
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Enar Jumaniyazova | -- | 4568 | 2023-11-29 15:12:36 | | | |
| 2 | Энар Джуманиязова | Meta information modification | 4568 | 2023-11-30 04:43:13 | | | | |
| 3 | Rita Xu | -62 word(s) | 4506 | 2023-11-30 04:51:12 | | |
Video Upload Options
Head and neck squamous cell cancer (HNSCC) is one of the ten most common malignant neoplasms, characterized by an aggressive course, high recurrence rate, poor response to treatment, and low survival rate. This creates the need for a deeper understanding of the mechanisms of the pathogenesis of this cancer. The tumor microenvironment (TME) of HNSCC consists of stromal and immune cells, blood and lymphatic vessels, and extracellular matrix. It is known that HNSCC is characterized by complex relationships between cancer cells and TME components. TME components and their dynamic interactions with cancer cells enhance tumor adaptation to the environment, which provides the highly aggressive potential of HNSCC and resistance to antitumor therapy.
1. Introduction
Head and neck squamous cell carcinoma (HNSCC) is the seventh most common cancer worldwide, causing more than 660,000 new cases and 325,000 deaths annually [1][2]. Modifiable risk factors for this pathology include tobacco use in one form or another, alcoholic beverages, human papillomavirus (HPV) infection (more commonly associated with oropharyngeal cancer), and Epstein–Barr virus (EBV) infection (especially for nasopharyngeal cancer). People in some countries eat areca nut, which also increases the risk of HNSCC. Previously, HNSCC was classified according to tumor location (tumor of the oral or nasal cavity, oropharynx, nasopharynx, larynx, or hypopharynx), TNM (tumor, nodus, and metastasis) stage, and histology, but the recognition of molecular genetic profiles now allows it to be more accurately classified into individual subtypes [3][4]. Today, the updated TNM classification (eighth edition, 2018), which has several differences from the previous seventh edition of 2010, should be used for staging this nosology. The main changes in HNSCC staging include adding depth of invasion for oral cavity tumors, introducing a pathomorphologic and clinical staging system for high-risk oropharyngeal tumors associated with papillomavirus infection (HPV+), and considering tumor extension beyond the lymph node capsule in high-risk HPV-negative oropharyngeal tumors and head and neck squamous cell carcinoma in other localizations, excluding nasopharyngeal cancer. Assessing the depth of invasion of oral cancer has prognostic value: deeper tumors show an increased risk of metastasis to lymph nodes and a decreased overall survival rate [5]. Extranodal extension, in turn, also serves as an unfavorable prognostic factor for HNSCC, with the exception of HPV+-related tumors [6]. A separate chapter in the eighth edition is devoted to nasopharyngeal cancer. The main changes are the inclusion of a T0 category for patients with metastatic cervical lymph nodes, EBV-positive patients with an unknown primary focus, and changes in the definition of regional lymph nodes. Unlike other localizations of head and neck cancer, for which surgery plays an important role in primary treatment, squamous cell cancer of the nasopharynx is primarily treated with radiation therapy, with or without chemotherapy. For this reason, pathologic classification is irrelevant in this disease of the nasopharyngeal region [7]. In 2022, the fifth edition of the World Health Organization (WHO) classification of head and neck tumors was published, focusing on the distinctive molecular genetic characteristics of head and neck tumors [8]. The delineation of the distinct molecular genetic signatures of HNSCC allows for a significant increase in diagnostic accuracy and also has prognostic value, which, in turn, leads to a personalized treatment approach for each patient.
There are many approaches to the treatment of HNSCC, but none of them are effective enough. In the early stages of the tumor (Stages I and II), surgical removal and radiation therapy are highly effective, but 70% of patients are diagnosed with later stages of the disease—III or IV, where the effectiveness of these methods of therapy sharply decreases [9][10]. The ineffectiveness of therapy in a number of patients dictates the need for a deeper understanding of the mechanisms of the pathogenesis of head and neck tumors. To solve this problem, many studies have been conducted to study the properties and relationships between tumor cells and the microenvironment. HNSCC is characterized by complex relationships between stromal, epithelial, and immune cells in the tumor microenvironment (TME) [11][12][13]. The HNSCC TME includes various cells, both stromal and immune, as well as the extracellular matrix (ECM), blood, and lymphatic vessels [12][14]. Recently, a number of authors have emphasized the role of stromal cells in the induction and maintenance of tumors [15][16][17] and have also shown the relationship stromal cells have with the resistance of tumor cells to therapy [18].
2. Vascular Component of the HNSCC TME
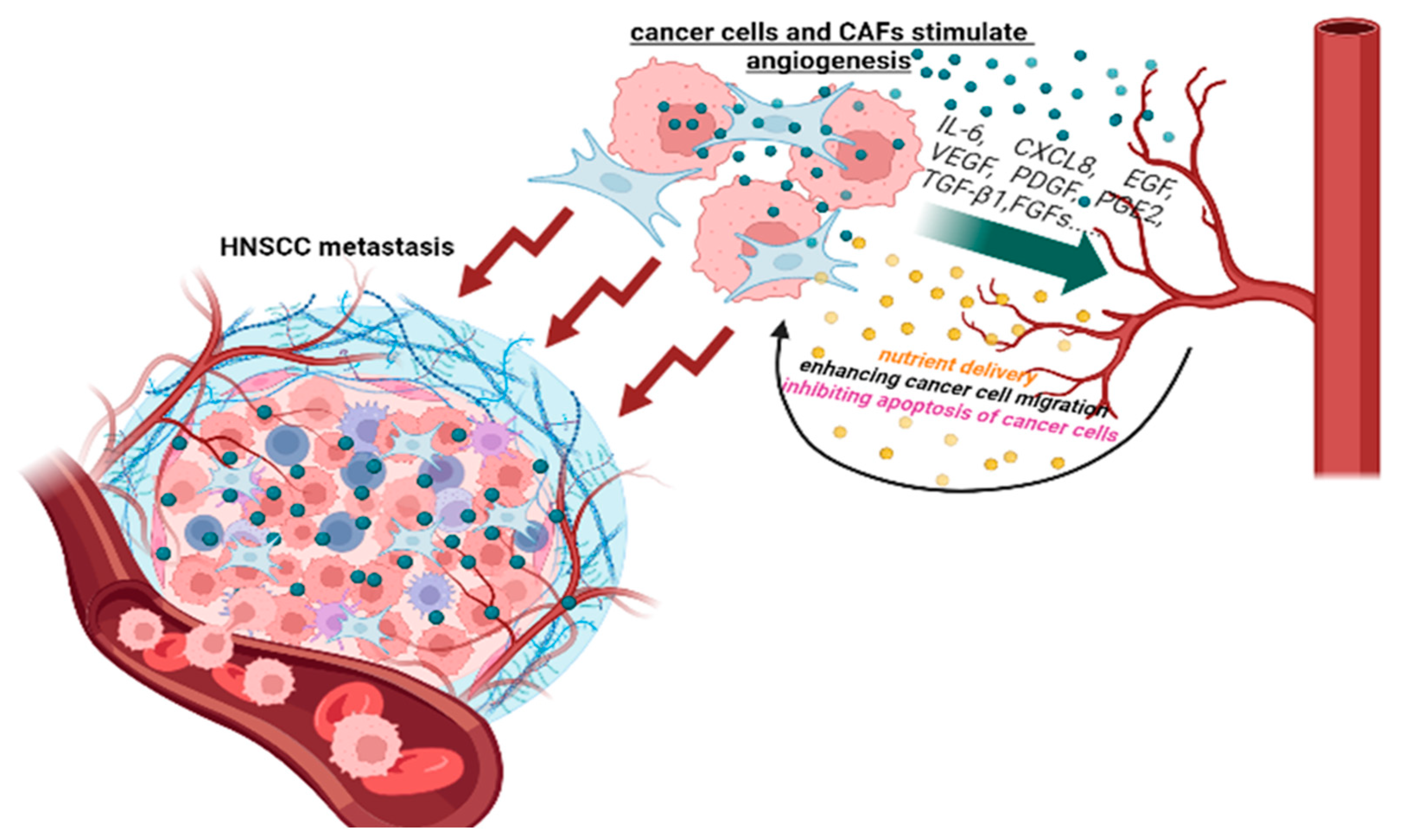
3. Role of the ECM in HNSCC
| Main Components of the HNSCC ECM | What Are the Effects on HNSCC Progression? | |
|---|---|---|
| Collagens |
|
|
| Elastin |
|
|
| Fibronectin |
|
|
| Laminins |
|
|
| Hyaluronan |
|
|
| Tenascin-C |
|
|
| Integrins |
|
|
| MMPs | MMP2 |
|
| MMP9 |
|
|
| MMP13 |
|
|
| MMP14 |
|
|
| MMP17 | ||
| MMP20 |
|
|
| Perlecan |
|
|
4. Conclusions
The TME is a key component of this cascade of events, as it is involved in phenotype changes (cancer cells acquire a stem cell phenotype, immune cells acquire a pro-tumor phenotype) and the modulation of the behavior of both tumor cells and all cellular components of tumor tissue. Analyzing the relationships between TME components, one gains an impression of some “vicious circles” that condition the aggressive potential of HNSCC. Understanding the peculiarities of heterotypic interactions between cancer cells, new vessels, CAF, ECM, and hypoxia will allow us to understand the mechanisms mediating rapid tumor progression and its resistance to antitumor drugs and radiation therapy. Basic research aimed at studying the role of TME components in HNSCC carcinogenesis may serve as a key to the discovery of both new biomarkers–predictors of prognosis and targets for new antitumor strategies.
References
- Nordemar, S.; Kronenwett, U.; Auer, G.; Högmo, A.; Lindholm, J.; Edström, S.; Tryggvasson, K.; Linder, S.; Munck-Wikland, E. Laminin-5 as a predictor of invasiveness in cancer in situ lesions of the larynx. Anticancer Res. 2001, 21, 509–512.
- Berndt, A.; Hyckel, P.; Könneker, A.; Katenkamp, D.; Kosmehl, H. Oral squamous cell carcinoma invasion is associated with a laminin-5 matrix re-organization but independent of basement membrane and hemidesmosome formation. Clues from an in vitro invasion model. Invasion Metastasis 1997, 17, 251–258.
- Meireles Da Costa, N.; Mendes, F.A.; Pontes, B.; Nasciutti, L.E.; Ribeiro Pinto, L.F.; Palumbo Júnior, A. Potential therapeutic significance of laminin in head and neck squamous carcinomas. Cancers 2021, 13, 1890.
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F.; Bsc, M.F.B.; Me, J.F.; Soerjomataram, M.I.; et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249.
- Johnson, D.E.; Burtness, B.; Leemans, C.R.; Lui, V.W.Y.; Bauman, J.E.; Grandis, J.R. Head and neck squamous cell carcinoma. Nat. Rev. Dis. Primers 2020, 6, 92.
- Lawrence, M.S.; Sougnez, C.; Lichtenstein, L.; Cibulskis, K.; Lander, E.; Gabriel, S.B.; Liu, W.; Lu, Y.; Mills, G.; Motter, T.; et al. Comprehensive genomic characterization of head and neck squamous cell carcinomas. Nature 2015, 517, 576–582.
- Zhang, P.; Li, S.; Zhang, T.; Cui, F.; Shi, J.H.; Zhao, F.; Sheng, X. Characterization of Molecular Subtypes in Head and Neck Squamous Cell Carcinoma with Distinct Prognosis and Treatment Responsiveness. Front. Cell Dev. Biol. 2021, 9, 711348.
- Ebrahimi, A.; Gil, Z.; Amit, M.; Yen, T.C.; Liao, C.T.; Chaturvedi, P.; Agarwal, J.P.; Kowalski, L.P.; Kreppel, M.; Cernea, C.R.; et al. Primary tumor staging for oral cancer and a proposed modification incorporating depth of invasion: An international multicenter retrospective study. JAMA Otolaryngol. Head Neck Surg. 2014, 140, 1138–1148.
- Zanoni, D.K.; Patel, S.G.; Shah, J.P. Changes in the 8th Edition of the American Joint Committee on Cancer (AJCC) Staging of Head and Neck Cancer: Rationale and Implications. Curr. Oncol. Rep. 2019, 21, 52.
- Nosé, V.; Lazar, A.J. Update from the 5th Edition of the World Health Organization Classification of Head and Neck Tumors: Familial Tumor Syndromes. Head Neck Pathol. 2022, 16, 143–157.
- Badoual, C. Update from the 5th Edition of the World Health Organization Classification of Head and Neck Tumors: Oropharynx and Nasopharynx. Head Neck Pathol. 2022, 16, 19–30.
- Lemaire, F.; Millon, R.; Young, J.; Cromer, A.; Wasylyk, C.; Schultz, I.; Muller, D.; Marchal, P.; Zhao, C.; Melle, D.; et al. Differential expression profiling of head and neck squamous cell carcinoma (HNSCC). Br. J. Cancer 2003, 89, 1940–1949.
- Saussez, S.; Duray, A.; Demoulin, S.; Hubert, P.; Delvenne, P. Immune suppression in head and neck cancers: A review. Clin. Dev. Immunol. 2010, 2010, 701657.
- Plzák, J.; Bouček, J.; Bandúrová, V.; Kolář, M.; Hradilová, M.; Szabo, P.; Lacina, L.; Chovanec, M.; Smetana, K. The head and neck squamous cell carcinoma microenvironment as a potential target for cancer therapy. Cancers 2019, 11, 440.
- Wang, G.; Zhang, M.; Cheng, M.; Wang, X.; Li, K.; Chen, J.; Chen, Z.; Chen, S.; Chen, J.; Xiong, G.; et al. Tumor microenvironment in head and neck squamous cell carcinoma: Functions and regulatory mechanisms. Cancer Lett. 2021, 507, 55–69.
- Elmusrati, A.; Wang, J.; Wang, C.Y. Tumor microenvironment and immune evasion in head and neck squamous cell carcinoma. Int. J. Oral Sci. 2021, 13, 24.
- Petrova, V.; Annicchiarico-Petruzzelli, M.; Melino, G.; Amelio, I. The hypoxic tumour microenvironment. Oncogenesis 2018, 7, 10.
- Castells, M.; Thibault, B.; Delord, J.P.; Couderc, B. Implication of tumor microenvironment in chemoresistance: Tumor-associated stromal cells protect tumor cells from cell death. Int. J. Mol. Sci. 2012, 13, 9545–9571.
- Almangush, A.; Alabi, R.O.; Troiano, G.; Coletta, R.D.; Salo, T.; Pirinen, M.; Mäkitie, A.A.; Leivo, I. Clinical significance of tumor-stroma ratio in head and neck cancer: A systematic review and meta-analysis. BMC Cancer 2021, 21, 480.
- Meads, M.B.; Gatenby, R.A.; Dalton, W.S. Environment-mediated drug resistance: A major contributor to minimal residual disease. Nat. Rev. Cancer 2009, 9, 665–674.
- Jumaniyazova, E.; Lokhonina, A.; Dzhalilova, D.; Kosyreva, A.; Fatkhudinov, T. Immune Cells in Head-and-Neck Tumor Microenvironments. J. Pers. Med. 2022, 12, 1521.
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674.
- Kyzas, P.A.; Cunha, I.W.; Ioannidis, J.P.A. Prognostic significance of vascular endothelial growth factor immunohistochemical expression in head and neck squamous cell carcinoma: A meta-analysis. Clin. Cancer Res. 2005, 11, 1434–1440.
- Rad, H.S.; Shiravand, Y.; Radfar, P.; Ladwa, R.; Perry, C.; Han, X.; Warkiani, M.E.; Adams, M.N.; Hughes, B.G.; O’Byrne, K.; et al. Understanding the tumor microenvironment in head and neck squamous cell carcinoma. Clin. Transl. Immunol. 2022, 11, e1397.
- Lee, P.; Chandel, N.S.; Simon, M.C. Cellular adaptation to hypoxia through hypoxia inducible factors and beyond. Nat. Rev. Mol. Cell Biol. 2020, 21, 268–283.
- Kato, Y.; Ozawa, S.; Miyamoto, C.; Maehata, Y.; Suzuki, A.; Maeda, T.; Baba, Y. Acidic extracellular microenvironment and cancer. Cancer Cell Int. 2013, 13, 89.
- Lugano, R.; Ramachandran, M.; Dimberg, A. Tumor angiogenesis: Causes, consequences, challenges and opportunities. Cell. Mol. Life Sci. 2020, 77, 1745–1770.
- Watanabe, S.; Kato, M.; Kotani, I.; Ryoke, K.; Hayashi, K. Lymphatic vessel density and vascular endothelial growth factor expression in squamous cell carcinomas of lip and oral cavity: A clinicopathological analysis with immunohistochemistry using antibodies to D2-40, VEGF-C and VEGF-D. Yonago Acta Med. 2013, 56, 29–37.
- Christopoulos, A.; Ahn, S.M.; Klein, J.D.; Kim, S. Biology of vascular endothelial growth factor and its receptors in head and neck cancer: Beyond angiogenesis. Head Neck 2010, 33, 1220–1229.
- Carla, C.; Daris, F.; Cecilia, B.; Francesca, B.; Francesca, C.; Paolo, F. Angiogenesis in head and neck cancer: A review of the literature. J. Oncol. 2011, 2012, 358472.
- Pradeep, C.R.; Sunila, E.S.; Kuttan, G. Expression of vascular endothelial growth factor (VEGF) and VEGF receptors in tumor angiogenesis and malignancies. Integr. Cancer Ther. 2005, 4, 315–321.
- Dumitru, C.S.; Raica, M. Vascular Endothelial Growth Factor Family and Head and Neck Squamous Cell Carcinoma. Anticancer Res. 2023, 43, 4315–4326.
- Kim, K.J.; Cho, C.S.; Kim, W.U. Role of placenta growth factor in cancer and inflammation. Exp. Mol. Med. 2011, 44, 10–19.
- Shibuya, M. Vascular Endothelial Growth Factor (VEGF) and Its Receptor (VEGFR) Signaling in Angiogenesis: A Crucial Target for Anti- and Pro-Angiogenic Therapies. Genes Cancer 2011, 2, 1097–1105.
- Apte, R.S.; Chen, D.S.; Ferrara, N. VEGF in Signaling and Disease: Beyond Discovery and Development. Cell 2019, 176, 1248–1264.
- Holmes, K.; Roberts, O.L.; Thomas, A.M.; Cross, M.J. Vascular endothelial growth factor receptor-2: Structure, function, intracellular signalling and therapeutic inhibition. Cell. Signal. 2007, 19, 2003–2012.
- Rizzolio, S.; Tamagnone, L. Multifaceted Role of Neuropilins in Cancer. Curr. Med. Chem. 2011, 18, 3563–3575.
- Ellis, L.M. The role of neuropilins in cancer. Mol. Cancer Ther. 2006, 5, 1099–1107.
- Wang, S.; Li, X.; Parra, M.; Verdin, E.; Bassel-Duby, R.; Olson, E.N. Control of endothelial cell proliferation and migration by VEGF signaling to histone deacetylase 7. Proc. Natl. Acad. Sci. USA 2008, 105, 7738–7743.
- Olsson, A.K.; Dimberg, A.; Kreuger, J.; Claesson-Welsh, L. VEGF receptor signalling—In control of vascular function. Nat. Rev. Mol. Cell Biol. 2006, 7, 359–371.
- Breslin, J.W.; Pappas, P.J.; Cerveira, J.J.; Hobson, R.W.; Duran, W.N. VEGF increases endothelial permeability by separate signaling pathways involving ERK-1/2 and nitric oxide. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H92–H100.
- Gupta, K.; Kshirsagar, S.; Li, W.; Gui, L.; Ramakrishnan, S.; Gupta, P.; Law, P.Y.; Hebbel, R.P. VEGF prevents apoptosis of human microvascular endothelial cells via opposing effects on MAPK/ERK and SAPK/JNK signaling. Exp. Cell Res. 1999, 247, 495–504.
- Reinmuth, N.; Liu, W.; Jung, Y.D.; Ahmad, S.A.; Shaheen, R.M.; Fan, F.; Bucana, C.D.; McMahon, G.; Gallick, G.E.; Ellis, L.M. Induction of VEGF in perivascular cells defines a potential paracrine mechanism for endothelial cell survival. FASEB J. 2001, 15, 1239–1241.
- Shigetomi, S.; Imanishi, Y.; Shibata, K.; Sakai, N.; Sakamoto, K.; Fujii, R.; Habu, N.; Otsuka, K.; Sato, Y.; Watanabe, Y.; et al. VEGF-C/Flt-4 axis in tumor cells contributes to the progression of oral squamous cell carcinoma via upregulating VEGF-C itself and contactin-1 in an autocrine manner. Am. J. Cancer Res. 2018, 8, 2046–2063.
- Campos, M.S.; Neiva, K.G.; Meyers, K.A.; Krishnamurthy, S.; Nör, J.E. Endothelial derived factors inhibit anoikis of head and neck cancer stem cells. Oral Oncol. 2012, 48, 26–32.
- Ziogas, A.C.; Gavalas, N.G.; Tsiatas, M.; Tsitsilonis, O.; Politi, E.; Terpos, E.; Rodolakis, A.; Vlahos, G.; Thomakos, N.; Haidopoulos, D.; et al. VEGF directly suppresses activation of T cells from ovarian cancer patients and healthy individuals via VEGF receptor Type 2. Int. J. Cancer 2011, 130, 857–864.
- Micaily, I.; Johnson, J.; Argiris, A. An update on angiogenesis targeting in head and neck squamous cell carcinoma. Cancers Head Neck 2020, 5, 5–7.
- Quail, D.F.; Joyce, J.A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 2013, 19, 1423–1437.
- Weis, S.M.; Cheresh, D.A. Tumor angiogenesis: Molecular pathways and therapeutic targets. Nat. Med. 2011, 17, 1359–1370.
- Curry, J.M.; Sprandio, J.; Cognetti, D.; Luginbuhl, A.; Bar-Ad, V.; Pribitkin, E.; Tuluc, M. Tumor microenvironment in head and neck squamous cell carcinoma. Semin. Oncol. 2014, 41, 217–234.
- Neiva, K.G.; Zhang, Z.; Miyazawa, M.; Warner, K.A.; Karl, E.; Nör, J.E. Cross talk initiated by endothelial cells enhances migration and inhibits anoikis of squamous cell carcinoma cells through STAT3/Akt/ERK signaling. Neoplasia 2009, 11, 583–593.
- Lu, S.L.; Herrington, H.; Reh, D.; Weber, S.; Bornstein, S.; Wang, D.; Li, A.G.; Tang, C.-F.; Siddiqui, Y.; Nord, J.; et al. Loss of transforming growth factor-β type II receptor promotes metastatic head-and-neck squamous cell carcinoma. Genes Dev. 2006, 20, 1331–1342.
- Smith, B.D.; Smith, G.L.; Carter, D.; Sasaki, C.T.; Haffty, B.G. Prognostic significance of Vascular Endothelial Growth Factor protein levels in oral and oropharyngeal squamous cell carcinoma. J. Clin. Oncol. 2000, 18, 2046–2052.
- Mineta, H.; Miura, K.; Ogino, T.; Takebayashi, S.; Misawa, K.; Ueda, Y.; Suzuki, I.; Dictor, M.; Borg, Å.; Wennerberg, J. Prognostic value of vascular endothelial growth factor (VEGF) in head and neck squamous cell carcinomas. Br. J. Cancer 2000, 83, 775–781.
- Tse, G.M.; Chan, A.W.H.; Yu, K.H.; King, A.D.; Wong, K.T.; Chen, G.G.; Tsang, R.K.Y.; Chan, A.B.W. Strong immunohistochemical expression of vascular endothelial growth factor predicts overall survival in head and neck squamous cell carcinoma. Ann. Surg. Oncol. 2007, 14, 3558–3565.
- El-Gazzar, R.; Macluskey, M.; Williams, H.; Ogden, G.R. Vascularity and expression of vascular endothelial growth factor in oral squamous cell carcinoma, resection margins, and nodal metastases. Br. J. Oral Maxillofac. Surg. 2006, 44, 193–197.
- Shang, Z.J.; Li, J.R.; Li, Z.B. Circulating levels of vascular endothelial growth factor in patients with oral squamous cell carcinoma. Int. J. Oral Maxillofac. Surg. 2002, 31, 495–498.
- Raudenská, M.; Svobodová, M.; Gumulec, J.; Falk, M.; Masařík, M. The importance of cancer-associated fibroblasts in the pathogenesis of head and neck cancers. Klin. Onkol. 2020, 33, 39–48.
- Zhang, X.; Dong, Y.; Zhao, M.; Ding, L.; Yang, X.; Jing, Y.; Song, Y.; Chen, S.; Hu, Q.; Ni, Y. ITGB2-mediated metabolic switch in CAFs promotes OSCC proliferation by oxidation of NADH in mitochondrial oxidative phosphorylation system. Theranostics 2020, 10, 12044.
- Kumar, D.; New, J.; Vishwakarma, V.; Joshi, R.; Enders, J.; Lin, F.; Dasari, S.; Gutierrez, W.R.; Leef, G.; Ponnurangam, S.; et al. Cancer-associated fibroblasts drive glycolysis in a targetable signaling loop implicated in head and neck squamous cell carcinoma progression. Cancer Res 2018, 78, 3769–3782.
- Pickup, M.W.; Mouw, J.K.; Weaver, V.M. The extracellular matrix modulates the hallmarks of cancer. Embo Rep. 2014, 15, 1243–1253.
- Rigoglio, N.N.; Rabelo, A.C.S.; Borghesi, J.; de Sá Schiavo Matias, G.; Fratini, P.; Prazeres, P.H.D.M.; Pimentel, C.M.M.M.; Birbrair, A.; Miglino, M.A. The tumor microenvironment: Focus on extracellular matrix. In Advances in Experimental Medicine and Biology; Springer: Cham, Switzerland, 2020.
- Clause, K.C.; Barker, T.H. Extracellular matrix signaling in morphogenesis and repair. Curr. Opin. Biotechnol. 2013, 24, 830–833.
- Walker, C.; Mojares, E.; Del Río Hernández, A. Role of extracellular matrix in development and cancer progression. Int. J. Mol. Sci. 2018, 19, 3028.
- Provenzano, P.P.; Inman, D.R.; Eliceiri, K.W.; Knittel, J.G.; Yan, L.; Rueden, C.T.; White, J.G.; Keely, P.J. Collagen density promotes mammary tumor initiation and progression. BMC Med. 2008, 6, 11.
- Mammoto, T.; Jiang, A.; Jiang, E.; Panigrahy, D.; Kieran, M.W.; Mammoto, A. Role of collagen matrix in tumor angiogenesis and glioblastoma multiforme progression. Am. J. Pathol. 2013, 183, 1293–1305.
- Provenzano, P.P.; Hingorani, S.R. Hyaluronan, fluid pressure, and stromal resistance in pancreas cancer. Br. J. Cancer 2013, 108, 1–8.
- Cox, T.R.; Erler, J.T. Remodeling and homeostasis of the extracellular matrix: Implications for fibrotic diseases and cancer. Dis. Model. Mech. 2011, 4, 165–178.
- Sala, M.; Ros, M.; Saltel, F. A Complex and Evolutive Character: Two Face Aspects of ECM in Tumor Progression. Front. Oncol. 2020, 10, 1620.
- Le, Q.T.; Harris, J.; Magliocco, A.M.; Kong, C.S.; Diaz, R.; Shin, B.; Cao, H.; Trotti, A.; Erler, J.T.; Chung, C.H.; et al. Validation of lysyl oxidase as a prognostic marker for metastasis and survival in head and neck squamous cell carcinoma: Radiation Therapy Oncology Group trial 90-03. J. Clin. Oncol. 2009, 27, 4281–4286.
- Kass, L.; Erler, J.T.; Dembo, M.; Weaver, V.M. Mammary epithelial cell: Influence of extracellular matrix composition and organization during development and tumorigenesis. Cell Biol. 2007, 39, 1987–1994.
- Huang, J.; Zhang, L.; Wan, D.; Zhou, L.; Zheng, S.; Lin, S.; Qiao, Y. Extracellular matrix and its therapeutic potential for cancer treatment. Signal Transduct. Target. Ther. 2021, 6, 153.
- Saint, A.; Van Obberghen-Schillinga, E. The role of the tumor matrix environment in progression of head and neck cancer. Curr. Opin. Oncol. 2021, 33, 168–174.
- Hynes, R.O. The extracellular matrix: Not just pretty fibrils. Science 2009, 326, 1216–1219.
- Puram, S.V.; Tirosh, I.; Parikh, A.S.; Patel, A.P.; Yizhak, K.; Gillespie, S.; Rodman, C.; Luo, C.L.; Mroz, E.A.; Emerick, K.S.; et al. Single-Cell Transcriptomic Analysis of Primary and Metastatic Tumor Ecosystems in Head and Neck Cancer. Cell 2017, 171, 1611–1624.e24.
- Peltanova, B.; Raudenska, M.; Masarik, M. Effect of tumor microenvironment on pathogenesis of the head and neck squamous cell carcinoma: A systematic review. Mol. Cancer 2019, 18, 63.
- Gong, Y.; Bao, L.; Xu, T.; Yi, X.; Chen, J.; Wang, S.; Pan, Z.; Huang, P.; Ge, M. The tumor ecosystem in head and neck squamous cell carcinoma and advances in ecotherapy. Mol. Cancer 2023, 22, 68.
- Naba, A.; Clauser, K.R.; Ding, H.; Whittaker, C.A.; Carr, S.A.; Hynes, R.O. The extracellular matrix: Tools and insights for the “omics” era. Matrix Biol. 2016, 49, 10–24.
- Gopal, S.; Veracini, L.; Grall, D.; Butori, C.; Schaub, S.; Audebert, S.; Camoin, L.; Baudelet, E.; Radwanska, A.; Beghelli-de la Forest Divonne, S.; et al. Fibronectin-guided migration of carcinoma collectives. Nat. Commun. 2017, 8, 14105.
- Tian, C.; Clauser, K.R.; Öhlund, D.; Rickelt, S.; Huang, Y.; Gupta, M.; Mani, D.R.; Carr, S.A.; Tuveson, D.A.; Hynes, R.O. Proteomic analyses of ECM during pancreatic ductal adenocarcinoma progression reveal different contributions by tumor and stromal cells. Proc. Natl. Acad. Sci. USA 2019, 116, 19609–19618.
- Acerbi, I.; Cassereau, L.; Dean, I.; Shi, Q.; Au, A.; Park, C.; Chen, Y.Y.; Liphardt, J.; Hwang, E.S.; Weaver, V.M. Human breast cancer invasion and aggression correlates with ECM stiffening and immune cell infiltration. Integr. Biol. 2015, 7, 1120–1134.
- Maller, O.; Drain, A.P.; Barrett, A.S.; Borgquist, S.; Ruffell, B.; Zakharevich, I.; Pham, T.T.; Gruosso, T.; Kuasne, H.; Lakins, J.N.; et al. Tumour-associated macrophages drive stromal cell-dependent collagen crosslinking and stiffening to promote breast cancer aggression. Nat. Mater. 2020, 20, 548–559.
- Provenzano, P.P.; Eliceiri, K.W.; Campbell, J.M.; Inman, D.R.; White, J.G.; Keely, P.J. Collagen reorganization at the tumor-stromal interface facilitates local invasion. BMC Med. 2006, 4, 38.
- Lai, S.L.; Tan, M.L.; Hollows, R.J.; Robinson, M.; Ibrahim, M.; Margielewska, S.; Parkinson, E.K.; Ramanathan, A.; Zain, R.B.; Mehanna, H.; et al. Collagen induces a more proliferative, migratory and chemoresistant phenotype in head and neck cancer via DDR1. Cancers 2019, 11, 1766.
- Itoh, Y. Discoidin domain receptors: Microenvironment sensors that promote cellular migration and invasion. Cell Adhes. Migr. 2018, 12, 378–385.
- Leitinger, B. Discoidin domain receptor functions in physiological and pathological conditions. Int. Rev. Cell Mol. Biol. 2014, 310, 39–87.
- Hidalgo-Carcedo, C.; Hooper, S.; Chaudhry, S.I.; Williamson, P.; Harrington, K.; Leitinger, B.; Sahai, E. Collective cell migration requires suppression of actomyosin at cell-cell contacts mediated by DDR1 and the cell polarity regulators Par3 and Par6. Nat. Cell Biol. 2010, 13, 49–59.
- Cox, T.R.; Gartland, A.; Erler, J.T. Lysyl oxidase, a targetable secreted molecule involved in cancer metastasis. Cancer Res 2016, 76, 188–192.
- Mayorca-Guiliani, A.; Erler, J.T. The potential for targeting extracellular LOX proteins in human malignancy. OncoTargets Ther. 2013, 6, 1729–1735.
- Sanada, T.; Islam, A.; Kaminota, T.; Kirino, Y.; Tanimoto, R.; Yoshimitsu, H.; Yano, H.; Mizuno, Y.; Okada, M.; Mitani, S.; et al. Elevated exosomal lysyl oxidase like 2 is a potential biomarker for head and neck squamous cell carcinoma. Laryngoscope 2019, 130, E327–E334.
- Mayorca-Guiliani, A.E.; Yano, H.; Nakashiro, K.I.; Hamakawa, H.; Tanaka, J. Premetastatic vasculogenesis in oral squamous cell carcinoma xenograft-draining lymph nodes. Oral Oncol. 2012, 48, 663–670.
- Zhou, W.H.; Du, W.D.; Li, Y.F.; Al-Aroomi, M.A.; Yan, C.; Wang, Y.; Zhang, Z.Y.; Liu, F.Y.; Sun, C.F. The Overexpression of Fibronectin 1 Promotes Cancer Progression and Associated with M2 Macrophages Polarization in Head and Neck Squamous Cell Carcinoma Patients. Int. J. Gen. Med. 2022, 15, 5027–5042.
- Marzeda, A.M.; Midwood, K.S. Internal Affairs: Tenascin-C as a Clinically Relevant, Endogenous Driver of Innate Immunity. J. Histochem. Cytochem. 2018, 66, 289–304.
- Phillips, G.R.; Krushel, L.A.; Crossin, K.L. Domains of tenascin involved in glioma migration. J. Cell Sci. 1998, 111, 1095–1104.
- Schenk, S.; Chiquet-Ehrismann, R.; Battegay, E.J. The fibrinogen globe of tenascin-C promotes basic fibroblast growth factor-induced endothelial cell elongation. Mol. Biol. Cell 1999, 10, 2933–2943.
- Tremble, P.; Chiquet-Ehrismann, R.; Werb, Z. The extracellular matrix ligands fibronectin and tenascin collaborate in regulating collagenase gene expression in fibroblasts. Mol. Biol. Cell 1994, 5, 439–453.
- Sternlicht, M.D.; Lochtest, A.; Sympson, C.J.; Huey, B.; Rougier, J.P.; Gray, J.W.; Pinkel, D.; Bissell, M.J.; Werb, Z. The stromal proteinase MMP3/stromelysin-1 promotes mammary carcinogenesis. Cell 1999, 98, 137–146.
- Spenle, C.; Loustau, T.; Murdamoothoo, D.; Erne, W.; Beghelli-De la Forest Divonne, S.; Veber, R.; Petti, L.; Bourdely, P.; Mörgelin, M.; Brauchle, E.-M.; et al. Tenascin-C orchestrates an immune-suppressive tumor microenvironment in oral squamous cell carcinoma. Cancer Immunol. Res. 2020, 8, 1122–1138.
- Marangon Junior, H.; Rocha, V.N.; Leite, C.F.; de Aguiar, M.C.F.; Souza, P.E.A.; Horta, M.C.R. Laminin-5 gamma 2 chain expression is associated with intensity of tumor budding and density of stromal myofibroblasts in oral squamous cell carcinoma. J. Oral Pathol. Med. 2013, 43, 199–204.
- Leitinger, B. Transmembrane collagen receptors. Annu. Rev. Cell Dev. Biol. 2011, 27, 265–290.
- Beaulieu, J.F. Integrin α6β4 in colorectal cancer: Expression, regulation, functional alterations and use as a biomarker. Cancers 2020, 12, 41.
- Li, H.X.; Zheng, J.H.; Fan, H.X.; Li, H.P.; Gao, Z.X.; Chen, D. Expression of αvβ6 integrin and collagen fibre in oral squamous cell carcinoma: Association with clinical outcomes and prognostic implications. J. Oral Pathol. Med. 2013, 42, 547–556.
- Regezi, J.A.; Ramos, D.M.; Pytela, R.; Dekker, N.P.; Jordan, R.C.K. Tenascin and β6 integrin are overexpressed in floor of mouth in situ carcinomas and invasive squamous cell carcinomas. Oral Oncol. 2002, 38, 332–336.
- Fan, Q.C.; Tian, H.; Wang, Y.; Liu, X.B. Integrin-α5 promoted the progression of oral squamous cell carcinoma and modulated PI3K/AKT signaling pathway. Arch. Oral Biol. 2019, 101, 85–91.
- Niu, J.; Li, Z. The roles of integrin αvβ6 in cancer. Cancer Lett. 2017, 403, 128–137.
- Xiong, H.; Hong, J.; Du, W.; Lin, Y.W.; Ren, L.L.; Wang, Y.C.; Su, W.Y.; Wang, J.L.; Cui, Y.; Wang, Z.H.; et al. Roles of STAT3 and ZEB1 proteins in E-cadherin down-regulation and human colorectal cancer epithelial-mesenchymal transition. J. Biol. Chem. 2012, 287, 5819–5832.
- Gómez-Herrera, Z.; Molina-Frechero, N.; Damián-Matsumura, P.; Bologna-Molina, R. Proteoglycans as potential biomarkers in odontogenic tumors. J. Oral Maxillofac. Pathol. 2018, 22, 98–103.
- Gubbiotti, M.A.; Neill, T.; Iozzo, R.V. A current view of perlecan in physiology and pathology: A mosaic of functions. Matrix Biol. 2017, 57–58, 285–298.
- Theocharis, A.D.; Karamanos, N.K. Proteoglycans remodeling in cancer: Underlying molecular mechanisms. Matrix Biol. 2019, 75–76, 220–259.
- Pereira, B.A.; Vennin, C.; Papanicolaou, M.; Chambers, C.R.; Herrmann, D.; Morton, J.P.; Cox, T.R.; Timpson, P. CAF Subpopulations: A New Reservoir of Stromal Targets in Pancreatic Cancer. Trends Cancer 2019, 5, 724–741.
- Netti, P.A.; Berk, D.A.; Swartz, M.A.; Grodzinsky, A.J.; Jain, R.K. Role of extracellular matrix assembly in interstitial transport in solid tumors. Cancer Res. 2000, 60, 2497–2503.
- Graham, K.; Unger, E. Overcoming tumor hypoxia as a barrier to radiotherapy, chemotherapy and immunotherapy in cancer treatment. Int. J. Nanomed. 2018, 13, 6049–6058.
- Hodi, F.S.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.J.; Rutkowski, P.; Cowey, C.L.; Lao, C.D.; Schadendorf, D.; Wagstaff, J.; Dummer, R.; et al. Nivolumab plus ipilimumab or nivolumab alone versus ipilimumab alone in advanced melanoma (CheckMate 067): 4-year outcomes of a multicentre, randomised, phase 3 trial. Lancet Oncol. 2018, 19, 1480–1492.
- Raavé, R.; van Kuppevelt, T.H.; Daamen, W.F. Chemotherapeutic drug delivery by tumoral extracellular matrix targeting. J. Control. Release 2018, 274, 1–8.
- Najafi, M.; Farhood, B.; Mortezaee, K. Extracellular matrix (ECM) stiffness and degradation as cancer drivers. J. Cell. Biochem. 2018, 120, 2782–2790.
- Piersma, B.; Hayward, M.K.; Weaver, V.M. Fibrosis and cancer: A strained relationship. Biochim. Biophys. Acta Rev. Cancer 2020, 1873, 188356.
- Henke, E.; Nandigama, R.; Ergün, S. Extracellular Matrix in the Tumor Microenvironment and Its Impact on Cancer Therapy. Front. Mol. Biosci. 2020, 6, 160.
- Hallmann, R.; Zhang, X.; Di Russo, J.; Li, L.; Song, J.; Hannocks, M.J.; Sorokin, L. The regulation of immune cell trafficking by the extracellular matrix. Curr. Opin. Cell Biol. 2015, 36, 54–61.
- Wei, J.; Wu, A.; Kong, L.Y.; Wang, Y.; Fuller, G.; Fokt, I.; Melillo, G.; Priebe, W.; Heimberger, A.B. Hypoxia potentiates glioma-mediated immunosuppression. PLoS ONE 2011, 6, e16195.
- Schaaf, M.B.; Garg, A.D.; Agostinis, P. Defining the role of the tumor vasculature in antitumor immunity and immunotherapy article. Cell Death Disease 2018, 9, 115.
- Ostroukhova, M.; Qi, Z.; Oriss, T.B.; Dixon-McCarthy, B.; Ray, P.; Ray, A. Treg-mediated immunosuppression involves activation of the Notch-HES1 axis by membrane-bound TGF-β. J. Clin. Investig. 2006, 116, 996–1004.
- Zhang, F.; Wang, H.; Wang, X.; Jiang, G.; Liu, H.; Zhang, G.; Wang, H.; Fang, R.; Bu, X.; Cai, S.; et al. TGF-β induces M2-like macrophage polarization via SNAILmediated suppression of a pro-inflammatory phenotype. Oncotarget 2016, 7, 52294–52306.
- Shibue, T.; Weinberg, R.A. EMT, CSCs, and drug resistance: The mechanistic link and clinical implications. Nat. Rev. Clin. Oncol. 2017, 14, 611–629.
- Fischer, K.R.; Durrans, A.; Lee, S.; Sheng, J.; Choi, H.; Li, F.; Wong, S.; Altorki, N.K.; Mittal, V.; Gao, D. Abstract 4721: Epithelial to mesenchymal transition is not required for breast to lung metastasis but contributes to chemoresistance. Cancer Res. 2015, 75 (Suppl. S15), 4721.
- Poltavets, V.; Kochetkova, M.; Pitson, S.M.; Samuel, M.S. The role of the extracellular matrix and its molecular and cellular regulators in cancer cell plasticity. Front. Oncol. 2018, 8, 431.
- Branch, K.M.; Hoshino, D.; Weaver, A.M. Adhesion rings surround invadopodia and promote maturation. Biol. Open 2012, 1, 711–722.
- Hu, C.; Zhang, Y.; Wu, C.; Huang, Q. Heterogeneity of cancer-associated fibroblasts in head and neck squamous cell carcinoma: Opportunities and challenges. Cell Death Discov. 2023, 9, 124.
- Leong, H.S.; Robertson, A.E.; Stoletov, K.; Leith, S.J.; Chin, C.A.; Chien, A.E.; Hague, M.N.; Ablack, A.; Carmine-Simmen, K.; McPherson, V.A.; et al. Invadopodia Are Required for Cancer Cell Extravasation and Are a Therapeutic Target for Metastasis. Cell Rep. 2014, 8, 1558–1570.
- Paz, H.; Pathak, N.; Yang, J. Invading one step at a time: The role of invadopodia in tumor metastasis. Oncogene 2014, 33, 4193–4202.
- Kumar, K.V.; Hema, K.N. Extracellular matrix in invasion and metastasis of oral squamous cell carcinoma. J. Oral Maxillofac. Pathol. 2019, 23, 10–16.
- Culhaci, N.; Metin, K.; Copcu, E.; Dikicioglu, E. Elevated expression of MMP-13 and TIMP-1 in head and neck squamous cell carcinomas may reflect increased tumor invasiveness. BMC Cancer 2004, 4, 42.
- Niland, S.; Riscanevo, A.X.; Eble, J.A. Matrix metalloproteinases shape the tumor microenvironment in cancer progression. Int. J. Mol. Sci. 2022, 23, 146.
- Zhang, Y.; Dong, P.; Yang, L. The role of nanotherapy in head and neck squamous cell carcinoma by targeting tumor microenvironment. Front. Immunol. 2023, 14, 1189323.
- Bates, A.M.; Hernandez, M.P.G.; Lanzel, E.A.; Qian, F.; Brogden, K.A. Matrix metalloproteinase (MMP) and immunosuppressive biomarker profiles of seven head and neck squamous cell carcinoma (HNSCC) cell lines. Transl. Cancer Res. 2018, 7, 533.
- Ginos, M.A.; Page, G.P.; Michalowicz, B.S.; Patel, K.J.; Volker, S.E.; Pambuccian, S.E.; Ondrey, F.G.; Adams, G.L.; Gaffney, P.M. Identification of a Gene Expression Signature Associated with Recurrent Disease in Squamous Cell Carcinoma of the Head and Neck. Cancer Res 2004, 64, 55–63.
- Liu, M.; Huang, L.; Liu, Y.; Yang, S.; Rao, Y.; Chen, X.; Nie, M.; Liu, X. Identification of the MMP family as therapeutic targets and prognostic biomarkers in the microenvironment of head and neck squamous cell carcinoma. J. Transl. Med. 2023, 21, 208.
- Peng, C.H.; Liao, C.T.; Peng, S.C.; Chen, Y.J.; Cheng, A.J.; Juang, J.L.; Tsai, C.Y.; Chen, T.C.; Chuang, Y.J.; Tang, C.Y.; et al. A novel molecular signature identified by systems genetics approach predicts prognosis in oral squamous cell carcinoma. PLoS ONE 2011, 6, e23452.
- Ye, H.; Yu, T.; Temam, S.; Ziober, B.L.; Wang, J.; Schwartz, J.L.; Mao, L.; Wong, D.T.; Zhou, X. Transcriptomic dissection of tongue squamous cell carcinoma. BMC Genom. 2008, 9, 69.
- Rosenthal, E.L.; Matrisian, L.M. Matrix metalloproteases in head and neck cancer. Head Neck 2006, 28, 639–648.
- Koontongkaew, S.; Amornphimoltham, P.; Monthanpisut, P.; Saensuk, T.; Leelakriangsak, M. Fibroblasts and extracellular matrix differently modulate MMP activation by primary and metastatic head and neck cancer cells. Med Oncol. 2011, 29, 690–703.
- Noda, Y.; Ishida, M.; Yamaka, R.; Ueno, Y.; Sakagami, T.; Fujisawa, T.; Iwai, H.; Tsuta, K. MMP14 expression levels accurately predict the presence of extranodal extensions in oral squamous cell carcinoma: A retrospective cohort study. BMC Cancer 2023, 23, 142.
- Yang, Q.; Liu, Y.; Huang, Y.; Huang, D.; Li, Y.; Wu, J.; Duan, M. Expression of COX-2, CD44v6 and CD147 and Relationship with Invasion and Lymph Node Metastasis in Hypopharyngeal Squamous Cell Carcinoma. PLoS ONE 2013, 8, e71048.
- Su, C.W.; Su, B.F.; Chiang, W.L.; Yang, S.F.; Chen, M.K.; Lin, C.W. Plasma levels of the tissue inhibitor matrix metalloproteinase-3 as a potential biomarker in oral cancer progression. Int. J. Med. Sci. 2017, 14, 37.
- Kudo, Y.; Kitajima, S.; Ogawa, I.; Hiraoka, M.; Sargolzaei, S.; Keikhaee, M.R.; Sato, S.; Miyauchi, M.; Takata, T. Invasion and metastasis of oral cancer cells require methylation of E-cadherin and/or degradation of membranous β-catenin. Clin. Cancer Res. 2004, 10, 5455–5463.
- Costanza, B.; Umelo, I.A.; Bellier, J.; Castronovo, V.; Turtoi, A. Stromal modulators of TGF-β in cancer. J. Clin. Med. 2017, 6, 7.
- Chen, J.M.; Chen, P.Y.; Lin, C.C.; Hsieh, M.C.; Lin, J.T. Antimetastatic effects of sesamin on human head and neck squamous cell Carcinoma through regulation of matrix metalloproteinase-2. Molecules 2020, 25, 2248.
- Utispan, K.; Niyomtham, N.; Yingyongnarongkul, B.E.; Koontongkaew, S. Ethanolic Extract of Ocimum sanctum Leaves Reduced Invasion and Matrix Metalloproteinase Activity of Head and Neck Cancer Cell Lines. Asian Pac. J. Cancer Prev. 2020, 21, 363.
- Juin, A.; Billotteta, C.; Moreau, V.; Destaing, O.; Albiges-Rizo, C.; Rosenbaum, J.; Génot, E.; Saltel, F. Physiological type I collagen organization induces the formation of a novel class of linear invadosomes. Mol. Biol. Cell 2012, 23, 297–309.
- Zhong, C.; Tao, B.; Tang, F.; Yang, X.; Peng, T.; You, J.; Xia, K.; Xia, X.; Chen, L.; Peng, L. Remodeling cancer stemness by collagen/fibronectin via the AKT and CDC42 signaling pathway crosstalk in glioma. Theranostics 2021, 11, 1991–2005.
- Hayashido, Y.; Kitano, H.; Sakaue, T.; Fujii, T.; Suematsu, M.; Sakurai, S.; Okamoto, T. Overexpression of integrin αv facilitates proliferation and invasion of oral squamous cell carcinoma cells via mek/erk signaling pathway that is activated by interaction of integrin αvβ8 with type I collagen. Int. J. Oncol. 2014, 45, 1875–1882.
- Ray, A.; Provenzano, P.P. Aligned forces: Origins and mechanisms of cancer dissemination guided by extracellular matrix architecture. Curr. Opin. Cell Biol. 2021, 72, 63–71.
- Levental, K.R.; Yu, H.; Kass, L.; Lakins, J.N.; Egeblad, M.; Erler, J.T.; Fong, S.F.T.; Csiszar, K.; Giaccia, A.; Weninger, W.; et al. Matrix Crosslinking Forces Tumor Progression by Enhancing Integrin Signaling. Cell 2009, 139, 891–906.
- Prabhudesai, S.A.; Carvalho, K.; Dhupar, A.; Spadigam, A. Elastin remodeling: Does it play a role in priming the malignant phenotype of oral mucosa? Indian J. Pathol. Microbiol. 2023, 66, 332.
- Von Au, A.; Vasel, M.; Kraft, S.; Sens, C.; Hackl, N.; Marx, A.; Stroebel, P.; Hennenlotter, J.; Todenhöfer, T.; Stenzl, A.; et al. Circulating fibronectin controls tumor growth. Neoplasia 2013, 15, 925–938.
- Zhang, Y.; Lu, H.; Dazin, P.; Kapila, Y. Squamous cell carcinoma cell aggregates escape suspension-induced, p53-mediated anoikis: Fibronectin and integrin αv mediate survival signals through focal adhesion kinase. J. Biol. Chem. 2004, 279, 48342–48349.
- Degen, M.; Natarajan, E.; Barron, P.; Widlund, H.R.; Rheinwald, J.G. MAPK/ERK-dependent translation factor hyperactivation and dysregulated laminin γ2 expression in oral dysplasia and squamous cell carcinoma. Am. J. Pathol. 2012, 180, 2462–2478.
- Bourguignon, L.Y.W.; Earle, C.; Shiina, M. Activation of matrix Hyaluronan-Mediated CD44 signaling, epigenetic regulation and chemoresistance in head and neck cancer stem cells. Int. J. Mol. Sci. 2017, 18, 1849.
- O’Connell, J.T.; Sugimoto, H.; Cooke, V.G.; MacDonald, B.A.; Mehta, A.I.; LeBleu, V.S.; Dewar, R.; Rocha, R.M.; Brentani, R.R.; Resnick, M.B.; et al. VEGF-A and Tenascin-C produced by S100A4 + stromal cells are important for metastatic colonization. Proc. Natl. Acad. Sci. USA 2011, 108, 16002–16007.
- Miller, S.E.; Veale, R.B. Environmental modulation of αv, α2 and β1 integrin subunit expression in human oesophageal squamous cell carcinomas. Cell Biol. Int. 2001, 25, 61–69.
- Xuan, S.H.; Zhou, Y.G.; Pan, J.Q.; Zhu, W.; Xu, P. Overexpression of integrin αv in the human nasopharyngeal carcinoma associated with metastasis and progression. Cancer Biomark. 2013, 13, 323–328.
- Ou, J.; Luan, W.; Deng, J.; Sa, R.; Liang, H. αV integrin induces multicellular radioresistance in human nasopharyngeal carcinoma via activating SAPK/JNK pathway. PLoS ONE 2012, 7, e38737.
- Terry, S.Y.A.; Abiraj, K.; Frielink, C.; Van Dijk, L.K.; Bussink, J.; Oyen, W.J.; Boerman, O.C. Imaging integrin αvβ3 on blood vessels with 111In-RGD2 in head and neck tumor xenografts. J. Nucl. Med. 2014, 55, 281–286.
- Zhang, W.; Liu, Y.; Wang, C.W. S100A4 promotes squamous cell laryngeal cancer Hep-2 cell invasion via NF-kB/MMP-9 signal. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 1361–1367.
- Vilen, S.T.; Salo, T.; Sorsa, T.; Nyberg, P. Fluctuating roles of matrix metalloproteinase-9 in oral squamous cell carcinoma. Sci. World J. 2013, 2013, 920595.
- Kudo, Y.; Iizuka, S.; Yoshida, M.; Tsunematsu, T.; Kondo, T.; Subarnbhesaj, A.; Deraz, E.M.; Siriwardena, S.B.S.M.; Tahara, H.; Ishimaru, N.; et al. Matrix metalloproteinase-13 (MMP-13) directly and indirectly promotes tumor angiogenesis. J. Biol. Chem. 2012, 287, 38716–38728.
- Yan, X.; Cao, N.; Chen, Y.; Lan, H.Y.; Cha, J.H.; Yang, W.H.; Yang, M.H. MT4-MMP promotes invadopodia formation and cell motility in FaDu head and neck cancer cells. Biochem. Biophys. Res. Commun. 2020, 522, 1009–1014.
- Aseervatham, J.; Ogbureke, K.U.E. Effects of DSPP and MMP20 silencing on adhesion, metastasis, angiogenesis, and epithelial-mesenchymal transition proteins in oral squamous cell carcinoma cells. Int. J. Mol. Sci. 2020, 21, 4734.
- Kawahara, R.; Granato, D.C.; Carnielli, C.M.; Cervigne, N.K.; Oliveria, C.E.; Martinez, C.A.R.; Yokoo, S.; Fonseca, F.P.; Lopes, M.; Santos-Silva, A.R.; et al. Agrin and perlecan mediate tumorigenic processes in oral squamous cell carcinoma. PLoS ONE 2014, 9, e115004.





