| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alexander Zhgun | -- | 3580 | 2023-12-18 11:07:08 | | | |
| 2 | Peter Tang | + 1 word(s) | 3581 | 2023-12-19 03:16:09 | | |
Video Upload Options
The natural fermentation of antibiotics, along with semi-synthetic and synthetic approaches, is one of the most important methods for their production. The majority of the antibiotic market comes from the fermentation of high-yielding (HY) fungal strains. These strains have been obtained since the 1950s from wild-type (WT) isolates as a result of classical strain improvement (CSI) programs primarily involving multi-round random mutagenesis and screening.
1. Introduction


2. Fermentation of Beta-Lactam Antibiotics in Fungi
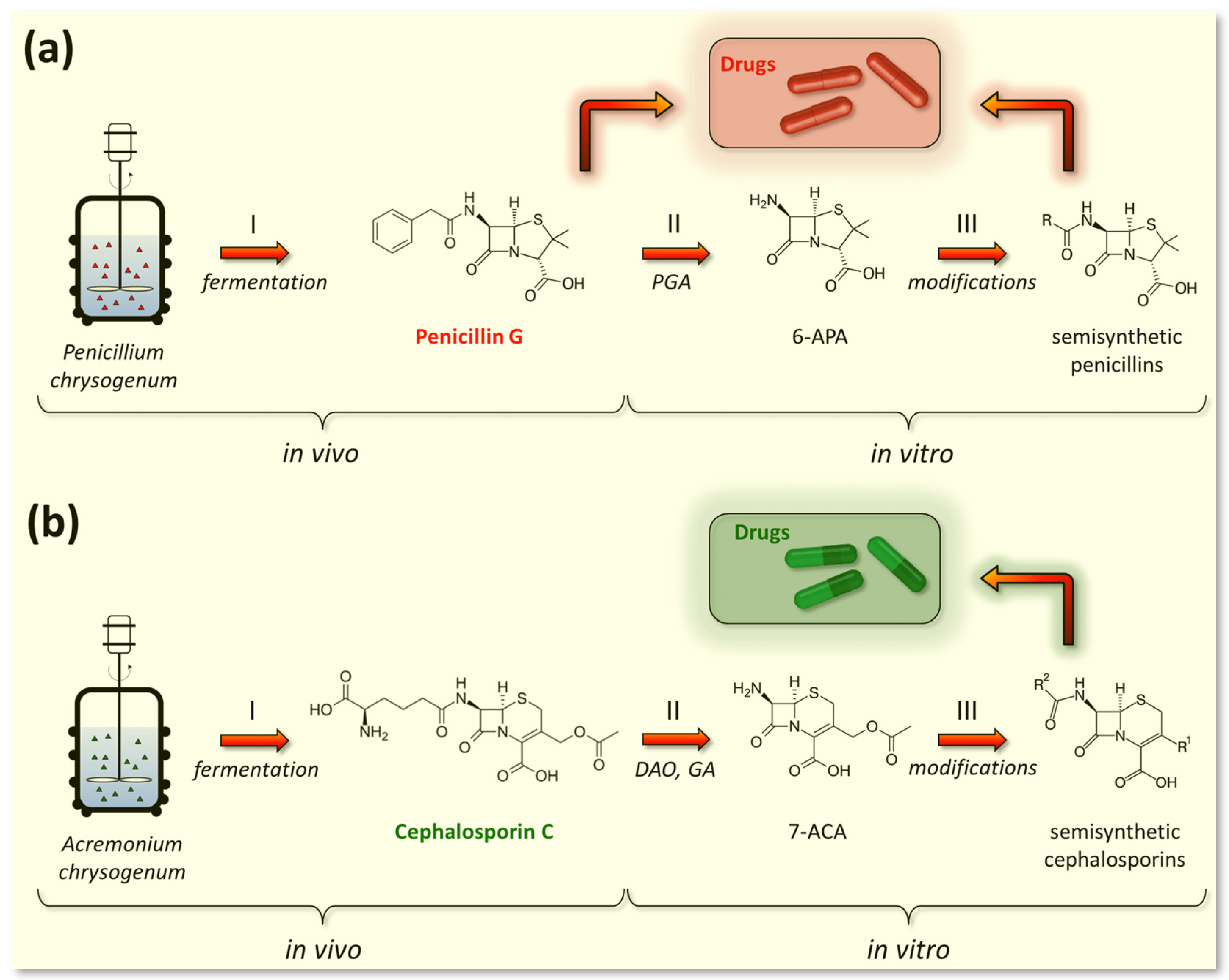
2.1. Fermentation of P. chrysogenum for Penicillin G (PenG) Production
|
Antibiotic |
Producer |
M |
Chemical Structure |
Production, mg/L |
References |
|
|---|---|---|---|---|---|---|
|
WT Strain |
HY Strain |
|||||
|
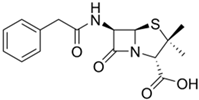
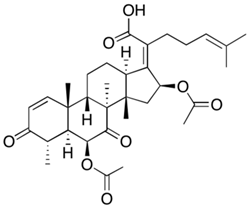
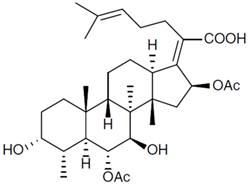
Penicillin G |
Penicillium chrysogenum |
334 |
|
50–70 |
87,650 |
[46] |
|
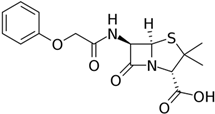
Penicillin V |
P. chrysogenum |
350 |
|
50–70 |
27,000 |
[47] |
|
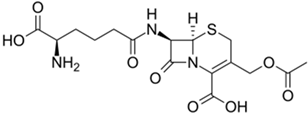
Cephalosporin C (for semisynthetic cephalosporins) |
Acremonium chrysogenum |
415 |
|
25–50 |
35,770 |
|
|
Fusidic acid (Fucidin®, Boehringer Ingelheim, Ingelheim, Germany) on market from 1962 |
Fusidium coccineum |
517 |
|
30–50 |
4540 |
|
|
Griseofulvin on market from 1959 |
Penicillium griseofulvum |
353 |
|
15–50 |
15,000+ |
|
|
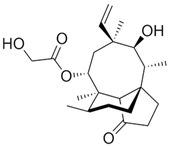
Pneumocandin B0 (for semisynthetic caspofungin; Cancidas®, Merck Research Laboratories, Rahway, NJ, USA; on market from 2001) |
Glarea lozoyensis |
1065 |
|
20–30 |
2131 |
|
|
Echinocandin B (for semisynthetic anidulafungin; Eraxis®, or Ecalta®, Pfizer, Inc., New York City, NY, USA; on market from 2006) |
Aspergillus nidulans |
1060 |
|
20–30 |
2426 |
[59] |
|
FR901379 (for semisynthetic micafungin; Mycamine®, Astellas Pharma Inc., Tokio, Japan; on market from 2005) |
Coleophoma empetri |
1197 |
|
20–50 |
4000 |
[60] |
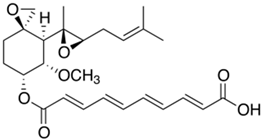
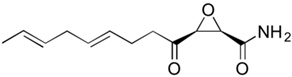
2.2. Fermentation of Penicillins Other Than PenG
2.3. Fermentation of A. chrysogenum for Cephalosporin C (CefC) Production
3. Fermentation of Non-Beta-Lactam Antibiotics in Fungi
3.1. Fusidanes
|
Antibiotic |
Producer |
M |
Chemical Structure |
Class of Antibiotic |
References |
|---|---|---|---|---|---|
|
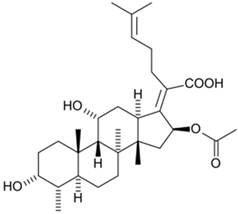
Helvolic acid |
Aspergillus fumigatus |
569 |
|
Fusidanes |
[100] |
|
Cephalosporin P1 |
A. chrysogenum |
575 |
|
Fusidanes |
[101] |
|
Pleuromutilin (for semisynthetic: (1) Retapamulin; Altabax®, or Altargo®, GSK plc, London, UK; on market from 2007 (2) Lefamulin; Xenleta®, Nabriva Therapeutics, Dublin, Ireland; on market from 2019) |
Clitophilus scyphoides |
379 |
|
Pleuromutilins |
|
|
Fusafungine (Locabiotal®, or Bioparox®, or Locabiosol®, Servier Laboratories, Suresnes, France; withdrawn from market in 2016 due to toxicity) |
Fusarium lateritium |
640 |
|
Enniatins |
[102] |
|
Enfumafungin (for semisynthetic Ibrexafungerp; Brexafemme®, GSK plc, London, UK) on market from 2021 |
Hormonema carpetanum |
709 |
|
Enfumafungins |
[103] |
|
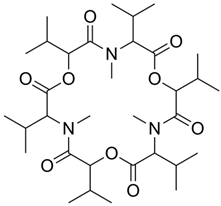
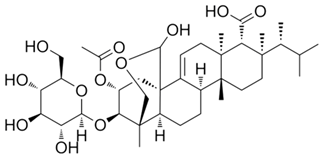
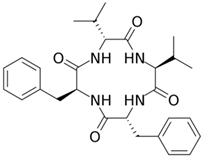
Fungisporin |
P. chrysogenum |
493 |
|
Antibiotic |
[104] |
|
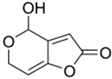
Patulin |
Aspergillus, Penicillium, Byssochlamys |
154 |
|
Antibiotic (discontinued due to high toxicity) |
[105] |
|
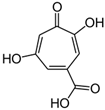
Stipitatic acid |
Talaromyces stipitatus (Penicillium stipitatum) |
182 |
|
Antibiotic activity |
[106] |
|
Ergokonin A |
Trichoderma sp. |
544 |
|
Antifungal activity |
[107] |
|
Chevalone E |
Aspergillus similanensis |
415 |
|
Antimicrobial |
[108] |
|
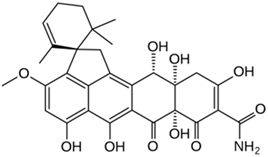
Viridicatumtoxin A |
Penicillium viridicatum |
566 |
|
Antibiotic (tetracycline-like) |
[109] |
|
Arthripenoid A |
Arthrinium sp. |
592 |
|
Antimicrobial, cytotoxic, and immunosuppressive |
[110] |
|
Fumagillin |
Aspergillus fumigatus |
459 |
|
Antibiotic (polyenes) and mycotoxin |
[111] |
|
Cerulenin |
Cephalosporium caerulens |
569 |
|
Antibiotic, also has an antitumor effect |
[100] |
|
Kojic acid |
Aspergillus oryzae |
142 |
|
Antimicrobial (used in pharmaceuticals, cosmetics, and food industry) |
[112] |
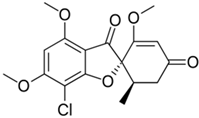
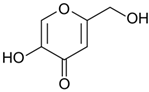
3.2. Griseofulvin
3.3. Pleuromutilins
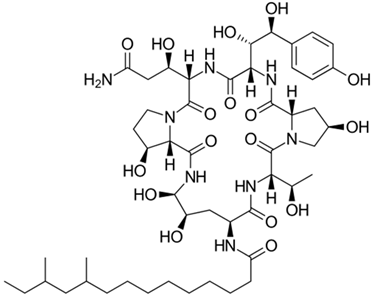
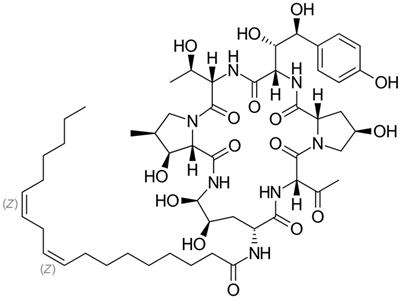
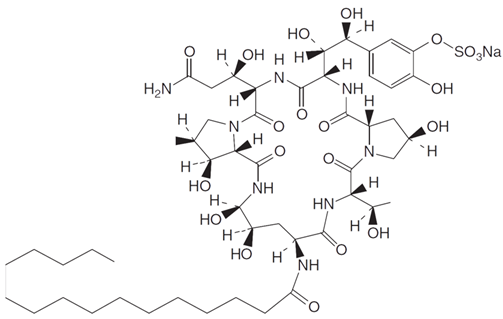
3.4. Echinocandins
3.5. Enfumafungins
3.6. Enniatins
4. Fungal Secondary Metabolites for the Development of Novel Drugs
References
- Aminov, R.I. A Brief History of the Antibiotic Era: Lessons Learned and Challenges for the Future. Front. Microbiol. 2010, 1, 134.
- Ehrlich, P. Experimental Researches on Specific Therapy: On Immunity with special Reference to the Relationship between Distribution and Action of Antigens: FIRST HARBEN LECTURE. In The Collected Papers of Paul Ehrlich; Elsevier: Amsterdam, The Netherlands, 1960; pp. 106–117.
- Bosch, F.; Rosich, L. The Contributions of Paul Ehrlich to Pharmacology: A Tribute on the Occasion of the Centenary of His Nobel Prize. Pharmacology 2008, 82, 171.
- Williams, K.J. The introduction of “chemotherapy” using arsphenamine—the first magic bullet. J. R. Soc. Med. 2009, 102, 343–348.
- Strebhardt, K.; Ullrich, A. Paul Ehrlich’s magic bullet concept: 100 years of progress. Nat. Rev. Cancer 2008, 8, 473–480.
- Lloyd, N.C.; Morgan, H.W.; Nicholson, B.K.; Ronimus, R.S. The composition of Ehrlich’s salvarsan: Resolution of a century-old debate. Angew. Chem. Int. Ed. Engl. 2005, 44, 941–944.
- Bayer, E.A.; Fierro, F.; Vaca, I.; Castillo, N.I.; Ovidio García-Rico, R.; Chávez, R. Penicillium chrysogenum, a Vintage Model with a Cutting-Edge Profile in Biotechnology. Microorganisms 2022, 10, 573.
- Sanchez, S.; Demain, A.L. Bioactive Products from Fungi. In Food Bioactives; Springer: Cham, Switzerland, 2017; p. 59.
- Hadacek, F.; Bachmann, G. Low-molecular-weight metabolite systems chemistry. Front. Environ. Sci. 2015, 3, 128574.
- Fouillaud, M.; Dufossé, L. Microbial Secondary Metabolism and Biotechnology. Microorganisms 2022, 10, 123.
- Pham, J.V.; Yilma, M.A.; Feliz, A.; Majid, M.T.; Maffetone, N.; Walker, J.R.; Kim, E.; Cho, H.J.; Reynolds, J.M.; Song, M.C.; et al. A review of the microbial production of bioactive natural products and biologics. Front. Microbiol. 2019, 10, 449147.
- Demain, A.L.; Fang, A. The natural functions of secondary metabolites. Adv. Biochem. Eng. Biotechnol. 2000, 69, 1–39.
- Maresca, M.; Muchagato Mauricio, E.; Brito, L.; Conrado, R.; Colombo Gomes, T.; Sales Calaço Roque, G.; Olívia De Souza, A. Overview of Bioactive Fungal Secondary Metabolites: Cytotoxic and Antimicrobial Compounds. Antibiotics 2022, 11, 1604.
- Keller, N.P. Translating biosynthetic gene clusters into fungal armor and weaponry. Nat. Chem. Biol. 2015, 11, 671–677.
- Gualerzi, C.O.; Brandi, L.; Fabbretti, A.; Pon, C.L. (Eds.) Antibiotics: Targets, Mechanisms and Resistance; Wiley: Hoboken, NJ, USA, 2013; Volume 1, ISBN 978-3-527-33305-9.
- Upmanyu, N.; Malviya, V.N. Antibiotics: Mechanisms of action and modern challenges. Microorg. Sustain. Environ. Health 2020, 1, 367–382.
- Ueda, K.; Beppu, T. Antibiotics in microbial coculture. J. Antibiot. 2016, 70, 361–365.
- Clardy, J.; Fischbach, M.A.; Currie, C.R. The natural history of antibiotics. Curr. Biol. 2009, 19, R437.
- Hutchings, M.; Truman, A.; Wilkinson, B. Antibiotics: Past, present and future. Curr. Opin. Microbiol. 2019, 51, 72–80.
- Moody, S.C. Microbial co-culture: Harnessing intermicrobial signaling for the production of novel antimicrobials. Future Microbiol. 2014, 9, 575–578.
- Zhang, C.; Straight, P.D. Antibiotic discovery through microbial interactions. Curr. Opin. Microbiol. 2019, 51, 64–71.
- Lyddiard, D.; Jones, G.L.; Greatrex, B.W. Keeping it simple: Lessons from the golden era of antibiotic discovery. FEMS Microbiol. Lett. 2016, 363, 84.
- Kbvresearch.com. Available online: https://www.kbvresearch.com/antibiotics-market/ (accessed on 17 November 2023).
- Latinoamericana, R.; Campos Muñiz, C.; Cuadra Zelaya, T.E.; Esquivel, G.R.; Fernández, F.J. Penicillin and cephalosporin production: A historical perspective. Rev. Latinoam. Microbiol. 2007, 49, 88–98.
- Pandey, N.; Cascella, M. Beta-Lactam Antibiotics, 1st ed.; StatPearls Publishing: St. Petersburg, FL, USA, 2023; Volume 1.
- De Rosa, M.; Verdino, A.; Soriente, A.; Marabotti, A. The Odd Couple(s): An Overview of Beta-Lactam Antibiotics Bearing More Than One Pharmacophoric Group. Int. J. Mol. Sci. 2021, 22, 617.
- Wu, M.; Crismaru, C.G.; Salo, O.; Bovenberg, R.A.L.; Driessena, A.J.M. Impact of Classical Strain Improvement of Penicillium rubens on Amino Acid Metabolism during β-Lactam Production. Appl. Environ. Microbiol. 2020, 86, e01561-19.
- Zhgun, A.; Dumina, M.; Valiakhmetov, A.; Eldarov, M. The critical role of plasma membrane H+-ATPase activity in cephalosporin C biosynthesis of Acremonium chrysogenum. PLoS ONE 2020, 15, e0238452.
- Martín, J.F. Insight into the Genome of Diverse Penicillium chrysogenum Strains: Specific Genes, Cluster Duplications and DNA Fragment Translocations. Int. J. Mol. Sci. 2020, 21, 3936.
- Why the Most Dreaded Injection is Called the “Peanut Butter” Shot. Available online: https://www.military.com/off-duty/2020/02/10/why-most-dreaded-injection-called-peanut-butter-shot.html (accessed on 13 September 2023).
- BICILLIN L-A-Penicillin g Benzathine Injection, Suspension. Available online: https://labeling.pfizer.com/showlabeling.aspx?id=691 (accessed on 13 September 2023).
- Elander, R.P. Strain Improvement and Preservation of β-Lactam-Producing Microorganisms. In Antibiotics: Containing the Beta-Lactam Structure; Demain, A.L., Solomon, N.A., Eds.; Springer: Berlin, Heidelberg, 1983; Volume 1, pp. 97–146.
- Laich, F.; Fierro, F.; Martín, J.F. Production of Penicillin by Fungi Growing on Food Products: Identification of a Complete Penicillin Gene Cluster in Penicillium griseofulvum and a Truncated Cluster in Penicillium verrucosum. Appl. Environ. Microbiol. 2002, 68, 1211.
- Aharonowitz, Y.; Cohen, G.; Martin, J.F. Penicillin and cephalosporin biosynthetic genes: Structure, organization, regulation, and evolution. Annu. Rev. Microbiol. 1992, 46, 461–495.
- Martín, J.F.; Liras, P. Transfer of Secondary Metabolite Gene Clusters: Assembly and Reorganization of the Β-Lactam Gene Cluster from Bacteria to Fungi and Arthropods. In Horizontal Gene Transfer: Breaking Borders between Living Kingdoms; Villa, T.G., Viñas, M., Eds.; Springer International Publishing: Cham, Switzerland, 2019; Volume 1, pp. 337–359. ISBN 9783030218621.
- Bud, R. Innovators, deep fermentation and antibiotics: Promoting applied science before and after the Second World War. Dynamis 2011, 31, 323–341.
- Sawant, A.M.; Vamkudoth, K.R. Biosynthetic process and strain improvement approaches for industrial penicillin production. Biotechnol. Lett. 2022, 44, 179–192.
- Weber, S.S.; Bovenberg, R.A.L.; Driessen, A.J.M. Biosynthetic concepts for the production of β-lactam antibiotics in Penicillium chrysogenum. Biotechnol. J. 2012, 7, 225–236.
- Newbert, R.W.; Barton, B.; Greaves, P.; Harper, J.; Turner, G. Analysis of a commercially improved Penicillium chrysogenum strain series: Involvement of recombinogenic regions in amplification and deletion of the penicillin biosynthesis gene cluster. J. Ind. Microbiol. Biotechnol. 1997, 19, 18–27.
- Penicillium Strain Named State Microbe of Illinois. Available online: https://www.ars.usda.gov/news-events/news/research-news/2021/penicillium-strain-named-state-microbe-of-illinois/ (accessed on 13 September 2023).
- Backus, M.P.; Stauffer, J.F. The Production and Selection of a Family of Strains in Penicillium chrysogenum. Mycologia 1955, 47, 429–463.
- Perlman, D. Some mycological aspects of penicillin production. Bot. Rev. 1950, 16, 449–523.
- Salo, O.V.; Ries, M.; Medema, M.H.; Lankhorst, P.P.; Vreeken, R.J.; Bovenberg, R.A.L.; Driessen, A.J.M. Genomic mutational analysis of the impact of the classical strain improvement program on β-lactam producing Penicillium chrysogenum. BMC Genom. 2015, 16, 937.
- Liu, L.; Chen, Z.; Liu, W.; Ke, X.; Tian, X.; Chu, J. Cephalosporin C biosynthesis and fermentation in Acremonium chrysogenum. Appl. Microbiol. Biotechnol. 2022, 106, 6413–6426.
- Elander, R.P. Industrial production of β-lactam antibiotics. Appl. Microbiol. Biotechnol. 2003, 61, 385–392.
- Wang, Z.; Wang, X.; Liu, C.; Hang, H.; Chu, J.; Guo, M.; Zhuang, Y. Method for Increasing Penicillin Fermentation Production Unit. China Patent CN113388658A, 14 September 2021.
- Jørgensen, H.; Nielsen, J.; Villadsen, J.; Møllgaard, H. Analysis of penicillin V biosynthesis during fed-batch cultivations with a high-yielding strain of Penicillium chrysogenum. Appl. Microbiol. Biotechnol. 1995, 43, 123–130.
- Duan, S.; Yuan, G.; Zhao, Y.; Ni, W.; Luo, H.; Shi, Z.; Liu, F. Simulation of computational fluid dynamics and comparison of cephalosporin C fermentation performance with different impeller combinations. Korean J. Chem. Eng. 2013, 30, 1097–1104.
- Yang, Y.; Xia, J.; Li, J.; Chu, J.; Li, L.; Wang, Y.; Zhuang, Y.; Zhang, S. A novel impeller configuration to improve fungal physiology performance and energy conservation for cephalosporin C production. J. Biotechnol. 2012, 161, 250–256.
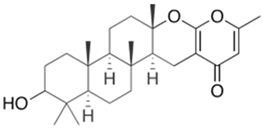
- Godtfredsen, W.O.; Jahnsen, S.; Lorck, H.; Roholt, K.; Tybring, L. Fusidic acid: A new antibiotic. Nature 1962, 193, 987.
- Verbist, L. The antimicrobial activity of fusidic acid. J. Antimicrob. Chemother. 1990, 25, 1–5.
- Huang, W.W.; Ge, X.Y.; Huang, Y.; Chai, X.T.; Zhang, L.; Zhang, Y.X.; Deng, L.N.; Liu, C.Q.; Xu, H.; Gao, J. High-yield strain of fusidic acid obtained by atmospheric and room temperature plasma mutagenesis and the transcriptional changes involved in improving its production in fungus Fusidium coccineum. J. Appl. Microbiol. 2021, 130, 405–415.
- Aris, P.; Yan, L.; Wei, Y.; Chang, Y.; Shi, B.; Xia, X. Conservation of griseofulvin genes in the gsf gene cluster among fungal genomes. G3 Genes|Genomes|Genet. 2022, 12, jkab39.
- Venkata Dasu, V.; Panda, T.; Chidambaram, M. Determination of significant parameters for improved griseofulvin production in a batch bioreactor by Taguchi’s method. Process Biochem. 2003, 38, 877–880.
- K1yoshi, U.; Abe, S. Production of Griseofulvin by Some Strains of the Genus Penicillum. J. Antibiot. Ser. A 1961, 14, 215–220.
- Nigam, P.; Singh, D. METABOLIC PATHWAYS|Production of Secondary Metabolites—Fungi. Encycl. Food Microbiol. 1999, 2, 1319–1328.
- Song, P.; Yuan, K.; Qin, T.; Zhang, K.; Ji, X.; Ren, L.; Guan, R.; Wen, J.; Huang, H. Metabolomics profiling reveals the mechanism of increased pneumocandin B0 production by comparing mutant and parent strains. J. Ind. Microbiol. Biotechnol. 2018, 45, 767–780.
- Song, P.; Zhang, K.; Zhang, S.; Huang, B.Q.; Ji, X.J.; Ren, L.J.; Gao, S.; Wen, J.P.; Huang, H. Enhancement of Pneumocandin B0 Production in Glarea lozoyensis by Low-Temperature Adaptive Laboratory Evolution. Front. Microbiol. 2018, 9, 2788.
- Hu, Z.C.; Li, W.J.; Zou, S.P.; Niu, K.; Zheng, Y.G. Mutagenesis of echinocandin B overproducing Aspergillus nidulans capable of using starch as main carbon source. Prep. Biochem. Biotechnol. 2020, 50, 745–752.
- Men, P.; Zhou, Y.; Xie, L.; Zhang, X.; Zhang, W.; Huang, X.; Lu, X. Improving the production of the micafungin precursor FR901379 in an industrial production strain. Microb. Cell Fact. 2023, 22, 44.
- Houbraken, J.; Frisvad, J.C.; Samson, R.A. Fleming’s penicillin producing strain is not Penicillium chrysogenum but P. rubens. IMA Fungus Glob. Mycol. J. 2011, 2, 87.
- Torres, M.J.; Doña, I.; Fernández, T.D.; Bogas, G. Penicillin. In Cutaneous Drug Hypersensitivity: Clinical Features, Mechanisms, Diagnosis, and Management; Bircher, A.J., Maibach, H.I., Brockow, K., Barbaud, A., Eds.; Springer Nature Switzerland AG: Cham, Switzerland, 2022; Volume 1, pp. 169–176. ISBN 9783030827434.
- Srirangan, K.; Orr, V.; Akawi, L.; Westbrook, A.; Moo-Young, M.; Chou, C.P. Biotechnological advances on penicillin G acylase: Pharmaceutical implications, unique expression mechanism and production strategies. Biotechnol. Adv. 2013, 31, 1319–1332.
- Ball, A.P.; Gray, J.A.; Murdoch, J.M. The Natural Penicillins—Benzylpenicillin (Penicillin G) and Phenoxymethylpenicillin (Penicillin V). In Antibacterial Drugs Today; Springer: Dordrecht, The Netherlands, 1978; Volume 1, pp. 6–18.
- Perron, Y.G.; Minor, W.F.; Holdrege, C.T.; Gottstein, W.J.; Godfrey, J.C.; Crast, L.B.; Babel, R.B.; Cheney, L.C. Derivatives of 6-Aminopenicillanic Acid. I. Partially Synthetic Penicillins Prepared from α-Aryloxyalkanoic Acids. J. Am. Chem. Soc. 1960, 82, 3934–3938.
- Güngör, Ö.Ö.N.; Gürkan, P.; Özçelik, B.; Oyardi, Ö. Synthesis and antimicrobial activities of new higher amino acid Schiff base derivatives of 6-aminopenicillanic acid and 7-aminocephalosporanic acid. J. Mol. Struct. 2016, 1106, 181–191.
- Cheptea, C.; Zara, A.; Dimitriu, D.G.; Sunel, V.; Dorohoi, D.O.; Cigu, T.A. New Semisynthetic Penicillins Obtained by Coupling of the 6-Aminopenicillanic Acid with 5-Mercapto-1,2,4-triazoles-3,4-disubstituted. Int. J. Mol. Sci. 2023, 24, 24.
- Bruggink, A.; Roy, P.D. Industrial Synthesis of Semisynthetic Antibiotics. In Synthesis of β-Lactam Antibiotics; Bruggink, A., Ed.; Springer: Dordrecht, The Netherlands, 2001; Volume 1, pp. 12–54.
- Möller, J.; Niehoff, J.; Dors, M.; Hiddessen, R.; Schügerl, K. Influence of medium composition on penicillin V production in a stirred tank reactor. J. Biotechnol. 1992, 25, 245–259.
- Chang, L.T.; McGrory, E.L.; Elander, R.P.; Hook, D.J. Decreased production of para-hydroxypenicillin V in penicillin V fermentations. J. Ind. Microbiol. 1991, 7, 175–179.
- Gutiérrez, S.; Velasco, J.; Fernandez, F.J.; Martín, J.F. The cefG gene of Cephalosporium acremonium is linked to the cefEF gene and encodes a deacetylcephalosporin C acetyltransferase closely related to homoserine O-acetyltransferase. J. Bacteriol. 1992, 174, 3056.
- Gutiérrez, S.; Fierro, F.; Casqueiro, J.; Martín, J.F. Gene organization and plasticity of the beta-lactam genes in different filamentous fungi. Antonie Leeuwenhoek 1999, 75, 81–94.
- Dumina, M.V.; Zhgun, A.A.; Domracheva, A.G.; Novak, M.I.; El’darov, M.A. Chromosomal polymorphism of Acremonium chrysogenum strains producing cephalosporin C. Russ. J. Genet. 2012, 48, 778–784.
- Hou, L.W.; Giraldo, A.; Groenewald, J.W.; Rämä, T.; Summerbell, R.C.; Huang, G.Z.; Cai, L.; Crous, P.W. Redisposition of acremonium-like fungi in Hypocreales. Stud. Mycol. 2023, 105, 23–203.
- Newton, G.G.; Abraham, E.P. Isolation of cephalosporin C, a penicillin-like antibiotic containing D-alpha-aminoadipic acid. Biochem. J. 1956, 62, 651–658.
- Acremonium chrysogenum (Thirumalachar et Sukapure) Gams—11550|ATCC. Available online: https://www.atcc.org/products/11550 (accessed on 22 September 2023).
- Brakhage, A.A. (Ed.) Molecular Biotechnolgy of Fungal Beta-Lactam Antibiotics and Related Peptide Synthetases. In Advances in Biochemical Engineering/Biotechnology, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2004; Volume 88, ISBN 978-3-540-22032-9.
- García-Estrada, C.; Martín, J.-F. Penicillins and Cephalosporins. In Comprehensive Biotechnology; Moo-Young, M., Ed.; Academic Press: Burlington, VT, USA, 2011; Volume 1, pp. 255–268. ISBN 978-0-08-088504-9.
- Ehl’darov, M.A.; Redo, V.A.; Zhgun, A.A.; Zejnalov, O.A.; Skrjabin, K.G. RU 231 68 C1 pETTvDAO2 RECOMBINANT PLASMID PROVIDING SYNTHESIS OF YEAST Trigonopsis variabilis D-AMINO ACID OXYDASE (DAO) IN Escherichia coli CELLS AND RECOMBINANT Escherichia coli STRAIN C41(DE3)/pETTvDAO2 AS PRODUCER OF DAO. Patent RU23168C1, 17 March 2006.
- Lee, S.K.; Park, S.W.; Kim, Y.I.; Chung, K.H.; Hong, S.I.; Kim, S.W. Immobilization of GL-7-ACA Acylase for the Production of 7-ACA. Korean J. Chem. Eng. 2002, 19, 261–266.
- Deaguero, A.L.; Blum, J.K.; Bommarius, A.S. Biocatalytic synthesis of β-lactam antibiotics. In Encyclopedia of Industrial Biotechnology: Bioprocess, Bioseparation, and Cell Technology; John Wiley Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 1–32.
- Eldarov, M.A.; Sklyarenko, A.V.; Dumina, M.; Medvedeva, N.V.; Jgoun, A.A.; Satarova, J.E.; Sidorenko, A.I.; Emperian, A.S.; Yarotsky, S.V. Recombinant cephalosporin-acid synthesase: Optimisation of expression in E. coli cells, immobilisation and application for biocatalytic cefazolin synthesis. Biomed. Khim. 2015, 61, 646–651.
- Falagas, M.E.; Grammatikos, A.P.; Michalopoulos, A. Potential of old-generation antibiotics to address current need for new antibiotics. Expert Rev. Anti. Infect. Ther. 2008, 6, 593–600.
- Beekman, A.M.; Barrow, R.A.; Beekman, A.M.; Barrow, R.A. Fungal Metabolites as Pharmaceuticals. Aust. J. Chem. 2014, 67, 827–843.
- Kavanagh, F.; And, A.H.; Robbins, W.J. Antibiotic Substances from Basidiomycetes: VIII. Pleurotus Multilus (Fr.) Sacc. and Pleurotus Passeckerianus Pilat*. Proc. Natl. Acad. Sci. USA 1951, 37, 570.
- Hartley, A.J.; De Mattos-Shipley, K.; Collins, C.M.; Kilaru, S.; Foster, G.D.; Bailey, A.M. Investigating pleuromutilin-producing Clitopilus species and related basidiomycetes. FEMS Microbiol. Lett. 2009, 297, 24–30.
- Watkins, R.R.; File, T.M. Lefamulin: A Novel Semisynthetic Pleuromutilin Antibiotic for Community-acquired Bacterial Pneumonia. Clin. Infect. Dis. 2020, 71, 2757–2762.
- Fischbach, M.A.; Walsh, C.T. Antibiotics for Emerging Pathogens. Science 2009, 325, 1089.
- Szymański, M.; Chmielewska, S.; Czyżewska, U.; Malinowska, M.; Tylicki, A. Echinocandins—Structure, mechanism of action and use in antifungal therapy. J. Enzyme Inhib. Med. Chem. 2022, 37, 876.
- Mapook, A.; Hyde, K.D.; Hassan, K.; Kemkuignou, B.M.; Čmoková, A.; Surup, F.; Kuhnert, E.; Paomephan, P.; Cheng, T.; de Hoog, S.; et al. Ten decadal advances in fungal biology leading towards human well-being. Fungal Divers. 2022, 116, 547–614.
- European Medicines Agency—Human Medicines—Fusafungine Containing Medicinal Products for Oromucosal and Nasal Use. Available online: https://web.archive.org/web/20180620220021/http://www.ema.europa.eu/ema//index.jsp?curl=pages%2Fmedicines%2Fhuman%2Freferrals%2FFusafungine_for_oromucosal_and_nasal_use%2Fhuman_referral_prac_000054.jsp&mid=WC0b01ac05805c516f (accessed on 26 September 2023).
- Mast, Y.; Stegmann, E. Actinomycetes: The Antibiotics Producers. Antibiotics 2019, 8, 105.
- Taurino, C.; Frattini, L.; Marcone, G.L.; Gastaldo, L.; Marinelli, F. Actinoplanes teichomyceticus ATCC 31121 as a cell factory for producing teicoplanin. Microb. Cell Fact. 2011, 10, 82.
- Stapley, E.O.; Jackson, M.; Hernandez, S.; Zimmerman, S.B.; Currie, S.A.; Mochales, S.; Mata, J.M.; Woodruff, H.B.; Hendlin, D. Cephamycins, a New Family of β-Lactam Antibiotics I. Production by Actinomycetes, Including Streptomyces lactamdurans sp. Antimicrob. Agents Chemother. 1972, 2, 122.
- Browne, K.; Chakraborty, S.; Chen, R.; Willcox, M.D.P.; Black, D.S.; Walsh, W.R.; Kumar, N. A New Era of Antibiotics: The Clinical Potential of Antimicrobial Peptides. Int. J. Mol. Sci. 2020, 21, 7047.
- Naeem, A.; Badshah, S.L.; Muska, M.; Ahmad, N.; Khan, K. The Current Case of Quinolones: Synthetic Approaches and Antibacterial Activity. Molecules 2016, 21, 268.
- Ovung, A.; Bhattacharyya, J. Sulfonamide drugs: Structure, antibacterial property, toxicity, and biophysical interactions. Biophys. Rev. 2021, 13, 259.
- Binod, P.; Sindhu, R.; Pandey, A. Production of antibiotics by filamentous fungi. In Current Developments in Biotechnology and Bioengineering: Filamentous Fungi Biorefinery; Taherzadeh, M.J., Ferreira, J.A., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2023; Volume 1, pp. 477–496. ISBN 9780323918725.
- Lv, J.M.; Hu, D.; Gao, H.; Kushiro, T.; Awakawa, T.; Chen, G.D.; Wang, C.X.; Abe, I.; Yao, X.S. Biosynthesis of helvolic acid and identification of an unusual C-4-demethylation process distinct from sterol biosynthesis. Nat. Commun. 2017, 8, 1644.
- Bills, G.F.; Platas, G.; Gams, W. Conspecificity of the cerulenin and helvolic acid producing ‘Cephalosporium caerulens’, and the hypocrealean fungus Sarocladium oryzae. Mycol. Res. 2004, 108, 1291–1300.
- Chou, T.S.; Eisenbraun, E.J.; Rapala, R.T. The chemistry of cephalosporin P1. Tetrahedron Lett. 1967, 8, 409–414.
- Sy-Cordero, A.A.; Pearce, C.J.; Oberlies, N.H. Revisiting the enniatins: A review of their isolation, biosynthesis, structure determination and biological activities. J. Antibiot. 2012, 65, 541–549.
- Pelaez, F.; Cabello, A.; Platas, G.; Díez, M.T.; Del Val, A.G.; Basilio, A.; Martán, I.; Vicente, F.; Bills, G.F.; Giacobbe, R.A.; et al. The discovery of enfumafungin, a novel antifungal compound produced by an endophytic Hormonema species biological activity and taxonomy of the producing organisms. Syst. Appl. Microbiol. 2000, 23, 333–343.
- Ali, H.; Ries, M.I.; Lankhorst, P.P.; Van Der Hoeven, R.A.M.; Schouten, O.L.; Noga, M.; Hankemeier, T.; Van Peij, N.N.M.E.; Bovenberg, R.A.L.; Vreeken, R.J.; et al. A non-canonical NRPS is involved in the synthesis of fungisporin and related hydrophobic cyclic tetrapeptides in Penicillium chrysogenum. PLoS ONE 2014, 9, e98212.
- Saleh, I.; Goktepe, I. The characteristics, occurrence, and toxicological effects of patulin. Food Chem. Toxicol. 2019, 129, 301–311.
- Saniewski, M.; Horbowicz, M.; Kanlayanarat, S. The biological activities of troponoids and their use in agriculture a review. J. Hortic. Res. 2014, 22, 5–19.
- Vicente, M.F.; Cabello, A.; Platas, G.; Basilio, A.; Díez, M.T.; Dreikorn, S.; Giacobbe, R.A.; Onishi, J.C.; Meinz, M.; Kurtz, M.B.; et al. Antimicrobial activity of ergokonin A from Trichoderma longibrachiatum. J. Appl. Microbiol. 2001, 91, 806–813.
- Prompanya, C.; Dethoup, T.; Bessa, L.J.; Pinto, M.M.M.; Gales, L.; Costa, P.M.; Silva, A.M.S.; Kijjoa, A. New Isocoumarin Derivatives and Meroterpenoids from the Marine Sponge-Associated Fungus Aspergillus similanensis sp. nov. KUFA 0013. Mar. Drugs 2014, 12, 5160.
- Chooi, Y.H.; Cacho, R.; Tang, Y. Identification of the viridicatumtoxin and griseofulvin gene clusters from Penicillium aethiopicum. Chem. Biol. 2010, 17, 483–494.
- Han, J.; Zhang, J.; Song, Z.; Zhu, G.; Liu, M.; Dai, H.; Hsiang, T.; Liu, X.; Zhang, L.; Quinn, R.J.; et al. Genome-based mining of new antimicrobial meroterpenoids from the phytopathogenic fungus Bipolaris sorokiniana strain 11134. Appl. Microbiol. Biotechnol. 2020, 104, 3835–3846.
- Yang, W.; Kim, W.S.; Fang, A.; Demain, A.L. Carbon and nitrogen source nutrition of fumagillin biosynthesis by Aspergillus fumigatus. Curr. Microbiol. 2003, 46, 275–279.
- Wu, L.; Zhang, L.; Li, X.; Lv, R.; Cao, W.; Gao, W.; Liu, J.; Xie, Z.; Liu, H. Effective production of kojic acid in engineered Aspergillus niger. Microb. Cell Fact. 2023, 22, 40.
- Aris, P.; Wei, Y.; Mohamadzadeh, M.; Xia, X. Griseofulvin: An Updated Overview of Old and Current Knowledge. Molecular 2022, 27, 7034.
- Oxford, A.E.; Raistrick, H.; Simonart, P. Studies in the biochemistry of micro-organisms: Griseofulvin, C(17)H(17)O(6)Cl, a metabolic product of Penicillium griseo-fulvum Dierckx. Biochem. J. 1939, 33, 240–248.
- Venkata Dasu, V.; Panda, T. Studies on production of griseofulvin. Bioprocess Eng. 1999, 21, 489–495.
- Zhgun, A.A. Fungal BGCs for Production of Secondary Metabolites: Main Types, Central Roles in Strain Improvement, and Regulation According to the Piano Principle. Int. J. Mol. Sci. 2023, 24, 11184.
- Drug and Biologic Approval and IND Activity Reports > NME Drug and New Biologic Approvals in 2007. Available online: https://wayback.archive-it.org/7993/20170406061415/https://www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/DrugandBiologicApprovalReports/ucm081690.htm (accessed on 26 September 2023).
- Jones, R.N.; Fritsche, T.R.; Sader, H.S.; Ross, J.E. Activity of Retapamulin (SB-275833), a Novel Pleuromutilin, against Selected Resistant Gram-Positive Cocci. Antimicrob. Agents Chemother. 2006, 50, 2583.
- Robert Daum, R.S.; Kar, S.; Kirkpatrick Retapamulin, P. Retapamulin. Nat. Rev. Drug Discov. 2007, 6, 865–866.
- Dillon, C.; Guarascio, A.J.; Covvey, J.R. Lefamulin: A promising new pleuromutilin antibiotic in the pipeline. Expert Rev. Anti. Infect. Ther. 2019, 17, 5–15.
- Wang, H.; Charles, C.V. A Review of Newly Approved Antibiotic Treatment for Community-Acquired Bacterial Pneumonia: Lefamulin. Sr. Care Pharm. 2020, 35, 349–354.
- Yuan, S.; Yu, B.; Liu, H.M. New drug approvals for 2019: Synthesis and clinical applications. Eur. J. Med. Chem. 2020, 205, 112667.
- Waites, K.B.; Crabb, D.M.; Duffy, L.B.; Jensen, J.S.; Liu, Y.; Paukner, S. In Vitro Activities of Lefamulin and Other Antimicrobial Agents against Macrolide-Susceptible and Macrolide-Resistant Mycoplasma pneumoniae from the United States, Europe, and China. Antimicrob. Agents Chemother. 2017, 61, 10-1128.
- Emri, T.; Majoros, L.; Tóth, V.; Pócsi, I. Echinocandins: Production and applications. Appl. Microbiol. Biotechnol. 2013, 97, 3267–3284.
- Denning, D.W. Echinocandins: A new class of antifungal. J. Antimicrob. Chemother. 2002, 49, 889–891.
- Debono, M.; Gordee, R.S. Antibiotics that inhibit fungal cell wall development. Annu. Rev. Microbiol. 1994, 48, 471–497.
- Yue, Q.; Chen, L.; Zhang, X.; Li, K.; Sun, J.; Liu, X.; An, Z.; Bills, G.F. Evolution of Chemical Diversity in Echinocandin Lipopeptide Antifungal Metabolites. Eukaryot. Cell 2015, 14, 698–718.
- Pharmacotherapy Update|New Antifungal Agents Additions to the Existing Armamentarium (Part 1). Available online: https://www.clevelandclinicmeded.com/medicalpubs/pharmacy/mayjune2003/antifungal.htm (accessed on 25 September 2023).
- Benz, F.; Knüsel, F.; Nüesch, J.; Treichler, H.; Voser, W.; Nyfeler, R.; Keller-Schierlein, W. Metabolites of microorganisms. 143. Echinocandin B, a novel polypeptide-antibiotic from Aspergillus nidulans var. echinulatus: Isolation and structural components. Helv. Chim. Acta 1974, 57, 2459–2477.
- Pfaller, M.A.; Boyken, L.; Hollis, R.J.; Messer, S.A.; Tendolkar, S.; Diekema, D.J. In Vitro Activities of Anidulafungin against More than 2,500 Clinical Isolates of Candida spp., Including 315 Isolates Resistant to Fluconazole. J. Clin. Microbiol. 2005, 43, 5425.
- Zida, A.; Bamba, S.; Yacouba, A.; Ouedraogo-Traore, R.; Guiguemdé, R.T. Anti-Candida albicans natural products, sources of new antifungal drugs: A review. J. Mycol. Med. 2017, 27, 1–19.
- Bouffard, F.A.; Zambias, R.A.; Dropinski, J.F.; Balkovec, J.M.; Hammond, M.L.; Abruzzo, G.K.; Bartizal, K.F.; Marrinan, J.A.; Kurtz, M.B.; McFadden, D.C.; et al. Synthesis and Antifungal Activity of Novel Cationic Pneumocandin Bo Derivatives. J. Med. Chem. 1994, 37, 222–225.
- Masurekar, P.S.; Fountoulakis, J.M.; Hallada, T.C.; Sosa, M.S.; Kaplan, L. Pneumocandins from Zalerion arboricola. II. Modification of product spectrum by mutation and medium manipulation. J. Antibiot. 1992, 45, 1867–1874.
- Tkacz, J.S.; Giacobbe, R.A.; Monaghan, R.L. Improvement in the titer of echinocandin-type antibiotics: A magnesium-limited medium supporting the biphasic production of pneumocandins A0 and B0. J. Ind. Microbiol. 1993, 11, 95–103.
- Qin, T.; Song, P.; Wang, X.; Ji, X.; Ren, L.; Huang, H. Protoplast mutant selection of Glarea Lozoyensis and statistical optimization of medium for pneumocandin B0 yield-up. Biosci. Biotechnol. Biochem. 2016, 80, 2241–2246.
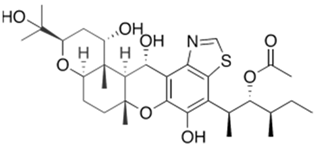
- Fujie, A. Discovery of micafungin (FK463): A novel antifungal drug derived from a natural product lead. Pure Appl. Chem. 2007, 79, 603–614.
- Ueda, S.; Sakamoto, K.; Oohata, N.; Tsuboi, M.; Yamashita, M.; Hino, M.; Yamada, M.; Hashimoto, S. Screening and characterization of microorganisms with FR901379 acylase activity. J. Antibiot. 2010, 63, 65–70.
- Kanda, M.; Tsuboi, M.; Sakamoto, K.; Shimizu, S.; Yamashita, M.; Honda, H. Improvement of FR901379 production by mutant selection and medium optimization. J. Biosci. Bioeng. 2009, 107, 530–534.
- Kanda, M.; Yamamoto, E.; Hayashi, A.; Yabutani, T.; Yamashita, M.; Honda, H. Scale-up fermentation of echinocandin type antibiotic FR901379. J. Biosci. Bioeng. 2010, 109, 138–144.
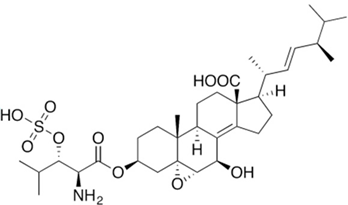
- Men, P.; Wang, M.; Li, J.; Geng, C.; Huang, X.; Lu, X. Establishing an Efficient Genetic Manipulation System for Sulfated Echinocandin Producing Fungus Coleophoma empetri. Front. Microbiol. 2021, 12, 734780.
- Schwartz, R.E.; Smith, S.K.; Onishi, J.C.; Meinz, M.; Kurtz, M.; Giacobbe, R.A.; Wilson, K.E.; Liesch, J.; Zink, D.; Horn, W.; et al. Isolation and structural determination of enfumafungin, a triterpene glycoside antifungal agent that is a specific inhibitor of glucan synthesis. J. Am. Chem. Soc. 2000, 122, 4882–4886.
- Kuhnert, E.; Li, Y.; Lan, N.; Yue, Q.; Chen, L.; Cox, R.J.; An, Z.; Yokoyama, K.; Bills, G.F. Enfumafungin synthase represents a novel lineage of fungal triterpene cyclases. Environ. Microbiol. 2018, 20, 3325.
- Jiménez-Ortigosa, C.; Perez, W.B.; Angulo, D.; Borroto-Esoda, K.; Perlin, D.S. De Novo Acquisition of Resistance to SCY-078 in Candida glabrata Involves FKS Mutations That both Overlap and Are Distinct from Those Conferring Echinocandin Resistance. Antimicrob. Agents Chemother. 2017, 61, 10-1128.
- Pfaller, M.A.; Messer, S.A.; Rhomberg, P.R.; Borroto-Esoda, K.; Castanheira, M. Differential Activity of the Oral Glucan Synthase Inhibitor SCY-078 against Wild-Type and Echinocandin-Resistant Strains of Candida Species. Antimicrob. Agents Chemother. 2017, 61, 10-1128.
- SCYNEXIS Announces FDA Approval of BREXAFEMME® (Ibrexafungerp Tablets) as the First and Only Oral Non-Azole Treatment for Vaginal Yeast Infections: SCYNEXIS, Inc. (SCYX). Available online: https://web.archive.org/web/20211231040808/https://www.scynexis.com/news-media/press-releases/detail/240/scynexis-announces-fda-approval-of-brexafemme (accessed on 9 October 2023).
- Olleik, H.; Nicoletti, C.; Lafond, M.; Courvoisier-Dezord, E.; Xue, P.; Hijazi, A.; Baydoun, E.; Perrier, J.; Maresca, M. Comparative Structure–Activity Analysis of the Antimicrobial Activity, Cytotoxicity, and Mechanism of Action of the Fungal Cyclohexadepsipeptides Enniatins and Beauvericin. Toxins 2019, 11, 514.
- Gäumann, E.; Roth, S.; Ettlinger, L.; Plattner, P.A.; Nager, U. Enniatin, a new antibiotic that works against mycobacteria. Experientia 1947, 3, 202–203.
- Osman, M.F. A clinical assessment of locabiotal in the treatment of infections of the nose and throat. J. Int. Med. Res. 1977, 5, 139–146.
- Hornbogen, T.; Glinski, M.; Zocher, R. Biosynthesis of Depsipeptide Mycotoxins in Fusarium. Eur. J. Plant Pathol. 2002, 108, 713–718.
- Zocher, R.; Keller, U.; Kleinkauf, H. Enniatin synthetase, a novel type of multifunctional enzyme catalyzing depsipeptide synthesis in Fusarium oxysporum. Biochemistry 1982, 21, 43–48.
- Herrmann, M.; Zocher, R.; Haese, A. Enniatin Production by Fusarium Strains and Its Effect on Potato Tuber Tissue. Appl. Environ. Microbiol. 1996, 62, 393.
- Hedenmalm, K.; Kurz, X.; Morales, D. Effect of withdrawal of fusafungine from the market on prescribing of antibiotics and other alternative treatments in Germany: A pharmacovigilance impact study. Eur. J. Clin. Pharmacol. 2019, 75, 979–984.
- Prescott, T.A.K.; Hill, R.; Mas-Claret, E.; Gaya, E.; Burns, E. Fungal Drug Discovery for Chronic Disease: History, New Discoveries and New Approaches. Biomolecules 2023, 13, 986.
- Aly, A.H.; Debbab, A.; Proksch, P. Fifty years of drug discovery from fungi. Fungal Divers. 2011, 50, 3–19.
- Keller, N.P. Fungal secondary metabolism: Regulation, function and drug discovery. Nat. Rev. Microbiol. 2019, 17, 167–180.
- Pela´ez, F. Biological Activities of Fungal Metabolites. In Handbook of Industrial Mycology; An, Z., Ed.; CRC Press: Boca Raton, FL, USA, 2004; Volume 1, pp. 68–111. ISBN 9780429224560.
- Schueffler, A.; Anke, T. Fungal natural products in research and development. Nat. Prod. Rep. 2014, 31, 1425–1448.
- Demain, A.L.; Martens, E. Production of valuable compounds by molds and yeasts. J. Antibiot. 2016, 70, 347–360.
- Butler, M.S.; Henderson, I.R.; Capon, R.J.; Blaskovich, M.A.T. Antibiotics in the clinical pipeline as of December 2022. J. Antibiot. 2023, 76, 431–473.
- Yang, H.; Zhang, M.; Chen, Y.; Ren, H.; Zhang, H.; Yu, C.; Lu, J.; You, L.; Yu, J.; Liang, H.; et al. Pharmacokinetics of benapenem for injection in subjects with mild to moderate renal impairment. Eur. J. Clin. Pharmacol. 2022, 78, 1079–1086.
- Iterum Therapeutics Receives Complete Response Letter from U.S. Food and Drug Administration for Oral Sulopenem: Iterum Therapeutics plc (ITRM). Available online: https://www.iterumtx.com/news/press-releases/detail/73/iterum-therapeutics-receives-complete-response-letter-from (accessed on 30 November 2023).
- Minamimura, M.; Taniyama, Y.; Inoue, E.; Mitsuhashi, S. In vitro antibacterial activity and beta-lactamase stability of CP-70,429 a new penem antibiotic. Antimicrob. Agents Chemother. 1993, 37, 1547.
- Doern, G.V.; Pierce, G.; Brueggemann, A.B. In vitro activity of sanfetrinem (GV104326), a new trinem antimicrobial agent, versus Streptococcus pneumonia, Haemophilus influenzae, and Moraxella catarrhalis. Diagn. Microbiol. Infect. Dis. 1996, 26, 39–42.
- Sanfetrinem|Working Group for New TB Drugs. Available online: https://www.newtbdrugs.org/pipeline/compound/sanfetrinem (accessed on 30 November 2023).
- Brian, P.W.; Curtis, P.J.; Hemming, H.G.; Norris, G.L.F. Wortmannin, an antibiotic produced by Penicillium wortmanni. Trans. Br. Mycol. Soc. 1957, 40, 365-IN3.
- Karve, S.; Werner, M.E.; Sukumar, R.; Cummings, N.D.; Copp, J.A.; Wang, E.C.; Li, C.; Sethi, M.; Chen, R.C.; Pacold, M.E.; et al. Revival of the abandoned therapeutic wortmannin by nanoparticle drug delivery. Proc. Natl. Acad. Sci. USA 2012, 109, 8230–8235.
- Pitz, M.W.; Eisenhauer, E.A.; MacNeil, M.V.; Thiessen, B.; Easaw, J.C.; Macdonald, D.R.; Eisenstat, D.D.; Kakumanu, A.S.; Salim, M.; Chalchal, H.; et al. Phase II study of PX-866 in recurrent glioblastoma. Neuro. Oncol. 2015, 17, 1270.
- Heiss, J.D.; Argersinger, D.P.; Theodore, W.H.; Butman, J.A.; Sato, S.; Khan, O.I. Convection-Enhanced Delivery of Muscimol in Patients with Drug-Resistant Epilepsy. Neurosurgery 2019, 85, E4.
- Davis, A.K.; Barrett, F.S.; May, D.G.; Cosimano, M.P.; Sepeda, N.D.; Johnson, M.W.; Finan, P.H.; Griffiths, R.R. Effects of Psilocybin-Assisted Therapy on Major Depressive Disorder: A Randomized Clinical Trial. JAMA Psychiatry 2021, 78, 1.
- Goodwin, G.M.; Aaronson, S.T.; Alvarez, O.; Arden, P.C.; Baker, A.; Bennett, J.C.; Bird, C.; Blom, R.E.; Brennan, C.; Brusch, D.; et al. Single-Dose Psilocybin for a Treatment-Resistant Episode of Major Depression. N. Engl. J. Med. 2022, 387, 1637–1648.
- Lee, T.H.; Lee, C.K.; Tsou, W.L.; Liu, S.Y.; Kuo, M.T.; Wen, W.C. A new cytotoxic agent from solid-state fermented mycelium of Antrodia camphorata. Planta Med. 2007, 73, 1412–1415.
- Ho, C.L.; Wang, J.L.; Lee, C.C.; Cheng, H.Y.; Wen, W.C.; Cheng, H.H.Y.; Chen, M.C.M. Antroquinonol blocks Ras and Rho signaling via the inhibition of protein isoprenyltransferase activity in cancer cells. Biomed. Pharmacother. 2014, 68, 1007–1014.
- Beheshtirouy, S.; Khani, E.; Khiali, S.; Entezari-Maleki, T. Investigational antiviral drugs for the treatment of COVID-19 patients. Arch. Virol. 2022, 167, 751–805.