
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kazuhiro Noguchi | -- | 1614 | 2023-12-24 06:16:38 | | | |
| 2 | Fanny Huang | Meta information modification | 1614 | 2023-12-29 09:49:12 | | |
Video Upload Options
Anaplastic lymphoma kinase (ALK)-positive anaplastic large cell lymphoma (ALCL) is an aggressive T-cell lymphoma characterized by large T-cells with strong CD30 and ALK expression. Although conventional chemotherapy is effective in most patients, approximately 30% experience a relapse or refractory disease and have a poor prognosis. Several risk factors associated with poor prognosis have been identified in pediatric ALK-positive ALCL. These include morphological patterns with the small cell variant or lymphohistiocytic variant, leukemic presentation, the presence of minimal disseminated disease, or involvement of the central nervous system. Relapsed or refractory ALK-positive ALCL is often resistant to conventional chemotherapy; therefore, salvage therapy is required. In recent years, targeted therapies such as ALK inhibitors and brentuximab vedotin (BV) have been developed. ALK inhibitors block the continuous activation of ALK kinase, a driver mutation that leads to cell proliferation in ALK-positive ALCL. Additionally, BV is an antibody–drug conjugate that targets CD30-positive cells. Both ALK inhibitors and BV have displayed dramatic effects in chemoresistant ALK-positive ALCL. Weekly vinblastine treatment and hematopoietic stem cell transplantation have also been reported to be effective therapies.
1. Introduction
2. Clinical Features
3. Oncogenic Mechanism
4. Risk Factors with Poor Prognosis
4.1. Morphological Pattern
4.2. Leukemic Presentation
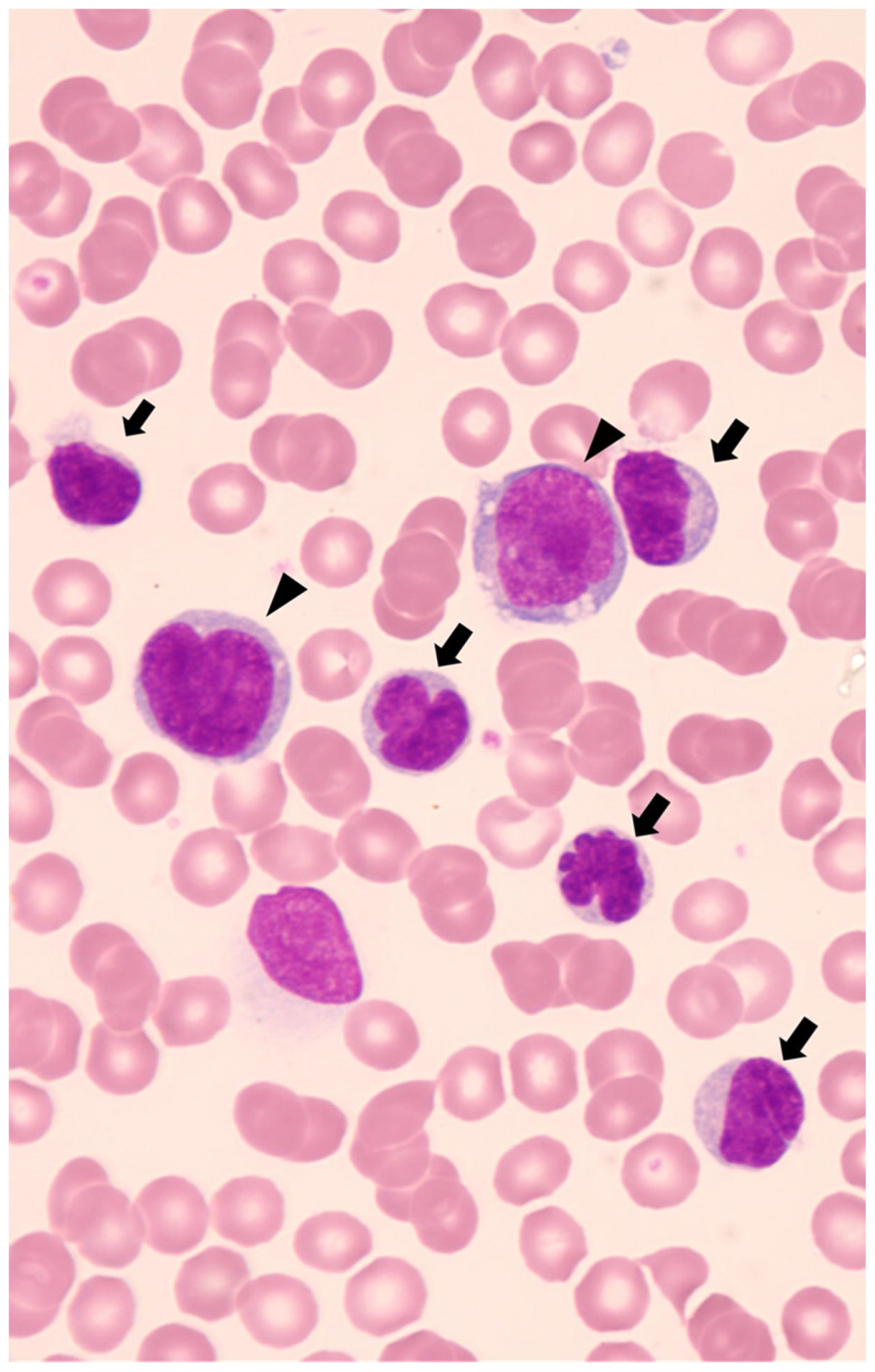
4.3. Minimal Disseminated Disease
4.4. CNS Involvement
References
- Stein, H.; Foss, H.-D. CD30+ Anaplastic Large Cell Lymphoma: A Review of Its Histopathologic, Genetic, and Clinical Features. Blood 2000, 96, 3681–3695.
- Perkins, S.L.; Pickering, D.; Lowe, E.J.; Zwick, D.; Abromowitch, M.; Davenport, G.; Cairo, M.S.; Sanger, W.G. Childhood Anaplastic Large Cell Lymphoma Has a High Incidence of ALK Gene Rearrangement as Determined by Immunohistochemical Staining and Fluorescent in Situ Hybridisation: A Genetic and Pathological Correlation. Br. J. Haematol. 2005, 131, 624–627.
- Swerdlow, S.H.; Campo, E.; Harris, N.L.; Jaffe, E.S.; Pileri, S.A.; Stein, H.; Thiele, J. (Eds.) WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues; IARC: Lyon, France, 2017; ISBN 978-92-832-4494-3.
- Falini, B.; Pileri, S.; Zinzani, P.L.; Carbone, A.; Zagonel, V.; Wolf-Peeters, C.; Verhoef, G.; Menestrina, F.; Todeschini, G.; Paulli, M.; et al. ALK+ Lymphoma: Clinico-Pathological Findings and Outcome. Blood 1999, 93, 2697–2706.
- Shiota, M.; Nakamura, S.; Ichinohasama, R.; Abe, M.; Akagi, T.; Takeshita, M.; Mori, N.; Fujimoto, J.; Miyauchi, J.; Mikata, A.; et al. Anaplastic Large Cell Lymphomas Expressing the Novel Chimeric Protein P80NPM/ALK. A Distinct Clinicopathologic Entity. Blood 1995, 86, 1954–1960.
- Tsuyama, N.; Sakamoto, K.; Sakata, S.; Dobashi, A.; Takeuchi, K. Anaplastic Large Cell Lymphoma: Pathology, Genetics, and Clinical Aspects. J. Clin. Exp. Hematop. 2017, 57, 120–142.
- Bohling, S.D.; Jenson, S.D.; Crockett, D.K.; Schumacher, J.A.; Elenitoba-Johnson, K.S.J.; Lim, M.S. Analysis of Gene Expression Profile of TPM3-ALK Positive Anaplastic Large Cell Lymphoma Reveals Overlapping and Unique Patterns with That of NPM-ALK Positive Anaplastic Large Cell Lymphoma. Leuk. Res. 2008, 32, 383–393.
- Mathew, P.; Sanger, W.G.; Weisenburger, D.D.; Valentine, M.; Valentine, V.; Pickering, D.; Higgins, C.; Hess, M.; Cui, X.; Srivastava, D.K.; et al. Detection of the t(2;5)(p23;q35) and NPM-ALK Fusion in Non-Hodgkin’s Lymphoma by Two-Color Fluorescence In Situ Hybridization. Blood 1997, 89, 1678–1685.
- Brugières, L.; Pacquement, H.; Le Deley, M.-C.; Leverger, G.; Lutz, P.; Paillard, C.; Baruchel, A.; Frappaz, D.; Nelken, B.; Lamant, L.; et al. Single-Drug Vinblastine As Salvage Treatment for Refractory or Relapsed Anaplastic Large-Cell Lymphoma: A Report From the French Society of Pediatric Oncology. J. Clin. Oncol. 2009, 27, 5056–5061.
- Lamant, L.; McCarthy, K.; d’Amore, E.; Klapper, W.; Nakagawa, A.; Fraga, M.; Maldyk, J.; Simonitsch-Klupp, I.; Oschlies, I.; Delsol, G.; et al. Prognostic Impact of Morphologic and Phenotypic Features of Childhood ALK-Positive Anaplastic Large-Cell Lymphoma: Results of the ALCL99 Study. J. Clin. Oncol. 2011, 29, 4669–4676.
- Locatelli, F.; Mauz-Koerholz, C.; Neville, K.; Llort, A.; Beishuizen, A.; Daw, S.; Pillon, M.; Aladjidi, N.; Klingebiel, T.; Landman-Parker, J.; et al. Brentuximab Vedotin for Paediatric Relapsed or Refractory Hodgkin’s Lymphoma and Anaplastic Large-Cell Lymphoma: A Multicentre, Open-Label, Phase 1/2 Study. Lancet Haematol. 2018, 5, e450–e461.
- Pro, B.; Advani, R.; Brice, P.; Bartlett, N.L.; Rosenblatt, J.D.; Illidge, T.; Matous, J.; Ramchandren, R.; Fanale, M.; Connors, J.M.; et al. Brentuximab Vedotin (SGN-35) in Patients With Relapsed or Refractory Systemic Anaplastic Large-Cell Lymphoma: Results of a Phase II Study. J. Clin. Oncol. 2012, 30, 2190–2196.
- Pro, B.; Advani, R.; Brice, P.; Bartlett, N.L.; Rosenblatt, J.D.; Illidge, T.; Matous, J.; Ramchandren, R.; Fanale, M.; Connors, J.M.; et al. Five-Year Results of Brentuximab Vedotin in Patients with Relapsed or Refractory Systemic Anaplastic Large Cell Lymphoma. Blood 2017, 130, 2709–2717.
- Woessmann, W.; Zimmermann, M.; Lenhard, M.; Burkhardt, B.; Rossig, C.; Kremens, B.; Lang, P.; Attarbaschi, A.; Mann, G.; Oschlies, I.; et al. Relapsed or Refractory Anaplastic Large-Cell Lymphoma in Children and Adolescents After Berlin-Frankfurt-Muenster (BFM)–Type First-Line Therapy: A BFM-Group Study. J. Clin. Oncol. 2011, 29, 3065–3071.
- Strullu, M.; Thomas, C.; Le Deley, M.-C.; Chevance, A.; Kanold, J.; Bertrand, Y.; Jubert, C.; Dalle, J.-H.; Paillard, C.; Baruchel, A.; et al. Hematopoietic Stem Cell Transplantation in Relapsed ALK+ Anaplastic Large Cell Lymphoma in Children and Adolescents: A Study on Behalf of the SFCE and SFGM-TC. Bone Marrow Transplant. 2015, 50, 795–801.
- Woessmann, W.; Peters, C.; Lenhard, M.; Burkhardt, B.; Sykora, K.-W.; Dilloo, D.; Kremens, B.; Lang, P.; Fuhrer, M.; Kuhne, T.; et al. Allogeneic Haematopoietic Stem Cell Transplantation in Relapsed or Refractory Anaplastic Large Cell Lymphoma of Children and Adolescents—A Berlin-Frankfurt-Munster Group Report. Br. J. Haematol. 2006, 133, 176–182.
- Bischof, D.; Pulford, K.; Mason, D.Y.; Morris, S.W. Role of the Nucleophosmin (NPM) Portion of the Non-Hodgkin’s Lymphoma-Associated NPM-Anaplastic Lymphoma Kinase Fusion Protein in Oncogenesis. Mol. Cell. Biol. 1997, 17, 2312–2325.
- Mussolin, L.; Le Deley, M.-C.; Carraro, E.; Damm-Welk, C.; Attarbaschi, A.; Williams, D.; Burke, A.; Horibe, K.; Nakazawa, A.; Wrobel, G.; et al. Prognostic Factors in Childhood Anaplastic Large Cell Lymphoma: Long Term Results of the International ALCL99 Trial. Cancers 2020, 12, 2747.
- Williams, D.; Mori, T.; Reiter, A.; Woessman, W.; Rosolen, A.; Wrobel, G.; Zsiros, J.; Uyttebroeck, A.; Marky, I.; Le Deley, M.-C.; et al. Central Nervous System Involvement in Anaplastic Large Cell Lymphoma in Childhood: Results from a Multicentre European and Japanese Study: CNS Involvement in Anaplastic Large Cell Lymphoma. Pediatr. Blood Cancer 2013, 60, E118–E121.
- Imamura, R.; Mouri, F.; Nomura, K.; Nakamura, T.; Oku, E.; Morishige, S.; Takata, Y.; Seki, R.; Osaki, K.; Hashiguchi, M.; et al. Successful Treatment of Small Cell Variant Anaplastic Large Cell Lymphoma with Allogeneic Peripheral Blood Stem Cell Transplantation, and Review of the Literature. Int. J. Hematol. 2013, 97, 139–143.
- Noguchi, K.; Ikawa, Y.; Takenaka, M.; Sakai, Y.; Fujiki, T.; Kuroda, R.; Wada, T. Characterisation of Two Tumour Cell Populations in the Small Cell Variant of Anaplastic Lymphoma Kinase-positive Anaplastic Large Cell Lymphoma. Br. J. Haematol. 2022, 196, 241–243.
- Onciu, M.; Behm, F.G.; Raimondi, S.C.; Moore, S.; Harwood, E.L.; Pui, C.-H.; Sandlund, J.T. ALK-Positive Anaplastic Large Cell Lymphoma With Leukemic Peripheral Blood Involvement Is a Clinicopathologic Entity With an Unfavorable Prognosis: Report of Three Cases and Review of the Literature. Am. J. Clin. Pathol. 2003, 120, 617–625.
- Spiegel, A.; Paillard, C.; Ducassou, S.; Perel, Y.; Plantaz, D.; Strullu, M.; Eischen, A.; Lutz, P.; Lamant, L.; Le Deley, M.-C.; et al. Paediatric Anaplastic Large Cell Lymphoma with Leukaemic Presentation in Children: A Report of Nine French Cases. Br. J. Haematol. 2014, 165, 545–551.
- Damm-Welk, C.; Busch, K.; Burkhardt, B.; Schieferstein, J.; Viehmann, S.; Oschlies, I.; Klapper, W.; Zimmermann, M.; Harbott, J.; Reiter, A.; et al. Prognostic Significance of Circulating Tumor Cells in Bone Marrow or Peripheral Blood as Detected by Qualitative and Quantitative PCR in Pediatric NPM-ALK–Positive Anaplastic Large-Cell Lymphoma. Blood 2007, 110, 670–677.
- Mussolin, L.; Pillon, M.; d’Amore, E.S.; Santoro, N.; Lombardi, A.; Fagioli, F.; Zanesco, L.; Rosolen, A. Prevalence and Clinical Implications of Bone Marrow Involvement in Pediatric Anaplastic Large Cell Lymphoma. Leukemia 2005, 19, 1643–1647.
- Iijima-Yamashita, Y.; Mori, T.; Nakazawa, A.; Fukano, R.; Takimoto, T.; Tsurusawa, M.; Kobayashi, R.; Horibe, K. Prognostic Impact of Minimal Disseminated Disease and Immune Response to NPM-ALK in Japanese Children with ALK-Positive Anaplastic Large Cell Lymphoma. Int. J. Hematol. 2018, 107, 244–250.
- Damm-Welk, C.; Lovisa, F.; Contarini, G.; Lüdersen, J.; Carraro, E.; Knörr, F.; Förster, J.; Zimmermann, M.; Sala, A.; Vinti, L.; et al. Quantification of Minimal Disease by Digital PCR in ALK-Positive Anaplastic Large Cell Lymphoma: A Step towards Risk Stratification in International Trials? Cancers 2022, 14, 1703.
- Damm-Welk, C.; Kutscher, N.; Zimmermann, M.; Attarbaschi, A.; Schieferstein, J.; Knörr, F.; Oschlies, I.; Klapper, W.; Woessmann, W. Quantification of Minimal Disseminated Disease by Quantitative Polymerase Chain Reaction and Digital Polymerase Chain Reaction for NPM-ALK as a Prognostic Factor in Children with Anaplastic Large Cell Lymphoma. Haematologica 2020, 105, 2141–2149.





