Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Dimitrios Savvas | -- | 4066 | 2024-01-05 11:30:06 | | | |
| 2 | Lindsay Dong | + 4 word(s) | 4070 | 2024-01-08 01:19:32 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Savvas, D.; Giannothanasis, E.; Ntanasi, T.; Karavidas, I.; Ntatsi, G. Crop Fertigation in Closed Soilless Systems. Encyclopedia. Available online: https://encyclopedia.pub/entry/53479 (accessed on 24 May 2026).
Savvas D, Giannothanasis E, Ntanasi T, Karavidas I, Ntatsi G. Crop Fertigation in Closed Soilless Systems. Encyclopedia. Available at: https://encyclopedia.pub/entry/53479. Accessed May 24, 2026.
Savvas, Dimitrios, Evangelos Giannothanasis, Theodora Ntanasi, Ioannis Karavidas, Georgia Ntatsi. "Crop Fertigation in Closed Soilless Systems" Encyclopedia, https://encyclopedia.pub/entry/53479 (accessed May 24, 2026).
Savvas, D., Giannothanasis, E., Ntanasi, T., Karavidas, I., & Ntatsi, G. (2024, January 05). Crop Fertigation in Closed Soilless Systems. In Encyclopedia. https://encyclopedia.pub/entry/53479
Savvas, Dimitrios, et al. "Crop Fertigation in Closed Soilless Systems." Encyclopedia. Web. 05 January, 2024.
Copy Citation
Inappropriate fertilisation results in the pollution of water resources with nitrates and phosphates, eutrophication in surface water, emission of greenhouse gasses, and unwanted N deposition in natural environments, thereby harming the whole ecosystem. Cultivation in closed soilless systems entails recycling of the fertigation effluents thus eliminating nutrient emissions to the environment and concomitantly pollution of water resources with nitrates and phosphates.
closed-loop soilless culture
drainage solution
water recycling
nutrient recirculation
decision support systems
NUTRISENSE
1. Introduction
Inappropriate crop fertilisation not only reduces yield and produce quality but also leads to nutrient emissions into the air and water resources, among which nitrogen and phosphorus result in serious environmental impacts. These emissions have serious negative consequences for the environment, including groundwater pollution rendering water undrinkable, eutrophication of aquatic ecosystems, and greenhouse gas emissions. The emission of nitrogen in agricultural crops occurs either in gaseous form due to denitrification and ammonia volatilisation, or in mineral form through the fertigation effluents [1]. Over-fertilisation with manure and inorganic P fertilisers for many years has resulted in saturation of the retention capacity for P in many agricultural soils and, consequently, P loss by run-off and leaching to surface water resources results, together with nitrate emissions, in eutrophication of surface water [2]. In view of these serious threats to the ecosystem, the European Union has set a strategic target in the Green Deal to reduce fertiliser consumption by 20% and nutrient losses by 50% by 2030 [3]. To mitigate the pollution of water resources caused by inappropriate fertilisation and irrigation practices, there is an urgent need to apply new agricultural production approaches and innovative technologies for crop management aiming to reduce inputs and extend the circularity of nutrients.
Greenhouse cultivation is an intensive form of plant production and thus inappropriate fertilisation of greenhouse crops can result in considerable emissions of nutrients to the environment. Soilless culture systems (SCSs) constitute the dominant cultivation technique in the modern greenhouse industry. The soilless crops are distinguished into open or closed-loop systems depending on the management of the drained solution, which can be either discharged to the environment or collected and recycled. Open soilless culture systems are easier to manage as the composition of the supplied nutrient solution is completely controlled by the grower. However, open systems have been criticised as inefficient from an environmental point of view because they result in the waste of nutrients and water, and pollution of the water resources by the discharged leachate. To minimise the pollution of water by nutrient emissions originating from the fertigation of greenhouse crops and to render greenhouse production more sustainable and environment-friendly, closed-loop SCSs have been developed, henceforth abbreviated as closed-loop soilless culture systems (CLSs). Nevertheless, crop nutrition is more complicated in CLSs than in open systems because the composition of the recycled DS is variable over time and unknown to the growers. Furthermore, in CLSs, the DS should be treated before its recycling to eliminate pathogens, thereby avoiding the spread of diseases from a small spot of infection to the whole crop. Despite these disadvantages, there is a shift from open soilless systems to CLSs due to their environmental benefits and the substantial reduction in fertilizer and water use without compromising yield performance.
2. Plant Nutrition in Soilless Cultivations
In soilless cultivations grown on inert substrates or in pure NS without any aggregate, the nutrition of the plants relies exclusively on fertigation, i.e., on the supply of NS to the crop via the irrigation system. The roots of the plants are in contact with the root solution and thus the satisfaction of their nutritional requirements depends directly on the nutrient levels in the root environment, irrespective of whether they grow in the soil or soilless systems. In soil-grown crops, the nutrient status in the root environment depends on the quantity of the nutrient reserves and the rate at which they are released to the soil solution. Hence, the nutrient status in the roots of soil-grown crops cannot be easily and quickly altered by crop fertilisation practices. However, the nutrient reserves in the root zone of soilless-grown crops are limited because the available substrate volume for root development is significantly less than in plants grown in soil. As a consequence, the nutrient status in the root environment of soilless crops may exhibit considerable fluctuations from day to day. Thus, the main objective of an efficient nutrient management system in soilless crops should be to maintain an optimal nutrient status in the root zone by properly modifying fertilisation practices whenever needed to achieve this goal. Consequently, to ensure optimal nutrition in a soilless crop, the composition of the NS supplied to a crop should be considered a variable that may change frequently, aiming to achieve the primary objective, which is the maintenance of a target nutrient status in the root environment [4].
The nutrients supplied via the NS to a soilless cropping system are removed from this system either via plant uptake or drainage solution [5]. If the removal of nutrients from the system via these two paths is lower or higher than the supply, the excessive or the missing nutrients will accumulate or deplete, respectively, in the root zone. Thus, to maintain optimal nutrient levels in the root zone of a soilless crop, which is the strategic target, the nutrient supply should be equal to the removal of nutrients via both paths, i.e., plant uptake and drainage. In CLSs, there is no output via drainage, while in open soilless cropping systems, the output of nutrients via the DS can be partly controlled by properly adjusting the drainage fraction. The only output of nutrients in CLSs is due to uptake by the crop, but this can be only roughly predicted if credible experimental data are available.
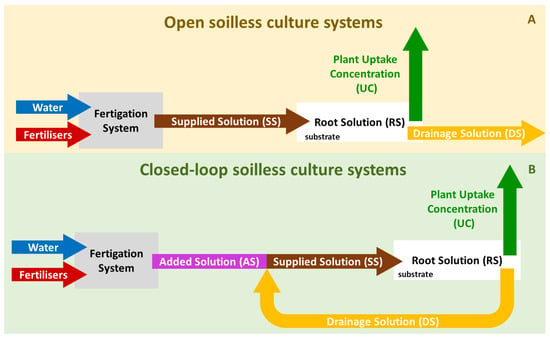
In Figure 1, the parameters involved in the supply of NS to soilless cultivations in both open and closed systems and the relevant terminologies are schematically outlined.

Figure 1. Schematic representation of the process of NS preparation in open (A) and closed-loop soilless cropping systems (B) and the terminologies used for the different parameters involved. Water: the raw water used for the preparation of NS; Fertilisers: liquid concentrated solutions (CSs) of water-soluble fertilisers used to add nutrients to the NS; Plant Uptake Concentrations (UCs): the ratios between each nutrient and the volume of water absorbed by the plant. Drainage solution (DS): the NS that drains out of the root zone after each irrigation event. Supplied solution (SS): the NS that is ultimately supplied to the crop, which in open SCSs is fresh NS prepared by mixing water with fertilisers, while in closed-loop SCSs it is the mixture of nutrients, water, and DS. Root solution (RS): the NS that is in immediate contact with the plant roots. Added solution (AS): an NS with concentrations corresponding to the ratios between the net nutrient input and the net water input (excluding the input of DS), when preparing SS in CLSs.
In both open and closed-loop SCSs, the NS that is ultimately supplied to the crop is termed supplied solution (SS). In substrate-grown crops, when the plants are irrigated through a drip irrigation system, the SS is frequently referenced as a “drip solution”. In open SCSs (Figure 1A), the SS is simply prepared by injecting liquid stock solutions of fertilisers into irrigation water at rates resulting in a pre-set electrical conductivity (EC) in this, using suitable equipment termed “fertigation system” (FS). In CLSs (Figure 1B), the SS is prepared by mixing raw water, stock solutions of fertilisers, and DS at suitable rates. In CLSs, the masses of plant nutrients added through the fertilisers and the volume of raw water added to the DS to prepare SS correspond to specific nutrient-to-water rations, which are considered concentrations of a solution termed added solution (AS) [5]. The AS constitutes the net input of nutrients and water to the SS, while the DS provides nutrients and water to the SS that are already in the system.
The ratio between the mass of a plant nutrient and the volume of water removed from soilless cultivation through plant uptake, expressed as mmol L−1 (mM), corresponds to a concentration of this nutrient, which is termed “uptake concentration” (UC) in the international scientific literature [5][6][7]. The UCs of all plant nutrients correspond to a theoretical NS that fully covers the plant’s needs but does not exist as a solution. In the international literature, there are experimental data for UCs of a wide range of plant species cultivated in SCSs [6][8][9][10][11][12].
The NS retained in the pores of the substrate, in troughs, or any other containers used for plant growth is termed “root solution” (RS), as it is in contact with the root system of the plants. When plants are grown out of the soil using a substrate as a rooting medium, part of the RS drains out of the root zone as DS and, consequently, the composition of the RS and the DS is almost identical. Therefore, in most cases, the management of nutrition in soilless crops is based on sampling and analysing DS and considering its composition as identical to that of the RS [13]. The indirect determination of the RS composition by measuring the composition of the DS is a common practice because the collection of RS samples directly from the substrates is difficult (except for rockwool), while the obtained samples are less representative of the whole crop than a sample of DS originating from a large crop area.
The mutual ratios between nutrients in the plant tissues are roughly similar in different crop species, although the differences are negligible. Based on this fact, a standard NS composition was developed by Hoagland and Arnon (1950) [14] and is used even today in plant nutrition studies. However, fine-tuning the composition of the NS supplied to crops to match the specific requirements of each cultivated species can substantially improve crop performance, especially in commercial greenhouses. The nutrients that should be essentially added to water to prepare NS include six macronutrients (K, Ca, Mg, N, P, S) and six micronutrients (Fe, Mn, Zn, Cu, B, Mo). Chloride is needed in very tiny amounts, which are always available in the NS, originating either from the mineral composition of the water or from fertiliser impurities.
Plants can selectively absorb most nutrients from the external solution to cover their specific nutritional requirements. Therefore, the nutrient concentrations in the root zone of plants grown in soilless cropping systems can fluctuate within a wide range. However, to maximize yield and quality in commercial soilless cultivations, it is important to provide the nutrients to the plants at rates that best meet their specific requirements.
To specify an optimal concentration of a certain nutrient in the SS for a crop species, two parameters must be determined experimentally: the mean UC and the optimal concentration of this nutrient in the RS [4][5]. As suggested from previous experimental results, the UC depends on the concentrations of the respective nutrients in the root zone [11][15]. The impact of the RS composition on the UCs may arise from differences in nutrient uptake mechanisms. Calcium is absorbed passively from the apical parts of the root hairs via mass-flow mechanisms [16], while potassium is taken up selectively [17][18].
In soilless culture, the total salt concentration in the root solution is considered one of the most important parameters determining yield and produce quality. The total salt concentration in the NS is expressed as electrical conductivity (EC, dS m−1) because this quantity can be easily and accurately measured in the greenhouse using portable EC meters. Many studies have shown that the crop yield in a soilless cultivated crop decreases as the EC in the RS increases, and the yield decrease is linearly or curvilinearly associated with the increase in the EC in the SS above a threshold value [6][19].
The pH of the RS also affects yield and fruit quality as it has a strong impact on the availability of some nutrients to the crop. More specifically, too high pH levels in the RS, i.e., values higher than 6.3–6.5, depending on the crop species, restrict the availability of Fe, Mn, Cu, and Zn due to partial conversion of the active bivalent cations into M(OH)2 (M = Fe, Mn, Zn, or Cu) which precipitate and are thus unavailable to plants. Nevertheless, due to the use of Fe chelates in NSs destined for soilless cultures, substantial amounts of Mn, Zn, and Cu are chelated as pH increases, and therefore their availability to plants with increasing pH of the RS is a more complex issue [6][20]. At pH levels below 5.0, there is a high risk of Mn and/or Al toxicity due to the partial conversion of their oxides into active Mn2+ and Al3+ ions.
3. Crop Fertigation in Closed Soilless Systems
3.1. Feasibility of Closed Soilless Systems
In CLSs, the fertigation effluents, henceforth termed drainage solution (DS), are collected and recycled. The main environmental advantage of CLSs is the elimination of surface and underground water pollution, as nitrogen and phosphorus emissions originating from DS discharge are eliminated or at least drastically reduced. Moreover, the recycling of fertigation effluents reduces irrigation water and fertiliser consumption by more than 20–35% and 40–50%, respectively [4]. CLSs are mandatory in northern European countries, such as The Netherlands, to avoid contamination of water resources by nutrient emissions [13]. However, CLSs are not common in southern European countries and many other countries in the world. The main bottleneck in the adoption of DS recycling in soilless cultures is the presence of sodium at relatively high concentrations in the water used to prepare nutrient solution (NS), which leads to Na+ accumulation in the recycled DS, and, concomitantly, in exposure of the crop to salinity stress [6][13][21].
3.2. Nutrient Solution Management in CLSs
Inappropriate composition of the SS can cause nutrient imbalances in the root environment, which may affect plant growth through nutrient deficiency, toxicity, or salinity stress [6][22][23][24]. Managing plant mineral nutrition by providing SS with optimal composition is a big challenge in CLSs, as the composition of the DS, which is a constituent of the SS, can vary with time due to temporal variations in nutrient uptake rates. Fluctuations in UCs have been observed not only between different plant species but also in the same species between different cultivars [9][25] or under different climatic conditions [26] or stress conditions, such as salinity stress due to Na+ accumulation [25]. To address the challenges associated with maintaining optimal nutrient supply to crops in the CLSs despite the unpredictable variations in the composition of the DS, two alternative concepts can be deployed. The first concept is based on a target nutrient composition for the SS, while the second concept is based on a target nutrient composition of the AS [8][27]. Both concepts are based on regularly monitoring the mineral composition of the DS.
Based on the concept of a target concentration of the SS, the composition of the DS is used to estimate the composition of the mix of DS and water. Subsequently, the nutrient concentrations in the mix are deducted from the target nutrient concentrations in the SS to obtain the rates of nutrient injection through fertilisers. Based on the concept of a target concentration in the AS, an AS with concentrations equal to the anticipated UCs is supplied. Thus, the rates of nutrient input (concentrations in the AS) are set equal to those of nutrient output from the system (UCs). Indeed, the only net nutrient input in CLSs is the AS, and the only nutrient output is the plant uptake because no DS is discharged. If the concentration of a nutrient in the AS is higher or lower than its UC, accumulation or depletion, respectively, of this nutrient in the root zone would gradually occur.
The UC of a nutrient fluctuates within a relatively narrow range under controlled greenhouse conditions when the concentration of this nutrient is roughly stable in the root zone of the plants [28]. However, some variation in the UC of a nutrient may occur during the cropping period due to changes in (i) the level of this nutrient in the root zone, (ii) microclimatic parameters, (iii) fruit load, and (iv) any other factor that can alter the nutrient needs of the plants [15][26].
Water culture systems such as the nutrient film technique (NFT), the deep flow technique (DFT), the floating system, and aeroponics are essentially operated as CLSs [10]. These systems are mostly used for the production of leafy vegetables. In these cropping systems, the nitrate content in the edible plant parts is much more effectively controlled without compromising the yield by substantially reducing the nitrate concentration in the added solution for some days before harvesting. Thus, the safety of leafy vegetables produced as hydroponic crops is higher than that of leafy vegetables produced as soil-grown crops. The reduction in the nitrate supply for a few days before harvesting can be compensated for by an equivalent increase in the sulphate or chloride concentration in the added solution [10]. Finally, the microelement content of vegetables produced in CLSs is much better regulated than in soil-grown crops by properly changing their concentrations in the added solution, taking into consideration their levels in the drainage solution, which are frequently determined.
3.3. Sodium Accumulation in Closed-Loop Soilless Culture Systems
The high Na+ concentration in the raw water used to prepare NS is the bottleneck in DS recycling. The UCs of Na+ by most cultivated plants are generally much lower than the Na+ concentrations in the water sources commonly used for irrigation [13][21][29][30][31]. Raw water with moderately high Na+ concentration can be used in CLSs if mixed with appropriate amounts of rainwater. However, this option is not available for growers in regions with low yearly rainfall, like those in many parts of the Mediterranean basin, who face serious problems with excessively high Na+ levels in the available water sources used for NS preparation [32]. The Na+ concentration in the AS is identical to that in the water used to prepare it, as no Na+ is added to NSs through fertilisers.
If the concentration of an ion in the AS exceeds that in the UC, this ion gradually accumulates in the RS, as the sole output of the ions from the RS in a CLS is plant uptake. The gradual increase in Na+ levels in the RS leads to elevated EC levels that restrict yield due to salinity stress [33][34]. To address this issue, a common practice is partial discharge of DS, which facilitates sodium output from the RS [35][36]. This technique is termed “semi-closed soilless culture system” (semi-CLS). This practice diminishes the environmental sustainability of CLSs and decreases water and nutrient use efficiency, and it is not allowed or is allowed under strict conditions in some northern European countries [13][37].
Controlled exposure of the crop to salinity eustress enhances the fruit quality of many vegetables such as tomatoes [38]. Thus, to obtain high-quality fruit, the standard recommended EC and nutrient concentrations in the root zones of soilless cultivated fruit vegetables are higher than the minimum concentrations required for optimal nutrition to impose salinity eustress [33]. If the Na+ concentration in the raw water used to prepare NS results in Na+ accumulation to levels that are not toxic to the plants but only impose salinity stress due to excessively high EC levels, a smart nutrient management strategy can alleviate or even eliminate yield losses. This strategy, which can be commercially applied, is based on the gradual decrease in nutrient input as Na+ accumulates, thus preventing an increase in EC in the RS [33][35][39]. This strategy accepts higher sodium levels in the RS without a commensurate increase in EC to harmful levels in the RS.
3.4. Technologies Used for Nutrient Solution Preparation in Closed Soilless Systems
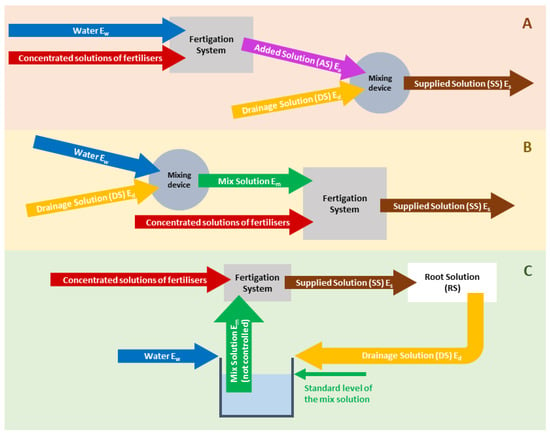
In commercial CLSs, the blending of DS with raw water and fertilisers to prepare SS can be performed by applying three alternative technologies, depending on the soilless cropping system. These alternative approaches differ mainly in the sequence of performing two distinct steps during the preparation of the SS for CLSs, i.e., the injection of stock solutions via the FS and mixing with the DS. In addition to the sequence of adding fertilisers and DS when preparing SS, the level of EC control during the whole process also differentiates the applied technical approach. The three alternative technical approaches are illustrated in Figure 2 and are described in more detail in the next section.

Figure 2. Different approaches for preparing NS for soilless crops when reusing the drainage solution. (A) First, the fertilisers are added to water to prepare AS with a target EC (Eu); subsequently, the DS is mixed with the AS to prepare SS with a target EC (Es). (B) First, the DS is mixed with raw water to obtain a mix with a pre-set EC (Em); subsequently, the fertilisers are added to this mix to prepare AS with a target EC (Es). (C) First, the DS is mixed with raw water without controlling the EC of the mix; subsequently, the fertilisers are added to this mix to prepare AS with a target EC (Es) [5].
3.4.1. Injecting Fertilisers into Water to Prepare AS with a Target EC (Ea), and Subsequently Mixing the DS with the AS to Prepare SS with a Target EC (Es)
This approach (Figure 2A) can be applied if the nutrient injection into the system is calculated according to the concept of target concentrations in the SS. According to this technical approach, an automated fertigation system injects stock solutions into the irrigation water, thus preparing the AS. The AS is subsequently blended with DS using a mixing device that automatically adjusts the mixing ratio with the aim of reaching a pre-set target EC in the outgoing SS (Es). This system is technically simple and understandable both in its technical implementation and in the calculation of the fertiliser needed to prepare stock solutions (or the nutrient injection rates if single-fertiliser stock solutions are used). However, a major drawback of this approach is the difficulty in properly adjusting the pH of the SS because the pH of the DS is usually higher than the target value. Consequently, the pH of the SS may increase to levels higher than the target value when the AS and the DS are mixed.
3.4.2. Mixing the DS with Raw Water to Obtain a Mix with a Pre-Set EC (Em), and Subsequently Injecting Fertiliser Stock Solutions into the Mix to Prepare SS with a Target EC (Es)
This technical approach (Figure 2B) is compatible with both concepts, i.e., the target composition of the AS and the target composition of the SS. The concept of a target composition in the SS better matches this technical approach because the fertiliser injection (composition or injection rates of stock solutions) is calculated following similar steps to those performed in open systems. However, the changes in the composition of the DS during the time interval between two determinations of its composition make this concept less reliable than the concept of a target composition of the AS, which involves standard rates of net nutrient supply based on the anticipated uptake concentrations.
3.4.3. Mixing the DS with Raw Water without Controlling the EC of the Mix, and Subsequently Injecting Fertiliser Stock Solutions into the Mix to Prepare SS with a Target EC (Es)
This concept (Figure 2C) is mainly applied in water culture systems (hydroponics). Due to the lack of a porous medium that can retain NS reserves in the root environment in water culture systems, the flow rate of supplied NS is dramatically higher than in substrate-grown crops. As a result, the DS fraction is only slightly lower than 1 and the irrigation of the crop with SS is frequent, if not continuous. Hence, it is much easier and thus more reasonable to initially collect the DS in a tank with a constant level maintained by controlling the inlet of raw water (e.g., using a floater). Since the mixing ratio of DS and water in this tank is controlled by maintaining a constant level and not by adjusting it automatically to achieve the target EC, the EC of the mix is variable, although no substantial fluctuations are anticipated. This mix can be pumped periodically into a fertigation system to inject fertilisers into it, thus maintaining its EC and pH close to preset target values and supplied to the crop either continuously or following a desired irrigation schedule. Furthermore, a similar approach is applied in floating systems, which are also water culture systems. The main tank where the roots are developed contains the RS, and AS is supplied to maintain RS at a constant level. This technical approach is only compatible with the concept of target nutrient composition in the AS.
References
- Qasim, W.; Xia, L.; Lin, S.; Wan, L.; Zhao, Y.; Butterbach-Bahl, K. Global greenhouse vegetable production systems are hotspots of soil N2O emissions and nitrogen leaching: A meta-analysis. Environ. Pollut. 2021, 272, 116372.
- Grenon, G.; Singh, B.; de Sena, A.; Madramootoo, C.A.; von Sperber, C.; Goyal, M.K.; Zhang, T. Phosphorus Fate, Transport and Management on Subsurface Drained Agricultural Organic Soils: A Review. Environ. Res. Lett. 2021, 16, 013004.
- European Green Deal. 2019. Available online: https://agriculture.ec.europa.eu/sustainability/environmental-sustainability/low-input-farming/nutrients_en (accessed on 13 October 2023).
- Savvas, D.; Giannothanasis, E.; Ntanasi, T.; Karavidas, I.; Drakatos, S.; Panagiotakis, I.; Neocleous, D.; Ntatsi, G. Improvement and Validation of a Decision Support System to Maintain Optimal Nutrient Levels in Crops Grown in Closed-Loop Soilless Systems. Agric. Water Manag. 2023, 285, 108373.
- Blok, C.; Voogt, W.; Barbagli, T. Reducing Nutrient Imbalance in Recirculating Drainage Solution of Stone Wool Grown Tomato. Agric. Water Manag. 2023, 285, 108360.
- Sonneveld, C.; Voogt, W. Plant Nutrition of Greenhouse Crops; Springer: Dordrecht, The Netherlands, 2009; ISBN 9789048125326.
- Thompson, R.B.; Gallardo, M.; Rodríguez, J.S.; Sánchez, J.A.; Magán, J.J. Effect of N Uptake Concentration on Nitrate Leaching from Tomato Grown in Free-Draining Soilless Culture under Mediterranean Conditions. Sci. Hortic. 2013, 150, 387–398.
- De Kreij, C.; Voogt, W.; Baas, R. Nutrient Solutions and Water Quality for Soilless Cultures; PBG: Naaldwijk, The Netherlands, 1999.
- Ropokis, A.; Ntatsi, G.; Kittas, C.; Katsoulas, N.; Savvas, D. Impact of Cultivar and Grafting on Nutrient and Water Uptake by Sweet Pepper (Capsicum annuum L.) Grown Hydroponically under Mediterranean Climatic Conditions. Front. Plant Sci. 2018, 9, 1244.
- Neocleous, D.; Nikolaou, G.; Ntatsi, G.; Savvas, D. Nitrate Supply Limitations in Tomato Crops Grown in a Chloride-Amended Recirculating Nutrient Solution. Agric. Water Manag. 2021, 258, 107163.
- Neocleous, D.; Savvas, D. Effect of Different Macronutrient Cation Ratios on Macronutrient and Water Uptake by Melon (Cucumis melo L.) Grown in Recirculating Nutrient Solution. J. Plant. Nutr. Soil Sci. 2015, 178, 320–332.
- Savvas, D.; Öztekin, G.B.; Tepecik, M.; Ropokis, A.; Tüzel, Y.; Ntatsi, G.; Schwarz, D. Impact of Grafting and Rootstock on Nutrient-to-Water Uptake Ratios during the First Month after Planting of Hydroponically Grown Tomato. J. Hortic. Sci. Biotechnol. 2017, 92, 294–302.
- Voogt, W.; Bar-Yosef, B. Water and Nutrient Management and Crops Response to Nutrient Solution Recycling in Soilless Growing Systems in Greenhouses. In Soilless Culture: Theory and Practice Theory and Practice; Elsevier: Amsterdam, The Netherlands, 2019; pp. 425–507. ISBN 9780444636966.
- Hoagland, D.R.; Arnon, D.I. The Water-Culture Method for Growing Plants without Soil. Calif. Agric. Exp. St. Circ. 1950, 347, 1–32.
- Ropokis, A.; Ntatsi, G.; Rouphael, Y.; Kotsiras, A.; Kittas, C.; Katsoulas, N.; Savvas, D. Responses of Sweet Pepper (Capsicum annum L.) Cultivated in a Closed Hydroponic System to Variable Calcium Concentrations in the Nutrient Solution. J. Sci. Food Agric. 2021, 101, 4342–4349.
- De Freitas, S.T.; Mitcham, E.J. Factors Involved in Fruit Calcium Deficiency Disorders. In Horticultural Reviews; Janick, J., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2012; Volume 40, pp. 107–146.
- Maathuis, F.J.M.; Sanders, D. Cell Biology Mechanism of High-Affinity Potassium Uptake in Roots of Arabidopsis Thaliana (Energized K+ Transport/K+-H+ CotaspWrt/Current/Voltage Analyis). Proc. Natl. Acad. Sci. USA 1994, 91, 9272–9276.
- Britto, D.T.; Kronzucker, H.J. Cellular Mechanisms of Potassium Transport in Plants. Physiol. Plant 2008, 133, 637–650.
- Bione, M.A.A.; Soares, T.M.; Cova, A.M.W.; Paz, V.P.d.S.; Gheyi, H.R.; Rafael, M.R.S.; Modesto, F.J.N.; Santana, J.d.A.; Neves, B.S.L. Hydroponic Production of ‘Biquinho’ Pepper with Brackish Water. Agric. Water Manag. 2021, 245, 106607.
- De Rijck, G.; Schrevens, E. Cationic Speciation in Nutrient Solutions as a Function of PH. J. Plant Nutr. 1998, 21, 861–870.
- Varlagas, H.; Savvas, D.; Mouzakis, G.; Liotsos, C.; Karapanos, I.; Sigrimis, N. Modelling Uptake of Na+ and Cl− by Tomato in Closed-Cycle Cultivation Systems as Influenced by Irrigation Water Salinity. Agric. Water Manag. 2010, 97, 1242–1250.
- Tzerakis, C.; Savvas, D.; Sigrimis, N.; Mavrogiannopoulos, G. Uptake of Mn and Zn by Cucumber Grown in Closed Hydroponic Systems as Influenced by the Mn and Zn Concentrations in the Supplied Nutrient Solution. Hortscience 2013, 48, 373–379.
- Neocleous, D.; Savvas, D. Response of Hydroponically-Grown Strawberry (Fragaria ananassa Duch.) Plants to Different Ratios of K:Ca:Mg in the Nutrient Solution. J. Hortic. Sci. Biotechnol. 2013, 88, 293–300.
- Massa, D.; Magán, J.J.; Montesano, F.F.; Tzortzakis, N. Minimizing Water and Nutrient Losses from Soilless Cropping in Southern Europe. Agric. Water Manag. 2020, 241, 106395.
- Ntanasi, T.; Karavidas, I.; Zioviris, G.; Ziogas, I.; Karaolani, M.; Fortis, D.; Conesa, M.À.; Schubert, A.; Savvas, D.; Ntatsi, G. Assessment of Growth, Yield, and Nutrient Uptake of Mediterranean Tomato Landraces in Response to Salinity Stress. Plants 2023, 12, 3551.
- Gallardo, M.; Cuartero, J.; Andújar de la Torre, L.; Padilla, F.M.; Segura, M.L.; Thompson, R.B. Modelling Nitrogen, Phosphorus, Potassium, Calcium and Magnesium Uptake, and Uptake Concentration, of Greenhouse Tomato with the VegSyst Model. Sci. Hortic. 2021, 279, 109862.
- Savvas, D. Automated Replenishment of Recycled Greenhouse Effluents with Individual Nutrients in Hydroponics by Means of Two Alternative Models. Biosyst. Eng. 2002, 83, 225–236.
- Voogt, W.; Sonneveld, C. Nutrient Management in Closed Growing Systems for Greenhouse Production. In Plant Production in Closed Ecosystems; Goto, E., Kurate, K., Hayashi, M., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1997; pp. 83–102.
- Magán, J.J.; Casas, E.; Gallardo, M.; Thompson, R.B.; Lorenzo, P. Uptake Concentrations of a Tomato Crop in Different Salinity Conditions. Acta Hortic. 2005, 697, 365–369.
- Neocleous, D.; Savvas, D. NaCl Accumulation and Macronutrient Uptake by a Melon Crop in a Closed Hydroponic System in Relation to Water Uptake. Agric. Water Manag. 2016, 165, 22–32.
- Neocleous, D.; Savvas, D. Simulating NaCl Accumulation in a Closed Hydroponic Crop of Zucchini: Impact on Macronutrient Uptake, Growth, Yield, and Photosynthesis. J. Plant. Nutr. Soil Sci. 2017, 180, 283–293.
- Ödemiş, B.; Bozkurt, S.; Ağca, N.; Yalçin, M. Quality of Shallow Groundwater and Drainage Water in Irrigated Agricultural Lands in a Mediterranean Coastal Region of Turkey. Environ. Monit. Assess. 2005, 115, 361–379.
- Sonneveld, C.; Van Der Burg, A.M.M. Sodium Chloride Salinity in Fruit Vegetable Crops in Soilless Culture. Neth. J. Agri. Sci. 1991, 39, 115–122.
- Rodríguez-Ortega, W.M.; Martínez, V.; Nieves, M.; Simón, I.; Lidón, V.; Fernandez-Zapata, J.C.; Martinez-Nicolas, J.J.; Cámara-Zapata, J.M.; García-Sánchez, F. Agricultural and Physiological Responses of Tomato Plants Grown in Different Soilless Culture Systems with Saline Water under Greenhouse Conditions. Sci. Rep. 2019, 9, 6733.
- Massa, D.; Incrocci, L.; Maggini, R.; Carmassi, G.; Campiotti, C.A.; Pardossi, A. Strategies to Decrease Water Drainage and Nitrate Emission from Soilless Cultures of Greenhouse Tomato. Agric. Water Manag. 2010, 97, 971–980.
- Katsoulas, N.; Savvas, D.; Kitta, E.; Bartzanas, T.; Kittas, C. Extension and Evaluation of a Model for Automatic Drainage Solution Management in Tomato Crops Grown in Semi-Closed Hydroponic Systems. Comput. Electron. Agric. 2015, 113, 61–71.
- van der Salm, C.; Voogt, W.; Beerling, E.; van Ruijven, J.; van Os, E. Minimising Emissions to Water Bodies from NW European Greenhouses; with Focus on Dutch Vegetable Cultivation. Agric. Water Manag. 2020, 242, 106398.
- Rouphael, Y.; Kyriacou, M.C. Enhancing Quality of Fresh Vegetables through Salinity Eustress and Biofortification Applications Facilitated by Soilless Cultivation. Front. Plant Sci. 2018, 9, 1254.
- Voogt, W.; Van Os, E.A. Strategies to Manage Chemical Water Quality Related Problems in Closed Hydroponic Systems. Acta Hortic. 2012, 927, 949–956.
More
Information
Subjects:
Horticulture
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
821
Revisions:
2 times
(View History)
Update Date:
08 Jan 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





