Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Lingjun Li | -- | 3569 | 2023-12-08 09:31:02 | | | |
| 2 | Jason Zhu | Meta information modification | 3569 | 2023-12-12 02:28:52 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Hu, Y.; Song, J.; Feng, A.; Li, J.; Li, M.; Shi, Y.; Sun, W.; Li, L. Nanotechnology-Based Tumor Tissue-Specific Drug Targeting Delivery Systems. Encyclopedia. Available online: https://encyclopedia.pub/entry/52519 (accessed on 24 May 2026).
Hu Y, Song J, Feng A, Li J, Li M, Shi Y, et al. Nanotechnology-Based Tumor Tissue-Specific Drug Targeting Delivery Systems. Encyclopedia. Available at: https://encyclopedia.pub/entry/52519. Accessed May 24, 2026.
Hu, Yu, Jizheng Song, Anjie Feng, Jieyu Li, Mengqi Li, Yu Shi, Wenxiu Sun, Lingjun Li. "Nanotechnology-Based Tumor Tissue-Specific Drug Targeting Delivery Systems" Encyclopedia, https://encyclopedia.pub/entry/52519 (accessed May 24, 2026).
Hu, Y., Song, J., Feng, A., Li, J., Li, M., Shi, Y., Sun, W., & Li, L. (2023, December 08). Nanotechnology-Based Tumor Tissue-Specific Drug Targeting Delivery Systems. In Encyclopedia. https://encyclopedia.pub/entry/52519
Hu, Yu, et al. "Nanotechnology-Based Tumor Tissue-Specific Drug Targeting Delivery Systems." Encyclopedia. Web. 08 December, 2023.
Copy Citation
Owing to high efficacy and safety, natural medicines have found their way into the field of cancer therapy over the past few decades. However, the effective ingredients of natural medicines have shortcomings of poor solubility and low bioavailability. Nanoparticles can not only solve the problems above but also have outstanding targeting ability. Targeting preparations can be classified into three levels, which are target tissues, cells, and organelles. On the premise of clarifying the therapeutic purpose of drugs, one or more targeting methods can be selected to achieve more accurate drug delivery and consequently to improve the anti-tumor effects of drugs and reduce toxicity and side effects.
nanoparticles
tumor targeting
natural medicines
1. EPR Effect-Mediated Drug Targeting
Owing to the high permeability of tumor blood vessels, nano-preparation with particles of a size less than 200 nm can enter the tumor stroma and be retained by impaired the lymphatic system. This phenomenon is known as the high permeability and retention effect of solid tumors (EPR effect) [1]. It is generally believed that the delivery system mediated by the EPR effect can effectively deliver nano-carriers to tumor tissues through passive transport [2][3]. In recent years, a series of studies on nano-preparation have been carried out at home and abroad, including micelle, liposome, nanoemulsion, and other dosage forms.
1.1. Micelle
When surfactant concentrations exceed the critical micelle concentrations (CMCs), soluble surfactants start to attract and associate with each other together to form micelles in an aqueous solution [4]. It has been repeatedly shown that the encapsulation of hydrophobic bioactive compounds inside this carrier system can improve their water solubility and bioavailability. According to the molecular weight of surfactants, micelles can be divided into low molecular micelles and polymer micelles.
As traditional excipients in the pharmaceutical field, low molecular surfactants are also good candidates for the preparation of micelles. In the early stage, phospholipids (amphoteric surfactants) and bile salts (anionic surfactants) were commonly used to prepare micelles, which could increase the solubility and anti-tumor efficacy of drugs [5][6][7]. Taking bile salts and phosphatidylcholine as carrier materials, Jiao et al. [5] prepared ISA (andrographolide derivative)-loaded mixed micelles with an encapsulation efficiency of 86.34%, a drug-loading rate of 4.87%, and an average particle size of 148.3 nm. The pharmacokinetic experiment displayed that compared with free drugs, the area under the curve (AUC0–12 h), in vivo retention time (MRT), and elimination half-life (t1/2) of micelles had increased by 2.62, 1.47, and 1.40 times, respectively, indicating that micelles could effectively improve the blood circulation time and bioavailability of ISA.
Despite all of these advantages, the application of micelles prepared from bile salts is frequently hampered because the alkaline micellar system is unsuitable for drugs that are unstable in alkaline environments. Non-ionic surfactants, which have been applied in establishing drug-carrying micelles, are a promising alternative carrier material for bile salts and can overcome the aforementioned drawback. Liang et al. [8] designed a paclitaxel (PTX)-loaded phospholipid-Tween-80 mixed micelle that exhibited stronger cytotoxic activity to cervical cancer cells HeLa and lung cancer cells A549 than free PTX (p < 0.01). The mechanism of action might be that Tween-80 could disrupt fatty molecules and bilayer membranes, evidently enhancing the permeability of cell membranes to PTX. An in vivo pharmacokinetic experiment demonstrated that the mixed micelle possessed higher bioavailability, with AUC0–t increasing by 1.3 times compared to the free PTX.
However, low molecular micelles have many inadequacies, including inflexibility in design and restricted cytotoxic effects on cancer cells and, which are more important, low molecular surfactants such as Tween-80 are the main anaphylactoid constituents of natural medicine injections. These factors have limited their clinical applications [9][10]. Therefore, in recent years, researchers have mostly switched to using amphiphilic polymers with low toxicities, excellent biodegradability, and satisfactory biocompatibility as carrier materials for the preparation of drug-carrying micelles.
Polymeric micelles, a disperse system with core–shell structure, are formed by a self-assembly of amphiphilic block copolymers in an aqueous solution. The average particle size of polymeric micelles ranges from 20 to 200 nm [11][12]. Amphiphilic copolymer carrier materials, including diblock, triblock, or pentablock copolymers (AB, ABA, ABC, or ABCBA block copolymers) [11][13][14][15], are commonly synthesized from two or more hydrophilic and hydrophobic copolymers through the esterification reaction, ring opening polymerization, or other methods [16][17]. The normally used raw materials can be divided into three categories: (1) hydrophilic polymers, such as polyethylene glycol (PEG), poly (vinyl pyrrolidone), poly(2-vinylpyridine), etc. [15][18][19]; (2) hydrophobic polymers, such as poly lactic-co-glycolic acid (PLGA), polycaprolactone (PCL), polylactic acid (PLA), etc. [15][20][21][22]; (3) amphiphilic block copolymers, mainly including D-alpha-tocopheryl polyethylene glycol succinate (TPGS), Soluplus®, and Pluronic® (F127, F68, and P123) [16][23][24][25][26][27][28][29]. During the micellization process, hydrophobic chain segments aggregate internally to form an inner core, serving as a reservoir for poorly water-soluble drugs. The hydrophilic outer corona is mainly composed of hydrophilic segments that can refrain from the clearance effect of the endothelial network system and prevent micellar particles from aggregating [4]. Such a core–shell structure not only enables the polymer to be well dispersed in aqueous solution but also provides a sufficient hydrophobic microenvironment for insoluble drugs due to its large relative molecular weight [25]. Therefore, compared with low molecular micelles, the encapsulation efficiency, drug loading, stability, anti-tumor effect, and bioavailability of polymer micelles are significantly improved [30]. Andrographolide (ADG) isolated from Andrographis paniculata (Burm. f.) Nees has anti-cancer and anti-inflammatory activities, but high hydrophobicity and poor bioavailability limit its clinical application [31]. Therefore, researchers prepared PLGA-PEG-PLGA/ADG polymer micelles with encapsulation efficiency and drug loading of 92% and 8.4%, respectively. The particle size was 124.3 ± 6.4 nm and could remain stable even after 15 days stored at 4 °C. ADG-loaded micelles had more outstanding anti-tumor ability and higher bioavailability compared to free ADG. In vitro tests proved that after 48 h of treatment, ADG micelles induced stronger cytotoxicity on breast cancer cell lines MAD-MB-231 than free drugs, with IC50 values of 7.45 ± 1.21 and 19.4 ± 2.52 µM, respectively. This might be related to the effect of inhibiting G2/M phase cell cycle and promoting cell apoptosis. In vivo experiments demonstrated that ADG micelles could continuously release within 48 h, and AUC0–∞ and MRT increased by 2.7- and 2.5-fold, respectively, compared to the original drug [32].
Polymer-mixed micelles (PMMs), formed by two or more different types of copolymers, can eliminate complex synthesis schemes of carrier materials, improve the stability of nano-micelles, enhance the compatibilization of hydrophobic compounds, and increase the anti-tumor efficacy of drugs [27][33]. In addition, PMMs have a smaller particle size (usually less than 100 nm), which is more conducive to cellular uptake and is a promising drug delivery system [33][34].
It is widely acknowledged that TPGS, which possesses inhibitory effects against P-glycoprotein (P-gp) overexpressing in multidrug-resistant cancer cells, has been a well-behaved carrier material for antineoplastic agents [35][36][37]. Nanoparticles utilizing TPGS individually to encapsulate CUR were regarded as an effective and safe delivery platform for oral administration, which were able to avoid degradation of the drug in the gastrointestinal tract and could be used for the treatment of colorectal cancer [36]. Despite its beneficial potential, the utilization of TPGS was still restricted due to high CMC and poor anti-dilution ability, making it difficult to maintain stability in blood circulation when administered intravenously. In view of these properties, F127 and P123 were added into the micellar system to prepare a CUR-loaded polymer-mixed micelle (CUR@NPT100), which held promise for the treatment of cervical cancer [35]. On the one hand, from the properties of the polymer carrier materials, the CMC values of the mixed copolymer and simplex TPGS were approximately 0.02 and 0.2 mg/mL, respectively, indicating that the copolymer micelles were expected to have great stability after dilution in the blood stream. On the other hand, from the perspective of efficacy, compared with non-cancerous cells NIH3T3, the mixed micelle significantly promoted the selective uptake of CUR by cervical cancer cells HeLa. Therefore, at the same drug concentration (2 μg/mL) for 48 h, CUR@NPT100 did not show evident cytotoxicity to NIH3T3 cells (the cell viability was approximately 85%) and showed a strong inhibitory effect on HeLa cells (the cell viability was about 55%). Moreover, the addition of TPGS to polymeric micelles can also significantly improve the encapsulation efficiency and bioavailability of drugs. When Soluplus/TPGS (3:2) were used as the carrier materials instead of Soluplus alone, the encapsulation efficiency of diosgenin increased from 66.7% to 92.6%, and the drug-loading rate increased from 3.3% to 4.6% [27]. When appropriate amounts of TPGS were added into HA-SS-PLA/PTX micelles, the t1/2, MRT, AUC0–∞, and peak concentration (Cmax) were raised by a factor of 1.33, 1.53, 2.05, and 1.33, respectively, indicating that mixed micelles could increase the retention time of simplex polymer micelles in vivo and improve the bioavailability of drugs [38]. Polymer micelles have attractive flexibility in design and can be composed of copolymers with multifarious physical and chemical properties, which is perfectly suitable for drugs with different degrees of hydrophobicity. Currently applied in establishing drug delivery system, polymer micelles have more practical meanings and can be considered as an ideal drug administration strategy against cancer.
1.2. Liposome
Liposomes, which can encapsulate or incorporate drugs into lipid bilayers, have many superiorities, such as sustained release, low toxicity, high stability, and strong permeability [39][40][41]. Once in the bloodstream, conventional liposomes would be coated with a series of plasma proteins, such as immunoglobulins and complements, giving rise to enhanced affinity with mononuclear macrophages, which would make it easy to be cleared in systemic circulation and prevent it from exerting long-lasting effects [42][43]. Therefore, researchers have been searching for more suitable carrier materials with the aim to obtain long-circulating function.
On the one hand, liposomes can achieve long-term circulation function through biological modifications. Erythrocytes, a type of circulating cell, has great biocompatibility, biodegradability, and long circulation properties. An increasing number of studies have confirmed that nanocarriers coated with erythrocyte membranes had preponderances in terms of long circulation and biocompatibility [44][45][46]. Zhong et al. [47] designed a novel biomimetic liposome coated with erythrocyte membranes and a co-loading of triptolide and celastrol (C + T/RBCm@Lip), which could effectively evade recognition and clearance by macrophages. Erythrocyte membrane coating could not only avoid the rapid clearance of an immune system and prolong the blood circulation of liposomes but also increase the uptake of liposomes by tumor cells and enhance the inhibitory effects of the drug on the growth of HepG2 cells (compared with free drugs, the inhibition activity of two drugs encapsulated in C + T/RBCm@Lip both decreased by a factor of 1.18). In addition to erythrocyte membranes, the coating of bovine serum albumin (BSA) also can endow ordinary liposomes with long circulation function. Wei et al. [48] prepared CUR liposomes with a BSA coating (BSA-CUR-Lips). It was found that the phagocytosis of BSA-CUR-Lips by the mouse macrophage Raw 264 was significantly reduced (p < 0.05), indicating that the liposomes could exert long-circulating effects. In addition, BSA-coated nano-carriers also have potential values for applications in bioimaging [49].
On the other hand, liposomes can be modified with structure-specific chemicals to obtain long-circulating effects. Utilizing 2-distearoyl-sn-glycero-3-phosphoethanolamine-N-methoxy-PEG2000 (DSPE-PEG2000) to modify liposomes has become a research hotspot over the years. DSPE-PEG2000, causing powerful steric hindrance and hydrophilicity in liposome systems, can prevent liposomes from binding to plasma opsonin or being ingested by monocytes and macrophages [42][50][51]. Long-circulating liposomes are much more suitable for diseases that require long-term and frequent administration and therefore have promising applications in cancer chemotherapy.
Despite the long-circulating effect and better pharmacological efficacy of such liposome, high doses of cholesterol, which play the key role of membrane stabilization, have severely hindered the clinical application of liposome in cancer patients who also suffer from concomitant hyperlipidemia and cardia-cerebrovascular diseases [52][53][54][55]. Moreover, the use of cholesterol also refers to religion and vegetarianism [55]. Numerous methods have been explored to address these issues. β-sitosterol succinic anhydride ester, a potential alternative drug delivery carrier for cholesterol, was linked to PEG2000 and applied to the preparation of a gambogic acid liposome [42]. While possessing more outstanding long-term circulation effects than ordinary long-circulating liposomes (compared with the ordinary long-circulating liposomes, the t1/2 and AUC of the novel liposomes were increased by 12.5% and 47.1%, respectively), this novel long-circulating liposome could also remedy the deficiency of cholesterol.
Ginsenosides are a class of compounds with both hydrophilicity and hydrophobicity, in which the hydrophobic domain is equipped with the same steroid structure as cholesterol, and the hydrophilic domain is constituted by two glucose groups [56][57]. First of all, from the perspective of carrier structure, the ginsenosides Rg3 and Rb2 can not only substitute cholesterol for exerting a membrane stabilizing effect but also act as long-circulating stealther instead of DSPE-PEG2000 [55][58]. Additionally, in the sight of therapeutic effects, when combined with chemotherapy drugs, the ginsenosides Rg3 and Rb2 can exert synergistic anti-cancer effects [58][59][60][61].
It can be seen that the design of nanocarriers is constantly being updated, and numerous attempts have been made to design for safer and more effective ways to prepare nanocarriers with excellent characteristics such as high stability, long circulation, more powerful efficacy, and so on. Natural medicines have found their way here, which can be used not only as an alternative or synergist for traditional chemotherapy agent but also a pharmaceutical excipient in the production of anti-cancer preparation. The exploration of nano-preparation of natural medicines is meaningful and worthy of further study.
1.3. Nanoemulsion
Nanoemulsions are thermodynamically stable colloidal solutions formed by droplets of the internal phase, with a particle size of 50 to 100 nm dispersed in in the external phase [62]. There are two major administration routes in the use of nanoemulsion: transdermal administration and oral administration [63][64]. As an ideal drug delivery carrier, nanoemulsions can increase the solubility, bioavailability, and anti-tumor activities of drugs [65]. At present, there are three main ways for nanoemulsions to carry natural medicines. Firstly, monomeric compounds with anti-tumor activities isolated from natural medicines were directly encapsulated using nanoemulsion technology [66][67]. In one study, luteolin was encapsulated into a nanoemulsion for the treatment of breast cancer. Due to its ability to improve the permeability of the skin stratum corneum, such nanoemulsions could directly deliver drugs to the tumor site through transdermal administration [66]. Secondly, aqueous solutions of polysaccharides in natural medicines are usually used as aqueous phases to prepare nanoemulsions. Li et al. [64] prepared a water-in-oil (W/O) nanoemulsion of shiitake mushroom polysaccharide (SMP), which was able to maintain relatively stable droplet size for 3 months (storage conditions were 4 °C or 37 °C), and the intestinal absorption and anti-tumor activity of SMP were clearly improved (anti-tumor activity for 18-fold, compared to non-treated SMP). Thirdly, using essential oils of natural medicines as the oil phase to prepare a nanoemulsion can not only improve the solubility and bio-accessibility of an essential oil, fully exert its anti-tumor effects, and expand its application range, but also avoid the potential toxicity caused by conventional oil phases [68][69][70]. Alam et al. [70] prepared a nanoemulsion system with high stability using cinnamon essential oil both as antineoplastic agent and as oil phase. Compared with cinnamon essential oil, the cytotoxic effect of nanoemulsion against A549 cells was significantly improved, with the IC50 decreased by a factor of 2.77.
In addition, compared with other forms of nanoemulsions, there are few studies on the preparation of nanoemulsions from aqueous polysaccharide solutions. It has been reported that polysaccharides in other natural medicines also possessed anti-tumor effects, such as Angelica sinensis polysaccharides [71], Dendrobium wardianum polysaccharides [72], and Poria cocos polysaccharides [73]. However, few researchers have made them into nanoemulsions. Therefore, it may be a novel and promising research direction to prepare nanoemulsions using polysaccharides in natural medicines, which is expected to expand the application scope of polysaccharides and improve their anti-tumor activities.
2. Active Ingredients in Natural Product-Mediated Drug Targeting
In natural medicines, there is a subset of drugs that are similar to targeting formulations in modern medicine and normally used to achieve “site-directed” effects [74]. As shown in Figure 1, a drug transportation system modified with active ingredients of natural medicines could change the action site of other drugs and increase the distribution of drugs in targeted tissues, which have important application values in nano-formulations.
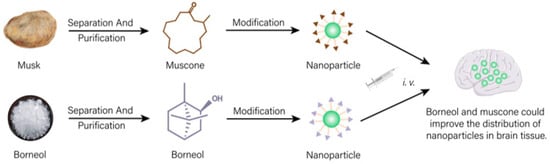
Figure 1. Drug delivery system modified with effective ingredients of natural medicines could increase distribution of other drugs in targeted tissue.
Borneol and musk, belonging to aromatic resuscitation herbs in natural medicines, can be used to modify nano-preparations acting on the brain, which can increase drug distribution in the brain and improve the brain targeting of the formulation [75][76][77]. Borneol (Bor) and transmembrane peptide Pep-1 co-modified micelles loaded with carmostine exhibited good therapeutic effects on brain glioma. Bor could effectively improve the blood–brain barrier (BBB) permeability for drugs. Cell experiments showed that the modification of Bor could not only remarkably enhance the cytotoxicity of drugs on human glioma BT325 cells (p < 0.01) but also evidently increase the uptake of micelles by brain microvascular endothelial cell lines HBMEC (fluorescence intensity increased 1.67 times). In vivo experiments showed that the fluorescence of untargeted micelles labeled with fluorescent probe DiD disappeared rapidly at 6 h post-injection. A single dose of Bor-modified micelles significantly increased the signal distribution in brain tissue (p < 0.01), suggesting that Bor modification enhanced its ability to penetrate the BBB. Additionally, the signal was still observed 24 h after treatment, indicating prolonged retention times in in brain tissue [75]. Other studies showed that similar to Bor, due to the brain targeting property of muscone, doxorubicin-loaded liposomes modified with muscone could cross the BBB. Compared with unmodified liposomes, muscone-modified liposomes showed concentrated accumulation in the glioma region of the brain and less distribution outside the glioma region. Thus, muskone modification could increase the distribution of drugs in brain tissue, then improving the curative effect for brain glioma of antineoplastic drug [77].
3. Ligand-Mediated Drug Targeting
3.1. APRPG Peptide Modified Nanocarrier
Vascular endothelial growth factor (VEGF) is highly expressed on tumor vascular endothelial cells but rarely on normal endothelial cells [78][79], making it a desirable target point for anti-tumor drug delivery. Ala-Pro-Arg-Pro-Gly (APRPG), the small molecule peptide sequence, is able to specifically bind to VEGF receptors (VEGFR). The modification of APRPG on the nano-formulations can considerably increase the effect of the first-level drug targeting by means of active targeting. It can actively deliver the drug to the tumor tissue and improve the effectiveness of chemotherapy [80][81][82]. After intravenous administration, APRPG-modified nanoparticles loaded with PTX and norethindrone were concentrated at the cancer site of mice and effectively inhibited the growth of ectopic solid tumors in tumor-bearing mice (the inhibition rate of APRPPG-modified nanoparticles group and non-targeted nanoparticles group were 78.67% and 62.98%, respectively) (p < 0.01), suggesting that APRPG could deliver chemotherapeutic drugs more effectively to tumor tissues through active targeting [82].
It is important to note that VEGF is not only a target for drug delivery but also the site of action of natural anti-tumor monomer components. For example, triptolide restrained breast cancer cell angiogenesis through inhibiting the ERK1/2-HIF1-α-VEGFA axis [83]. Honokiol inhibited the NF-κB pathway, which, in turn, led to the down-regulation of VEGF expression and reduced the viability and angiogenesis of human lung cancer cell lines [84]. Cantharidin inhibited tumor angiogenesis by suppressing VEGF-induced signaling pathways [85]. The development of angiogenesis inhibitors targeting VEGF/VEGFR has become a vital field in anti-tumor research.
3.2. NGR Peptide-Modified Nanocarrier
NGR peptide (NGR), a peptide containing an asparagine-glycine-arginine (Asn-Gly-Arg) motif, is capable of specifically recognizing aminopeptidase N (APN/CD13), which is highly expressed on tumor vascular cells [86][87][88]. NGR is considered as a potential targeting ligand that can target tumor blood vessels [86]. Therefore, similar to APRPG, NGR ligands can be used for surface modifications of nano-formulations to enhance the targeting ability of drug delivery systems to tumor tissues [89][90].
This targeted ligand can not only increase the effect of first-level drug targeting depending on the means of active targeting, so that chemotherapeutic drugs or nano-agents with anti-tumor effects can accumulate more effectively and selectively in cancer tissues and thus be more fully taken up by tumor cells. It can also make drugs that have the ability to regulate the tumor environment accurately locate in tumor tissue and act as an anti-tumor agent indirectly by improving the tumor environment. A dual-targeted micelle-liposome bilayer delivery platform triggered by matrix metalloproteinases was designed by Duan et al. [89] for simultaneous loading of the anti-fibrotic drug quercetin (Que) and the herbal chemotherapeutic drug PTX. Owing to the first-level drug targeting function of NGR, the liposomes carrying Que on the outer layer of the formulation specifically accumulate at the tumor tissue to exert the anti-fibrotic effect of Que and ameliorate the tumor microenvironment. Modification of NGR evidently facilitated intracellular accumulation of liposomes in human umbilical vein cell lines (HUVEC), with an uptake 1.3 times higher than that of unmodified liposomes. This suggested that the NGR-modified formulation could function to target tumor tissue through CD13 receptor-mediated endocytosis. The results of in vivo experiments exhibited that non-targeted agents were rapidly eliminated after injection, whereas NGR modification not only significantly increased the accumulation of the agents at the cancer site (p < 0.05) but also distinctly prolonged the residence time of nanoparticles at the tumor site (fluorescent signals were still detectable after intravenous injection for 24 h), suggesting that NGR played a crucial role in mediating the accumulation of nano-agents in tumor tissue.
It is worth noting that the above two targets and pathways initially are not used for the first-level drug targeting but for specifically delivering anti-angiogenic drugs to tumor blood vessels for the purpose of tumor therapy by inhibiting the generation of new blood vessels [86][91]. But now, NGR and APRPG have found their new way to achieve a tumor tissue-specific targeting function as an active targeting ligand. This also provides inspiration and ideas for future research: due to the difficulty in discovering new targets and pathways, when designing new targeting carriers, the flexibility of a nanocarrier can be fully utilized. Starting from known targets and pathways, new carrier forms can be explored to obtain more novel and powerful targeting strategies and expand the application range of natural medicines’ nano-formulations.
References
- Wu, J. The Enhanced Permeability and Retention (EPR) Effect: The Significance of the Concept and Methods to Enhance Its Application. J. Pers. Med. 2021, 11, 771.
- Park, J.; Choi, Y.; Chang, H.; Um, W.; Ryu, J.H.; Kwon, I.C. Alliance with EPR Effect: Combined Strategies to Improve the EPR Effect in the Tumor Microenvironment. Theranostics 2019, 9, 8073–8090.
- Shi, Y.; van der Meel, R.; Chen, X.; Lammers, T. The EPR effect and beyond: Strategies to improve tumor targeting and cancer nanomedicine treatment efficacy. Theranostics 2020, 10, 7921–7924.
- Vaidya, F.U.; Sharma, R.; Shaikh, S.; Ray, D.; Aswal, V.K.; Pathak, C. Pluronic micelles encapsulated curcumin manifests apoptotic cell death and inhibits pro-inflammatory cytokines in human breast adenocarcinoma cells. Cancer Rep. 2019, 2, e1133.
- Jiao, W.W.; Zhang, S.J.; Zhang, Y.; Gao, X.R.; Han, G. Preparation of Andrographolide Derivative ISA-Loaded Bile Salt-Phosphatidy-I Choline-Mixed Micelles and Pharmacokinetics Evaluation in Rats. Chin. Pharm. J. 2012, 47, 1643–1648.
- Zhang, Z.; Huang, Y.; Gao, F.; Gao, Z.; Bu, H.; Gu, W.; Li, Y. A self-assembled nanodelivery system enhances the oral bioavailability of daidzein: In vitro characteristics and in vivo performance. Nanomedicine 2011, 6, 1365–1379.
- Duan, Y.; Wang, J.; Yang, X.; Du, H.; Xi, Y.; Zhai, G. Curcumin-loaded mixed micelles: Preparation, optimization, physicochemical properties and cytotoxicity in vitro. Drug Deliv. 2015, 22, 50–57.
- Liang, H.; Yang, Q.; Deng, L.; Lu, J.; Chen, J. Phospholipid-Tween 80 mixed micelles as an intravenous delivery carrier for paclitaxel. Drug Dev. Ind. Pharm. 2011, 37, 597–605.
- Yang, R.; Lao, Q.C.; Yu, H.P.; Zhang, Y.; Liu, H.C.; Luan, L.; Sun, H.M.; Li, C.Q. Tween-80 and impurity induce anaphylactoid reaction in zebrafish. J. Appl. Toxicol. 2015, 35, 295–301.
- Xu, Y.; Liu, C.; Dou, D.; Wang, Q. Evaluation of anaphylactoid constituents in vitro and in vivo. Int. Immunopharmacol. 2017, 43, 79–84.
- Guo, Y.; Gao, T.; Fang, F.; Sun, S.; Yang, D.; Li, Y.; Lv, S. A novel polymer micelle as a targeted drug delivery system for 10-hydroxycamptothecin with high drug-loading properties and anti-tumor efficacy. Biophys. Chem. 2021, 279, 106679.
- Ren, Q.; Li, M.; Deng, Y.; Lu, A.; Lu, J. Triptolide delivery: Nanotechnology-based carrier systems to enhance efficacy and limit toxicity. Pharmacol. Res. 2021, 165, 105377.
- Alami-Milani, M.; Zakeri-Milani, P.; Valizadeh, H.; Fathi, M.; Salatin, S.; Salehi, R.; Jelvehgari, M. PLA-PCL-PEG-PCL-PLA based micelles for improving the ocular permeability of dexamethasone: Development, characterization, and in vitro evaluation. Pharm. Dev. Technol. 2020, 25, 704–719.
- Cho, H.; Gao, J.; Kwon, G.S. PEG-b-PLA micelles and PLGA-b-PEG-b-PLGA sol-gels for drug delivery. J. Control Release 2016, 240, 191–201.
- Ferrentino, N.; Romano, M.P.; Zappavigna, S.; Abate, M.; Del Vecchio, V.; Romano, D.; Germinario, C.; Grifa, C.; Filosa, R.; Pappalardo, D. Poly(ε-caprolactone)-poly(ethylene glycol) Tri-Block Copolymer as Quercetin Delivery System for Human Colorectal Carcinoma Cells: Synthesis, Characterization and In Vitro Study. Polymers 2023, 15, 1179.
- Song, J.; Liu, Y.; Lin, L.; Zhao, Y.; Wang, X.; Zhong, M.; Xie, T.; Luo, Y.; Li, S.; Yang, R.; et al. Glycyrrhetinic acid modified and pH-sensitive mixed micelles improve the anticancer effect of curcumin in hepatoma carcinoma cells. RSC Adv. 2019, 9, 40131–40145.
- Cui, Y.; Jin, R.; Zhou, Y.; Yu, M.; Ling, Y.; Wang, L.Q. Crystallization enhanced thermal-sensitive hydrogels of PCL-PEG-PCL triblock copolymer for 3D printing. Biomed. Mater. 2021, 16, 035006.
- Brewer, K.; Bai, F.; Blencowe, A. pH-Responsive Poly(ethylene glycol)-b-poly(2-vinylpyridine) Micelles for the Triggered Release of Therapeutics. Pharmaceutics 2023, 15, 977.
- Liang, R.; Ma, L.; Zhang, L.; Li, C.; Liu, W.; Wei, M.; Yan, D.; Evans, D.G.; Duan, X. A monomeric photosensitizer for targeted cancer therapy. Chem. Commun. 2014, 50, 14983–14986.
- Repp, L.; Skoczen, S.L.; Rasoulianboroujeni, M.; Stern, S.T.; Kwon, G.S. Plasma Stability and Plasma Metabolite Concentration-Time Profiles of Oligo(Lactic Acid)(8)-Paclitaxel Prodrug Loaded Polymeric Micelles. AAPS J. 2023, 25, 39.
- Wang, S.; Xie, L.; Liu, Y.; Yang, Q.; Jia, W.; Zhao, D.; Zhao, X. Study on the preparation and activity of intelligent response poly(lactic-co-glycolic acid)-ss-polyethylene glycol copolymer micelles. J. Biomater. Appl. 2022, 37, 259–274.
- Zhang, Q.; Bao, J.; Duan, T.; Hu, M.; He, Y.; Wang, J.; Hu, R.; Tang, J. Nanomicelle-Microsphere Composite as a Drug Carrier to Improve Lung-Targeting Specificity for Lung Cancer. Pharmaceutics 2022, 14, 510.
- Sun, S.; Du, X.; Fu, M.; Khan, A.R.; Ji, J.; Liu, W.; Zhai, G. Galactosamine-modified PEG-PLA/TPGS micelles for the oral delivery of curcumin. Int. J. Pharm. 2021, 595, 120227.
- Zhang, Y.; Zhou, Q.; Jia, S.; Lin, K.; Fan, G.; Yuan, J.; Yu, S.; Shi, J. Specific Modification with TPGS and Drug Loading of Cyclodextrin Polyrotaxanes and the Enhanced Antitumor Activity Study in Vitro and in Vivo. ACS Appl. Mater. Interfaces 2019, 11, 46427–46436.
- Liu, Y.; Fu, S.; Lin, L.; Cao, Y.; Xie, X.; Yu, H.; Chen, M.; Li, H. Redox-sensitive Pluronic F127-tocopherol micelles: Synthesis, characterization, and cytotoxicity evaluation. Int. J. Nanomed. 2017, 12, 2635–2644.
- Patil, S.; Ujalambkar, V.; Rathore, A.; Rojatkar, S.; Pokharkar, V. Galangin loaded galactosylated pluronic F68 polymeric micelles for liver targeting. Biomed. Pharmacother. 2019, 112, 108691.
- Zhao, J.; Xu, Y.; Wang, C.; Ding, Y.; Chen, M.; Wang, Y.; Peng, J.; Li, L.; Lv, L. Soluplus/TPGS mixed micelles for dioscin delivery in cancer therapy. Drug Dev. Ind. Pharm. 2017, 43, 1197–1204.
- Nicoud, M.B.; Ospital, I.A.; Taquez Delgado, M.A.; Riedel, J.; Fuentes, P.; Bernabeu, E.; Rubinstein, M.R.; Lauretta, P.; Martinez Vivot, R.; Aguilar, M.L.A.; et al. Nanomicellar Formulations Loaded with Histamine and Paclitaxel as a New Strategy to Improve Chemotherapy for Breast Cancer. Int. J. Mol. Sci. 2023, 24, 3546.
- de Oliveira, A.C.V.; de Morais, F.A.P.; Campanholi, K.; Bidoia, D.L.; Balbinot, R.B.; Nakamura, C.V.; Caetano, W.; Hioka, N.; Monteiro, O.D.S.; da Rocha, C.Q.; et al. Melanoma-targeted photodynamic therapy based on hypericin-loaded multifunctional P123-spermine/folate micelles. Photodiagnosis. Photodyn. Ther. 2022, 40, 103103.
- Li, B.; Shao, H.; Gao, L.; Li, H.; Sheng, H.; Zhu, L. Nano-drug co-delivery system of natural active ingredients and chemotherapy drugs for cancer treatment: A review. Drug Deliv. 2022, 29, 2130–2161.
- Ren, X.; Xu, W.; Sun, J.; Dong, B.; Awala, H.; Wang, L. Current Trends on Repurposing and Pharmacological Enhancement of Andrographolide. Curr. Med. Chem. 2021, 28, 2346–2368.
- Zhang, J.; Li, Y.; Gao, W.; Repka, M.A.; Wang, Y.; Chen, M. Andrographolide-loaded PLGA-PEG-PLGA micelles to improve its bioavailability and anticancer efficacy. Expert Opin. Drug Deliv. 2014, 11, 1367–1380.
- Manjappa, A.S.; Kumbhar, P.S.; Patil, A.B.; Disouza, J.I.; Patravale, V.B. Polymeric Mixed Micelles: Improving the Anticancer Efficacy of Single-Copolymer Micelles. Crit. Rev. Ther. Drug Carrier Syst. 2019, 36, 1–58.
- Giuliani, C. The Flavonoid Quercetin Induces AP-1 Activation in FRTL-5 Thyroid Cells. Antioxidants 2019, 8, 112.
- Wang, J.; Liu, Q.; Yang, L.; Xia, X.; Zhu, R.; Chen, S.; Wang, M.; Cheng, L.; Wu, X.; Wang, S. Curcumin-Loaded TPGS/F127/P123 Mixed Polymeric Micelles for Cervical Cancer Therapy: Formulation, Characterization, and InVitro and InVivo Evaluation. J. Biomed. Nanotechnol. 2017, 13, 1631–1646.
- Li, H.; Yan, L.; Tang, E.K.Y.; Zhang, Z.; Chen, W.; Liu, G.; Mo, J. Synthesis of TPGS/Curcumin Nanoparticles by Thin-Film Hydration and Evaluation of Their Anti-Colon Cancer Efficacy In Vitro and In Vivo. Front. Pharmacol. 2019, 10, 769.
- Chen, X.P.; Li, Y.; Zhang, Y.; Li, G.W. Formulation, Characterization And Evaluation Of Curcumin- Loaded PLGA- TPGS Nanoparticles For Liver Cancer Treatment. Drug Des. Devel. Ther. 2019, 13, 3569–3578.
- Du, X.; Yin, S.; Zhou, F.; Du, X.; Xu, J.; Gu, X.; Wang, G.; Li, J. Reduction-sensitive mixed micelles for selective intracellular drug delivery to tumor cells and reversal of multidrug resistance. Int. J. Pharm. 2018, 550, 1–13.
- Mozafari, M.R. Nanoliposomes: Preparation and analysis. Methods Mol. Biol. 2010, 605, 29–50.
- Talens-Visconti, R.; Diez-Sales, O.; de Julian-Ortiz, J.V.; Nacher, A. Nanoliposomes in Cancer Therapy: Marketed Products and Current Clinical Trials. Int. J. Mol. Sci. 2022, 23, 4249.
- Feng, X.; Pi, C.; Fu, S.; Yang, H.; Zheng, X.; Hou, Y.; Wang, Y.; Zhang, X.; Zhao, L.; Wei, Y. Combination of Curcumin and Paclitaxel Liposomes Exhibits Enhanced Cytotoxicity Towards A549/A549-T Cells and Unaltered Pharmacokinetics. J. Biomed. Nanotechnol. 2020, 16, 1304–1313.
- Yu, F.; Tang, X. Novel Long-Circulating Liposomes Consisting of PEG Modified beta-Sitosterol for Gambogic Acid Delivery. J. Nanosci. Nanotechnol. 2016, 16, 3115–3121.
- Tahara, Y.; Yoshikawa, T.; Sato, H.; Mori, Y.; Zahangir, M.H.; Kishimura, A.; Mori, T.; Katayama, Y. Encapsulation of a nitric oxide donor into a liposome to boost the enhanced permeation and retention (EPR) effect. Medchemcomm 2017, 8, 415–421.
- Chu, Y.; Zhang, J.; Pan, H.; Shi, J.; Wang, J.; Chen, L. Preparation and evaluation of long circulating erythrocyte membrane-cloaked anti-cancer drug delivery system. Drug Deliv. Transl. Res. 2020, 10, 1278–1287.
- Marshall, S.K.; Angsantikul, P.; Pang, Z.; Nasongkla, N.; Hussen, R.S.D.; Thamphiwatana, S.D. Biomimetic Targeted Theranostic Nanoparticles for Breast Cancer Treatment. Molecules 2022, 27, 6473.
- Ferrel, C.; Rayamajhi, S.; Nguyen, T.; Marasini, R.; Saravanan, T.; Deba, F.; Aryal, S. Re-engineering a Liposome with Membranes of Red Blood Cells for Drug Delivery and Diagnostic Applications. ACS Appl. Bio Mater. 2021, 4, 6974–6981.
- Zhong, X.; Xian, J.; Shi, J.; Wu, Y.; Chen, J.; Lin, J. Preparation and Characterization of Biomimetic Liposomes Coated with Erythrocyte Membrane Co-Loading Triptolide and Celastrol. Acta Pharm. Sin. 2021, 56, 3252–3260.
- Wei, X.Q.; Ba, K. Construction a Long-Circulating Delivery System of Liposomal Curcumin by Coating Albumin. ACS Omega 2020, 5, 16502–16509.
- Wang, R.; Zou, L.; Yi, Z.; Zhang, Z.; Zhao, M.; Shi, S. PLGA nanoparticles loaded with curcumin produced luminescence for cell bioimaging. Int. J. Pharm. 2023, 639, 122944.
- Song, J.W.; Liu, Y.S.; Guo, Y.R.; Zhong, W.X.; Guo, Y.P.; Guo, L. Nano-Liposomes Double Loaded with Curcumin and Tetrandrine: Preparation, Characterization, Hepatotoxicity and Anti-Tumor Effects. Int. J. Mol. Sci. 2022, 23, 6858.
- Alemi, A.; Zavar Reza, J.; Haghiralsadat, F.; Zarei Jaliani, H.; Haghi Karamallah, M.; Hosseini, S.A.; Haghi Karamallah, S. Paclitaxel and curcumin coadministration in novel cationic PEGylated niosomal formulations exhibit enhanced synergistic antitumor efficacy. J. Nanobiotechnol. 2018, 16, 28.
- Carson, J.A.S.; Lichtenstein, A.H.; Anderson, C.A.M.; Appel, L.J.; Kris-Etherton, P.M.; Meyer, K.A.; Petersen, K.; Polonsky, T.; Van Horn, L.; American Heart Association Nutrition Committee of the Council on, L; et al. Dietary Cholesterol and Cardiovascular Risk: A Science Advisory From the American Heart Association. Circulation 2020, 141, e39–e53.
- Bae, Y.; Heo, J.; Chung, Y.; Shin, S.Y.; Lee, S.W. Effect of Total Cholesterol Level Variabilities on Cerebrovascular Disease. Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 544–557.
- Wu, Q.; Wang, Q.; Fu, J.; Ren, R. Polysaccharides derived from natural sources regulate triglyceride and cholesterol metabolism: A review of the mechanisms. Food Funct. 2019, 10, 2330–2339.
- Hong, C.; Liang, J.; Xia, J.; Zhu, Y.; Guo, Y.; Wang, A.; Lu, C.; Ren, H.; Chen, C.; Li, S.; et al. One Stone Four Birds: A Novel Liposomal Delivery System Multi-functionalized with Ginsenoside Rh2 for Tumor Targeting Therapy. Nanomicro Lett. 2020, 12, 129.
- Hong, C.; Wang, D.; Liang, J.; Guo, Y.; Zhu, Y.; Xia, J.; Qin, J.; Zhan, H.; Wang, J. Novel ginsenoside-based multifunctional liposomal delivery system for combination therapy of gastric cancer. Theranostics 2019, 9, 4437–4449.
- Zhu, Y.; Wang, A.; Zhang, S.; Kim, J.; Xia, J.; Zhang, F.; Wang, D.; Wang, Q.; Wang, J. Paclitaxel-loaded ginsenoside Rg3 liposomes for drug-resistant cancer therapy by dual targeting of the tumor microenvironment and cancer cells. J. Adv. Res. 2022, 49, 159–173.
- Chen, C.; Xia, J.; Ren, H.; Wang, A.; Zhu, Y.; Zhang, R.; Gan, Z.; Wang, J. Effect of the structure of ginsenosides on the in vivo fate of their liposomes. Asian J. Pharm. Sci. 2022, 17, 219–229.
- Ma, J.; Zhao, D.; Yu, D.; Song, W.; Yang, X.; Yin, H. Ginsenoside Rh2 attenuates the progression of non-small cell lung cancer by sponging miR-28-5p/STK4 axis and inactivating Wnt/beta-catenin signaling. Cancer Med. 2023, 12, 12653–12667.
- Tang, M.; Deng, H.; Zheng, K.; He, J.; Yang, J.; Li, Y. Ginsenoside 3beta-O-Glc-DM (C3DM) suppressed glioma tumor growth by downregulating the EGFR/PI3K/AKT/mTOR signaling pathway and modulating the tumor microenvironment. Toxicol. Appl. Pharmacol. 2023, 460, 116378.
- Li, J.; Li, F.; Jin, D. Ginsenosides are Promising Medicine for Tumor and Inflammation: A Review. Am. J. Chin. Med. 2023, 51, 883–908.
- Asasutjarit, R.; Sooksai, N.; Fristiohady, A.; Lairungruang, K.; Ng, S.F.; Fuongfuchat, A. Optimization of Production Parameters for Andrographolide-Loaded Nanoemulsion Preparation by Microfluidization and Evaluations of Its Bioactivities in Skin Cancer Cells and UVB Radiation-Exposed Skin. Pharmaceutics 2021, 13, 1290.
- Hsu, H.Y.; Chen, B.H. A Comparative Study on Inhibition of Breast Cancer Cells and Tumors in Mice by Carotenoid Extract and Nanoemulsion Prepared from Sweet Potato (Ipomoea batatas L.) Peel. Pharmaceutics 2022, 14, 980.
- Li, T.; Han, X.; Bao, R.; Hao, Y.; Li, S. Preparation and properties of water-in-oil shiitake mushroom polysaccharide nanoemulsion. Int. J. Biol. Macromol. 2019, 140, 343–349.
- Gupta, A.; Eral, H.B.; Hatton, T.A.; Doyle, P.S. Nanoemulsions: Formation, properties and applications. Soft Matter 2016, 12, 2826–2841.
- Altamimi, M.A.; Hussain, A.; Alshehri, S.; Imam, S.S.; Alnemer, U.A. Development and Evaluations of Transdermally Delivered Luteolin Loaded Cationic Nanoemulsion: In Vitro and Ex Vivo Evaluations. Pharmaceutics 2021, 13, 1218.
- Ahmadi Oskooei, F.; Mehrzad, J.; Asoodeh, A.; Motavalizadehkakhky, A. Olive oil-based quercetin nanoemulsion (QuNE)’s interactions with human serum proteins (HSA and HTF) and its anticancer activity. J. Biomol. Struct. Dyn. 2023, 41, 778–791.
- Panyajai, P.; Chueahongthong, F.; Viriyaadhammaa, N.; Nirachonkul, W.; Tima, S.; Chiampanichayakul, S.; Anuchapreeda, S.; Okonogi, S. Anticancer activity of Zingiber ottensii essential oil and its nanoformulations. PLoS ONE 2022, 17, e0262335.
- Chen, Y.; Wang, S.; Hu, Q.; Zhou, L. Self-emulsifying System Co-loaded with Paclitaxel and Coix Seed Oil Deeply Penetrated to Enhance Efficacy in Cervical Cancer. Curr. Drug Deliv. 2023, 20, 919–926.
- Alam, A.; Ansari, M.J.; Alqarni, M.H.; Salkini, M.A.; Raish, M. Antioxidant, Antibacterial, and Anticancer Activity of Ultrasonic Nanoemulsion of Cinnamomum Cassia L. Essential Oil. Plants 2023, 12, 834.
- Zhou, W.-J.; Wang, S.; Hu, Z.; Zhou, Z.-Y.; Song, C.-J. Angelica sinensis polysaccharides promotes apoptosis in human breast cancer cells via CREB-regulated caspase-3 activation. Biochem. Biophys. Res. Commun. 2015, 467, 562–569.
- Ye, G.; Li, J.; Zhang, J.; Liu, H.; Ye, Q.; Wang, Z. Structural characterization and antitumor activity of a polysaccharide from Dendrobium wardianum. Carbohydr. Polym. 2021, 269, 118253.
- Li, X.; Ma, L.; Zhang, L. Molecular basis for Poria cocos mushroom polysaccharide used as an antitumor drug in China. Prog. Mol. Biol. Transl Sci. 2019, 163, 263–296.
- Feng, L.; Liu, L.; Zhao, Y.; Zhao, R. Saikosaponins A, C and D enhance liver-targeting effects of anticancer drugs by modulating drug transporters. Oncotarget 2017, 8, 110092–110102.
- Kang, S.; Duan, W.; Zhang, S.; Chen, D.; Feng, J.; Qi, N. Muscone/RI7217 co-modified upward messenger DTX liposomes enhanced permeability of blood-brain barrier and targeting glioma. Theranostics 2020, 10, 4308–4322.
- Wang, L.; Xu, L.; Du, J.; Zhao, X.; Liu, M.; Feng, J.; Hu, K. Nose-to-brain delivery of borneol modified tanshinone IIA nanoparticles in prevention of cerebral ischemia/reperfusion injury. Drug Deliv. 2021, 28, 1363–1375.
- Guo, X.; Wu, G.; Wang, H.; Chen, L. Pep-1&borneol-Bifunctionalized Carmustine-Loaded Micelles Enhance Anti-Glioma Efficacy Through Tumor-Targeting and BBB-Penetrating. J. Pharm. Sci. 2019, 108, 1726–1735.
- Frezzetti, D.; Gallo, M.; Maiello, M.R.; D’Alessio, A.; Esposito, C.; Chicchinelli, N.; Normanno, N.; De Luca, A. VEGF as a potential target in lung cancer. Expert. Opin. Ther. Targets 2017, 21, 959–966.
- Zhao, Y.; Adjei, A.A. Targeting Angiogenesis in Cancer Therapy: Moving Beyond Vascular Endothelial Growth Factor. Oncologist 2015, 20, 660–673.
- Lai, K.; Li, Y.; Gong, Y.; Li, L.; Huang, C.; Xu, F.; Zhong, X.; Jin, C. Triptolide-nanoliposome-APRPG, a novel sustained-release drug delivery system targeting vascular endothelial cells, enhances the inhibitory effects of triptolide on laser-induced choroidal neovascularization. Biomed. Pharmacother. 2020, 131, 110737.
- Wang, Y.; Liu, P.; Duan, Y.; Yin, X.; Wang, Q.; Liu, X.; Wang, X.; Zhou, J.; Wang, W.; Qiu, L.; et al. Specific cell targeting with APRPG conjugated PEG-PLGA nanoparticles for treating ovarian cancer. Biomaterials 2014, 35, 983–992.
- Xie, M.H.; Fu, Z.L.; Hua, A.L.; Zhou, J.F.; Chen, Q.; Li, J.B.; Yao, S.; Cai, X.J.; Ge, M.; Zhou, L.; et al. A new core-shell-type nanoparticle loaded with paclitaxel/norcantharidin and modified with APRPG enhances anti-tumor effects in hepatocellular carcinoma. Front. Oncol. 2022, 12, 932156.
- Liu, H.; Tang, L.; Li, X.; Li, H. Triptolide inhibits vascular endothelial growth factor-mediated angiogenesis in human breast cancer cells. Exp. Ther. Med. 2018, 16, 830–836.
- Cheng, X.; Wang, F.; Qiao, Y.; Chen, T.; Fan, L.; Shen, X.; Yu, D.; Huang, Y.; Wei, M. Honokiol inhibits interleukin-induced angiogenesis in the NSCLC microenvironment through the NF-κB signaling pathway. Chem.-Biol. Interact. 2023, 370, 110295.
- Wang, T.; Liu, J.; Xiao, X.-Q. Cantharidin inhibits angiogenesis by suppressing VEGF-induced JAK1/STAT3, ERK and AKT signaling pathways. Arch. Pharmacal Res. 2014, 38, 282–289.
- Cai, X.J.; Fei, W.D.; Xu, Y.Y.; Xu, H.; Yang, G.Y.; Cao, J.W.; Ni, J.J.; Wang, Z. Combination of metronomic administration and target delivery strategies to improve the anti-angiogenic and anti-tumor effects of triptolide. Drug Deliv. Transl. Res. 2020, 10, 93–107.
- Conte, C.; Longobardi, G.; Barbieri, A.; Palma, G.; Luciano, A.; Dal Poggetto, G.; Avitabile, C.; Pecoraro, A.; Russo, A.; Russo, G.; et al. Non-covalent strategies to functionalize polymeric nanoparticles with NGR peptides for targeting breast cancer. Int. J. Pharm. 2023, 633, 122618.
- Tripodi, A.A.P.; Randelovic, I.; Biri-Kovacs, B.; Szeder, B.; Mezo, G.; Tovari, J. In Vivo Tumor Growth Inhibition and Antiangiogenic Effect of Cyclic NGR Peptide-Daunorubicin Conjugates Developed for Targeted Drug Delivery. Pathol. Oncol. Res. 2020, 26, 1879–1892.
- Duan, H.; Liu, C.; Hou, Y.; Liu, Y.; Zhang, Z.; Zhao, H.; Xin, X.; Liu, W.; Zhang, X.; Chen, L.; et al. Sequential Delivery of Quercetin and Paclitaxel for the Fibrotic Tumor Microenvironment Remodeling and Chemotherapy Potentiation via a Dual-Targeting Hybrid Micelle-in-Liposome System. ACS Appl. Mater. Interfaces 2022, 14, 10102–10116.
- Kunjachan, S.; Pola, R.; Gremse, F.; Theek, B.; Ehling, J.; Moeckel, D.; Hermanns-Sachweh, B.; Pechar, M.; Ulbrich, K.; Hennink, W.E.; et al. Passive versus active tumor targeting using RGD- and NGR-modified polymeric nanomedicines. Nano Lett. 2014, 14, 972–981.
- Luo, L.M.; Huang, Y.; Zhao, B.X.; Zhao, X.; Duan, Y.; Du, R.; Yu, K.F.; Song, P.; Zhao, Y.; Zhang, X.; et al. Anti-tumor and anti-angiogenic effect of metronomic cyclic NGR-modified liposomes containing paclitaxel. Biomaterials 2013, 34, 1102–1114.
More
Information
Subjects:
Materials Science, Biomaterials
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
679
Revisions:
2 times
(View History)
Update Date:
12 Dec 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





