
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Rahni Hossain | -- | 3175 | 2023-11-19 07:50:43 | | | |
| 2 | Jason Zhu | -1 word(s) | 3174 | 2023-11-20 04:02:38 | | |
Video Upload Options
Kratom (Mitragyna speciosa Korth. Havil) has been considered a narcotic drug for years, barred by the law in many parts of the world, while extensive research over the past few decades proves its several beneficial effects, some of which are still in ambiguity. In many countries, including Thailand, the indiscriminate use and abuse of kratom have led to the loss of life. Nonetheless, researchers have isolated almost fifty pure compounds from kratom, most of which are alkaloids. The most prevalent compounds, mitragynine and 7-hydroxy mitragynine, are reported to display agonist morphine-like effects on human μ-opioid receptors and antagonists at κ- and δ-opioid receptors with multimodal effects at other central receptors.
1. Introduction
2. Kratom, an Indole-like Alkaloid for Neurological Effects
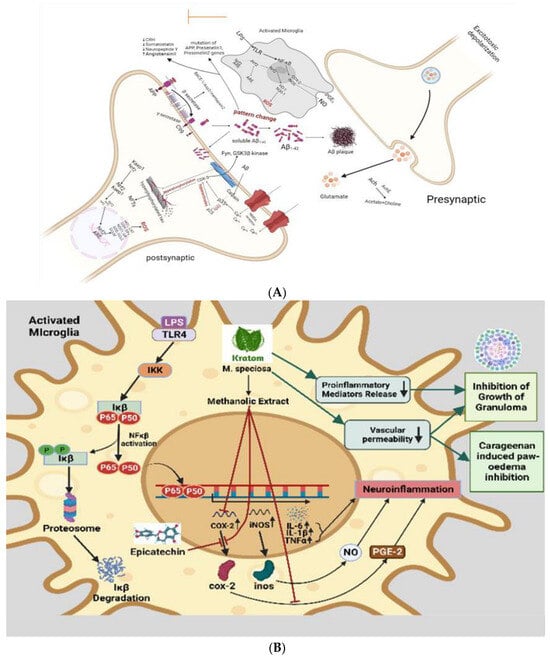
3. Anti-Inflammatory Effects Leading to Neuroprotective Effects
4. Analgesic and Anti-Nociceptive Effects
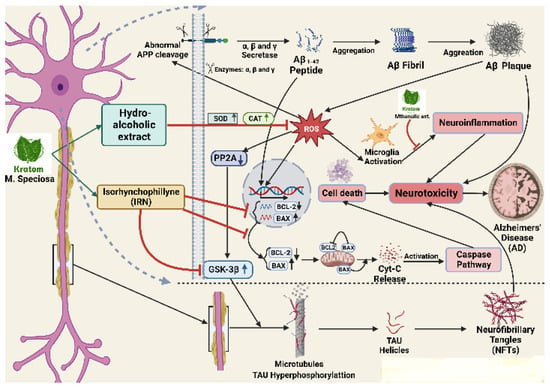
5. Neurological Effects by Gene Regulation
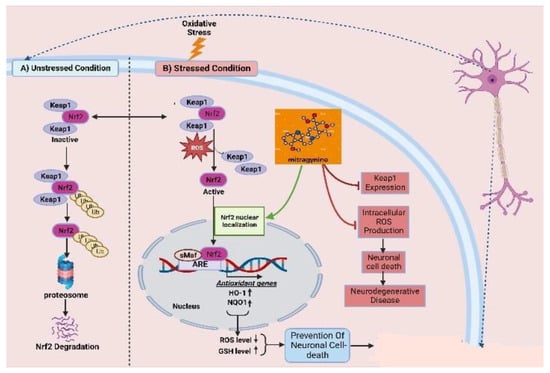
6. Antioxidative Effects
References
- Grewal, K.S. Observations OX the Pharmacology of Mitragynine. J. Pharmacol. 1932, 46, 251–271.
- Kumarnsit, E.; Keawpradub, N.; Nuankaew, W. Effect of Mitragyna speciosa aqueous extract on ethanol withdrawal symptoms in mice. Fitoterapia 2007, 78, 182–185.
- Mohammad Yusoff, N.H.; Mansor, S.M.; Visweswaran, N.; Muller, C.P.; Hassan, Z. GABAB receptor system modulates mitragynine-induced conditioned place preference in rats. In Proceedings of the 14th Meeting of the Asian-Pacific Society for Neurochemistry, Kuala Lumpur, Malaysia, 27–30 August 2016.
- Effendy, M.A.; Yunusa, S.; Mat, N.H.; Has, A.T.C.; Müller, C.P.; Hassan, Z. The role of AMPA and NMDA receptors in mitragynine effects on hippocampal synaptic plasticity. Behav. Brain Res. 2023, 13, 114169.
- Kruegel, A.C.; Grundmann, O. The medicinal chemistry and neuropharmacology of kratom: A preliminary discussion of a promising medicinal plant and analysis of its potential for abuse. Neuropharmacology 2018, 134 Pt A, 108–120.
- Apryani, E.; Hidayat, M.T.; Moklas, M.; Fakurazi, S.; Idayu, N.F. Effects of mitragynine from Mitragyna speciosa Korth leaves on working memory. J. Ethnopharmacol. 2010, 129, 357–360.
- Ammar, I.H.; Muzaimi, M.; Sharif, M.M. The effects on motor behaviour and short-term memory tasks in mice following an acute administration of Mitragyna speciosa alkaloid extract and mitragynine. J. Med. Plants Res. 2011, 5, 5810–5817.
- Singh, D.; Narayanan, S.; Müller, C.P.; Vicknasingam, B.; Yücel, M.; Ho, E.T.W.; Hassan, Z.; Mansor, S.M. Long-Term Cognitive Effects of Kratom (Mitragyna speciosa Korth.) Use. J. Psychoact. Drugs 2019, 51, 19–27.
- Ismail, N.I.W.; Jayabalan, N.; Mansor, S.M.; Müller, C.P.; Muzaimi, M. Chronic mitragynine (kratom) enhances punishment resistance in natural reward seeking and impairs place learning in mice. Addict. Biol. 2017, 22, 967–976.
- Senik, M.; Mansor, S.; Rammes, G.; Tharakan, J.; Abdullah, J. Mitragyna speciosa Korth standardized methanol extract induced short-term potentiation of CA1 subfield in rat hippocampal slices. J. Med. Plants Res. 2012, 6, 1234–1243.
- Raymond-Hamet, A. Les alcaloïdes du Mitragyna speciosa Korthals . Ann. Pharm. Françaises 1950, 8, 482–490.
- Fluyau, D.; Revadigar, N. Biochemical benefits, diagnosis, and clinical risks evaluation of kratom. Front. Psychiatry 2017, 8, 62.
- Federico, A.; Morgillo, F.; Tuccillo, C.; Ciardiello, F.; Loguercio, C. Chronic inflammation and oxidative stress in human carcinogenesis. Int. J. Cancer 2007, 121, 2381–2386.
- Hussain, S.P.; Harris, C.C. Inflammation and cancer: An ancient link with novel potentials. Int. J. Cancer 2007, 121, 2373–2380.
- Utar, Z.; Majid, M.I.A.; Adenan, M.I.; Jamil, M.F.A.; Lan, T.M. Mitragynine inhibits the COX-2 mRNA expression and prostaglandin E2 production induced by lipopolysaccharide in RAW264.7 macrophage cells. J. Ethnopharmacol. 2011, 136, 75–82.
- Otero-Losada, M.; Capani, F.; Perez Lloret, S. Neuroprotection—New Approaches and Prospects; IntechOpen: London, UK, 2020; p. 77918.
- Mossadeq, W.S.; Sulaiman, M.; Mohamad, T.T.; Chiong, H.; Zakaria, Z.; Jabit, M.; Baharuldin, M.; Israf, D. Anti-inflammatory and antinociceptive effects of Mitragyna speciosa Korth methanolic extract. Med. Princ. Pract. 2009, 18, 378–384.
- Vermaire, D.J.; Skaer, D.; Tippets, W. Kratom and general anesthesia: A case report and review of the literature. AA Pract. 2019, 12, 103–105.
- Takayama, H.; Ishikawa, H.; Kurihara, M.; Kitajima, M.; Aimi, N.; Ponglux, D.; Koyama, F.; Matsumoto, K.; Moriyama, T.; Yamamoto, L.T.; et al. Studies on the synthesis and opioid agonistic activities of mitragynine-related indole alkaloids: Discovery of opioid agonists structurally different from other opioid ligands. J. Med. Chem. 2002, 45, 1949–1956.
- Horie, S.; Koyama, F.; Takayama, H.; Ishikawa, H.; Aimi, N.; Ponglux, D.; Matsumoto, K.; Murayama, T. Indole alkaloids of a Thai medicinal herb, Mitragyna speciosa, that has opioid agonistic effect in guinea-pig ileum. Planta Medica 2005, 71, 231–236.
- Matsumoto, K.; Takayama, H.; Ishikawa, H.; Aimi, N.; Ponglux, D.; Watanabe, K.; Horie, S. Partial agonistic effect of 9-hydroxycorynantheidine on mu-opioid receptor in the guinea-pig ileum. Life Sci. 2005, 78, 2265–2271.
- Reanmongkol, W.; Keawpradub, N.; Sawangjaroen, K. Effects of the extracts from Mitragyna speciosa Korth. leaves on analgesic and behavioral activities in experimental animals. Songklanakarin J. Sci. Technol. 2007, 29 (Suppl. S1), 39–48.
- Sabetghadam, A.; Ramanathan, S.; Mansor, S.M. The evaluation of antinociceptive activity of alkaloid, methanolic, and aqueous extracts of Malaysian Mitragyna speciosa Korth leaves in rats. Pharmacogn. Res. 2010, 2, 181.
- Idid, S.; Saad, L.; Yaacob, H.; Shahimi, M. Evaluation of analgesia induced by mitragynine, morphine and paracetamol on mice. ASEAN Rev. Biodivers. Environ. Conserv. 1998, 4, 1–7.
- Raffa, R.B. Kratom and Other Mitragynines: The Chemistry and Pharmacology of Opioids from a Non-Opium Source; CRC Press: Boca Raton, FL, USA, 2014.
- Botpiboon, O. Effects of Caffeine and Codeine on Pharmacokinetics and Antinociceptive Activity of Alkaloid Extract from Leaves of Kratom (Mitragyna speciosa Korth.). Ph.D. Thesis, Prince of Songkla University, Hat Yai, Thailand, 2010.
- Matsumoto, K.; Hatori, Y.; Murayama, T.; Tashima, K.; Wongseripipatana, S.; Misawa, K.; Kitajima, M.; Takayama, H.; Horie, S. Involvement of μ-opioid receptors in antinociception and inhibition of gastrointestinal transit induced by 7-hydroxymitragynine, isolated from Thai herbal medicine Mitragyna speciosa. Eur. J. Pharmacol. 2006, 549, 63–70.
- Watanabe, K.; Yano, S.; Horie, S.; Yamamoto, L.T. Inhibitory effect of mitragynine, an alkaloid with analgesic effect from Thai medicinal plant Mitragyna speciosa, on electrically stimulated contraction of isolated guinea-pig ileum through the opioid receptor. Life Sci. 1997, 60, 933–942.
- Matsumoto, K.; Mizowaki, M.; Suchitra, T.; Takayama, H.; Sakai, S.-I.; Aimi, N.; Watanabe, H. Antinociceptive action of mitragynine in mice: Evidence for the involvement of supraspinal opioid receptors. Life Sci. 1996, 59, 1149–1155.
- Suwanlert, S. A study of kratom eaters in Thailand. Bull. Narc. 1975, 27, 21–27.
- Boyer, E.W.; Babu, K.M.; Adkins, J.E.; McCurdy, C.R.; Halpern, J.H. Self-treatment of opioid withdrawal using kratom (Mitragynia speciosa korth). Addiction 2008, 103, 1048–1050.
- Takayama, H. Chemistry and pharmacology of analgesic indole alkaloids from the rubiaceous plant, Mitragyna speciosa. Chem. Pharm. Bull. 2004, 52, 916–928.
- Thongpradichote, S.; Matsumoto, K.; Tohda, M.; Takayama, H.; Aimi, N.; Sakai, S.-I.; Watanabe, H. Identification of opioid receptor subtypes in antinociceptive actions of supraspinally-admintstered mitragynine in mice. Life Sci. 1998, 62, 1371–1378.
- Yamamoto, L.T.; Horie, S.; Takayama, H.; Aimi, N.; Sakai, S.-I.; Yano, S.; Shan, J.; Pang, P.K.; Ponglux, D.; Watanabe, K. Opioid receptor agonistic characteristics of mitragynine pseudoindoxyl in comparison with mitragynine derived from Thai medicinal plant Mitragyna speciosa. Gen. Pharmacol. Vasc. Syst. 1999, 33, 73–81.
- White, C.M. Pharmacologic and clinical assessment of kratom. Bull. Am. Soc. Hosp. Pharm. 2018, 75, 261–267.
- Stolt, A.-C.; Schröder, H.; Neurath, H.; Grecksch, G.; Höllt, V.; Meyer, M.R.; Maurer, H.H.; Ziebolz, N.; Havemann-Reinecke, U.; Becker, A. Behavioral and neurochemical characterization of kratom (Mitragyna speciosa) extract. Psychopharmacology 2014, 231, 13–25.
- Hemby, S.E.; McIntosh, S.; Leon, F.; Cutler, S.J.; McCurdy, C.R. Abuse liability and therapeutic potential of the Mitragyna speciosa (kratom) alkaloids mitragynine and 7-hydroxymitragynine. Addict. Biol. 2019, 24, 874–885.
- Havemann-Reinecke, U. P01-50-Kratom and alcohol dependence: Clinical symptoms, withdrawal treatment and pharmacological mechanisms-A case report. Eur. Psychiatry 2011, 26 (Suppl. S2), 50.
- Obeng, S.; Leon, F.; Patel, A.; Gonzalez, J.D.Z.; Da Silva, L.C.; Restrepo, L.F.; Gamez-Jimenez, L.R.; Ho, N.P.; Calvache, M.P.G.; Pallares, V.L.; et al. Interactive Effects of µ-Opioid and Adrenergic-α (2) Receptor Agonists in Rats: Pharmacological Investigation of the Primary Kratom Alkaloid Mitragynine and Its Metabolite 7-Hydroxymitragynine. J. Pharmacol. Exp. Ther. 2022, 383, 182–198.
- Vijeepallam, K.; Pandy, V.; Kunasegaran, T.; Murugan, D.D.; Naidu, M. Mitragyna speciosa leaf extract exhibits antipsychotic-like effect with the potential to alleviate positive and negative symptoms of psychosis in mice. Front. Pharmacol. 2016, 7, 464.
- Lu, J.; Wei, H.; Wu, J.; Jamil, M.F.A.; Tan, M.L.; Adenan, M.I.; Wong, P.; Shim, W. Evaluation of the cardiotoxicity of mitragynine and its analogues using human induced pluripotent stem cell-derived cardiomyocytes. PLoS ONE 2014, 9, e115648.
- Sandager, M.; Nielsen, N.D.; Stafford, G.I.; van Staden, J.; Jäger, A.K. Alkaloids from Boophane disticha with affinity to the serotonin transporter in rat brain. J. Ethnopharmacol. 2005, 98, 367–370.
- Elgorashi, E.E.; Stafford, G.I.; Jäger, A.K.; Van Staden, J. Inhibition of citalopram binding to the rat brain serotonin transporter by Amaryllidaceae alkaloids. Planta Medica 2006, 72, 470–473.
- Neergaard, J.S.; Andersen, J.; Pedersen, M.E.; Stafford, G.I.; Van Staden, J.; Jäger, A.K. Alkaloids from Boophone disticha with affinity to the serotonin transporter. S. Afr. J. Bot. 2009, 75, 371–374.
- Kong, L.; Cheng, C.H.; Tan, R. Inhibition of MAO A and B by some plant-derived alkaloids, phenols and anthraquinones. J. Ethnopharmacol. 2004, 91, 351–355.
- Oboh, G.; Oyeleye, S.; Ademiluyi, A. The food and medicinal values of indigenous leafy vegetables. Afr. Veg. Forum 2017, 1238, 137–156.
- Nwanna, E.; Oyeleye, S.; Ogunsuyi, O.; Oboh, G.; Boligon, A.; Athayde, M. In vitro neuroprotective properties of some commonly consumed green leafy vegetables in Southern Nigeria. NFS J. 2016, 2, 19–24.
- Oboh, G.; Akinyemi, A.J.; Adeleye, B.; Oyeleye, S.I.; Ogunsuyi, O.B.; Ademosun, A.O.; Ademiluyi, A.O.; Boligon, A.A. Polyphenolic compositions and in vitro angiotensin-I-converting enzyme inhibitory properties of common green leafy vegetables: A comparative study. Food Sci. Biotechnol. 2016, 25, 1243–1249.
- Oboh, G.; Ogunruku, O.O.; Oyeleye, S.I.; Olasehinde, T.A.; Ademosun, A.O.; Boligon, A.A. Phenolic extracts from Clerodendrum volubile leaves inhibit cholinergic and monoaminergic enzymes relevant to the management of some neurodegenerative diseases. J. Diet. Suppl. 2017, 14, 358–371.
- Lühr, S.; Vilches-Herrera, M.; Fierro, A.; Ramsay, R.R.; Edmondson, D.E.; Reyes-Parada, M.; Cassels, B.K.; Iturriaga-Vásquez, P. 2-Arylthiomorpholine derivatives as potent and selective monoamine oxidase B inhibitors. Bioorganic Med. Chem. 2010, 18, 1388–1395.
- Chen, L.; Fei, S.; Olatunji, O.J. LC/ESI/TOF-MS Characterization, Anxiolytic and Antidepressant-like Effects of Mitragyna speciosa Korth Extract in Diabetic Rats. Molecules 2022, 27, 2208.
- Innok, W.; Hiranrat, A.; Chana, N.; Rungrotmongkol, T.; Kongsune, P. In silico and in vitro anti-AChE activity investigations of constituents from Mytragyna speciosa for Alzheimer’s disease treatment. J. Comput.-Aided Mol. Des. 2021, 35, 325–336.
- Tsuji, M.; Takeuchi, T.; Miyagawa, K.; Ishii, D.; Imai, T.; Takeda, K.; Kitajima, M.; Takeda, H. Yokukansan, a traditional Japanese herbal medicine, alleviates the emotional abnormality induced by maladaptation to stress in mice. Phytomedicine 2014, 21, 363–371.
- Neef, D.W.; Jaeger, A.M.; Thiele, D.J. Heat shock transcription factor 1 as a therapeutic target in neurodegenerative diseases. Nat. Rev. Drug Discov. 2011, 10, 930–944.
- Gomez-Pastor, R.; Burchfiel, E.T.; Thiele, D.J. Regulation of heat shock transcription factors and their roles in physiology and disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 4–19.
- Steinkraus, K.A.; Smith, E.D.; Davis, C.; Carr, D.; Pendergrass, W.R.; Sutphin, G.L.; Kennedy, B.K.; Kaeberlein, M. Dietary restriction suppresses proteotoxicity and enhances longevity by an hsf-1-dependent mechanism in Caenorhabditis elegans. Aging Cell 2008, 7, 394–404.
- Gülçin, I.; Elmastaş, M.; Aboul-Enein, H.Y. Antioxidant activity of clove oil–A powerful antioxidant source. Arab. J. Chem. 2012, 5, 489–499.
- Sadeli, R.A. Uji Aktivitas Antioksidan Dengan Metode DPPH (1, 1-Diphenyl-2-Picrylhydrazyl) Ekstrak Bromelain Buah Nanas (Ananas comosus (L.) Merr.). Ph.D. Thesis, Fakultas Farmasi, Universitas Sanata Dharma, Yogyakarta, Indonesia, 2016.
- Rusmarilin, H.; Lubis, Z.; Lubis, L.M.; Barutu, Y.A.P. Potential of natural antioxidants of black cumin seed (Nigella sativa) and sesame seed (Sesamum indicum) extract by microencapsulation methods. IOP Conf. Ser. Earth Environ. Sci. IOP Publ. 2019, 260, 012097.
- Suhaling, S. Uji Aktivitas Antioksidan Ekstrak Metanol Kacang Merah (Phaseolus vulgaris L.) Dengan Metode DPPH. Master’s Thesis, Universitas Islam Negeri Alauddin Makassar, Sulawesi Selatan, Indonesia, 2010.
- Ikhlas, N. Uji Aktivitas Antioksidan Ekstrak Herba Kemangi (Ocimum americanum Linn) dengan Metode DPPH (2, 2-Difenil-1-Pikrilhidrazil). Master’s Thesis. 2013. Available online: http://repository.uinjkt.ac.id/dspace/handle/123456789/25905 (accessed on 7 September 2023).
- Azizi, J.; Ismail, S.; Mordi, M.N.; Ramanathan, S.; Said, M.I.M.; Mansor, S.M. In vitro and in vivo effects of three different Mitragyna speciosa Korth leaf extracts on phase II drug metabolizing enzymes—Glutathione transferases (GSTs). Molecules 2010, 15, 432–441.
- Parthasarathy, S.; Bin Azizi, J.; Ramanathan, S.; Ismail, S.; Sasidharan, S.; Said, M.I.M.; Mansor, S.M. Evaluation of antioxidant and antibacterial activities of aqueous, methanolic and alkaloid extracts from Mitragyna speciosa (Rubiaceae family) leaves. Molecules 2009, 14, 3964–3974.
- Yuniarti, R.; Nadia, S.; Alamanda, A.; Zubir, M.; Syahputra, R.A.; Nizam, M. Characterization, phytochemical screenings and antioxidant activity test of kratom leaf ethanol extract (Mitragyna speciosa Korth) using DPPH method. J. Phys. Conf. Ser. IOP Publ. 2020, 1462, 012026.
- Chae, H.S.; Park, H.J.; Hwang, H.R.; Kwon, A.; Lim, W.H.; Yi, W.J.; Han, D.H.; Kim, Y.H.; Baek, J.H. The effect of antioxidants on the production of pro-inflammatory cytokines and orthodontic tooth movement. Mol. Cells 2011, 32, 189–196.





