
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Fabio Peluso | -- | 9169 | 2023-11-28 04:10:35 | | | |
| 2 | Fabio Peluso | + 1 word(s) | 9174 | 2023-11-29 14:23:00 | | |
Video Upload Options
The Dual Model of Liquids (DML) is a new mesoscopic model of liquids, whose validity and applicability was demonstrated in several cases. It is shown here that DML may even explain crossed effects of Non-Equilibrium Thermodynamics (NET). According to DML, liquids are arranged on a mesoscopic scale by means of aggregates of molecules, or liquid particles. These structures share the liquid world with a population of lattice particles, i.e., elastic waves that interact with the liquid particles by means of an inertial force, allowing the mutual exchange of energy and momentum between the two populations. The hit particle relaxes the acquired energy and momentum due to the interaction, giving them back to the system a step forward and a time-lapse later, alike in a tunnel effect. The transport phenomena in liquids out of equilibrium have been studied since their discoveries, however, no firm theoretical interpretation exists yet. It is demonstrated that the DML may correctly model the thermodiffusion, in particular getting formal expressions for positive and negative Soret coefficient, and another “unexpected” mechano-thermal effect recently discovered in liquids submitted to shear strain, for which the first-ever theoretical interpretation is provided. Both applications of the DML are supported by the comparison with experimental data. The generality of the approach allows us to customize it for other non-equilibrium phenomena of NET.
1. Introduction
Non-Equilibrium Thermodynamics (NET) is a very robust method and an extensive theory of physics to describe fluid systems, either gases or liquids (with appropriate restrictions also solids), subjected to boundary conditions that may vary vs. time and/or space [1]. NET follows from the Non-Equilibrium Statistical Mechanics that in turn, other than those of the equilibrium theory, is based on the additional relevant postulate of the time reversibility of the physical laws. Typical phenomena described with the methods of NET are those in which transport processes take place, such as for instance the heat flow crossing a medium (described by the Fourier law), the mass flow due to a concentration gradient (Fick’s law), the volume flow due to a pressure gradient (Poiseuille law), or also the electrical current due to a voltage difference applied to an electrical load (Ohm’s law). The approach most largely adopted to describe such phenomenology in NET is due to Onsager [2][3], in which it is supposed that the gradients of some physical quantities, such as temperature, mass, pressure, voltage, etc, behave as driving forces (sometimes named affinities) producing associated fluxes, such as the flux of heat, mass, volume, electrical charge, etc, similarly to the “cause-effect” relationship adopted in classical mechanics.
Apart from the phenomena listed above, there is a long list of “crossed” phenomena, in which a driving force is responsible also for other fluxes not strictly related to the applied force. Depending on which are the forces involved and the fluxes generated, such crossed effects are named, for instance, mechano-thermal, thermo-mechanical, thermo-electric, etc. Typical examples are the Soret, the Dufour, the Peltier, the Seebeck, the Thomson effects, etc. We will deal with in this paper mainly two of such coupled effects, one is the Soret effect [4][5] (with reference also to the Dufour effect), and the other is an “unexpected” mechano-thermal effect recently revealed in confined liquids under shear geometry [6][7][8][9][10][11].
The Ludwig–Soret effect, or simply Soret effect, is named after the German and Swiss researchers who in the XIX century independently observed that a temperature gradient applied to an isotropic mixture gave origin to a concentration gradient of the mixture components (other than the heat flow crossing the mixture). This phenomenon, generally named thermodiffusion, has been extensively experimentally investigated and many theoretical models have been proposed to interpret the experimental results, although none of them has provided yet a definitive explanation of all the characteristics of the Soret effect ( [12][13][14] and references therein.)
The “unexpected” mechano-thermal effect has been discovered a few years ago by the group of Noirez and co-workers [6][7][8][9][10][11]. It is highlighted in two types of similar experiments, whose difference is the motion law of a movable disk, oscillating or following a Heaviside function. The mechano-thermal effect consists in both cases of the occurrence in a liquid in shear geometry of a temperature gradient due to the momentum transferred to it by a moving plate. Therefore, this mechano-thermal effect reveals a coupling between a mechanical force induced to the liquid by the shear strain which generates in turn the thermal gradient. Many interesting papers have been published showing, for instance, that the universal law for the low-frequency shear modulus is confirmed also in confined liquids [15][16]; however, no firm theoretical explanation of such phenomenology at the mesoscopic level has been advanced yet.
In the frame of the liquid modeling at the mesoscopic scale, the Dual Model of Liquids (DML) has been validated and applied in several cases [17][18][19] finding good correspondence with the experimental data for the thermal conductivity , the order-of-magnitude of the relaxation times governing the interaction process [17], the liquid specific heat [17][18]. It has also allowed providing a physical interpretation of the memory term appearing in the hyperbolic form of the heat propagation equation (Cattaneo) [19].
The three major ingredients of the DML that make it new with respect to other liquid models are: (i) The dual character of liquids, supposedly composed by quasi-solid liquid particles and wave-packets, or lattice particles. (ii) The interaction between these two populations, in particular, its inelastic character and the time reversibility; the first allows modeling the mutual transfer of momentum other than energy between the lattice and the liquid particles, and the second makes this interaction as a good candidate for the elementary interaction at the base of the Onsager’s time-reversibility that characterizes the NET. (iii) The so-called “tunnel effect” of the interactions between lattice and liquid particles, whose effects deeply influence the transient phases of a thermodynamic perturbation on the system.
The aim of this paper is to show that the crossed effects typical of NET may be fruitfully explained by means of the DML.
2. The Thermodiffusion and the Soret Equilibrium in the DML
Readers interested in the details of the Dual Model of Liquids are invited to refer to the literature [17][18][19] where the theoretical developments, numerical simulations, experimental results, and applications are collected and discussed in depth. The DML considers liquid molecules arranged at the mesoscopic level on metastable solid-like local lattices, as ideally shown in Figure 1. Propagation of perturbations occurs at characteristic timescales typical of solids within these local domains of coherence, while mutual interactions of local clusters with inelastic wave-packets allow exchanging with them energy and momentum. The liquid is then assumed as a Dual System, the two subsystems being the liquid particles (i.e., the clusters of molecules) and the lattice particles.

Figure 1. Icebergs of solid lattice fluctuating and interacting within the liquid global system at equilibrium.
Solid-like icebergs, the liquid particles, are dispersed in the amorphous phase of liquid, while thermo-elastic waves, the lattice particles, travel through them. As far as the perturbations travel inside a solid-like structure, they show a solid-like character, i.e., they are (quasi) harmonic waves traveling at speeds close to that of the corresponding solid phase (3200 m/s in the case of water, [17][18][20]). When however they leave the liquid particle and travel through the amorphous environment, lose the harmonic outfit and become wave-packets. The interaction itself between these waves and the solid-like icebergs has an anharmonic character, allowing momentum Δp𝑤𝑝 and energy Δ𝜀𝑤𝑝 to be exchanged between the two reservoirs, the liquid particles and the lattice particles. The momentum transferred generates the force

responsible for the energy and mass diffusion in liquids [17][18][19]; 〈𝜏𝑝〉 represents the (finite) duration of the lattice particle ↔ liquid particle interaction and 𝜙𝑡ℎ is the anharmonic interaction potential between the lattice particle and the liquid particle, given by [17][19]:

𝑢𝜙 is the phase velocity associated with the wave-packet and 𝜎𝑝 is the cross-section of the “obstacle”, the solid-like cluster, on the surface of which 𝑓𝑡ℎ is applied; 𝐽𝑤𝑝𝑞 the energy flux carried by wave-packets [17] (about the functional dependence of 𝜙𝑡ℎ upon distance and about its quantum/classical form, the reader may refer to the Discussion in [17]). 𝜙𝑡ℎ is supposed to be anharmonic because of the inelastic character of the interaction, which is not instantaneous but lasts 〈𝜏𝑝〉 during which the particle is displaced by 〈Λ𝑝〉 (here and in the rest of the article, the two brackets 〈 〉 indicate the average over a statistical ensemble of the quantity inside them). 𝑓𝑡ℎ can either be positive or negative depending on whether the quantity (𝐽𝑤𝑝𝑞/𝑢𝜙) increases or decreases as a consequence of the interaction. This point is connected with the last equality in Equation (2). It is indeed well known that a wave impinging on an obstacle exerts on it a pressure. The radiation pressure was introduced for the first time by Balfour Stewart in 1871 [21] for the case of thermal radiation. Maxwell took up this theme in Arts. 792–793 of his Treatise in 1873 [22], arguing on the basis of his stress tensor. The first convincing experimental evidence for the radiation pressure of light was given by Lebedev in 1901 [23] (the Crookes radiometer does not demonstrate electromagnetic radiation pressure!). We have shown by means of the radiant vector 𝑅→=(ΔΠ⋅𝜉˙)𝑟→=𝐽el→ [17] that this concept may be generalized and used for any type of wave ( 𝜉˙ is the oscillation velocity of the particles, 𝑟→ the unit vector along the direction of propagation and the 𝐽el→ flux of elastic energy generating the pressure ). Here we exploit again this concept and apply it to the momentum transferred by elastic, or thermal, waves that impinge on an obstacle, such as for instance a solute molecule in a liquid mixture. Accordingly, we replace the gradient of elastic pressure with that of thermal energy, yielding:

Equation (3) contains two supplementary information; first, because thermal energy is carried by elastic wave-packets, the velocity of energy propagation remains the same. Second, the gradient of pressure gives rise to an inertial term, ψth, dimensionally a force per unit of volume, responsible for momentum transport associated with the wave-packets propagation. Equation (3) is valid when the characteristics of the medium change continuously along the wave-packets propagation direction. The same argument may be used when wave-packets impinge on an “obstacle”, as for instance a liquid particle, either of solute or of solvent: the radiant vector changes and a thermal radiation pressure Πth is produced on the boundary. Observing that the inelastic character of the collision allows the exchange of momentum, this leads to the appearance of the force fth acting on molecular clusters. By rewriting Equation (3) in terms of discrete quantities, one yields the following expression for the net Πth:

We conclude that the last equality in Equation (2) provides the algebraic sign for 𝑓𝑡ℎ according to the sign of the quantity in square brackets of Equation (4), i.e., on whether the energy associated with the propagation of elastic waves in the medium “1” is larger or smaller than that in the medium “2”. Expressions analogous to Equation (4) have been previously derived by many authors following different approaches [17][21][22][23][24][25][26][27]. It derives from the Boltzmann–Ehrenfest’ Adiabatic Theorem [28], later generalized by Smith [29] and by Gaeta et al. [30]; another approach gave analogous results by calculating the radiation pressure produced by acoustic waves on an idealized liquid-liquid interface [31].
The DML rests on a few basic theoretical concepts, which are hereafter explained. The internal energy per unit of volume of a liquid at temperature 𝑇 is 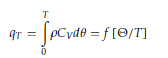
where 𝜌 is the medium density, 𝐶𝑉 the specific heat at constant volume per unit mass, Θ the Debye temperature of the liquid at temperature 𝑇. Since this dynamics occurs mainly at high frequencies and involves only the DoF of the lattice, we introduce the parameter 𝑚 to account for the average number of DoF participating in the collective dynamics. Defining m as the ratio between the number of collective DoF surviving at temperature 𝑇 and the total number of available collective DoF, the fraction
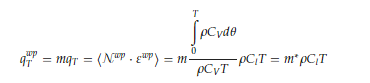
of 𝑞𝑇 accounts for that part of the thermal energy transported by the wave-packets (the definition of 𝑚* is easily deduced). Of course, it holds 0≤𝑚≤1 [17]. 𝒩𝑤𝑝 is the number of wave-packets per unit of volume and 𝜀𝑤𝑝 their average energy. 𝑞𝑤𝑝𝑇 is propagated through the liquid by means of inelastic interactions between the liquid particles and the lattice particles, i.e., the wave-packets that transport the thermal or, generally, the elastic energy.

Figure 2. Schematic representation of inelastic collisions between wave-packets and liquid particles. The dots represent the molecules, arranged in a metastable liquid particle, the small springs indicate the forces involving the internal DoF and responsible for the temporary stability of the cluster. In the event represented in (a), an energetic wave-packet transfers energy and momentum to a liquid particle; it is commuted upon time reversal into the one represented in (b), where a liquid particle transfers energy and momentum to a wave-packet. The particle changes velocity and the frequency of wave-packet is shifted by the amount (𝜈2−𝜈1). Due to its time symmetry, this mechanism has been assumed the equivalent of Onsager’s reciprocity law at microscopic level [17][19]. This elementary interaction has an activation threshold for potential energy and, depending on how much energy is absorbed by internal DoF, the rest will become kinetic energy of the liquid particle. When a system is subjected to an external temperature gradient, the first effect that occurs is the establishment of the internal temperature gradient, according to the Cattaneo–Fourier equation. Once the internal DoF have reached the statistical equilibrium in terms of distribution of their degree of excitation depending on the local temperature, the phenomenon of matter diffusion develops due to the temperature gradient and at the expense of the kinetic energy reservoir. In other words, inelastic effects dominate the dynamics during the transient phase, leaving the control to the kinetic reservoir at the steady-state, where only the elastic effects of the elementary interactions matter.
Figure 2 exemplifies the elementary physical mechanism behind the DML. The lattice particle ↔ liquid particle interaction allows the exchange of both energy and momentum between the phonon and the liquid particle. To make more intuitive the model, we have ideally divided the elementary liquid particle ↔ lattice particle interaction into two parts: one in which the lattice particle collides with the liquid particle and transfers to it momentum and energy (both kinetic and potential) and the other in which the liquid particle relaxes returning the energy to the thermal reservoir through a lattice particle, alike in a tunnel effect. Considering an event of type (a) of Figure 2, an energetic wave-packet collides with a liquid particle that acquires the momentum Δ𝑝𝑤𝑝 and the energy Δ𝜀𝑤𝑝:
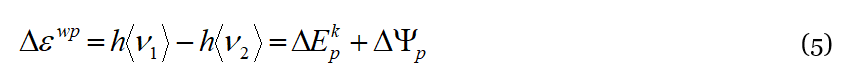

In Equation (5) 〈𝜈1〉 and 〈𝜈2〉 represent the wave-packet (central) frequency [17] before and after the collision, respectively. Part of Δ𝜀𝑤𝑝 becomes the kinetic energy Δ𝐸𝑘𝑝 acquired by the liquid particle, and the remaining part becomes ΔΨ𝑝, i.e., the potential energy of internal DoF of the solid-like cluster. As a consequence of the collision (a), the wave-packet loses energy and momentum, which are acquired by the cluster and converted into kinetic and potential energy. The kinetic energy is dissipated as friction against the liquid. The potential energy ΔΨ𝑝 is relaxed by the liquid particle once the interaction is completed, its dissipation lasts 〈𝜏𝑅〉 during which the liquid particle travels by 〈Λ𝑅〉, at the end of which the residual energy stored into internal DoF returns to the liquid reservoir. The total displacement of the particle, 〈Λ〉, and the total duration of the process, 〈𝜏〉 , are:


There are several interesting observations at this point that we want to propose to the reader’s attention, however, those readers keenly interested are warmly addressed to the related literature [17][18][19]. First, as pointed out before, the elementary interaction looks like a tunnel effect, inasmuch as the energy subtracted from the phonon’s reservoir returns to it a time interval 〈𝜏〉 later and a step 〈Λ〉 forward. It is the case to clarify however that the purely classical “tunnel effect” occurring in the liquid particle ↔ lattice particle interaction should not be confused with tunneling in physics, which is ordinarily associated with quantum phenomenology. We have adopted such terminology only to give the reader the idea of what happens in a liquid upon the interaction, but there are no hidden quantum phenomena beyond such a word. What matters here to the scope of DML is the dynamics linked to the relaxation time, i.e., to recognize that 〈𝜏〉 is the time interval during which the energy disappears from the liquid thermal reservoir because it is trapped in the internal DoF; once 〈𝜏〉 has elapsed, it reappears in a different place. In this way, relaxation times, introduced ad hoc by Frenkel [32], find a clear physical interpretation. The second point recalls us to the mind that in this manuscript we are dealing with the Soret equilibrium, i.e., a phenomenon characterized by a dynamical equilibrium between the diffusion of particles due to the heat crossing the mixture, and the Fick back diffusion due to the concentration gradient that is being established by the thermodiffusion. In the papers published so far [17][18][19], we have mainly been concerned with that part of the energy lost by the wave-packets and converted into internal (potential) energy of liquid particles, ΔΨ𝑝, and with the consequences of its propagation. At equilibrium, the variation of potential energy ΔΨ𝑝 is zero in the average because the thermal content of the system does not vary anymore, as well as the number of DoF excited. Accordingly, the only term surviving in the third member of Equation (5) is that accounting for the average kinetic energy acquired by the liquid particle, Δ𝐸𝑘𝑝. Apart from this, in the frame of DML, we are instead concerned with a flux of momentum, in fact, we are analyzing the mechanism responsible for the solute, or solvent, molecules flux pushed by a flux of thermal (elastic) energy crossing the liquid and due to an external source, the temperature gradient applied to the system (a liquid mixture). We are then concerned here with that part of energy lost by wave-packets and converted into kinetic energy of liquid particles.
We want now to draw the attention of the reader to the event of type (b) of Figure 2 which is the time-reversal of (a): an energetic cluster interacts with a wave-packet transferring to it energy and momentum. The outgoing wave-packet has momentum and energy larger than the incoming one so that the net effect of the interaction is to increase the liquid thermal energy carried by phonons at the expense of the kinetic and internal energy of the liquid particle. Another crucial point relevant to our purposes is that at (thermal or concentration) equilibrium conditions, events (a) will alternate over time with events (b), to keep statistically equivalent the balance of the two energy reservoirs. The effect of the interactions is null if integrated over the entire surface of the particle since its thermal regime does not change, nor does it move; consequently the mesoscopic equilibrium induces the macroscopic equilibrium, following the Onsager postulate [1][2][3][17]: events like those of Figure 2 will be equally probable along any direction, with a null average over time and space. On the contrary, when the symmetry is broken by an external force field, for instance by a temperature (Soret) or a concentration (Dufour) gradient, type (a) events will prevail over type (b), or vice-versa, along the direction of the gradient; the gradient will generate an imbalance of the collisions and possibly also a variation of the energy of the phonons (their number does not change as a first approximation) and an imbalance of the number of collisions. We will return to this point when discussing the non-equilibrium phenomena which are the topic of the present manuscript.
A question that the reader is certainly wondering at this point is “what pushes and drives the wave-packets in liquids between two successive collisions?”. To answer such a question we should keep in mind that in DML the wave-packet current is always present in liquid, even in isothermal conditions. During their free-flight between two successive interactions (Figure 1) with the liquid particle, the wave-packets are considered in the DML as local microscopic heat currents. Each lattice particle propagates along an average distance 〈Λ𝑤𝑝〉, during a time interval 〈𝜏𝑤𝑝〉, which is its average life-time. Accordingly, the ratio 〈Λ𝑤𝑝〉/〈𝜏𝑤𝑝〉 defines the wave-packet average propagation velocity between two successive interactions, 𝑢𝑤𝑝=〈Λ𝑤𝑝〉/〈𝜏𝑤𝑝〉=𝜆𝑤𝑝⋅𝜈𝑤𝑝 , where 𝜈𝑤𝑝 and 𝜆𝑤𝑝 are the (central) frequency and wave-length 𝜆𝑤𝑝 of the wave-packet 𝜈𝑤𝑝. Considering a surface S arbitrarily oriented within the liquid, at thermal equilibrium an equal number of wave-packets will flow through it in opposite directions. Let’s assume now that locally every heat current, although isotropically distributed in the liquid, is driven by a virtual temperature gradient 〈𝛿𝑇/𝛿𝑧〉 applied over 〈Λ𝑤𝑝〉. The variation of the energy density within the liquid along the “+𝑧” direction is:
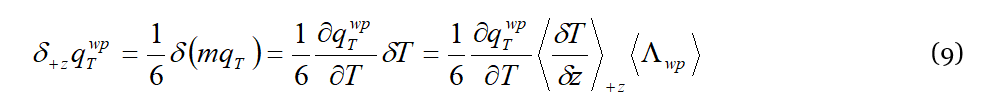
where the local virtual temperature gradient 〈𝛿𝑇/𝛿𝑧〉 in the last member represents the thermodynamic “generalized” force driving the diffusion of thermal excitations along z. The factor 1/6 accounts for the isotropy of the phonon current through a liquid at equilibrium. By applying this driving force over the distance 〈Λ𝑤𝑝〉, a wave-packet diffusion, i.e., the heat current 𝑗𝑤𝑝, is generated along the “+𝑧” direction:

where 𝐷𝑤𝑝+=1/6 * 𝑢𝑤𝑝〈Λ𝑤𝑝〉 is the wave packets diffusion coefficient along “+𝑧”. Therefore, the propagation of an elementary heat current lasts the time interval 〈𝜏𝑤𝑝〉 and is driven by a virtual temperature gradient 〈𝛿𝑇/𝛿𝑧〉 over the distance 〈Λ𝑤𝑝〉. Of course, in an isothermal medium, the number of scatterings along any direction is balanced by that in the opposite one, to get a null diffusion over time. On the contrary, when an external thermodynamic (generalized) force is applied to the system, for instance, a temperature gradient, a heat flux (in the opposite direction) is generated, and therefore, an increase of the number of “wave-packet ↔ liquid particle” interactions in the same direction. In this case (Soret effect), events of type (a) of Figure 2 have a larger probability to occur than events of type (b). Analogously, if the symmetry is broken by a particle concentration gradient (Dufour effect), the events having a larger probability of occurrence will be the (b). To quantify such reasoning, let 𝑑𝑇/𝑑𝑧=(𝑇1−𝑇2)/𝐿 be the temperature gradient externally applied to a system. As before, we assume here as a first approximation that the application of an external temperature gradient does not change the average total number of the wave-packets, (small gradient assumption). Let then 〈𝜈𝑝〉 be the average number per second of “wave-packet ↔ liquid particle” collisions at 𝑇0≡ (𝑇2+𝑇1)/2 in the isothermal liquid; due to the presence of the virtual temperature gradient, which is equally present in all three directions, for each direction this number is 〈𝜈𝑝〉/6. If an external (small) temperature gradient 𝑑𝑇/𝑑𝑧 is applied to the liquid, the total number of collisions per second 〈𝜈𝑝〉/6 is assumed to stay constant because we have supposed that 𝒩𝑤𝑝 does not change, but for collisions occurring in the direction of heat propagation, there will be 𝛿〈𝜈𝑝〉 excess of collisions per second and an equal defect −𝛿〈𝜈𝑝〉 in the opposite direction. If the intensity of the temperature gradient is not too high, we may assume that there will be no non-linear effects and that the initial state of the system at the microscopic level locally continues to be similar to the one at uniform temperature, except for thermal excitations along z. Therefore a liquid particle at a given place in the liquid experiences the same number of collisions per second as if the temperature was uniform; the only difference with the isothermal case is that there is an imbalance in the number of collisions with wave-packets that originated in the two half-spaces along z, one hotter and the other cooler. In other words, while phonons in liquids are pushed by the local virtual temperature gradient 〈𝛿𝑇/𝛿𝑧〉, the external temperature gradient 𝑑𝑇/𝑑𝑧 has the duty of orienting them along its own direction. In the linear range, we may assume that 2⋅𝛿〈𝜈𝑝〉 is proportional to the temperature gradient 𝑑𝑇/𝑑𝑧 externally applied to the liquid. Consequently, there is a heat flux 𝐽𝑒𝑥𝑡𝑞=−𝐾𝑙(𝑑𝑇/𝑑𝑧) superimposed to two equal and opposite heat fluxes +𝑗𝑤𝑝+𝑧=−𝐾𝑤𝑝〈𝛿𝑇/𝛿𝑧〉+ and 𝑗𝑤𝑝−𝑧=−𝐾𝑤𝑝〈𝛿𝑇/𝛿𝑧〉− ( 𝐾𝑙 represents the thermal conductivity of the macroscopic system, 𝐾𝑤𝑝 is that related to the collective DoF, [17][19]) giving:

Accordingly, an external temperature gradient 𝑑𝑇/𝑑𝑧 increases by (𝑑𝑇/𝑑𝑧)/〈𝛿𝑇/𝛿𝑧〉 the phonon flux with respect to that corresponding to the local microscopic heat currents 𝑗𝑤𝑝 due to spontaneous phonon diffusivity. Equation (11) gives the ratio of the number of “wave-packet ↔ liquid particle” collisions per second due to the applied gradient, to that due to the random motions of collective thermal excitations. The clusters therefore will experience 2𝛿〈𝜈𝑝〉 collisions in excess along the direction of the heat flux and will execute as many jumps per second of average length 〈Λ〉 in excess in the same direction. Consequently, every particle travels the distance 2〈Λ〉𝛿〈𝜈𝑝〉 per second along the direction of heat flux due to the external temperature gradient; this represents the drift velocity 〈𝜐𝑡ℎ𝑝〉 of the liquid particle along 𝑧 due to the external temperature gradient:
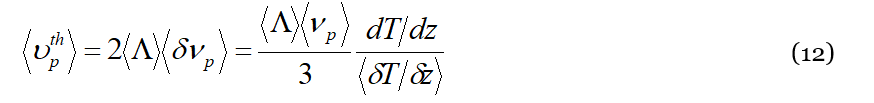
Equation (13) allows defining the thermal diffusion coefficient 𝐷𝑡ℎ𝑝 that, by definition, is the drift velocity of molecules in a unitary temperature gradient:

Because 𝐷𝑡ℎ𝑝 is a signature of the “solute + solvent” mixture, it is correctly given in Equation (13) in terms of the liquid particle drift 〈Λ〉 caused by the 〈𝜈𝑝〉 collisions with the lattice particle. Even more relevant to the purpose of the manuscript is its (reciprocal) dependence upon 〈𝛿𝑇/𝛿𝑧〉. In fact, 〈𝛿𝑇/𝛿𝑧〉 is the only term in Equation (13) that intrinsically contains an algebraic sign, determining in such a way the possibility for 𝐷𝑡ℎ𝑝 to change sign when some particular conditions occur in a liquid mixture. The expression for the Soret coefficient 𝑆𝑇 is easily obtained from its definition given by the ratio between the thermodiffusion coefficient and that of ordinary diffusion, for which we exploit the well-known Einstein relation (as specified before, we suppose the mixture sufficiently dilute so that the motion of molecules, either of solute or of solvent, does not interfere each other):
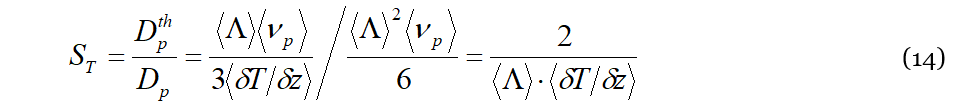
As for 𝐷𝑡ℎ𝑝, 𝑆𝑇 in Equation (15) contains its algebraic sign hidden in the term 〈𝛿𝑇/𝛿𝑧〉. A direct evaluation of 𝑆𝑇 through Equation (15) is not a simple task, of course, for this reason, dedicated numerical evaluations are in due course. However, Equation (14) contains very useful and important information about the meaning of 𝑆𝑇 in the DML framework. Recalling that 〈Λ〉 is the liquid particle drift caused by 〈𝑣𝑝〉 the collisions per second with the lattice particle, Equation (14) becomes:

Here 〈𝛿𝑇〉𝑧 is the average temperature difference experienced by a solute liquid particle over the distance 〈Λ〉 along 𝑧 covered after a collision with a phonon. This is a direct consequence of the dynamics characterizing the DML and fully in line with the macroscopic meaning of the Soret coefficient in NET.
We now have all the ingredients to calculate the flux of solute (or solvent) molecules 𝐽𝑚 due to the collisions with phonons in the presence of an external temperature gradient. If all solute particles have mass 𝑚𝑝 and their density is 𝑁𝑝, we have:
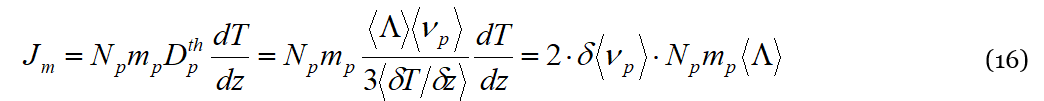
To recover from the present lack of a numerical evaluation of Equation (14), we propose here an alternative analysis of thermodiffusion, still in the DML framework, namely that of the variation of Π𝑤𝑝, or Πth, along the gradient. Besides being an energy density, 𝑞𝑤𝑝𝑇 represents in fact also the pressure Πwp exerted by elastic wave-packets on the liquid particles. Compiling Equation (4) with Equations (9) and (10) we get:
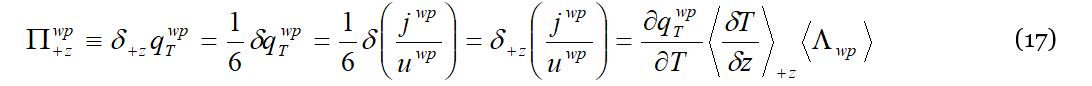
Of course also for Π𝑤𝑝 the time-averaged pressure generated by the ubiquitous wave-packets streams in a system without external gradients is zero. This is no longer true in the presence of a temperature (or a concentration) gradient working on the system. Assuming as always that the external gradient is small enough that linear deviations suffice to describe the induced variations due to its application, Equation (11) still holds for 〈𝛿𝜈𝑝〉, i.e., the increase in the number of collisions due to the external gradient. Such an increase in the number of collisions causes in turn an increase in the energy and momentum exchanged upon the collisions, namely 𝛿𝑊𝑡ℎ=〈𝛿𝜈𝑝〉⋅Δ𝜀𝑤𝑝 and 𝛿𝐹𝑡ℎ=〈𝛿𝜈𝑝〉⋅Δ𝑝𝑤𝑝. The reader has certainly guessed that 𝛿𝑊𝑡ℎ and 𝛿𝐹𝑡ℎ are respectively the total power dissipated by collisions and the force exerted on the collision target, i.e., the liquid particle or the lattice particle, depending on which situation prevails in the system, (a) or (b) of Figure 2. At the Soret equilibrium, 𝛿𝑊𝑡ℎ is the power needed to sustain the thermal and concentration gradients of the mixture (the Soret equilibrium is characterized by a positive rate of entropy production, the ratio 𝛿𝑞/𝑇 at the cold side being higher than that at the hot side, Δ𝑠=𝛿𝑞(1/𝑇𝑐−1/𝑇ℎ)>0), while 𝛿𝐹𝑡ℎ is the net force responsible for the separation of the two chemical species. Adapting Equation (17) to the “discrete” situation involving the two distinct liquid particles “1” and “2”, we get that:
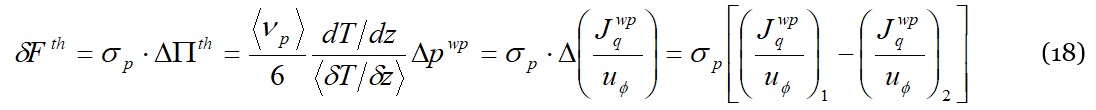
Equation (18) provides a comparison between two expressions for the total force, or the pressure, exerted on the targets following the application of an external temperature gradient. Comparing in fact its third and last members, we understand how the imbalance of the number of collisions influences the dynamical behavior of the mixture. In other words, Equation (18) tells us which of the two species, “1” or “2”, is pushed by the collisions to the cold side and why: the chemical species which is pushed towards the cold side by the external thermal gradient is the one for which the difference Δ(𝐽𝑤𝑝𝑞/𝑢𝜙) is positive, and therefore generates a virtual thermal gradient 〈𝛿𝑇/𝛿𝑧〉 parallel to the external one, determining in turn the algebraic sign of 〈𝛿𝑇/𝛿𝑧〉 in Equation (14) or Equation (15). Because the concentration of the solute, i.e., of the less concentrated species, is usually observed in solutions, one refers to positive or negative Soret depending on its concentration variation. Actually, when there is a negative Soret coefficient, according to the DML it happens that 𝛿𝐹𝑡ℎ, or ΔΠ𝑡ℎ, is positive for the solvent molecules, which will therefore concentrate on the cold side, while the solute will concentrate on the hot side due to Newton’s third law. In general, saying that 𝛿𝐹𝑡ℎ, or ΔΠ𝑡ℎ, is positive (negative) means in the DML that the radiation pressure due to the lattice particle stream on species “1” is higher (lesser) than on species “2”. Because the system is closed, it will obviously happen that only one of the two species will be pushed towards the cold side by the stream of lattice particles, while the other chemical species will move to the hot side due to Newton’s third law (a bit like cork floats on water while also being attracted by gravity, or a foam floating on the liquid surface will concentrate towards the axis of rotation in a closed rotating system.)
In order to get an expression for 𝑆𝑇 that can be easily evaluated on an experimental or numerical basis, we write Equation (18) at equilibrium replacing the expression for the thermal flux 𝐽𝑞 using the Fourier law; we get then:
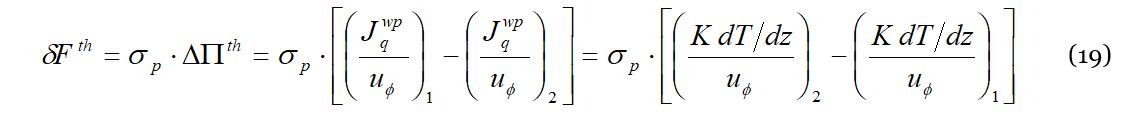
where 𝐾 is the thermal conductivity. Proceeding now in the reasoning, what happens at the equilibrium, i.e., when also the concentration gradient is established in the mixture, is that the power generated by the thermal engine is dissipated against the viscous forces, 𝑊𝑝ℎ≡𝑊𝑡ℎ=𝑊𝜂. The velocity of the liquid particle is 〈𝜐𝑡ℎ𝑝〉 defined by Equation (13); accordingly:
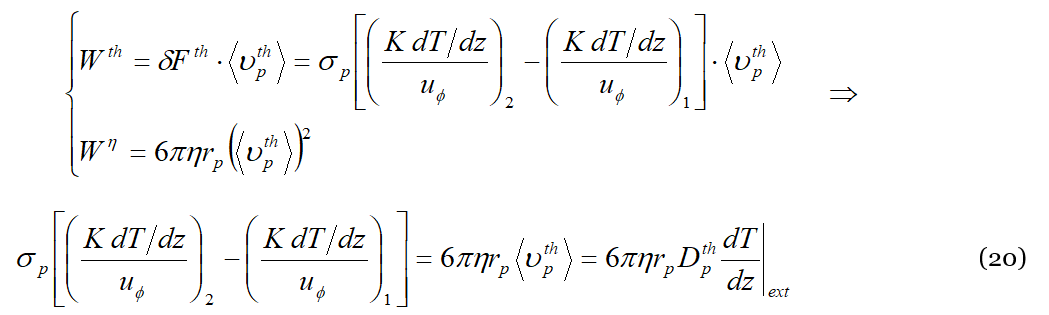
It is trivial now to get:
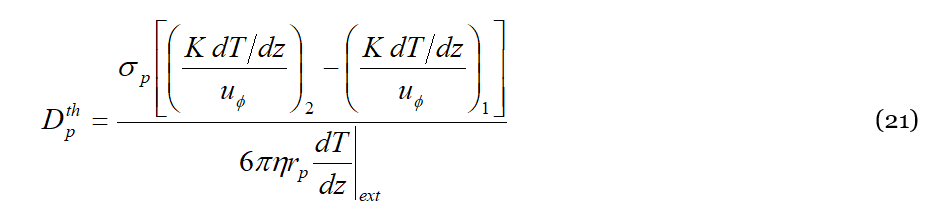
Introducing the Stokes–Einstein expression for the diffusion coefficient 𝐷𝑝=𝐾𝐵𝑇𝑎𝑣/6𝜋𝜂𝑟𝑝 eventually we get for 𝑆𝑇:
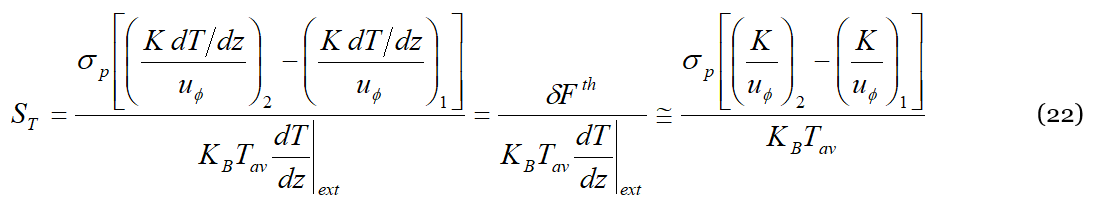
Equations (14) and (22) provide for the first time a simple interpretation of the Soret effect at the mesoscopic level, they are without matching parameters, are falsifiable (in the Popperian meaning) and can be verified by means of experiments. Equation (14) is based on elementary principles applied at the mesoscopic level, while Equation (22) is based on macroscopic quantities. A question that the reader certainly wonders is what one expects from their comparison. Let’s then consider the third member of Equation (22); the numerator is the force exerted by the thermal field to displace the solute, or solvent, molecules to generate and sustain the concentration gradient that gives origin to the Soret effect. At equilibrium the (average) energy available in the system is 𝐾𝐵𝑇𝑎𝑣. The ratio between such two quantities is just the mean displacement of the solute (solvent) particle, 〈Λ〉 , due to the collisions with the phonons,

Consequently, Equations (14) and (22) formally represent two similar expressions for the Soret coefficient (apart from a numerical factor due to statistical averages).
The separation of the components of a liquid mixture upon the application of a thermal gradient is due to the capability of the forces acting at the mesoscopic level in a liquid to convert the heat flux into a momentum flux. An external temperature gradient has the effect of orienting the local gradients along the same direction. When instead ST or Dth are negative means that the local virtual gradients are oriented along the direction opposite to that of the external gradient. This circumstance produces a thermodiffusive drift along the direction opposite to that of the external gradient, resulting in a negative value of 𝑆𝑇. In other words, the thermodiffusive drift of the solvent prevails on that of the solute, consequently we will observe the solute migrating to the hot side and the solvent to the cold one because the force due to the radiation pressure exerted by wave-packets on the solvent particles is higher than that produced on the solute particles. In Table 1 some examples of Soret coefficients evaluated from Equation (22) are provided for both positive and negative 𝑆𝑇. Table 2 collects data for the rations 𝐾/𝑢 elaborated from thermodiffusion in liquid mixtures performed with a Klusius–Dickel device. Original data report only which of the two species involved in each single experiment concentrate on the hot or cold side, however, this is enough to verify how the direction of the thermodiffusion drift is correctly predicted from Equation (22).
Table 1. Experimental values for Soret coefficient (last column, units 10−3K−1) for mixtures of polyvinylpyrrolidone K90 of 360,000 amu in various solvents as listed in the first column, measured and reported in [33]. Thermodiffusion of this macromolecule was experimentally studied in several solvents; in particular, an inversion of the sign of the Soret coefficient was detected in buthanol and propanol. Columns 2 and 3 report the values of the ratios 𝐾/𝑢 for the listed substances (units 10−3 Jm−2K−1). Their differences are reported in the fourth column. It is interesting to note the sign inversion for the same two solvents as experimentally detected. Data for 𝐾′𝑠 and 𝑢′s are deduced from [34].
|
Mixture |
|
|
|
|
|
K-90 in water |
0.4031 |
0.137 |
0.266 |
19.82 |
|
K-90 in methanol |
0.1830 |
0.137 |
0.046 |
0.38 |
|
K-90 in ethanol |
0.1440 |
0.137 |
0.007 |
2.31 |
|
K-90 in buthanol |
0.1205 |
0.137 |
−0.016 |
−5.78 |
|
K-90 in propanol |
0.1197 |
0.137 |
−0.017 |
−6.01 |
Table 2. Values of the ratios 𝐾/𝑢 elaborated from experimental data of thermodiffusion in liquid mixtures obtained with the Klusius-Deckel device [35]. Columns 2 and 3 report the values of the ratios 𝐾/𝑢 for the substances (units 10−3 Jm−2K−1). Their differences are reported in the fourth column. It is interesting to note the sign inversion for the same two solvents as experimentally detected. Data for 𝐾′𝑠 and 𝑢′𝑠 are deduced from [34]. Data from the two Tables, in particular for the ratios 𝐾/𝑢, must however be taken cum grano salis and considered with caution, a thorough evaluation of 𝑆𝑇 in the DML should be elaborated from Equation (14).
|
Mixture |
|
|
|
Component Drifting to Cold Plate |
|
Hexane-Carbontetrachloride |
0.1130 |
0.1120 |
0.0010 |
2 |
|
Hexane-Cyclohexane |
0.1130 |
0.0978 |
0.0152 |
2 |
|
Toluene-Cyclohexane |
0.1100 |
0.0978 |
0.0122 |
2 |
|
Toluene-Benzene |
0.1100 |
0.1028 |
0.0072 |
2 |
|
Benzene-Chlorobenzene |
0.1028 |
0.1021 |
0.0007 |
2 |
|
Benzene-Nitrobenzene |
0.1028 |
0.0990 |
0.0038 |
2 |
|
Bromobenzene-Carbontetrachloride |
0.0973 |
0.1120 |
−0.0157 |
1 |
|
m-Xylene-o-Xylene |
0.0978 |
0.0974 |
0.0004 |
2 |
Figure 3 shows another way to conceptually validate the expression of 𝑆𝑇 given by Equation (22). Figure 3a collects the data obtained by Bierlein, Finch, and Bowers for several mixtures of benzene and n-heptane [36]. The interesting point is that 𝑆𝑇 for such liquid mixtures increases with the average temperature for all the relative concentration values of the two components. Even more interesting is the fact that the three plots obtained as a function of temperature cross the “zero” value for 𝑆𝑇 at about the same temperature, around 60 °C. More precisely, 𝑆𝑇 is negative for temperature below such threshold, goes through zero, and becomes positive for average temperature above the threshold. In Figure 3b we have plotted the values of the ratio 𝐾/𝑢 for the two components; it is straightforward to note that such a ratio assumes the same value around the temperature of 60 °C. Because the sign of 𝑆𝑇 in Equation (22) depends just upon the difference of the ratios 𝐾/𝑢 for the two components, this observation confirms that Equation (22) is a good candidate to foresee even the sign inversion of 𝑆𝑇. Equation (14) is in principle the correct expression for 𝑆𝑇 in the DML, contains the sign dependence hidden into the term 〈𝛿𝑇/𝛿𝑧〉, more precisely in the orientation of 〈𝛿𝑇/𝛿𝑧〉 with respect to the external temperature gradient. Supporting the above reasoning is the dependence of 𝐷𝑡ℎ𝑝 in Equation (4) upon 〈𝛿𝑇/𝛿𝑧〉 instead of the external gradient. This is not surprising because the thermodiffusion is a signature of the mixture, therefore it shall be dependent only upon the mixture’s parameters. The reader must not be fooled by the inverse proportionality with 〈𝛿𝑇/𝛿𝑧〉: it tells us that as large the local virtual gradient 〈𝛿𝑇/𝛿𝑧〉, as difficult will be to observe the thermodiffusion, as one would have expected. 〈𝛿𝑇/𝛿𝑧〉 as well 〈Λ〉 or 〈𝜈𝑝〉 will change according to the relative concentration of the components, or to the temperature of the system. A more extensive collection of experimental data on thermodiffusion is in due course and their comparison with the theoretical predictions of Equations (14) and (22) will be shown elsewhere. Because we do not have access to a lab, the only possibility to provide further quantitative evaluations of Equation (22) is through experimental data available in the literature for which enough data should even be available for K’s and uϕ’s.

Figure 3. (a) Values of Soret coefficient deduced from [36] and their best-fit lines. (b) Values of the ratios 𝐾/𝑢 and their best-fit lines for the same couple of liquids as in (a). The two plots cross at about the same temperature as where 𝑆𝑇 crosses the “zero” in (a).
Recently [37], experiments performed in Newtonian fluids, such as water and glycerol, revealed for the first time the presence and long-lasting permanence of elasticity on a mesoscopic scale. The elastic response in such liquids persisted indeed for time laps of microseconds, hence four orders of magnitude longer than the typical intermolecular relaxation time. Such rubber-like elasticity and large strain response in fluid glycerol were interpreted by the authors as due to the relaxation processes of collective modes in metastable groups of molecules (those we have dubbed icebergs in the DML). Consequently, one may argue that the lifetime of such icebergs is equal to, if not larger than, the relaxation time of elastic propagation, as already hypothesized in a previous paper [17]. The authors also point out that such modes require the existence of a transient state with solid-like long-range correlations, although different from the bulk state. This study has then revealed that liquid shear elasticity could be very underestimated on such a mesoscopic scale and high-frequency domain with respect to what is measured with the classical methods on a macroscopic scale. The same effect could affect also the viscosity value on the same scale, hence it is arguable that measuring viscosity at a mesoscopic scale in the high-frequency domain may reveal a response very different with respect to that typical of liquids. Once again, the DML is potentially able to provide the basis for the viscosity modeling in Newtonian fluids.
Finally, some considerations could be dedicated to a comparison between the capabilities of DML applied to cross effects in NET and the previous theoretical models for thermodiffusion. We do not want here to enter into the debates of the models or make a historical excursus, that is, however, the topic of other interesting papers [12][13], but only propose a critical comparison of the DML with some of the most relevant alternative models. The first comment is dedicated to the interesting paper by Najafi and Golestanian [38], in which they suppose the thermodiffusion to be due to the Soret-Casimir effect. They in fact calculate such force in several configurations, however, this method is intrinsically unable to foresee negative values for 𝑆𝑇. Besides, they have also argued that in some cases the Soret effect violates the III Principle of dynamics, a circumstance that has been definitively excluded in the present manuscript because of the liquid’s duality. Duhr and Braun [39] have modeled the thermodiffusion due to the gradients of thermodynamic potentials inside mixtures. They have applied the model to macromolecular solutions, in particular DNA, finding good correspondence also with the mass and size of the solute, however only in those cases with positive 𝑆𝑇.
Two very interesting historical reviews are represented by the works of Eslamian and Saghir [12] and Rahman and Saghir [13], from which a large amount of models of thermodiffusion jumps to the eye, very different from one another and often from experimental data. These reviews are united by similar conclusions on the several models they deal with. In particular, it is affirmed that many theoretical models, initially accepted by the scientific community, were found wrong after more careful analysis [40][41]; many others correctly reproduce the thermodiffusion only in gases, while “in liquids, reliable and satisfactory predictive theories are still lacking”; furthermore “the largest part of models necessitate of external matching parameters to be defined, making the models dependent on the experiments” [12]. A relevant point is the fact that quite all the models either do not predict negative 𝑆𝑇 or fail in its prediction [14].
All the above points of weakness are overcome by the DML, as discussed above.
3. An “Unexpected” Mechano-Thermal Effect in Pure Liquids Explained by Means of DML
The cross-effect discovered by the group of Noirez a few years ago [6][7][8][9][10][11] results from the coupling between an acceleration field and a thermal field in a confined liquid layer submitted to shear strain. The system consists of a pure liquid layer confined in between two coaxial disc plates, one is fixed while the other one is mobile following a Heaviside function or an oscillatory motion, see Figure 4. The system is observed by far by means of a thermocamera, which allows for measurement of the thermal field that is being established inside the liquid, as well its time evolution. When the Heaviside motion law is applied to the mobile plate, typically the lower one, it undergoes a sudden acceleration in one direction, performs the expected movement, and then stops for a few seconds. Then it goes back to the starting point with an acceleration lower than that of the outward path. In the second type of experiment, the rotating plate oscillates according to a periodic law. The thermal behavior of the liquid in the two cases is not exactly the same, although the dependence of the intensity of the mechano-thermal phenomenon, in particular of the temperature difference that originates with respect to the acceleration field, remains almost the same. From the classical point of view, even a hardly detectable (faint) heating of the liquid layer facing the moving plate is not expected. Indeed, at these low frequencies and strain amplitudes, the energy stored cannot generate viscous heating following the classical empirical evaluation of the Nahme number (also known as the Brinkman number).
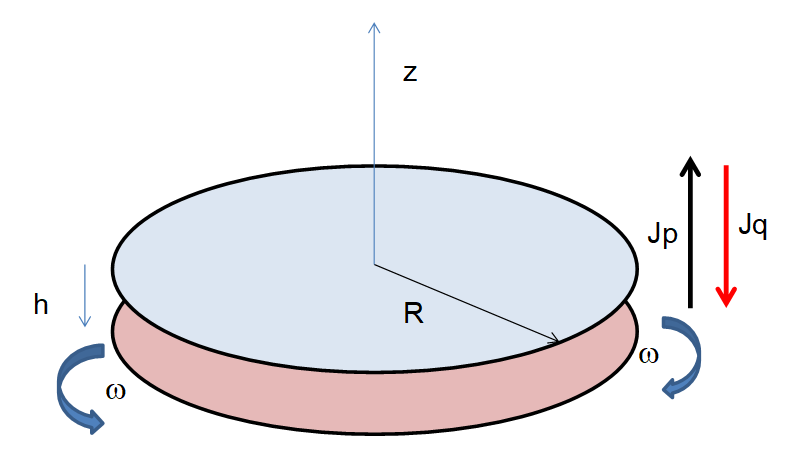
Figure 4. Schematic (drawing not to scale) of the experimental device adopted in [11]. The liquid, glycerol in this case, lies in between the two plates, one of which may rotate, the other is fixed.
Much faster velocities are needed for viscous liquids, closer to sound velocities. Experiments performed by Noirez and co-workers revealed instead that unexpectedly liquids confined to sub-millimeter scale show a thermal response consisting of the cooling of the layer facing the moving plate and a corresponding heating of that facing the fixed plate.
Generally, the non-uniform temperature shear bands observed along the strain direction indicate the formation of a faint temperature gradient in phase with the deformation. The synchronization of the thermal field with the mechanical strain is a signature of a mechano-thermal effect of elastic origin indicating that viscous liquids might exhibit thermoelastic behavior, as pointed out also by the authors of the experiments [42][43].
Noirez and co-workers pointed out that this response to low-frequency mechanical stress is typical of amorphous solids, exhibiting a storage shear modulus 𝐺′ much higher than the loss viscous modulus 𝐺″. This is a key point in both of the experiments and the theoretical interpretation of the DML framework that we are going to show in what follows. In fact, being the geometry of these experiments of shear strain type, it is implicitly demonstrated by the experimental results that liquids possess shear elasticity as solids, contrary to what is generally supposed due to the absence of rigidity, but in line with the duality of liquids as supposed in the DML. By increasing the frequency of the mechanical stress and/or the scale on which such stress is applied, the effect disappears, and the liquid returns to its classical full-viscous behavior (𝐺′=0). Nonaffine framework has provided the physical interpretation of such behavior, consisting of the cut-off of low-frequency transverse modes, which in turn allows the high-frequency modes to maximize their effect at the mesoscopic level [16]. In this framework, the authors have assumed that transverse modes are carried by phonons. Accordingly, the DML may provide the missing ingredient to nonaffine treatment by means of the phonon ↔ liquid particle interactions, where phonons assume the role of transforming the momentum they receive from the liquid particles in a thermal field. Therefore, 𝑓𝑡ℎ is the best candidate for the nonaffine force introduced in the equation of motion of the microscopic building block [16]. In fact, in the nonaffine framework, there is a competition between the rigid-like behavior of coordinated clusters and the relaxations via nonaffine motions due to unbalanced forces in the disordered molecular environment.
Let’s then proceed with the DML interpretation. The key is provided indeed by the elementary lattice particle ↔ liquid particle interaction, described in Figure 2b. The acceleration of the moving plate generates a velocity gradient through the liquid, which in turn gives rise to a momentum flux, directed towards the fixed plate, following the relation
𝐽𝑝=−𝜂𝑙*(𝑑𝜐/𝑑𝑧), with 𝜂𝑙 the liquid viscosity. According to the DML, and also following Rayleigh’s reasoning, the momentum flux is dimensionally an energy density and generates a temperature gradient. The liquid particles, pushed by the momentum flux, transfer their excess impulse to the phonons of the liquid, following the (b) event of Figure 2; the phonons emerge from each single impact with an increased energy, being frequency-shifted. They are then pushed and collected towards the fixed plate, giving rise to a temperature gradient directed towards the fixed plate. The rationale is the same adopted to justify Equation (22), where however, in this case, the imbalance of collisions 〈𝛿𝜈𝑤𝑝〉 affects the wave packets instead of the liquid particles, and is due to the ratio between velocity gradients instead of temperature 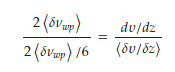 The supposed linear dependence this time is between the external velocity gradient 𝑑𝜐/𝑑𝑧 and that virtual, normally present in mechanical equilibrium for a liquid particle between two successive collisions with the lattice particles, 〈𝛿𝜐/𝛿𝑧〉. Each wave packet executes 2〈𝛿𝜈𝑤𝑝〉 jumps per second in the direction of the momentum flux, each long 〈Λ𝑤𝑝〉; they accumulate towards the fixed plate and decrease near the moving one, generating the temperature field observed. The product 𝜐𝑤𝑝∇𝜐=2〈Λ𝑤𝑝〉〈𝛿𝜈𝑤𝑝〉 defines the drift velocity of the thermal front due to the velocity gradient. The phenomenon is therefore very rapid because it is determined by a succession of (almost) elastic collisions, which propagate very quickly. Obviously, the higher thermal content of the liquid in proximity to the fixed plate compensates for the lower thermal content near the rotating plate, keeping the thermal balance unchanged (neglecting the losses toward the environment). It is important to highlight that the lattice particle ↔ liquid particle collision also explains why a temperature gradient (i.e., a generalized flux-force crossed effect) is generated inside the liquid instead of a simple heating due to the energy stored as a consequence of the mechanical stress (𝐺′>>𝐺″), as one would have expected on the basis of a classical interpretation of the elastic modulus. At the same time, the phenomenon can be observed only on the small length scale as that adopted in the experiments, the faintness of the thermal gradient would be in fact destroyed by the thermal unrest of the molecules in larger liquid volumes.
The supposed linear dependence this time is between the external velocity gradient 𝑑𝜐/𝑑𝑧 and that virtual, normally present in mechanical equilibrium for a liquid particle between two successive collisions with the lattice particles, 〈𝛿𝜐/𝛿𝑧〉. Each wave packet executes 2〈𝛿𝜈𝑤𝑝〉 jumps per second in the direction of the momentum flux, each long 〈Λ𝑤𝑝〉; they accumulate towards the fixed plate and decrease near the moving one, generating the temperature field observed. The product 𝜐𝑤𝑝∇𝜐=2〈Λ𝑤𝑝〉〈𝛿𝜈𝑤𝑝〉 defines the drift velocity of the thermal front due to the velocity gradient. The phenomenon is therefore very rapid because it is determined by a succession of (almost) elastic collisions, which propagate very quickly. Obviously, the higher thermal content of the liquid in proximity to the fixed plate compensates for the lower thermal content near the rotating plate, keeping the thermal balance unchanged (neglecting the losses toward the environment). It is important to highlight that the lattice particle ↔ liquid particle collision also explains why a temperature gradient (i.e., a generalized flux-force crossed effect) is generated inside the liquid instead of a simple heating due to the energy stored as a consequence of the mechanical stress (𝐺′>>𝐺″), as one would have expected on the basis of a classical interpretation of the elastic modulus. At the same time, the phenomenon can be observed only on the small length scale as that adopted in the experiments, the faintness of the thermal gradient would be in fact destroyed by the thermal unrest of the molecules in larger liquid volumes.
When the plate stops, the system relaxes and reaches the temperature it had at the beginning. During the return of the rotating plate to the starting position, the liquid is in exactly the same condition it had at the beginning of the experiment, and the temperature gradient forms again. This interpretation is in line with all the basic hypotheses made by the authors, including the one on the number of Grüneisen. In this particular case, the expansion/compression effect of the material medium would be a consequence of the density of the phononic contribution.
We will now try and get an expression for the (rate of) temperature variation due to the occurrence of events of type (b) of Figure 2 which are at the base of the observed phenomenon. In fact, what happens inside the liquid is that the mechanical energy injected into the liquid by the rotating plate is (partially) commuted in thermal energy carried by wave-packets. This energy, as we have seen, accumulates close to the fixed plate. To do this, we begin by considering that the momentum flux 𝐽𝑝 through the liquid due to the plate rotation represents also the pressure ΔΠ𝑙 exerted by the liquid molecules on the wave-packets, and is given by [44]:

where 𝜌𝑙 and 𝜐𝑙 are the density and shear speed of liquid, respectively. Neglecting dissipation effects due to the liquid viscosity, and remembering Equations (17) and (18), the momentum flux is converted into the pressure that pushes the phonon’s stream and gives rise to a heat flux along the opposite direction:
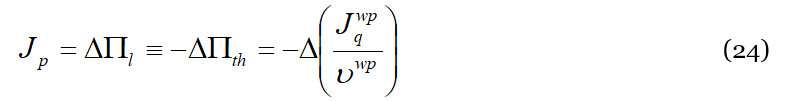
where 𝜐𝑤𝑝 is the wave-packet average speed. As is well known, the momentum flux corresponds even to the energy density Δ𝑊𝑡ℎ injected into the system by the mechanical stress. The duality of the DML provides us a key to (partially) convert at a mesoscopic level such mechanical energy in thermal energy, i.e., that carried by phonons that are excited at higher frequencies upon interactions of type (b) of Figure 2 with the liquid particles. Neglecting variations with respect to the temperature, we may use the average macroscopic value given by the ratio 𝜐𝑤𝑝=ℎ/Δ𝑡, where ℎ is the height of the liquid sample (parallel to the rotation axis), and Δ𝑡 the time necessary to establish the observed temperature difference across the liquid. Such value will be a multiple of the ratio 〈Λ〉/〈𝜏〉. Getting now the temperature difference Δ𝑇 (or the temperature gradient Δ𝑇/ℎ) is trivial, being enough to divide Equation (24) by the specific heat (per unit of volume) of the substance, i.e.,:
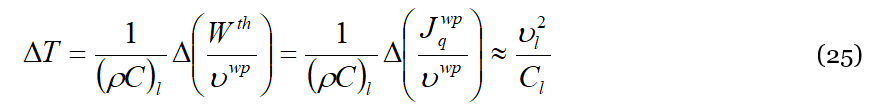
We evaluate now Equation (25) using the data contained in [11]. First, we use the value of the momentum flux averaged over the disc surface,
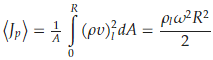
where 𝜔 is the plate's angular velocity and its radius. The liquid used in the experiment was glycerol, for which 𝜌𝑙=1.26 g/cm3 and 𝑐𝑝=2.39 J/g·K. Using a value of 100 μm for ℎ and 10 Hz for 𝜔, one gets Δ𝑇≈0.06 K of the correct order of magnitude as measured and reported in [11]. It is relevant to point out that the above interpretation is made possible because the DML foresees the presence of wave-packets that, interacting with the liquid particles, behave in this case not only as carriers of the thermal energy but also as converters of mechanical energy into thermal energy.
Equation (25), with all the limitations and simplifications adopted, shows that the temperature difference is proportional to the square of the shear speed, making the ΔT orientation independent upon the shear versus, as expected. The energy and momentum input depend only upon the momentum of the moving disc, and the ΔT observed is independent of the amount of substance or of the gap thickness of the layer (𝐶𝑙 is the specific heat per unit of volume). In the comparison of our results with the experimental observation, we should on one side keep in mind either the simplifications adopted in obtaining Equation (25), and the experimental difficulties in getting a stable and well-thermally isolated experimental setup. Nevertheless, on the other side, we could speculate on those aspects that may appear contradictory. One of these is that in some cases an apparent inversion of the thermal gradient was observed upon inversion of the shear motion. This effect could have the same origin as that observed in those cases where more than a single gradient was observed in the liquid, a sort of alternation between hot and cold layers. A possible explanation is that the stationary plate reacts to the inversion of the shear direction with a sort of “mirror” effect, pushing back the thermal excitations. The liquid near the stationary plate perceives the shear inversion as a sort of mirror effect of the stationary plate.
4. Conclusions
In previous papers [17][18][19], the DML has shown its capabilities in the calculation of the specific heat of liquids, thermal conductivity, the modeling of diffusion, the relevance, and the calculation of the relaxation times involved in the liquid dynamics at the mesoscopic scale, the justification of the Cattaneo equation for heat propagation, the self-diffusion in pure liquids, etc. In the present manuscript, its application field has been extended to the modeling of thermodiffusion and of the Soret equilibrium, as well as the Dufour effect. In particular, the DML provides a simple model not only for positive but also for negative values of 𝑆𝑇. Moreover, DML has been used to provide the first-ever theoretical interpretation of a new unexpected mechano-thermal effect discovered in liquids in shear geometry; at the same time, DML represents, to the author's knowledge, the first theory able to explain the conversion of mechanical energy into thermal energy, either in pure liquids or in liquid mixtures. Comparison with experimental results has been provided for all the cases considered.
Although there are still many aspects characterizing the liquid structure and their dynamics to be verified, a so large field of applications where the DML has shown its capabilities represents a good test bed for such a model, also in view of further developments. The next big challenge will certainly be the modeling of viscosity, it will not be a surprise if many insights and similarities are found with the works of Eyring [45][46][47][48][49][50]. This shall be the topic of future works.
Understanding how liquid parameters, as correlation lengths, sound velocity, thermal conductivity, etc., evolve from equilibrium to non-equilibrium conditions is an interesting topic that could be achieved by performing experiments in non-stationary temperature gradients. Also very interesting is to understand whether and how a temperature gradient affects the viscous coupling. One point that deserves deepening is certainly that of the evaluation of the parameter m, i.e., the collective DoF available at a given temperature and pressure for a liquid particle. In one of the previous papers [17], generic numerical results on dual models have already been commented on, although the calculation of the parameter necessitates to be supported by specific calculations. This and other simulations will be the core of future works currently in preparation.
We want to close the article with another consideration of the capabilities of the DML, trying to answer the question of why an approach based on non-equilibrium statistical mechanics of a dual system succeeds whereas other molecular, traditional approaches fail. The gold example is the Boltzmann equation and its application to transport processes in dense fluids. Prigogine [51] highlighted that it relates the derivatives of the one-particle velocity distribution function with the collision operator. Such collisions are characterized by their instantaneity and localization at a point in space. This simplification may be fruitful in gases but can fail when applied to liquids, where the duration of the interactions can be of the same order of magnitude as the interval between two successive interactions or of the relaxation times. The DML approach, although considering the interactions in a scheme that is simple and powerful at the same time, bypasses this weak point by means of the tunnel effect and considers the interactions as inelastic. The tunnel effect allows introducing the relaxation time(s) and moreover separates the interactions one from the other. The inelastic character ensures the capability of exchanging momentum other than energy between the two reservoirs, that of the liquid particles and of the lattice particles. This last point will be the core of future works.
References
- Callen, H.B. Thermodynamics and an Introduction to Thermostatics, 2nd ed.; Wiley: Philadelphia, PA, USA, 1991; ISBN: 978-0-471-86256-7.
- Onsager, L. Reciprocal relations in irreversible processes I. Phys. Rev. 1931, 37, 405–426.
- Onsager, L. Reciprocal relations in irreversible processes II. Phys. Rev. 1931, 38, 2265–2279.
- Ludwig, C. Sitzungsber. Akad. Wiss. Wien Math.-Naturwiss. Kl. 1856, 20, 539.
- Soret, M.C. Sur l’état d’équilibre que prend au point de vue de sa concentration une dissolution saline primitivement homohéne dont deux parties sont portées à des températures différentes Ann. Chim. Phys. 1881, 22, 293-297.
- Kume, E.; Zaccone, A.; Noirez, L. Unexpected thermoelastic effects in liquid glycerol by mechanical deformation. Phys. Fluids 2021, 33, 072007. https://doi.org/10.1063/5.0051587.
- Kume, E.; Noirez, L. Identification of thermal response of mesoscopic liquids under mechanical excitations: From harmonic to nonharmonic thermal wave. J. Phys. Chem. B 2021, 125, 8652–8658. https://doi.org/10.1021/acs.jpcb.1c04362.
- Noirez, L.; Baroni, P. Identification of a low-frequency elastic behaviour in liquid water. J. Phys. Condens. Matter 2012, 24, 372101–372107. https://doi.org/10.1088/0953-8984/24/37/372101.
- Kume, E.; Baroni, P.; Noirez, L. Strain-induced violation of temperature uniformity in mesoscale liquids. Sci. Rep. 2020, 10, 13340. https://doi.org/10.1038/s41598-020-69404-1.
- Kume, E.; Noirez, L. Thermal Shear Waves Induced in Mesoscopic Liquids at Low Frequency Mechanical Deformation. J. Non-Equilib. Thermodyn. 2022, 47, 155–163. https://doi.org/10.1515/jnet-2021-0091.
- Noirez, L.; Baroni, P. Revealing the solid-like nature of glycerol at ambient temperature. J. Mol. Struct. 2010, 972, 16–21. https://doi.org/10.1016/j.molstruc.2010.02.013.
- Eslamian, M.; Saghir, Z. A critical review of the thermodiffusion model: Role and significance of the heat of transport and the activation energy of viscous flow. Non-Equilib. Thermodyn. 2009, 34, 97–131. https://doi.org/10.1515/JNETDY.2009.007.
- Rahman, M.A.; Saghir, M.Z. Thermodiffusion or Soret effect: Historical review. Int. J. Heat Mass Tranfer 2014, 73, 693–705. https://doi.org/10.1016/j.ijheatmasstransfer.2014.02.057.
- Gittus, O.R.; Bresme, F. On the microscopic origin of the Soret coefficient minima in liquid mixtures. arXiv 2022, arXiv:2207.12864vl.
- Zaccone, A.; Noirez, L. Universal law for the low-frequency shear modulus of confined liquids. J. Phys. Chem. Lett. 2021, 12, 650–657.
- Phillips, A.E.; Baggioli, M.; Sirk, T.W.; Trachenko, K.; Zaccone, A. Universal finite-size effects in the viscoelasticity of amorphous systems. Phys. Rev. Mater. 2021, 5, 035602.
- Peluso, F. Mesoscopic collective dynamics in liquids and the Dual Model ASME. J. Heat Transf. 2022, 144, 112502. https://doi.org/10.1115/1.4054988.
- Peluso, F. Isochoric specific heat in the Dual Model of Liquids. Liquids 2021, 1, 77–95. https://doi.org/10.3390/liquids1010007.
- Peluso, F. How Does Heat Propagate in Liquids? Liquids 2023, 3, 92–117. https://doi.org/10.3390/liquids3010009.
- Ruocco, G.; Sette, F.; Bergmann, U.; Krisch, M.; Masciovecchio, C.; Mazzacurati, V.; Signorelli, G.; Verbeni, R. Equivalence of the sound velocity in water and ice at mesoscopic lengths. Nature 1996, 379, 521–523.
- Stewart, B. Temperature Equilibrium of an Enclosure in which there is a Body in Visible Motion. In British Association Reports; 41st Meeting, Notes and Abstracts; John Murray, Albemare st., London, UK, 1871; p. 45.
- Maxwell, J.C. A Treatise on Electricity and Magnetism; Clarendon Press, London, UK: 1873; Volume 2.
- Lebedev, P. Untersuchen ueber die Druckkraefte des Lichtes. Ann. Phys. 1901, 6, 433.
- Joyce, W.B. Classical-particle description of photons and phonons. Phys. Rev. D 1974, 9, 3234–3256.
- Westervelt, P.J. Acoustic Radiation Pressure. J. Acoust. Soc. Am. 1957, 29, 26–29.
- Mercier, J. De la pression de radiation dans le fluides. J. Phys. Rad. 1956, 17, 401–404.
- Joyce, W.B. Radiation force and the classical mechanics of photons and phonons. Am. J. Phys. 1974, 43, 245–255.
- Brillouin, L. Tensors in Mechanics and Elasticity; Academic Press: New York, NY, USA, 1964; pp. 240–243.
- Smith, W.E. Generalization of the Boltzmann-Ehrenfest Adiabatic Theorem in Acoustics. J. Acoust. Soc. Am. 1971, 50, 386–388.
- Gaeta, F.S.; Ascolese, E.; Tomicki, B. Radiation forces associated with heat propagation in nonisothermal systems. Phys. Rev. A 1991, 44, 5003–5017.
- Gaeta, F.S.; Mita, D.G. Nonisothermal mass transport in porous media. J. Membr. Sci. 1978, 3, 191–214.
- Frenkel, J. Kinetic Theory of Liquids; Oxford University Press: Oxford, UK, 1946.
- Gaeta, F.S.; Scala, G.; Brescia, G.; Di Chiara, A. Characterization of macromolecules in liquid solutions by thermal diffusion. I. Dependence of the Soret coefficient on the nature of the dispersing medium. J. Polym. Sci. Polym. Phys. Ed. 1975, 13, 177–202. https://doi.org/10.1002/pol.1975.180130115.
- Experimental data for ’s and ’s have been collected from several fonts, in particular: (a) Landolt-Bornstein, Numerical data and Functional Relationships in Science and Technology-Group II. Atomic and Molecular Physics, Vol. 5, Molecular Acoustics, W. Schaafs, FA.; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1967. 2. (b) Tsederberg, N.V. Thermal Conductivity of Gases and Liquids; for the thermal conductivities of liquid mixtures and solutions, see 220–221; M.I.T. Press: Cambridge, MA, USA, 1965. 3. (c) Hodgman, C.H.; Weast, R.C.; Shankland, R.S.; Selby, S.M. (Eds.) Handbook of Chemistry and Physics; The Chemical Rubber Publishing Co.: Cleveland, OH, USA, 1962. 4. (d) Lide, D.R., (Ed.) Handbook of Chemistry and Physics; 80th ed.; CRC Publisher: New York, NY, USA, 1999. 5. (e) (c) Available online: theengineeringtoolbox.com (accessed on August 2023).
- Prigogine, I.; De Brouckere, L.; Amand, M.R. Recherche sur la thermodiffusion en phase liquid. Physica 1950, 16, 577–598.
- Bierlein, A.; Finch, C.R.; Bowers, H.E. Coefficients de soret dans le système benzène=heptane normal. J. Chim. Phys. 1957, 54, 872–878. https://doi.org/10.1051/jcp/1957540872.
- Kayanattil, M.; Huang, Z.; Gitaric, D.; Epp, S.W. Rubber-like elasticity in laser-driven free surface flow of a Newtonian fluid, Proc. Natl. Acad. Sci. USA 2023, 120, e2301956120. https://doi.org/10.1073/pnas.2301956120.
- Najafi, A.; Golestanian, R. Forces Induced by Non-Equilibrium Fluctuations: The Soret-Casimir Effect. Europhys. Lett. 2004, 68, 776.
- Duhr, S.; Braun, D. Why molecules move along a temperature gradient. Proc. Natl. Acad. Sci. USA 2006, 103, 19678–19682. https://doi.org/10.1073/pnas.0603873103.
- Kempers, L.J.T.M. A comprehensive thermodynamic theory of the Soret effect in a multicomponent gas, liquid or solid. J. Chem. Phys. 2001, 115, 6330–6341. https://doi.org/10.1063/1.1398315.
- Eslamian, M.; Saghir, Z. Dynamic thermodiffusion theory for ternary liquid mixtures. J. Non-Equilibr. Thermodyn. 2010, 35, 51–73.
- Baroni, P.; Bouchet, P.; Noirez, L. Highlighting a cooling regime in liquids under submillimeter flows. J. Phys. Chem. Lett. 2013, 4, 2026–2029. https://doi.org/10.1021/jz400673d.
- Noirez, L.; Baroni, P. Identification of thermal shear bands in a low molecular weight polymer melt using oscillatory strain field. Colloid Polym. Sci. 2017, 296, 713–720. https://doi.org/10.1007/s00396-018-4264-4.
- Landau, L.D. Mechanique des Fluides; Edition MIR: Moscow, Russia, 1971; Volume Tome VI, p. §7.
- Glasstone, S.; Laidler, K.J.; Eyring, H. The theory of rate processes. In The Kinetics of Chemical Reactions, Viscosity, Diffusion and Electrochemical Phenomena; McGraw-Hill: New York, NY, USA; London, UK, 1941.
- Ewell, R.H.; Eyring, H. The viscosity of liquids as function of temperature and pressure. J. Chem. Phys. 1937, 5, 726–736.
- Kauzmann, W.; Eyring, H. The viscous flow of large molecules. J. Am. Chem. Soc. 1940, 62, 3113.
- Andrade, J.N., The Viscosity of Liquids, Nature 1930, 125, 582-584.
- Bondi, A. Notes on the rate process theory of flow. J. Chem. Phys. 1946, 14, 591.
- Macedo, P.B.; Litovitz,T.A. On the relative roles of free volume and activation energy in the viscosity of liquids. J. Chem. Phys. 1965, 42, 245.
- Prigogine, I. Non Equilibrium Statistical Mechanics; Wiley Interscience: New York, NY, USA, 1962.





