Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Rodolfo Ippoliti | -- | 5551 | 2023-12-30 10:40:53 | | | |
| 2 | Catherine Yang | Meta information modification | 5551 | 2024-01-02 02:24:03 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Di Leandro, L.; Colasante, M.; Pitari, G.; Ippoliti, R. Plant Toxins: Recombinant Production as Components of Immunotoxins. Encyclopedia. Available online: https://encyclopedia.pub/entry/53285 (accessed on 26 May 2026).
Di Leandro L, Colasante M, Pitari G, Ippoliti R. Plant Toxins: Recombinant Production as Components of Immunotoxins. Encyclopedia. Available at: https://encyclopedia.pub/entry/53285. Accessed May 26, 2026.
Di Leandro, Luana, Martina Colasante, Giuseppina Pitari, Rodolfo Ippoliti. "Plant Toxins: Recombinant Production as Components of Immunotoxins" Encyclopedia, https://encyclopedia.pub/entry/53285 (accessed May 26, 2026).
Di Leandro, L., Colasante, M., Pitari, G., & Ippoliti, R. (2023, December 30). Plant Toxins: Recombinant Production as Components of Immunotoxins. In Encyclopedia. https://encyclopedia.pub/entry/53285
Di Leandro, Luana, et al. "Plant Toxins: Recombinant Production as Components of Immunotoxins." Encyclopedia. Web. 30 December, 2023.
Copy Citation
Plant toxins that target eukaryotic 28S ribosomal RNA (Ribosome Inactivating Proteins, RIPs), such as the catalytic (A) chain of ricin (a dimeric A-B toxin from Ricinus communis) and saporin (a single chain A toxin from Saponaria officinalis), have found widespread use in the construction of immunotoxins. Both these toxins depurinate ribosomal 28S RNA at a specific site (GAGA loop) inhibiting the binding of elongation factor 2 and blocking protein synthesis. Their high toxicity towards eukaryotic cells has thus justified their use as components of chimeric toxins in cancer treatment. A brief description of recombinant RIPs production is given.
toxin
immunotoxins
plant toxin
recombinant production
1. Saporin SO6
One of the most widely used type I ribotoxins (RIPs) for the development of therapeutics is saporin, which is obtained from the seeds of the Saponaria officinalis plant [1]. In the production of immunotoxins or chimeric toxins, saporin-6 (SO6) was chosen for toxin synthesis among the variety of saporins that Saponaria officinalis expresses because, when compared to other type I ribotoxins [2], it is stable and resistant to several modifications, such as denaturation and proteolysis [3], and for the maintenance of its enzymatic activity after conjugation procedures [4].
Saporin-S6 (Figure 1) has a full length of 253 amino acids. The sequence was identified in 1990, and lysine residues account for about 10% of the total amino acids, which allows the protein to have an exceptionally high pI (around 10) [5].
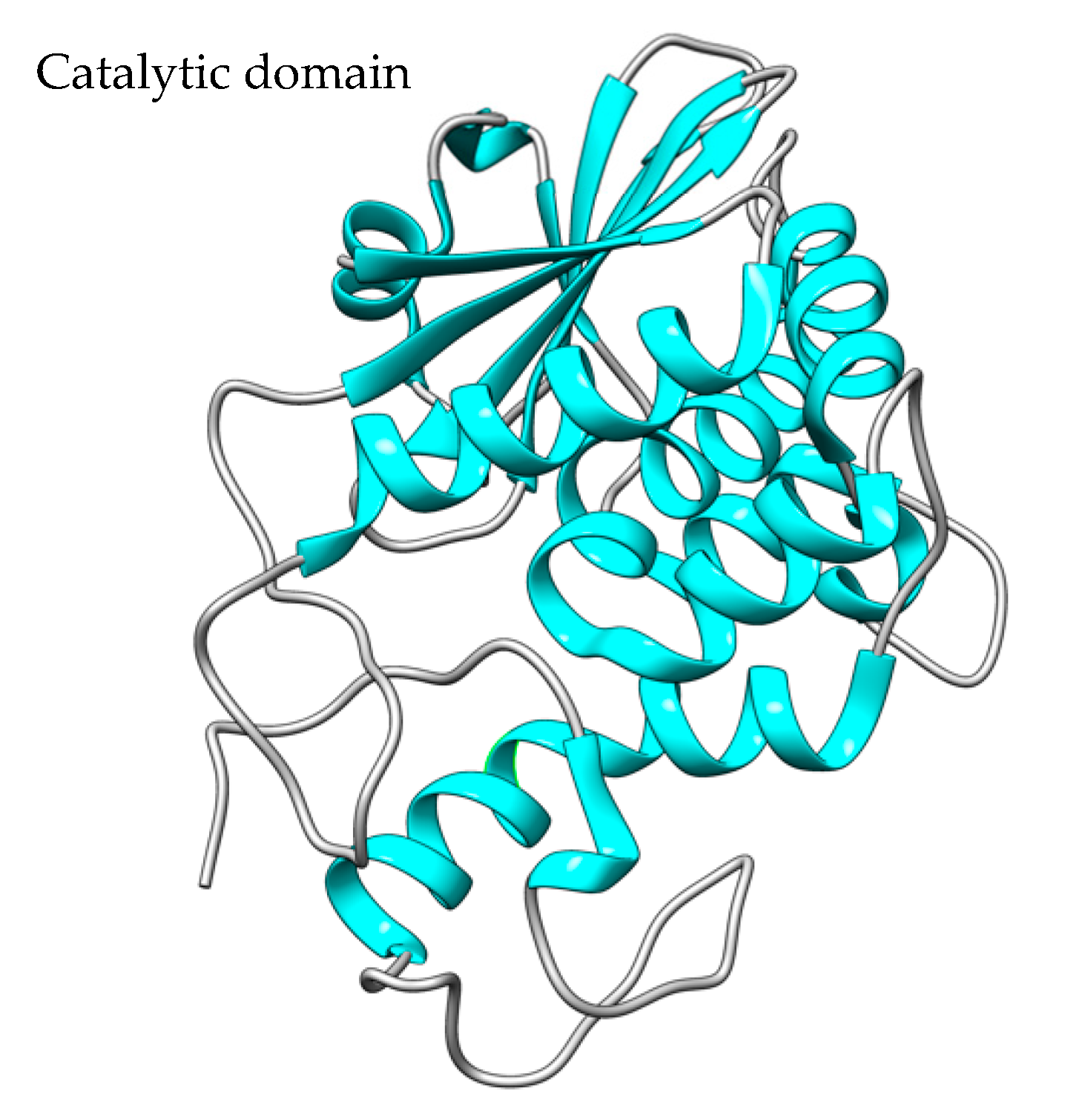
Figure 1. The crystal structure of saporin SO6 (PDB code: 1QI7). Saporin contains a single N-glycosidase catalytic A domain. The structure was created using the UCSF Chimera Software (1.17.3).
The saporin-S6 molecule contains no neutral sugars, notwithstanding the presence of glycosylation sites in the precursor pro-saporin C-terminal sequence, which is removed to produce the mature protein. Saporin and all plant RIPs have N-glycosidase activity (EC 3.2.2.22), which removes an adenine (A4324 in the 28S ribosomal rRNA) from the sarcin/ricin loop, a universally conserved GAGA-tetra loop [6]. This depurination process permanently deactivates the major ribosomal subunit by preventing the recognition and binding of the elongation factor EF-2 and influencing both EF1 and EF2 GTPase activity. This prevents tRNA translocation from the A to P sites, irreversibly stopping protein synthesis [7][8][9]. Saporin-S6 has demonstrated harmful effects in both in vivo studies using animal models and in vitro experiments involving various cell lines.
Saporin-S6 has an LD50 of 4.0 mg of RIP/Kg of body weight in mice. It can activate apoptosis (both caspase-dependent and -independent apoptosis), oxidative stress, autophagy, necroptosis, and protein synthesis inhibition once it enters the cytosol, ER, or nucleus. When full-length antibodies are used, the presence of the antibody itself can trigger cell death through apoptosis, complement-dependent cytotoxicity (CDC), or antibody-dependent cellular cytotoxicity (ADCC) in saporin-based ITs [10].
Type 1 RIPs have low inherent toxicity and lack a cellular entrance mechanism. Type 1 RIPs become highly toxic when linked to a cell recognition and entrance element, and they have shown significant action against hematological and solid malignancies [11][12][13][14][15][16][17][18][19][20]. Saporin S6 constructs have been shown to go straight from endosomes to the cytoplasm, whereas ricin conjugates have been shown to move to the Golgi from endosomes, then to the endoplasmic reticulum, and finally to the cytosol [9].
In the literature, there are a lot of works that describe the design and construction of RIP-based immunotoxins for treating cancer, HIV, and other infectious diseases [10][21][22][23][24], including saporin. Classical immunotoxins were first produced via chemical cross-linking [4], but the need for more homogeneous and reproducible products has led to the study of recombinant forms.
The effective production and appropriate folding in a host cell is an important feature for the development of therapeutic drugs for saporin chimeras (as well as that of plant RIPs chimeric toxins). To express plant protein toxins, particularly saporin or Type I RIPs recombinant fusion chimeras, a eukaryotic expression system would thus be desirable to drive their expression as secretory proteins so that correct folding with the cellular quality control system would be easiest in the endoplasmic reticulum (ER) microenvironment. These expression systems, however, face important problems mainly due to ER stress and the intrinsic toxicities of plant toxins to eukaryotic ribosomes.
Saporin Expression Hosts
-
Saporin Bacterial expression systems
While Ricinus communis [25][26], Mirabilis jalapa [27], Phytolacca americana [28], Dianthus leaves [29], Trichosanthes kirilowii [30], Maize [31], and Gelonium multiflorum [32] RIPs have been cloned and expressed in E. coli, saporin expression in bacteria is more difficult because of the direct action on the host ribosomes that may cause autointoxication; in contrast to type II RIPs, type I RIPs, and particularly saporin, are active against both eukaryotic and prokaryotic ribosomal RNA [33].
Additionally, although several E. coli vector designs for the expression of saporins were tried, they all showed severe toxicity in E. coli non-expression strains during plasmid maintenance and strain selection. Toxin expression might be finely controlled using the E. coli strain BL21 (DE3) pLysS, but it is not possible to prevent endotoxin contamination and the expression of toxins in insoluble form [3].
In a 1993 study, the group of Barthelemy described the amplification of genomic DNA from the leaves of S. officinalis and the expression in E. coli of a PCR-amplified genomic clone of saporin S3 and S6 [34]. The saporin coding sequence was inserted into the periplasmic secretion vector PIN-IIIompA2, resulting in a gene fusion between the mature saporin coding sequence and the plasmid ompA leader peptide segment to direct membrane translocation in E. coli and assist proper protein folding of the expressed protein. Most of the saporin expressed showed similar protein synthesis inhibition activity to the native saporin but remained inside the cells, and approximately only 10% was found in the periplasm [34].
Fabbrini et al. expressed for the first time in E. coli five isoforms of saporin: SAP1, SAP3, SAP4, SAP6, and SAP-C [35]. For the expression of recombinant proteins, the authors used the BL21 (DE3) pLysS strain and the pET11d plasmid to engineer the saporin genes. The saporin-coding leaf cDNA [36] has been modified to include a stop codon before the transcribed C-terminal propeptide [37]. After that, the resulting construct, pET-11d-SAP-C, was completely sequenced to ensure that no modifications were introduced during the amplification stage. The protein expression in the soluble fraction has been possible with the use of a tightly regulated host/vector inducible system; there was no evidence of leaky expression of saporin genes prior to T7 RNA polymerase activation, and yields of soluble recombinant seed-type saporin isoforms were between 1 and 3 mg/liter of culture [35].
The substitution of Glu 176 with Lys and the deletion of 19 amino acids at the C-terminal of the saporin sequence have been used to express two saporin mutants in E. coli to reduce its toxicity to bacterial cells and affect its enzymatic activity on polynucleotide substrates. Bacterial cultures carrying wild-type recombinant saporin (pET-Sap) slowed to a halt, whereas cultures having pET-E176K and pET-K234stop mutant variants grew normally and at the same rate as control cultures, but the majority of recombinant saporin mutants appeared as inclusion bodies [38].
Günhan et al., in 2008, described the expression and purification of saporin derivatives in E. coli BL21(DE3)pLysS cells, a strain used for the expression of toxic proteins because it encodes T7 lysozyme, which suppresses the basal expression of toxic target proteins prior to induction. The authors introduced a mutation at the C-terminus (Ser255Cys) of the gene to permit the conjugation reaction of a heterobifunctional crosslinking agent to antibodies, cytokines, peptides, and the construction of conjugates. The Cys255-Sap was efficiently recovered from the soluble fraction (12.5 mg/120 mg total proteins), reaching 95% purity and a yield of 2.7 mg/L after the size-exclusion chromatography step [39]. As previously indicated, Sap S6 shows great resistance to chemical modification, denaturation, and proteolysis, so it is a very good candidate for the construction of conjugates for therapeutic purposes.
Giansanti et al. have developed, generated, and characterized a RIP saporin (SapVSAV) engineered form with an additional C-terminal sequence (SEVSAV) that is recognized using the PDZ2 domain of mouse protein tyrosine phosphatase [40]. The co-expression of PDZ2 and the mutated saporin gene boosts toxin production in E. coli BL21 strain cells and gains an expression level similar to what is found in the PlysS-protected system. The authors accomplished co-expression using pET28 and pET11 vectors but also achieved sustained co-expression using a bi-cistronic pETDuet plasmid containing both the two gene sequences. The induction of SapVSAV expression did not affect bacterial growth, maybe because it was produced in an inactive form stabilized through the PDZ2 interaction, but when SapVSAV was evaluated in cellular systems (Daudi or U937 cells), the authors found that it had cytotoxic activity comparable to wt saporin, implying a putative activation mechanism induced in mammalian cells.
The same group in 2015 produced recombinant chimeric toxins composed of the PDZ-hCASK (serine kinase calcium/calmodulin-dependent of the MAGUK family binding to CD98) sequence coupled to saporin S6 as monovalent (hCASK-SAP) or bivalent (hCASK(2SAP) toxins and tested their toxicity towards human glioblastoma cell lines (GL15 and U87) [41]. The synthetic genes hCASK and SAP S6 were fused, cloned, and expressed in Rosetta GamiTM B pLysS(DE3) E. coli strains, which combine enhanced disulfide bond formation with increased expression of eukaryotic proteins containing rare E. coli codons. The expression yields of both chimeras were not as efficient as those of the hCASK domain alone, but they showed higher toxicity on two glioblastoma cell lines (U87 and GL15), especially in the presence of saponin as a “coadiuvant” for the endo/lysosomal compartment escape of the toxins [42].
Recently, a new saporin conjugate production has been described in the literature. The saporin gene has been genetically fused to the ACDCRGDCFCG peptide (RGD-4C), an αv-integrin ligand, and expressed in E. coli with a histidine tag at the C-terminus to promote endosomal escape. This conjugate was tested in in vivo studies in different orthotopic mouse models of bladder cancer and was able to reduce tumor growth and significantly prolong animal survival. The RGD-SAP was easily recovered from the bacterial soluble fraction as a monomer with no need for renaturation steps [43].
-
Saporin Yeasts expression systems
Although the E. coli strain BL21 (DE3) pLysS could be used to regulate RIP expression more tightly, their high toxicity and the processing of inclusion bodies remain problematic, and recombinant saporin-IT production has been extensively described in yeasts [44]. Yeasts exhibit diminished nutritional needs in comparison to insect and mammalian cell lines and furthermore combine the simplicity of a unicellular organism with the capacity to carry out most post-translational modifications needed for a physiologically active recombinant protein. P. pastoris is an obligate aerobic yeast that can obtain carbon from methanol. This last feature enabled the construction of an expression system exploiting the methanol-inducible AOX1 promoter. When compared to S. cerevisiae, P. pastoris is recognized for producing a higher number of recombinants because it is Crabtree-negative, avoiding loss of carbon through the production of ethanol under respiratory circumstances, resulting in more biomass creation and, subsequently, more recombinant protein [45].
In the aim of resolving some important issues of the first-generation RIP-based ITs and imposing several advantages, such as a defined toxin-ligand interface and the possibility to genetically edit mutations in the recombinant toxin to boost potency and reduce nonspecific toxicity [46][47], recombinant ITs were created using single-chain variable fragments (scFvs) as the carrier moiety, later replaced by disulfide-stabilized Fvs (dsFvs) [48][49].
To achieve optimal expression of saporin and relative fusion chimeras in P. pastoris, an important factor that must be considered is the use of optimization of the coding sequence on the basis of yeast codon usage. Saporin S6 was initially expressed by the Fabbrini group, both as a standalone toxin and as part of a fusion immunotoxin in P. pastoris [50], and then the same group produced a chimera consisting of the ATF of human uPA fused to the saporin S3 isoform that specifically kills uPAR (urokinase plasminogen activator receptor) over-expressing cancer cells [51].
In an interesting paper from 2015, Della Cristina et al. [52] showed the construction and recombinant production of two various constructs containing the identical recombinant anti-CD22 single-chain variable fragment (scFv) fused to two different toxin domains: PE40 or saporin. Both immunotoxins were expressed in E. coli and in P. pastoris. On a small scale, the PE40 chimeras expressed in E. coli accumulated in inclusion bodies, and no measurable recombinant immunotoxin (rIT) could be retrieved in a soluble form, either within the cytoplasmic or periplasmic spaces. The renaturation of inclusion bodies in a larger culture volume showed a recovery of about 3 mg/L of the immunotoxin, corresponding to 80% of the total expressed protein [2]. In the case of the saporin domain containing rIT, the authors found a lower level of immunotoxin production than that observed for the rIT containing PE40 following IPTG induction in E. coli, not due to host auto-intoxication effects. Even though this saporin chimera could be recovered from inclusion bodies at a concentration of 4 mg/L, most (>90%) of the toxin was lost during the renaturation process as a result of aggregation and precipitation events. Having obtained low- and non-functional quantities of this saporin-based IT in bacteria, the authors decided to design some constructs suitable for P. pastoris expression, fusing the sequences coding for the anti-CD22 VH and VL domains to a saporin yeast-optimized for codon usage, either engineered to have an N- or C-terminal His-tag. Codon optimization has previously been demonstrated to significantly reduce the toxicity challenges related to saporin expression in P. pastoris, as well as to be required for producing clones that express high levels of active recombinant saporin [44]. In particular, it has been shown that misfolding may occur when saporin is fused to an “unfavorable” domain, such as the scFv, resulting in increased host toxicity and lowering expression levels. Codon-usage optimization could counteract such an effect, improving the expression yield in Pichia even in the presence of the scFv domain. The estimated secretion yields of secreting colonies were about 1–2 mg/L, and the proteins have full cytotoxic activity, suggesting that P. pastoris is a better host for saporin-based rITs than E. coli. Codon usage optimization is a strategy also used for the design and production of the ATF-SAP chimera in the fermentation process [53]. Between the 14 (fully optimized ATF-SAP) and 15 (only optimized SAP) GS115 clones producing ATF-SAP, the expression levels vary from 1 to 5 mg/L for the partially optimized chimera and from 3 to 7 mg/L for the fully optimized chimera. A feeding strategy based on the fed-batch slow addition of methanol and on the oxygen transfer rate increase allows the yeast cells to adapt smoothly to methanol after induction, so the concentration of the secreted recombinant ATF-SAP reached approximately 6 mg/L, demonstrating the tolerance of this strain towards SAP. Further advancements would be achievable by improving pre-induction biomass formation, as is possible in industrial-scale bioreactors.
-
Saporin expression in Tobacco protoplasts
The use of plant tobacco protoplasts has been investigated as an alternative approach for producing recombinant plant RIPs. However, it was discovered that the saporin precursor is extremely harmful to this expression system [54]. Interestingly, tobacco protoplasts were able to express the native preproricin construct, and normal processing, glycosylation, and targeting of the vacuole took place with no harmful effects found [55]. In contrast, when an orphan secretory ricin toxic A chain (RTA) polypeptide was expressed, RTA was retro-translocated to the cytosol, and protein synthesis was thereafter inhibited [55]. This further suggests that RIP toxicity may have negative effects on the tobacco protoplast expression system. The purified toxin showed protein translation inhibitory activity similar to the native one only when the expressed precursor contained the C-terminal propeptide [56], suggesting a fundamental role of the latter in the segregation of this RIP. It was demonstrated in other RIPs, pre-pro-trichosanthin, that proper processing during the expression of transgenic tobacco plants was dependent on the presence of the two peptides:
-
Gene therapy with saporin gene
Suicide saporin-gene therapy has been studied on various cell lines (B16, Hela, U87, and MDA-MB-435), using the toxin gene carried in plasmids and expressed under the control of cytomegalovirus (CMV) or simian virus 40 (SV40) promoters [57]. B16 melanoma cells transfected with the pCI-SAP plasmid showed a dramatic reduction in growth of about 70% with respect to controls, and also in B16 melanoma-bearing mice, direct intra-tumoral injection of pCI-SAP resulted in a significant reduction in tumor growth [58]. Co-transfection treatment of HeLa, U87, 9L, and MDA-MB-435 cells with two distinct mammalian gWIZ plasmids, pGEL (gWIZgelonin) and pSAP (gWIZ-saporin), demonstrated strong cytotoxicity on all the cell lines tested using a DNA concentration of only 2 µg/mL. However, due to the non-selective DNA delivery system, the toxin expression caused cytotoxicity in both malignant and non-cancer cells [59]. Some examples of selective delivery systems have been described more recently in the literature. The antitumoral activity of a plasmid harboring the saporin gene bound to lipid-protamine DNA nanoparticles coated with a peptide directed against human urokinase (U11) was established in mouse models [60]. With a similar strategy, di Leandro L. et al. tested an aptamer-mediated (AS1411 targeting surface nucleolin) saporin gene delivery system against U87 glioblastoma cells. A 50% reduction in U87 viability was achieved using an average saporin DNA concentration of 24–30 μg/mL, demonstrating selective cytotoxicity on glioblastoma U87 cells without toxic effect in 3T3 control cells [61].
2. Ricin A Chain (RTA)
Ricin, a type II RIP mainly purified from the seeds of Ricinus communis, is made up of two different polypeptides (the A and B chains) held together by a disulfide bond. The B chain is a galactose-binding lectin that ricin uses to bind to cell membranes, whereas the A chain is a N-glycosidase (EC 3.2.2.22) that, once delivered to the cytoplasm via retrograde transport through the Golgi and ER, kills the cell by catalytically inactivating the 60 S ribosomal subunits [62][63].
Numerous scientists have attempted to use ricin’s high cytotoxicity to kill cancerous cells for medical needs. Despite having extremely effective cell-killing capabilities, ricin is not selective for cell targets. The prospect of coupling ricin to carriers specialized for targets on undesirable cells has been extensively investigated in an effort to improve selectivity. In a cell culture test system, the ricin A chain (RTA) shows less than 0.01% toxicity of the naturally occurring protein, has no effect on both non-infected and TMV-infected tobacco protoplasts, and is unable to enter the cell without the help of the B chain [64]. But due to its great potency in blocking protein synthesis, the A chain (RTA) is extensively used to build cytotoxic conjugates effective against tumor cells as immunotoxins and fusion proteins [65][66][67].
There are many studies in which immunotoxins and RTA-based chimeras were proposed and designed against various cancer conditions, in particular against refractory hematologic malignancies [68][69]. A recombinant anti-IL-2R IT carrying deglycosylated dgRTA has obtained approval from the US Food and Drug Administration for the treatment in adults of cutaneous T-cell lymphoma [69], and several others are in clinical trials (Combotox, 3A1-dgRTA, IgG-HD37-dgA, Xomozyme-791, RTF-5-dgA, 260F9-rA, H65-RTA) [70][71][72]. For a more exhaustive view of the clinical evaluations of ricin-based immunotoxins, see also the review of de Virgilio M. et al. [73].
Native RTA must be effectively separated from the ricin B chain (RB), which binds to structures on the cell surface that contain galactose and facilitates the entry of RA into the cytoplasm, in order to be purified from the entire toxin [47]. One issue with employing A chains produced from fully native ricin is that laborious and extensive methods are required to remove all the contaminating B chains, procedures that are required to avoid non-specific toxicity [74]. A second issue is that the ricin A chain is N-glycosylated and must be deglycosylated to prevent immunotoxins from being cleared quickly in vivo by liver cells that carry mannose receptors. To prevent even minute levels of contaminating toxin or RB that can hide the hybrid toxin’s target selectivity and raise overall toxicity, preparations of RA and its conjugates must be closely regulated. Additionally, dealing with a lot of ricin and castor beans puts your health at risk [74].
To circumvent these issues and to more affordably obtain large quantities of RTA for therapeutic development, genetic methods are used to express RTA or deglycosylated RTA (dgRTA) genes in E. coli, as reported below.
To date, recombinant RTA and dgRTA have been used to obtain immunoconjugates in which the antibody is chemically coupled to the toxin [67].
Moreover, in the literature, few examples of RTA immunotoxins produced by genetic fusions in E. coli are reported [75][76]; instead, there are several examples of RTA chimeras that have been successfully expressed and tested for anticancer activities.
RTA Expression Hosts
-
RTA Bacterial expression systems
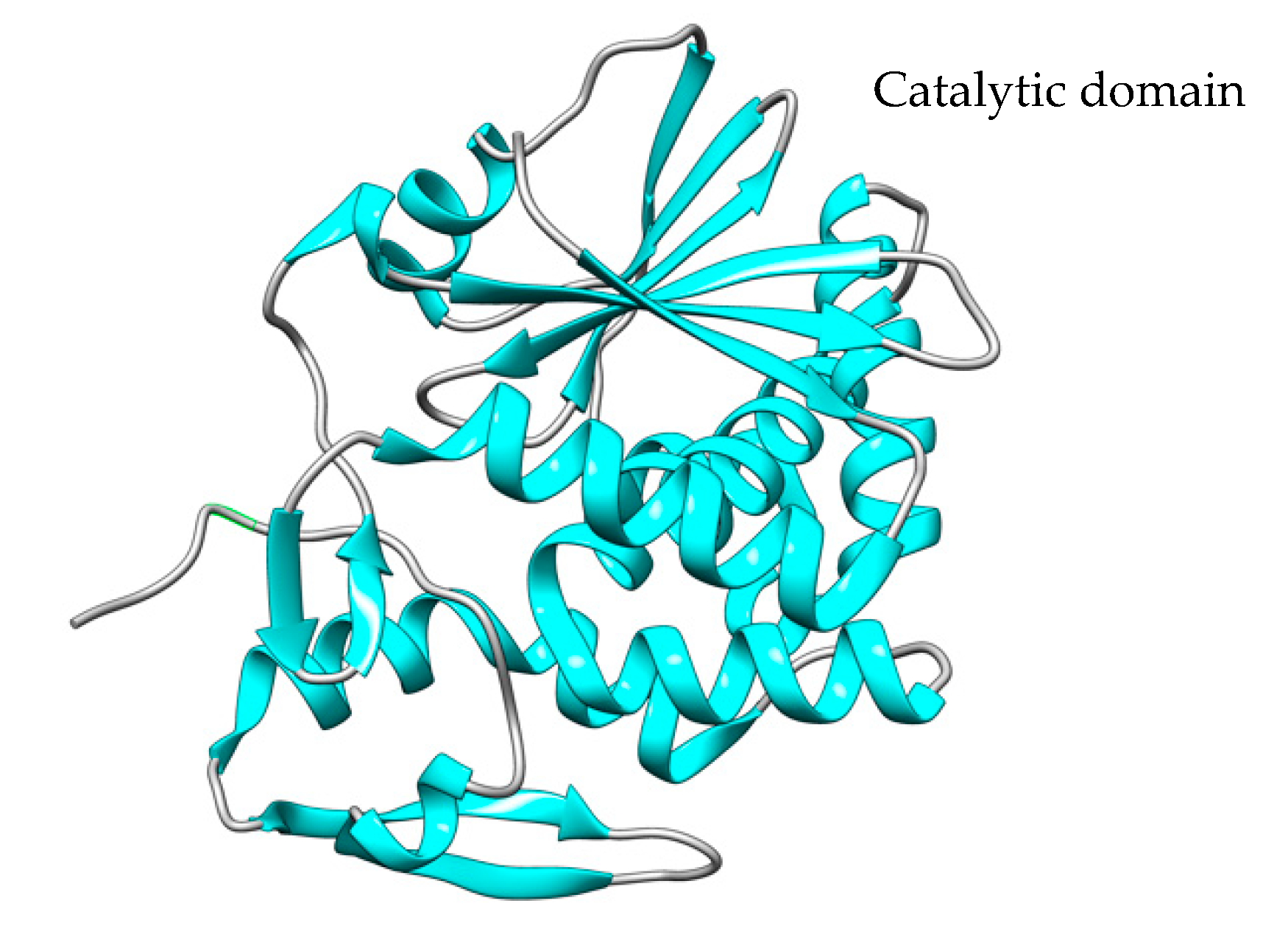
Figure 2. The crystal structure of the A chain (PDB code: 4Q2V). The catalytic domain is represented in blue. The structure was created using the UCSF Chimera Software (1.17.3).
The ricin A chain was expressed for the first time in E. coli in 1987 [78]. In the same year, it was also described as the construction of the first recombinant immunotoxin based on RTA: in a nude mouse model of human ovarian cancer, the growth of OVCAR-3 tumors was shown to be inhibited by an immunotoxin made up of an antibody to the human transferrin receptor (454A12) and the ricin A chain (RTA) [79]. Similarly, one of the first examples of recombinant chimeras containing RTA was reported in a 1994 study in which the expression in E. coli of a chimeric toxin prepared via genetic fusion of RTA and DTA produced a partially soluble toxin; in fact, only 20% of RTA-DTA was found in the lysate supernatant, while most were insoluble. The chimera was then tested for cytotoxic activity against human ovarian cancer cells, OVCA433 [80]. To enhance the cytotoxicity of recombinant RTA, several methods have been optimized, such as the addition of a TGN retention signal YQRL to the C-terminus of RTA [81] or an endoplasmic reticulum retention sequence KDEL [82][83]. Furthermore, in cases of some RTA-based chimeric toxins, a protease-sensitive cleavage site has been inserted between the RTA sequence and the other protein sequences [84][85].
Recombinant immunotoxins are chimeric proteins made of a single-chain antibody fragment (scFv) and a shortened, binding-deficient, catalytically active toxin. These fusion gene products are more readily modifiable, more readily produced, and more homogeneous than chemical conjugates [86]. Single-chain immunotoxins produced by bacteria vary greatly in terms of stability, and some have shown a marked propensity for aggregation [87]. To increase solubility, different strategies can be used, some involving modification of the target (as the use of tRNA complementation plasmids and stabilization of mRNA), others involving modification of the growth conditions (pH, temperature, media, addition of molecular chaperones, etc.) [88].
To develop novel anticancer Ribosome Inactivating Toxin-Affibody Fusions (RTA RITs), Park et al. genetically combined an affibody coding sequence directed either to HER2 (ZHER2:342; HER2Afb) or EGFR (ZEGFR:1907; EGFRAfb) with the RTA N-terminus (residues 36–302). This fusion was engineered to carry the KDEL signal peptide and expressed in E. coli as HER2Afb-RTA-KDEL and EGFRAfb-RTA-KDEL [76]. To generate both HER2Afb-RTA and EGFRAfb-RTA, the RTA gene was cloned into a pETDuet-1 vector carrying either the HER2Afb or EGFRAfb gene, adding a C-terminal histidine tag for easier purification. Each of the recombinant protein-encoding plasmids was separately introduced into the BL21 (DE3) strain of E. coli, and the recombinant proteins were then overexpressed via induction with IPTG at 18 °C overnight. In this way, the proteins were recovered from the soluble fraction of the cell extract.
A new immunotoxin (Figure 3) has been newly developed by combining the single-chain variable fragment (scFv) obtained from panitumumab. This scFv comprises the VH and VL regions and is fused with the catalytic domain of ricin (RTA). The new construct sequence was optimized for expression in E. coli strain BL21 (DE3) and inserted into the pET32a (+) expression vector. The fusion protein has been refolded through dialysis from inclusion bodies and purified at a concentration of about 0.18 mg/mL. This immunotoxin is designed against the Epidermal Growth Factor Receptor (EGFR)-induced cytotoxicity and apoptosis in HCT-116 and MDA-MB-468 cells [75].
Figure 3. Schematic representation of the EGFR-specific recombinant ricin-panitumumab (scFv) immunotoxin. The figure has been adapted from [75].
In an effort to target infected cells and recognize viral components specifically, fusion and hybrid proteins of RTA and PAPs have also been produced [89][90]. A fusion protein between ricin A chain (RTA) and Pokeweed antiviral protein (PAP) isoform S1 (from the seeds of Phytolacca americana) was produced in an E. coli expression system and assayed for its anti-HBV inhibitory function and cytotoxic effect in the chronically infected cell line AD38 [91]. The chimeric toxin was purified from inclusion bodies with a yield of about 200 mg/L of culture with 90% purity. To improve its production in E. coli, the authors also produced an RTA mutant-Pokeweed antiviral protein isoform 1 from the leaves of Phytolacca americana (RTAM-PAP1). Two specific point mutations were incorporated into the RTA moiety as well as in the flexible linker to substitute the cysteine (Cys) residues with alanine residues. This modification was implemented to prevent the unwanted formation of disulfide bonds at positions 171 and 259 entirely (C171A and C259A). Additionally, to reduce any potential impact on its structure and function, a 6-His tag was added at the N-terminal of the RTAM-PAP1 protein. These modifications really made a difference in solubility and activity: the fusion proteins RTAM-PAP1 were produced exclusively with great solubility (a few were found in inclusion bodies), and using a three-step purification approach, soluble proteins with >90% homogeneity were obtained. Nevertheless, from 1 L of culture, 0.1 mg of protein with >95% purity and 0.22 mg of protein with >90% purity were recovered, but at the same time, the anti-HBV bioactivity of RTAM-PAP1 was increased with respect to RTA-PAP1 protein.
As previously discussed, the potential toxicity of RIPs towards the host cells may prevent or decrease recombinant protein production and their use as such or included in chimeric proteins or in ITs, but since RTA does not exhibit toxicity towards prokaryotic ribosomes, it could be easily produced in E. coli with high yields and with minimal difficulty. Consideration should be given to how this catalytic domain is released into the cytoplasm. Chimeric toxins containing retinoic acid (RA) necessitate intracellular proteolytic cleavage for the liberation of the RA component, enabling them to exhibit cytotoxic effects on target cells. If the recombinant chimeric toxin undergoes extracellular cleavage, it fails to selectively target cells in a specific manner [92].
In order to enable appropriate and distinct folding of both domains of the chimeric toxin, this issue may be addressed by inserting a flexible peptide linker between the targeting domain and the toxic moiety. The 218 linker GSTSGSGKPGSGEGSTKG and the G4S peptide linker are used to improve the chimeric proteins’ resistance to intracellular proteases and even further decrease scFv (antibody single chain variable fragment) aggregation when expressed in bacterial systems [52][93].
3. Other Plant Toxins
It is worth noting that besides the ricin A chain and saporin, widely studied RIPs, a couple of examples should be mentioned for the recombinant expression of toxins from plants.
3.1. Bouganin
In the last few years, the single-chain ribosome-inactivating protein bouganin (from Bougainvillea spectabilis Willd.) has attracted renewed interest due to the possibility of engineering its amino acid sequence to decrease the immunogenic properties of these toxins, thus increasing its attractivity as a component of immunotoxins [94]. Bouganin is a type 1 ribosome-inactivating protein purified from the leaf extracts of Bougainvillea spectabilis Willd [95] as a 26 kDa single-chain protein. Bouganin exhibits characteristics typical of type 1 ribosome-inactivating proteins (RIPs), such as N-glycosylase activity and antiviral properties; it blocks protein synthesis in a cell-free system (with an IC50 of 10 ng/mL), but its efficacy on whole cells is comparatively lower than other type 1 RIPs. It was noted that the concentrations of bouganin needed to inhibit protein synthesis in human cell lines were significantly higher than those required in a rabbit reticulocyte lysate assay and did not exhibit toxicity in mice at the highest tested dose of 32 mg/kg [96]. Moreover, bouganin possesses distinctive characteristics, including a higher activity on DNA compared to ribosomal RNA, low systemic toxicity, and immunological properties that differ significantly from other RIPs. The limited non-specific toxicity of this protein to animals, when compared to all other identified and characterized toxins, enhances the therapeutic possibilities associated with bouganin; in particular, it represents a highly appealing tool for incorporation into immunotoxins and fusion chimeras [94].
-
Bacterial expression systems
The first report of recombinant bouganin expression in E. coli was described in 2002 by den Hartog and co-workers [97]. The authors synthesized bouganin cDNA from total RNA isolated from the leaves of B. spectabilis Willd, and the full-length bouganin gene was directly amplified with PCR. The PCR product was cloned into a PG212 plasmid containing the pElB leader peptide for periplasmic expression, and the E. coli strain BL21-CodonPlus(DE3) was used as the host. Despite the presence of the pELB leader peptide, the majority of the recombinant protein was found in inclusion bodies, and only a small portion present in the periplasmic extract was purified through affinity chromatography. The most interesting aspect of this work was that the activity of recombinant bouganin was comparable to that of the native protein in living cells. This suggested that the recombinant production of bouganin does not alter its inherent difficulty in binding to cells, as evidenced by the high IC50 values shown in living cells compared to the cell-free assay. Bouganin’s 3D structure was solved with X-ray crystallography (Figure 4) and revealed a conserved structure typical of this class of toxins [98]. Interestingly, bouganin possesses two cysteine groups that can be employed to create a disulfide bond directly with an activated antibody thiol group through a disulfide exchange reaction. This method simplifies the generation of immunotoxin molecules with bouganin, suitable for use in novel therapeutic strategies.
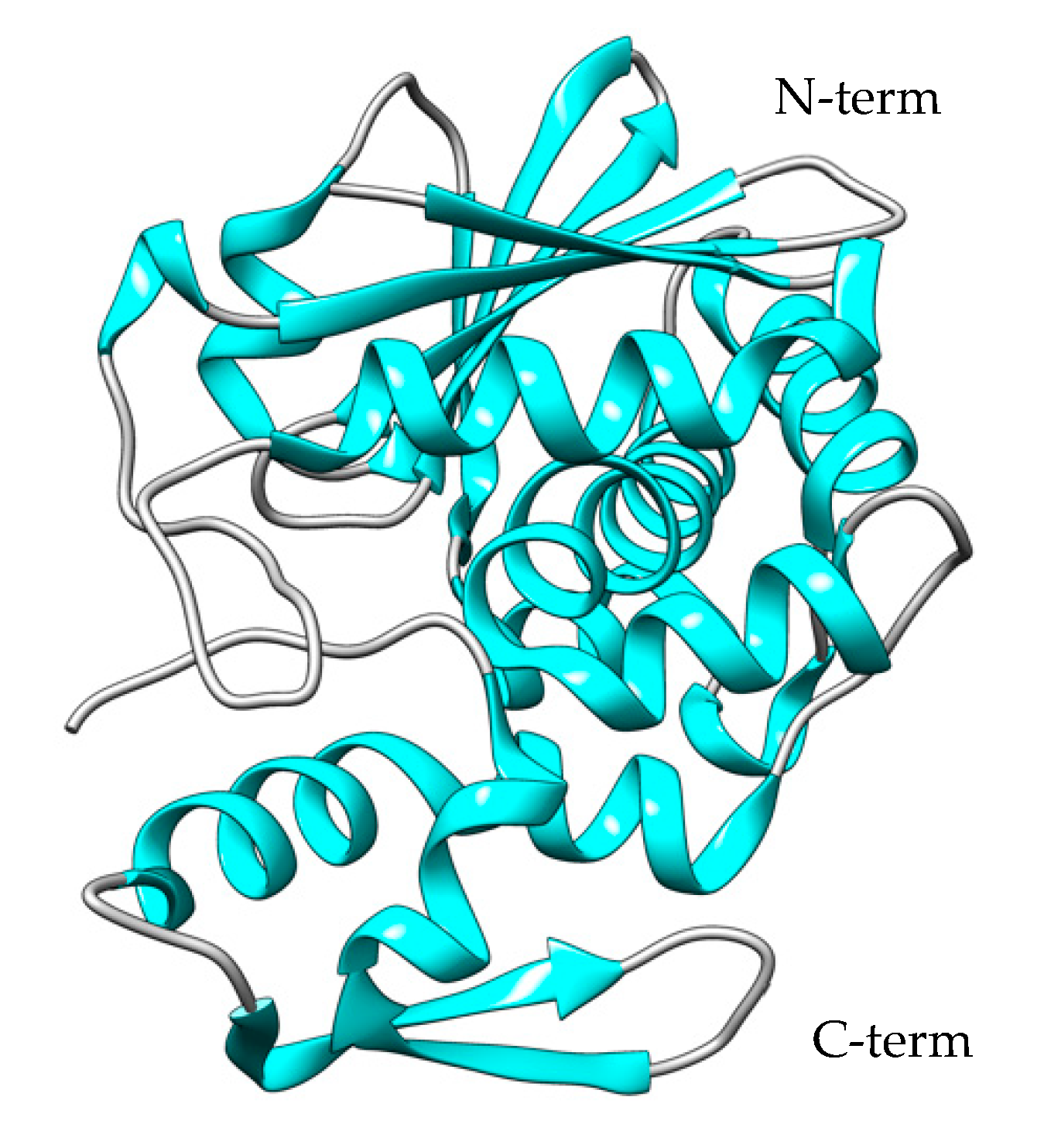
Figure 4. The crystal structure of bouganin (PDB code: 3CTK). The N-terminal domain consists of a mixed β-sheet of seven filaments (β1–β9). The C-terminal domain consists mainly of eight α-helices. The structure was created using the UCSF Chimera Software (1.17.3).
On the basis of knowledge of the structural features of bouganin, it was possible to change some specific amino acid residues to impact the immunological properties of the toxin without affecting its catalytic activity [99]. Cizeau et al. mutated the bouganin gene to remove the T-cell epitopes to create a T-cell epitope-depleted variant of bouganin, named de-bouganin. By genetically combining de-bouganin with an anti-epithelial cell adhesion molecule (EpCAM) Fab moiety, the VB6-845 immunotoxin was obtained.
To test the most effective antibody–de-bouganin orientation, multiple versions of dicistronic expression units were created, expressed, and evaluated for potency. In all instances, the dicistronic unit was inserted into the pING3302 vector, regulated with the arabinose-inducible araBAD promoter, and introduced into the E104 E. coli strain. Upon induction, the PelB leader sequence facilitated the secretion of the Fab–de-bouganin fusion protein into the periplasmic space. The conjugate biological activity was tested against cell lines expressing EpCAM, CAL-27, and OVCAR-3; the immunotoxin displayed higher potency compared to numerous widely used chemotherapeutic agents. In vivo, effectiveness was validated through an EpCAM-positive human tumor xenograft model in severe combined immunodeficiency (SCID) mice, with the majority of treated mice remaining tumor-free [99].
Some other recombinant de-bouganin-based ITs have been designed and expressed in E. coli. The immunotoxin Trastuzumab (anti-HER2 mAb)–bouganin was used as a complement to the treatment of mammary gland ductal carcinoma cells resistant to treatment with maytansinoids [100]. An anti-HER2 C6.5 diabody–de-bouganin/de-bouganin–C6.5 diabody fusion protein was expressed and purified from E. coli with a yield of about 0.15–0.5 mg/L and then characterized for their activity on HCC1419 and BT-474 cells, where the fusion-carrying bouganin at the N-terminus proved to be more efficient in killing cells [101]. Recently, a phase I trial has been initiated using an antiEPCAM/de-bouganin fusion for the treatment of epithelial tumors [102]. The absence of immune reactivity to bouganin in patients underscores the effectiveness of the T cell epitope-depletion strategy in mitigating the immune response and confirms the feasibility of utilizing bouganin in therapeutic strategies.
3.2. Pulchellin
Pulchellin is a ricin-like lectin (type II RIP) obtained from Abrus pulchellus. Pulchellin consists of a RIP A chain and a B chain linked by a disulfide bond [103]. The B chain, serving as a non-toxic carbohydrate-binding component, shows a crucial role in facilitating the endocytosis of the A chain and could be utilized as a mechanism for drug delivery. The catalytic residues within the pulchellin A chain are positioned identically to those found in the Ricin and Abrin A chains. Pulchellin exists in four isoforms, with isoform II being the most potent, exhibiting an LD50 toxicity of 15 µg/kg in mice [104].
The DNA fragment that encodes pulchellin A chain was cloned and introduced into the pGEX-5X plasmid for the expression of recombinant pulchellin A chain (rPAC) in Escherichia coli [103], with a good final yield after purification of about 3 mg/L.
The recombinant pulchellin A chain (rPAC) contains a single free cysteine situated in the C-terminal region, allowing it to readily engage in a disulfide-exchange reaction with an activated antibody thiol group. This characteristic facilitates the easy production of rPAC in a heterologous system. Consequently, rPAC holds promise for the development of immunoconjugates with significant potential as chemotherapeutic agents. The first example of using pulchellin in targeted therapy is described by the work of Sadraeian et al., in which the authors showed the expression and purification of immunotoxins made by chemically linking the toxin to antibodies directed against gp120 and gp41 expressed on the surface of HIV-envelope-producing cells [105]. The recombinant toxins were produced in E. coli Rosetta (DE3) and conjugated to HIV MAbs 924 and 7B2 with the single-free cysteine on the A chain toxin. The recombinant pulchellin was internalized by these cells, suggesting its possible use as a therapeutic agent against HIV-infected cells.
References
- Lappi, D.A.; Esch, F.S.; Barbieri, L.; Stirpe, F.; Soria, M. Characterization of a Saponaria officinalis seed ribosome-inactivating protein: Immunoreactivity and sequence homologies. Biochem. Biophys. Res. Commun. 1985, 129, 934–942.
- Pizzo, E.; Di Maro, A. A new age for biomedical applications of Ribosome Inactivating Proteins (RIPs): From bioconjugate to nanoconstructs. J. Biomed. Sci. 2016, 23, 54.
- Santanché, S.; Bellelli, A.; Brunori, M. The Unusual Stability of Saporin, a Candidate for the Synthesis of Immunotoxins. Biochem. Biophys. Res. Commun. 1997, 234, 129–132.
- Bolognesi, A.; Tazzari, P.L.; Tassi, C.; Gromo, G.; Gobbi, M.; Stirpe, F. A comparison of anti-lymphocyte immunotoxins containing different ribosoma-inactivating proteins and antibodies. Clin. Exp. Immunol. 1992, 89, 341–346.
- Maras, B.; Ippoliti, R.; De Luca, E.; Lendaro, E.; Bellelli, A.; Barra, D.; Bossa, F.; Brunori, M. The amino acid sequence of a ribosome-inactivating protein from Saponaria officinalis seeds. Biochem. Int. 1990, 21, 831–838.
- Endo, Y.; Tsurugi, K. The RNA N-glycosidase activity of ricin A-chain. The characteristics of the enzymatic activity of ricin A-chain with ribosomes and with rRNA. J. Biol. Chem. 1988, 263, 8735–8739.
- Barbieri, L.; Battelli, M.G.; Stirpe, F. Ribosome-inactivating proteins from plants. Biochim. Biophys. Acta 1993, 1154, 237–282.
- Bagga, S.; Seth, D.; Batra, J.K. The cytotoxic activity of ribosome-inactivating protein saporin-6 is attributed to its rRNA N-glycosidase and internucleosomal DNA fragmentation activities. J. Biol. Chem. 2003, 278, 4813–4820.
- Stirpe, F. Ribosome-inactivating proteins: From toxins to useful proteins. Toxicon 2013, 67, 12–16.
- Polito, L.; Bortolotti, M.; Farini, V.; Battelli, M.G.; Barbieri, L.; Bolognesi, A. Saporin induces multiple death pathways in lymphoma cells with different intensity and timing as compared to ricin. Int. J. Biochem. Cell Biol. 2009, 41, 1055–1061.
- Thorpe, P.E.; Brown, A.N.; Bremner, J.A.G., Jr.; Foxwell, B.M.; Stirpe, F. An immunotoxin composed of monoclonal anti-Thy 1.1 antibody and a ribosome-inactivating protein from Saponaria officinalis: Potent antitumor effects in vitro and in vivo. J. Natl. Cancer Inst. 1985, 75, 151–159.
- Glennie, M.J.; McBride, H.M.; Stirpe, F.; Thorpe, P.E.; Worth, A.T.; Stevenson, G.T. Emergence of immunoglobulin variants following treatment of a B cell leukemia with an immunotoxin composed of antiidiotypic antibody and saporin. J. Exp. Med. 1987, 166, 43–62.
- Siena, S.; Lappi, D.A.; Bregni, M.; Formosa, A.; Villa, S.; Soria, M.; Bonadonna, G.; Gianni, A.M. Synthesis and characterization of an antihuman T-lymphocyte saporin immunotoxin (OKT1-SAP) with in vivo stability into nonhuman primates. Blood 1988, 72, 756–765.
- Tazzari, P.L.; Bolognesi, A.; De Totero, D.; Lemoli, R.M.; Fortuna, A.; Conte, R.; Crumpton, M.J.; Stirpe, F. Immunotoxins containing saporin linked to different CD2 monoclonal antibodies: In vitro evaluation. Br. J. Haematol. 1994, 86, 97–105.
- Morland, B.J.; Barley, J.; Boehm, D.; Flavell, S.U.; Ghaleb, N.; Kohler, J.A.; Okayama, K.; Wilkins, B.; Flavell, D.J. Effectiveness of HB2 (anti-CD7)-Saporin immunotoxin in an in vivo model of human T-cell leukaemia developed in severe combined immunodeficient mice. Br. J. Cancer 1994, 69, 279–285.
- Flavell, D.J.; Boehm, D.A.; Okayama, K.; Kohler, J.A.; Flavell, S.U. Therapy of human T-cell acute lymphoblastic leukaemia in severe combined immunodeficient mice with two different anti-CD7-saporin immunotoxins containing hindered or non-hindered disulphide cross-linkers. Int. J. Cancer 1994, 58, 407–414.
- Flavell, D.J.; Flavell, S.U.; Boehm, D.; Emery, L.; Noss, A.; Ling, N.R.; Richardson, P.R.; Hardie, D.; Wright, D.H. Preclinical studies with the anti-CD19-saporin immunotoxin BU12-SAPORIN for the treatment of human-B-cell tumours. Br. J. Cancer 1995, 72, 1373–1379.
- Flavell, D.J.; Boehm, D.A.; Noss, A.M.; Flavell, S.U. Comparison of the potency and therapeutic efficacy of the anti-CD7 immunotoxin HB2-saporin constructed with one or two saporin moieties per immunotoxin molecule. Br. J. Cancer 1997, 75, 1035–1043.
- Flavell, D.J.; Warnes, S.; Noss, A.; Flavell, S.U. Host-mediated antibody-dependent cellular cytotoxicity contributes to the in vivo therapeutic efficacy of an anti-CD7-saporin immunotoxin in a severe combined immunodeficient mouse model of human T-cell acute lymphoblastic leukemia. Cancer Res. 1998, 58, 5787–5794.
- Flavell, D.J.; Warnes, S.L.; Noss, A.L.; Flavell, S.U. Anti-CD7 antibody and immunotoxin treatment of human CD7(+)T-cell leukaemia is significantly less effective in NOD/LtSz-scid mice than in CB.17 scid mice. Br. J. Cancer 2000, 83, 1755–1761.
- Polito, L.; Bortolotti, M.; Mercatelli, D.; Battelli, M.G.; Bolognesi, A. Saporin-S6: A useful tool in cancer therapy. Toxins 2013, 5, 1698–1722.
- Puri, M.; Kaur, I.; Perugini, M.A.; Gupta, R.C. Ribosome-inactivating proteins: Current status and biomedical applications. Drug Discov. Today 2012, 17, 774–783.
- Barbieri, L.; Bolognesi, A.; Valbonesi, P.; Polito, L.; Olivieri, F.; Stirpe, F. Polynucleotide: Adenosine glycosidase activity of immunotoxins containing ribosome-inactivating proteins. J. Drug Target. 2000, 8, 281–288.
- Qi, L.; Nett, T.M.; Allen, M.C.; Sha, X.; Harrison, G.S.; Frederick, B.A.; Crawford, E.D.; Glode, L.M. Binding and cytotoxicity of conjugated and recombinant fusion proteins targeted to the gonadotropin-releasing hormone receptor. Cancer Res. 2004, 64, 2090–2095.
- Frankel, A.; Welsh, P.; Richardson, J.; Robertus, J.D. Role of arginine 180 and glutamic acid 177 of ricin toxin A chain in enzymatic inactivation of ribosomes. Mol. Cell. Biol. 1990, 10, 6257–6263.
- Robertus, J.D.; Piatak, M.; Ferris, R.; Houston, L.L. Crystallization of ricin A chain obtained from a cloned gene expressed in Escherichia coli. J. Biol. Chem. 1987, 262, 19–20.
- Habuka, N.; Akiyama, K.; Tsuge, H.; Miyano, M.; Matsumoto, T.; Noma, M. Expression and secretion of Mirabilis antiviral protein in Escherichia coli and its inhibition of in vitro eukaryotic and prokaryotic protein synthesis. J. Biol. Chem. 1990, 265, 10988–10992.
- Kataoka, J.; Habuka, N.; Furuno, M.; Miyano, M.; Takanami, Y.; Koiwai, A. Expression of a pokeweed antiviral protein in Escherichia coli and its characterization. J. Biol. Chem. 1991, 266, 8426–8430.
- Legname, G.; Fossati, G.; Monzini, N.; Gromo, G.; Marcucci, F.; Mascagni, P.; Modena, D. Heterologous expression, purification, activity and conformational studies of different forms of dianthin 30. Biomed. Pept. Proteins Nucleic Acids 1995, 1, 61–68.
- Shaw, P.C.; Yun, M.H.; Zhu, R.H.; Ho, W.K.K.; Ng, T.B.; Yeung, H.W. Cloning of trichosanthin cDNA and its expression in Escherichia coli. Gene 1991, 97, 267–272.
- Bass, H.W.; Krawetz, J.E.; OBrian, G.R.; Zinselmeier, C.; Habben, J.E.; Boston, R.S. Maize ribosome-inactivating proteins (RIPs) with distinct expression patterns have similar requirements for proenzyme activation. J. Exp. Bot. 2004, 55, 2219–2233.
- Ding, G.B.; Wu, G.; Li, B.; Yang, P.; Li, Z. High-yield expression in Escherichia coli, biophysical characterization, and biological evaluation of plant toxin gelonin. 3 Biotech 2019, 9, 19.
- Hartley, M.R.; Legname, G.; Osborn, R.; Chen, Z.; Lord, J.M. Single-chain ribosome inactivating proteins from plants depurinate Escherichia coli 23S ribosomal RNA. FEBS Lett. 1991, 290, 65–68.
- Barthelemy, I.; Martineau, D.; Ong, M.; Matsunami, R.; Ling, N.; Benatti, L.; Cavallaro, U.; Soria, M.; Lappi, D.A. The expression of saporin, a ribosome-inactivating protein from the plant Saponaria officinalis, in Escherichia coli. J. Biol. Chem. 1993, 268, 6541–6548.
- Fabbrini, M.S.; Rappocciolo, E.; Carpani, D.; Solinas, M.; Valsasina, B.; Breme, U.; Cavallaro, U.; Nykjaer, A.; Rovida, E.; Legname, G.; et al. Characterization of a saporin isoform with lower ribosome-inhibiting activity. Biochem. J. 1997, 322, 719–727.
- Benatti, L.; Saccardo, M.B.; Dani, M.; Nitti, G.P.; Sassano, M.; Lorenzetti, R.; Lappi, D.A.; Soria, M. Nucleotide sequence of cDNA coding for saporin-6, a type-1 ribosome-inactivating protein from Saponaria officinalis. Eur. J. Biochem. 1989, 183, 465–470.
- Benatti, L.; Nitti, G.; Solinas, M.; Valsasina, B.; Vitale, A.; Ceriotti, A.; Soria, M.R. A Saporin-6 cDNA containing a precursor sequence coding for a carboxyl-terminal extension. FEBS Lett. 1991, 291, 285–288.
- Pittaluga, E.; Poma, A.; Tucci, A.; Spanò, L. Expression and characterisation in E. coli of mutant forms of saporin. J. Biotechnol. 2005, 117, 263–266.
- Günhan, E.; Swe, M.; Palazoglu, M.; Voss, J.C.; Chalupa, L.M. Expression and purification of cysteine introduced recombinant saporin. Protein Expr. Purif. 2008, 58, 203–209.
- Giansanti, F.; di Leandro, L.; Koutris, I.; Pitari, G.; Fabbrini, M.S.; Lombardi, A.; Flavel, D.J.; Flavell, S.U.; Gianni, S.; Ippoliti, R. Engineering a switchable toxin: The potential use of PDZ domains in the expression, targeting and activation of modified saporin variants. Protein Eng. Des. Sel. 2010, 23, 61–68.
- Giansanti, F.; Sabatini, D.; Pennacchio, M.R.; Scotti, S.; Angelucci, F.; Dhez, A.C.; Antonosante, A.; Cimini, A.; Giordano, A.; Ippoliti, R. PDZ Domain in the Engineering and Production of a Saporin Chimeric Toxin as a Tool for targeting Cancer Cells. J. Cell. Biochem. 2015, 116, 1256–1266.
- Weng, A.; Thakur, M.; von Mallinckrodt, B.; Beceren-Braun, F.; Gilabert-Oriol, R.; Wiesner, B.; Eichhorst, J.; Böttger, S.; Melzig, M.F.; Fuchs, H. Saponins modulate the intracellular trafficking of protein toxins. J. Control Release 2012, 164, 74–86.
- Zuppone, S.; Assalini, C.; Minici, C.; Botrugno, O.A.; Curnis, F.; Degano, M.; Corti, A.; Montorsi, F.; Salonia, A.; Vago, R.A. Novel RGD-4C-Saporin Conjugate Inhibits Tumor Growth in Mouse Models of Bladder Cancer. Front. Oncol. 2022, 12, 846958.
- Lombardi, A.; Bursomanno, S.; Lopardo, T.; Traini, R.; Colombatti, M.; Ippoliti, R.; Flavell, D.J.; Flavell, S.U.; Ceriotti, A.; Fabbrini, M.S. Pichia pastoris as a host for secretion of toxic saporin chimeras. FASEB J. 2010, 24, 253–265.
- Mattanovich, D.; Branduardi, P.; Dato, L.; Gasser, B.; Sauer, M.; Porro, D. Recombinant protein production in yeasts. In Methods in Molecular Biology (Clifton, N.J.); Humana Press: Totowa, NJ, USA, 2012; Volume 824, pp. 329–358.
- Heisler, I.; Keller, J.; Tauber, R.; Sutherland, M.; Fuchs, H. A cleavableadapter to reduce nonspecific cytotoxicity of recombinant immuno-toxins. Int. J. Cancer 2003, 103, 277–282.
- Vitetta, E.S. Immunotoxins and vascular leak syndrome. Cancer J. 2000, 6, 218–224.
- Chaudhary, V.K.; Gallo, M.G.; FitzGerald, D.J.; Pastan, I. A recombinant single-chain immunotoxin composed of anti-Tac variable regions and a truncated diphtheria toxin. Proc. Natl. Acad. Sci. USA 1990, 87, 9491–9494.
- Madhumathi, J.; Verma, R.S. Therapeutic targets and recent advances in protein immunotoxins. Curr. Opin. Microbiol. 2012, 15, 300–309.
- Fabbrini, M.S.; Flavell, D.J.; Ippoliti, R. Plant protein toxins: Structure, function and biotechnological applications. In Bacterial, Plant and Animal Toxins; Ascenzi, P.P.F., Visca, P., Eds.; Research Signpost: Kerala, India, 2003; pp. 66–69.
- Fabbrini, M.S.; Carpani, D.; Bello-Rivero, I.; Soria, M.R. The amino-terminal fragment of human urokinase directs a recombinant chimeric toxin to target cells: Internalization is toxin mediated. FASEB J. 1997, 11, 1169–1176.
- Della Cristina, P.; Castagna, M.; Lombardi, A.; Barison, E.; Tagliabue, G.; Ceriotti, A.; Koutris, I.; Di Leandro, L.; Giansanti, F.; Vago, R.; et al. Systematic comparison of single-chain Fv antibody-fusion toxin constructs containing Pseudomonas Exotoxin A or saporin produced in different microbial expression systems. Microb. Cell Fact. 2015, 14, 19.
- Errico Provenzano, A.; Posteri, R.; Giansanti, F.; Angelucci, F.; Flavell, S.U.; Flavell, D.J.; Fabbrini, M.S.; Porro, D.; Ippoliti, R.; Ceriotti, A.; et al. Optimization of construct design and fermentation strategy for the production of bioactive ATF-SAP, a saporin based anti-tumoral uPAR-targeted chimera. Microb. Cell Fact. 2016, 15, 194.
- Marshall, R.S.; D’Avila, F.; Di Cola, A.; Traini, R.; Spano, L.; Fabbrini, M.S.; Ceriotti, A. Signal peptide-regulated toxicity of a plant ribosome-inactivating protein during cell stress. Plant J. 2011, 65, 218–229.
- Frigerio, L.; Vitale, A.; Lord, J.M.; Ceriotti, A.; Roberts, L.M. Free ricin A chain, proricin, and native toxin have different cellular fates when expressed in tobacco protoplasts. J. Biol. Chem. 1998, 273, 14194–14199.
- Krishnan, R.; McDonald, K.A.; Dandekar, A.M.; Jackman, A.P.; Falk, B. Expression of recombinant trichosanthin, a ribosome-inactivating protein, in transgenic tobacco. J. Biotechnol. 2002, 97, 69–88.
- Zarovni, N.; Vago, R.; Soldà, T.; Monaco, L.; Fabbrini, M.S. Saporin as a novel suicide gene in anticancer gene therapy. Cancer Gene Ther. 2007, 14, 165–173.
- Zarovni, N.; Vago, R.; Fabbrini, M.S. Saporin suicide gene therapy. Methods Mol. Biol. 2009, 542, 261–283.
- Min, K.A.; He, H.; Yang, V.C.; Shin, M.C. Construction and characterization of gelonin and saporin plasmids for toxic gene-based cancer therapy. Arch. Pharm. Res. 2016, 39, 677–686.
- Salvioni, L.; Zuppone, S.; Andreata, F.; Monieri, M.; Mazzucchelli, S.; Di Carlo, C.; Morelli, L.; Cordiglieri, C.; Donnici, L.; De Francesco, R.; et al. Nanoparticle-Mediated Suicide Gene Therapy for Triple Negative Breast Cancer Treatment. Adv. Therap. 2020, 3, 2000007.
- di Leandro, L.; Giansanti, F.; Mei, S.; Ponziani, S.; Colasante, M.; Ardini, M.; Angelucci, F.; Pitari, G.; d’Angelo, M.; Cimini, A.; et al. Aptamer-Driven Toxin Gene Delivery in U87 Model Glioblastoma Cells. Front. Pharmacol. 2021, 12, 588306.
- Zhou, Y.; Li, X.-P.; Kahn, J.N.; Tumer, N.E. Functional Assays for Measuring the Catalytic Activity of Ribosome Inactivating Proteins. Toxins 2018, 10, 240.
- Shi, W.W.; Tang, Y.S.; Sze, S.Y.; Zhu, Z.N.; Wong, K.B.; Shaw, P.C. Crystal Structure of Ribosome-Inactivating Protein Ricin A Chain in Complex with the C-Terminal Peptide of the Ribosomal Stalk Protein P2. Toxins 2016, 8, 296.
- Watanabe, K.; Kawasaki, T.; Sako, N.; Funatsu, G. Actions of pokeweed antiviral protein on virus-infected protoplasts. Biosci. Biotechnol. Biochem. 1997, 61, 994–997.
- Gilabert-Oriol, R.; Weng, A.; Mallinckrodt, B.; Melzig, M.F.; Fuchs, H.; Thakur, M. Immunotoxins constructed with ribosome-inactivating proteins and their enhancers: A lethal cocktail with tumor specific efficacy. Curr. Pharm. Des. 2014, 20, 6584–6643.
- Bortolotti, M.; Polito, L.; Bolognesi, A. Toxin and Immunotoxin Based Therapeutic Approaches. Toxins 2022, 14, 63.
- Polito, L.; Djemil, A.; Bortolotti, M. Plant Toxin-Based Immunotoxins for Cancer Therapy: A Short Overview. Biomedicines 2016, 4, 12.
- Słomińska-Wojewódzka, M.; Sandvig, K. Ricin and Ricin-Containing Immunotoxins: Insights into Intracellular Transport and Mechanism of action in Vitro. Antibodies 2013, 2, 236–269.
- Schnell, R.; Borchmann, P.; Staak, J.O.; Schindler, J.; Ghetie, V.; Vitetta, E.S.; Engert, A. Clinical evaluation of ricin A-chain immunotoxins in patients with Hodgkin’s lymphoma. Ann. Oncol. 2003, 14, 729–736.
- Messmann, R.A.; Vitetta, E.S.; Headlee, D.; Senderowicz, A.M.; Figg, W.D.; Schindler, J.; Michiel, D.F.; Creekmore, S.; Steinberg, S.M.; Kohler, D.; et al. A phase I study of combination therapy with immunotoxins IgG-HD37-deglycosylated ricin A chain (dgA) and IgG-RFB4-dgA (Combotox) in patients with refractory CD19(+), CD22(+) B cell lymphoma. Clin. Cancer Res. 2000, 6, 1302–1313.
- LoRusso, P.M.; Lomen, P.L.; Redman, B.G.; Poplin, E.; Bander, J.J.; Valdivieso, M. Phase I study of monoclonal antibody-ricin A chain immunoconjugate Xomazyme-791 in patients with metastatic colon cancer. Am. J. Clin. Oncol. 1995, 18, 307–312.
- Engert, A.; Diehl, V.; Schnell, R.; Radszuhn, A.; Hatwig, M.T.; Drillich, S.; Schön, G.; Bohlen, H.; Tesch, H.; Hansmann, M.L.; et al. A phase-I study of an anti-CD25 ricin A-chain immunotoxin (RFT5-SMPT-dgA) in patients with refractory Hodgkin’s lymphoma. Blood 1997, 89, 403–410.
- de Virgilio, M.; Degryse, B. Harnessing the Destructive Power of Ricin to Fight Human Cancer. In Ricin Toxin; John, W.C., Ed.; Bentham Science Publishers: Soest, The Netherlands, 2014; pp. 208–237.
- Olsnes, S.; Pihl, A. Molecular Action of Toxins and Viruses; Cohen, P., Van Heyningen, S., Eds.; Elsevier Scientific Publishing Co., Inc.: New York, NY, USA, 1982; pp. 51–105.
- Naemi, A.A.; Salmanian, A.H.; Noormohammadi, Z.; Amani, J. A novel EGFR-specific recombinant ricin-panitumumab (scFv) immunotoxin against breast and colorectal cancer cell lines; in silico and in vitro analyses. Eur. J. Pharmacol. 2023, 955, 175894.
- Park, S.G.; Kim, H.; Jun, H.; Choi, S.Y.; Kim, E.; Kang, S. Directing ricin-based immunotoxins with targeting affibodies and KDEL signal peptide to cancer cells effectively induces apoptosis and tumor suppression. J. Nanobiotechnology 2022, 20, 387.
- Mlsna, D.; Monzingo, A.F.; Katzin, B.J.; Ernst, S.; Robertus, J.D. Structure of recombinant ricin A chain at 2.3 Å. Protein Sci. 1993, 2, 429–435.
- O’Hare, M.; Roberts, L.M.; Thorpe, P.E.; Watson, G.J.; Prior, B.; Lord, J.M. Expression of ricin a chain in Escherichia coli. FEBS Lett. 1987, 216, 73–78.
- FitzGerald, D.J.; Bjorn, M.J.; Ferris, R.J.; Winkelhake, J.L.; Frankel, A.E.; Hamilton, T.C.; Ozols, R.F.; Willingham, M.C.; Pastan, I. Antitumor activity of an immunotoxin in a nude mouse model of human ovarian cancer. Cancer Res. 1987, 47, 1407–1410.
- Li, B.Y.; Ramakrishnan, S. Recombinant hybrid toxin with dual enzymatic activities. Potential use in preparing highly effective immunotoxins. J. Biol. Chem. 1994, 269, 2652–2658.
- Zhan, J.; Ge, L.; Shen, J.; Wang, K.; Zheng, S. A trans-Golgi network retention signal YQRL fused to ricin A chain significantly enhances its cytotoxicity. Biochem. Biophys. Res. Commun. 2004, 313, 1053–1057.
- Zhan, J.; Stayton, P.; Press, O.W. Modification of ricin A chain, by addition of endoplasmic reticulum (KDEL) or Golgi (YQRL) retention sequences, enhances its cytotoxicity and translocation. Cancer Immunol. Immunother. 1998, 46, 55–60.
- Tagge, E.; Chandler, J.; Tang, B.L.; Hong, W.; Willingham, M.C.; Frankel, A. Cytotoxicity of KDEL-terminated ricin toxins correlates with distribution of the KDEL receptor in the Golgi. J. Histochem. Cytochem. 1996, 44, 159–165.
- Mahmoudi, R.; Dianat-Moghadam, H.; Poorebrahim, M.; Siapoush, S.; Poortahmasebi, V.; Salahlou, R.; Rahmati, M. Recombinant immunotoxins development for HER2-based targeted cancer therapies. Cancer Cell Int. 2021, 21, 470.
- Westby, M.; Argent, R.H.; Pitcher, C.; Lord, J.M.; Roberts, L.M. Preparation and characterization of recombinant proricin containing an alternative protease-sensitive linker sequence. Bioconjugate Chem. 1992, 3, 375–381.
- Kreitman, R.J.; Pastan, I. Recombinant toxins. Adv. Pharmacol. 1994, 28, 193–219.
- Benhar, I.; Pastan, I. Cloning, expression and characterization of the Fv fragments of the anti-carbohydrate mAbs B1 and B5 as single-chain immunotoxins. Protein Eng. 1994, 7, 1509–1515.
- Sørensen, H.P.; Mortensen, K.K. Soluble expression of recombinant proteins in the cytoplasm of Escherichia coli. Microb. Cell Fact. 2005, 4, 1.
- Hassan, Y.; Ogg, S. Gene cloning and construction of prokaryotic and plant expression vectors of RICIN-A-Chain/PAP-S1 fusion protein and its inhibition of protein synthesis. bioRxiv 2016.
- Hassan, Y.; Ogg, S. Expression of Pokeweed Antiviral Protein Isoform S1 (PAP-S1) And of Ricin-A-Chain/PAP-S1 Novel Fusion Protein (RTA/PAP-S1) In Escherichia coli And Their Comparative Inhibition of Protein Synthesis In Vitro. bioRxiv 2017.
- Hassan, Y.; Ogg, S.; Ge, H. Expression of novel fusion antiviral proteins ricin a chain-pokeweed antiviral proteins (RTA-PAPs) in Escherichia coli and their inhibition of protein synthesis and of hepatitis B virus in vitro. BMC Biotechnol. 2018, 18, 47.
- Cook, J.P.; Savage, P.M.; Lord, J.M.; Roberts, L.M. Biologically active interleukin 2-ricin A chain fusion proteins may require intracellular proteolytic cleavage to exhibit a cytotoxic effect. Bioconjugate Chem. 1993, 4, 440–447.
- Asrorov, A.M.; Gu, Z.; Min, K.A.; Shin, M.C.; Huang, Y. Advances on Tumor-Targeting Delivery of Cytotoxic Proteins. ACS Pharmacol. Transl. Sci. 2019, 3, 107–118.
- Bortolotti, M.; Bolognesi, A.; Polito, L. Bouganin, an Attractive Weapon for Immunotoxins. Toxins 2018, 10, 323.
- Petrosini, L.; De Bartolo, P.; Cutuli, D. Neurotoxic Effects, Mechanisms, and Outcome of 192-IgG Saporin. In Handbook of Neurotoxicity; Kostrzewa, R., Ed.; Springer: New York, NY, USA, 2014.
- Bolognesi, A.; Polito, L.; Olivieri, F.; Valbonesi, P.; Barbieri, L.; Battelli, M.G.; Carusi, M.V.; Benvenuto, E.; Del Vecchio Blanco, F.; Di Maro, A.; et al. New Ribosome-Inactivating Proteins with Polynucleotide: Adenosine Glycosidase and Antiviral Activities from Basella Rubra, L. and Bougainvillea Spectabilis Willd. Planta 1997, 203, 422–429.
- Den Hartog, M.T.; Lubelli, C.; Boon, L.; Heerkens, S.; Ortiz Buijsse, A.P.; de Boer, M.; Stirpe, F. Cloning and Expression of cDNA Coding for Bouganin. Eur. J. Biochem. 2002, 269, 1772–1779.
- Fermani, S.; Tosi, G.; Farini, V.; Polito, L.; Falini, G.; Ripamonti, A.; Barbieri, L.; Chambery, A.; Bolognesi, A. Structure/Function Studies on Two Type 1 Ribosome Inactivating Proteins: Bouganin and Lychnin. J. Struct. Biol. 2009, 168, 278–287.
- Cizeau, J.; Grenkow, D.M.; Brown, J.G.; Entwistle, J.; MacDonald, G.C. Engineering and Biological Characterization of VB6-845, an Anti-EpCAM Immunotoxin Containing a T-Cell Epitope-Depleted Variant of the Plant Toxin Bouganin. J. Immunother. 2009, 32, 574–584.
- Dillon, R.L.; Chooniedass, S.; Premsukh, A.; Adams, G.P.; Entwistle, J.; MacDonald, G.C.; Cizeau, J. Trastuzumab-deBouganin Conjugate Overcomes Multiple Mechanisms of T-DM1 Drug Resistance. J. Immunother. 2016, 39, 117–126.
- Chooniedass, S.; Dillon, R.L.; Premsukh, A.; Adams, G.P.; MacDonald, G.C.; Cizeau, J. Abstract 79: Trastuzumab and C6.5 Diabody Armed with deBouganin Overcome Drug Resistance to ADCs Comprised of Anti-Microtubule Agents. Cancer Res. 2017, 77, 79.
- Kowalski, M.; Brazas, L.; Zaretsky, R.; Rasamoelisolo, M.; MacDonald, G.; Cuthbert, W.; Glover, N. A Phase I Study of VB6–845, an Anti-EpCAM Fusion Protein Targeting Advanced Solid Tumours of Epithelial Origin: Preliminary Results. J. Clin. Oncol. 2008, 26, 14663.
- Silva, A.L.; Goto, L.S.; Dinarte, A.R.; Hansen, D.; Moreira, R.A.; Beltramini, L.M.; Araújo, A.P. Pulchellin, a Highly Toxic Type 2 Ribosome-inactivating Protein from Abrus Pulchellus: Cloning, Heterologous Expression of A-chain and Structural Studies. FEBS J. 2005, 272, 1201–1210.
- Castilho, P.V.; Goto, L.S.; Roberts, L.M.; Araújo, A.P.U. Isolation and Characterization of Four Type 2 Ribosome Inactivating Pulchellin Isoforms from Abrus Pulchellus Seeds. FEBS J. 2008, 275, 948–959.
- Sdraeian, M.; Guimarães, F.E.G.; Araújo, A.P.U.; Worthylake, D.K.; LeCour, L.J.; Pincus, S.H. Selective Cytotoxicity of a Novel Immunotoxin Based on Pulchellin A Chain for Cells Expressing HIV Envelope. Sci. Rep. 2017, 7, 7579.
More
Information
Subjects:
Biochemistry & Molecular Biology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
949
Revisions:
2 times
(View History)
Update Date:
02 Jan 2024
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





