoptosis [42]. It also decreased the expression of survivin [42].
A selenium-containing polysaccharide from Ziyang green tea (Se-ZGTP) or short hairpin RNA (shRNA) for neuron-glia 2 inhibited the proliferation of KFs [43]. Se-ZGTP or NG2 shRNA induced apoptosis mediated by an increase in pro-apoptotic BAX expression, the activation of caspase-3, the subsequent cleavage and inactivation of poly (ADP-ribose) polymerase (PARP), and a decrease in the expression levels of anti-apoptotic BCL-2 [43]. Se-ZGTP or neuron-glia 2 shRNA reduced collagen 1 and protein expression in KFs following TGF-β1 stimulation [43].
As a photodynamic therapy, the combined treatment of hypocrellin A with a light-emitting diode (LED)’s red light irradiation increased ROS production [51] and decreased KF viability, proliferation, invasion, collagen production, and the expression of collagen 1/3, α-SMA, and fibronectin, while increasing cell apoptosis and the expression of BAX and caspase-3 [44]. The combined photodynamic therapy reduced autophagy, the protein expression of Beclin-1, and the conversion of LC3-I to LC3-II [44]. It inhibited the expression of TGF-β and the downstream signaling pathways mediated by ERK1/2 and SMD2/3 [44].
References
- Lee, C.C.; Tsai, C.H.; Chen, C.H.; Yeh, Y.C.; Chung, W.H.; Chen, C.B. An updated review of the immunological mechanisms of keloid scars. Front. Immunol. 2023, 14, 1117630.
- Betarbet, U.; Blalock, T.W. Keloids: A Review of Etiology, Prevention, and Treatment. J. Clin. Aesthet. Dermatol. 2020, 13, 33–43.
- Limandjaja, G.C.; Niessen, F.B.; Scheper, R.J.; Gibbs, S. Hypertrophic scars and keloids: Overview of the evidence and practical guide for differentiating between these abnormal scars. Exp. Dermatol. 2021, 30, 146–161.
- Mokos, Z.B.; Jovic, A.; Grgurevic, L.; Dumic-Cule, I.; Kostovic, K.; Ceovic, R.; Marinovic, B. Current Therapeutic Approach to Hypertrophic Scars. Front. Med. 2017, 4, 83.
- Jeschke, M.G.; Wood, F.M.; Middelkoop, E.; Bayat, A.; Teot, L.; Ogawa, R.; Gauglitz, G.G. Scars. Nat. Rev. Dis. Primers 2023, 9, 64.
- Gauglitz, G.G.; Korting, H.C.; Pavicic, T.; Ruzicka, T.; Jeschke, M.G. Hypertrophic scarring and keloids: Pathomechanisms and current and emerging treatment strategies. Mol. Med. 2011, 17, 113–125.
- Tan, S.; Khumalo, N.; Bayat, A. Understanding Keloid Pathobiology From a Quasi-Neoplastic Perspective: Less of a Scar and More of a Chronic Inflammatory Disease With Cancer-Like Tendencies. Front. Immunol. 2019, 10, 1810.
- Biazus Soares, G.; Mahmoud, O.; Yosipovitch, G. Pruritus in keloid scars: Mechanisms and treatments. Ital. J. Dermatol. Venerol. 2023, 158, 401–407.
- Al-Attar, A.; Mess, S.; Thomassen, J.M.; Kauffman, C.L.; Davison, S.P. Keloid pathogenesis and treatment. Plast. Reconstr. Surg. 2006, 117, 286–300.
- Barone, N.; Safran, T.; Vorstenbosch, J.; Davison, P.G.; Cugno, S.; Murphy, A.M. Current Advances in Hypertrophic Scar and Keloid Management. Semin. Plast. Surg. 2021, 35, 145–152.
- Ojeh, N.; Bharatha, A.; Gaur, U.; Forde, A.L. Keloids: Current and emerging therapies. Scars Burn. Health 2020, 6, 2059513120940499.
- Walsh, L.A.; Wu, E.; Pontes, D.; Kwan, K.R.; Poondru, S.; Miller, C.H.; Kundu, R.V. Keloid treatments: An evidence-based systematic review of recent advances. Syst. Rev. 2023, 12, 42.
- Ekstein, S.F.; Wyles, S.P.; Moran, S.L.; Meves, A. Keloids: A review of therapeutic management. Int. J. Dermatol. 2021, 60, 661–671.
- Long, X.; Zeng, X.; Zhang, F.Q.; Wang, X.J. Influence of quercetin and x-ray on collagen synthesis of cultured human keloid-derived fibroblasts. Chin. Med. Sci. J. 2006, 21, 179–183.
- Phan, T.T.; See, P.; Tran, E.; Nguyen, T.T.; Chan, S.Y.; Lee, S.T.; Huynh, H. Suppression of insulin-like growth factor signalling pathway and collagen expression in keloid-derived fibroblasts by quercetin: Its therapeutic potential use in the treatment and/or prevention of keloids. Br. J. Dermatol. 2003, 148, 544–552.
- Phan, T.T.; Lim, I.J.; Sun, L.; Chan, S.Y.; Bay, B.H.; Tan, E.K.; Lee, S.T. Quercetin inhibits fibronectin production by keloid-derived fibroblasts. Implication for the treatment of excessive scars. J. Dermatol. Sci. 2003, 33, 192–194.
- Phan, T.T.; Lim, I.J.; Chan, S.Y.; Tan, E.K.; Lee, S.T.; Longaker, M.T. Suppression of transforming growth factor beta/smad signaling in keloid-derived fibroblasts by quercetin: Implications for the treatment of excessive scars. J. Trauma. 2004, 57, 1032–1037.
- Phan, T.T.; Sun, L.; Bay, B.H.; Chan, S.Y.; Lee, S.T. Dietary compounds inhibit proliferation and contraction of keloid and hypertrophic scar-derived fibroblasts in vitro: Therapeutic implication for excessive scarring. J. Trauma. 2003, 54, 1212–1224.
- Park, G.; Yoon, B.S.; Moon, J.H.; Kim, B.; Jun, E.K.; Oh, S.; Kim, H.; Song, H.J.; Noh, J.Y.; Oh, C.; et al. Green tea polyphenol epigallocatechin-3-gallate suppresses collagen production and proliferation in keloid fibroblasts via inhibition of the STAT3-signaling pathway. J. Investig. Dermatol. 2008, 128, 2429–2441.
- Syed, F.; Bagabir, R.A.; Paus, R.; Bayat, A. Ex vivo evaluation of antifibrotic compounds in skin scarring: EGCG and silencing of PAI-1 independently inhibit growth and induce keloid shrinkage. Lab. Investig. 2013, 93, 946–960.
- Zhang, Q.; Kelly, A.P.; Wang, L.; French, S.W.; Tang, X.; Duong, H.S.; Messadi, D.V.; Le, A.D. Green tea extract and (-)-epigallocatechin-3-gallate inhibit mast cell-stimulated type I collagen expression in keloid fibroblasts via blocking PI-3K/AkT signaling pathways. J. Investig. Dermatol. 2006, 126, 2607–2613.
- Jurzak, M.; Adamczyk, K.; Antonczak, P.; Garncarczyk, A.; Kusmierz, D.; Latocha, M. Evaluation of genistein ability to modulate CTGF mRNA/protein expression, genes expression of TGFbeta isoforms and expression of selected genes regulating cell cycle in keloid fibroblasts in vitro. Acta Pol. Pharm. 2014, 71, 972–986.
- Zhang, X.; Liu, W.; Wei, S. Luteolin affects keloid fibroblast proliferation and apoptosis by regulating FRAT1 gene expression. Cell Mol. Biol. 2020, 66, 185–190.
- Zhang, Q.; Qian, D.; Tang, D.D.; Liu, J.; Wang, L.Y.; Chen, W.; Wu, C.J.; Peng, W. Glabridin from Glycyrrhiza glabra Possesses a Therapeutic Role against Keloid via Attenuating PI3K/Akt and Transforming Growth Factor-beta1/SMAD Signaling Pathways. J. Agric. Food Chem. 2022, 70, 10782–10793.
- Pu, X.; Cao, X.; Liu, H.; Huang, W.; Zhang, L.; Jiang, T. Isorhamnetin attenuates the proliferation, invasion, migration and fibrosis of keloid fibroblasts by targeting S1PR1. Exp. Ther. Med. 2023, 26, 310.
- Hsu, Y.C.; Chen, M.J.; Yu, Y.M.; Ko, S.Y.; Chang, C.C. Suppression of TGF-beta1/SMAD pathway and extracellular matrix production in primary keloid fibroblasts by curcuminoids: Its potential therapeutic use in the chemoprevention of keloid. Arch. Dermatol. Res. 2010, 302, 717–724.
- Ikeda, K.; Torigoe, T.; Matsumoto, Y.; Fujita, T.; Sato, N.; Yotsuyanagi, T. Resveratrol inhibits fibrogenesis and induces apoptosis in keloid fibroblasts. Wound Repair. Regen. 2013, 21, 616–623.
- Tang, B.; Zhu, B.; Liang, Y.Y.; Bi, L.K.; Hu, Z.C.; Chen, B.; Zhang, K.; Zhu, J.Y. Asiaticoside suppresses collagen expression and TGF-beta/Smad signaling through inducing Smad7 and inhibiting TGF-beta RI and TGF-beta RII in keloid fibroblasts. Arch. Dermatol. Res. 2011, 303, 563–572.
- Wu, X.; Bian, D.; Dou, Y.; Gong, Z.; Tan, Q.; Xia, Y.; Dai, Y. Asiaticoside hinders the invasive growth of keloid fibroblasts through inhibition of the GDF-9/MAPK/Smad pathway. J. Biochem. Mol. Toxicol. 2017, 31, e21922.
- Bian, D.; Zhang, J.; Wu, X.; Dou, Y.; Yang, Y.; Tan, Q.; Xia, Y.; Gong, Z.; Dai, Y. Asiatic acid isolated from Centella asiatica inhibits TGF-beta1-induced collagen expression in human keloid fibroblasts via PPAR-gamma activation. Int. J. Biol. Sci. 2013, 9, 1032–1042.
- Tang, M.; Bian, W.; Cheng, L.; Zhang, L.; Jin, R.; Wang, W.; Zhang, Y. Ginsenoside Rg3 inhibits keloid fibroblast proliferation, angiogenesis and collagen synthesis in vitro via the TGF-beta/Smad and ERK signaling pathways. Int. J. Mol. Med. 2018, 41, 1487–1499.
- Ranti, I.; Wahyuningsih, M.S.H.; Wirohadidjojo, Y.W. The antifibrotic effect of isolate tagitinin C from tithonia diversifolia (Hemsley) A. Gray on keloid fibroblast cell. Pan Afr. Med. J. 2018, 30, 264.
- De Felice, B.; Manfellotto, F.; Garbi, C.; Santoriello, M.; Nacca, M. miR-34 modulates apoptotic gene expression in Ingenol mebutate treated keloid fibroblasts. Mol. Med. Rep. 2018, 17, 7081–7088.
- Jeon, Y.R.; Roh, H.; Jung, J.H.; Ahn, H.M.; Lee, J.H.; Yun, C.O.; Lee, W.J. Antifibrotic Effects of High-Mobility Group Box 1 Protein Inhibitor (Glycyrrhizin) on Keloid Fibroblasts and Keloid Spheroids through Reduction of Autophagy and Induction of Apoptosis. Int. J. Mol. Sci. 2019, 20, 4134.
- Luo, Y.; Wang, D.; Yuan, X.; Jin, Z.; Pi, L. Oleanolic acid regulates the proliferation and extracellular matrix of keloid fibroblasts by mediating the TGF-beta1/SMAD signaling pathway. J. Cosmet. Dermatol. 2023, 22, 2083–2089.
- Zhang, G.Y.; Gao, W.Y.; Li, X.; Yi, C.G.; Zheng, Y.; Li, Y.; Xiao, B.; Ma, X.J.; Yan, L.; Lu, K.H.; et al. Effect of camptothecin on collagen synthesis in fibroblasts from patients with keloid. Ann. Plast. Surg. 2009, 63, 94–99.
- Gao, Y.; Cheng, X.; Wang, Z.; Wang, J.; Gao, T.; Li, P.; Kong, M.; Chen, X. Transdermal delivery of 10,11-methylenedioxycamptothecin by hyaluronic acid based nanoemulsion for inhibition of keloid fibroblast. Carbohydr. Polym. 2014, 112, 376–386.
- Fan, D.L.; Zhao, W.J.; Wang, Y.X.; Han, S.Y.; Guo, S. Oxymatrine inhibits collagen synthesis in keloid fibroblasts via inhibition of transforming growth factor-beta1/Smad signaling pathway. Int. J. Dermatol. 2012, 51, 463–472.
- Song, N.; Wu, X.; Gao, Z.; Zhou, G.; Zhang, W.J.; Liu, W. Enhanced expression of membrane transporter and drug resistance in keloid fibroblasts. Hum. Pathol. 2012, 43, 2024–2032.
- Wang, M.; Chen, L.; Huang, W.; Jin, M.; Wang, Q.; Gao, Z.; Jin, Z. Improving the anti-keloid outcomes through liposomes loading paclitaxel-cholesterol complexes. Int. J. Nanomed. 2019, 14, 1385–1400.
- Song, R.; Li, G.; Li, S. Aspidin PB, a novel natural anti-fibrotic compound, inhibited fibrogenesis in TGF-beta1-stimulated keloid fibroblasts via PI-3K/Akt and Smad signaling pathways. Chem. Biol. Interact. 2015, 238, 66–73.
- Chen, G.; Liang, Y.; Liang, X.; Li, Q.; Liu, D. Tanshinone IIA Inhibits Proliferation and Induces Apoptosis through the Downregulation of Survivin in Keloid Fibroblasts. Ann. Plast. Surg. 2016, 76, 180–186.
- Lu, L.; Chai, L.; Wang, W.; Yuan, X.; Li, S.; Cao, C. A Selenium-Enriched Ziyang Green Tea Polysaccharide Induces Bax-Dependent Mitochondrial Apoptosis and Inhibits TGF-beta1-Stimulated Collagen Expression in Human Keloid Fibroblasts via NG2 Inactivation. Biol. Trace Elem. Res. 2017, 176, 270–277.
- Niu, T.; Tian, Y.; Shi, Y.; Guo, G.; Tong, Y.; Wang, G. Antifibrotic effects of Hypocrellin A combined with LED red light irradiation on keloid fibroblasts by counteracting the TGF-beta/Smad/autophagy/apoptosis signalling pathway. Photodiagn. Photodyn. Ther. 2021, 34, 102202.
- Jurzak, M.; Adamczyk, K. Influence of genistein on c-Jun, c-Fos and Fos-B of AP-1 subunits expression in skin keratinocytes, fibroblasts and keloid fibroblasts cultured in vitro. Acta Pol. Pharm. 2013, 70, 205–213.
- Ito, S.; Nagata, K. Roles of the endoplasmic reticulum-resident, collagen-specific molecular chaperone Hsp47 in vertebrate cells and human disease. J. Biol. Chem. 2019, 294, 2133–2141.
- Bellaye, P.S.; Burgy, O.; Bonniaud, P.; Kolb, M. HSP47: A potential target for fibrotic diseases and implications for therapy. Expert. Opin. Ther. Targets. 2021, 25, 49–62.
- Khaiwa, N.; Maarouf, N.R.; Darwish, M.H.; Alhamad, D.W.M.; Sebastian, A.; Hamad, M.; Omar, H.A.; Orive, G.; Al-Tel, T.H. Camptothecin’s journey from discovery to WHO Essential Medicine: Fifty years of promise. Eur. J. Med. Chem. 2021, 223, 113639.
- Skubnik, J.; Pavlickova, V.S.; Ruml, T.; Rimpelova, S. Vincristine in Combination Therapy of Cancer: Emerging Trends in Clinics. Biology 2021, 10, 849.
- Tu, Y.; Cheng, S.X.; Zhang, S.; Sun, H.T.; Xu, Z.W. Vincristine induces cell cycle arrest and apoptosis in SH-SY5Y human neuroblastoma cells. Int. J. Mol. Med. 2013, 31, 113–119.
- Niu, T.H.; Tian, Y.; Wang, G.Y.; Guo, G.J.; Tong, Y.; Shi, Y. Inhibition of ROS-NF-κB-dependent autophagy enhances Hypocrellin A united LED red light-induced apoptosis in squamous carcinoma A431 cells. Cell. Signal. 2020, 69, 109550.
More
Information
Subjects:
Dermatology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
652
Revisions:
2 times
(View History)
Update Date:
20 Feb 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





