
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ana Cláudia Pereira | -- | 3389 | 2023-11-16 13:37:25 | | | |
| 2 | Peter Tang | -1 word(s) | 3388 | 2023-11-17 02:47:13 | | |
Video Upload Options
This research presents the virulence factors, clinical relevance, and current treatments of C. acnes, highlighting its association with AV, post-surgical infections, and other diseases. It also explores alternative innovative therapies such as phage therapy in development/research that are gaining prominence, with a growing focus on personalized medical approaches.
1. Taxonomy and Nomenclature
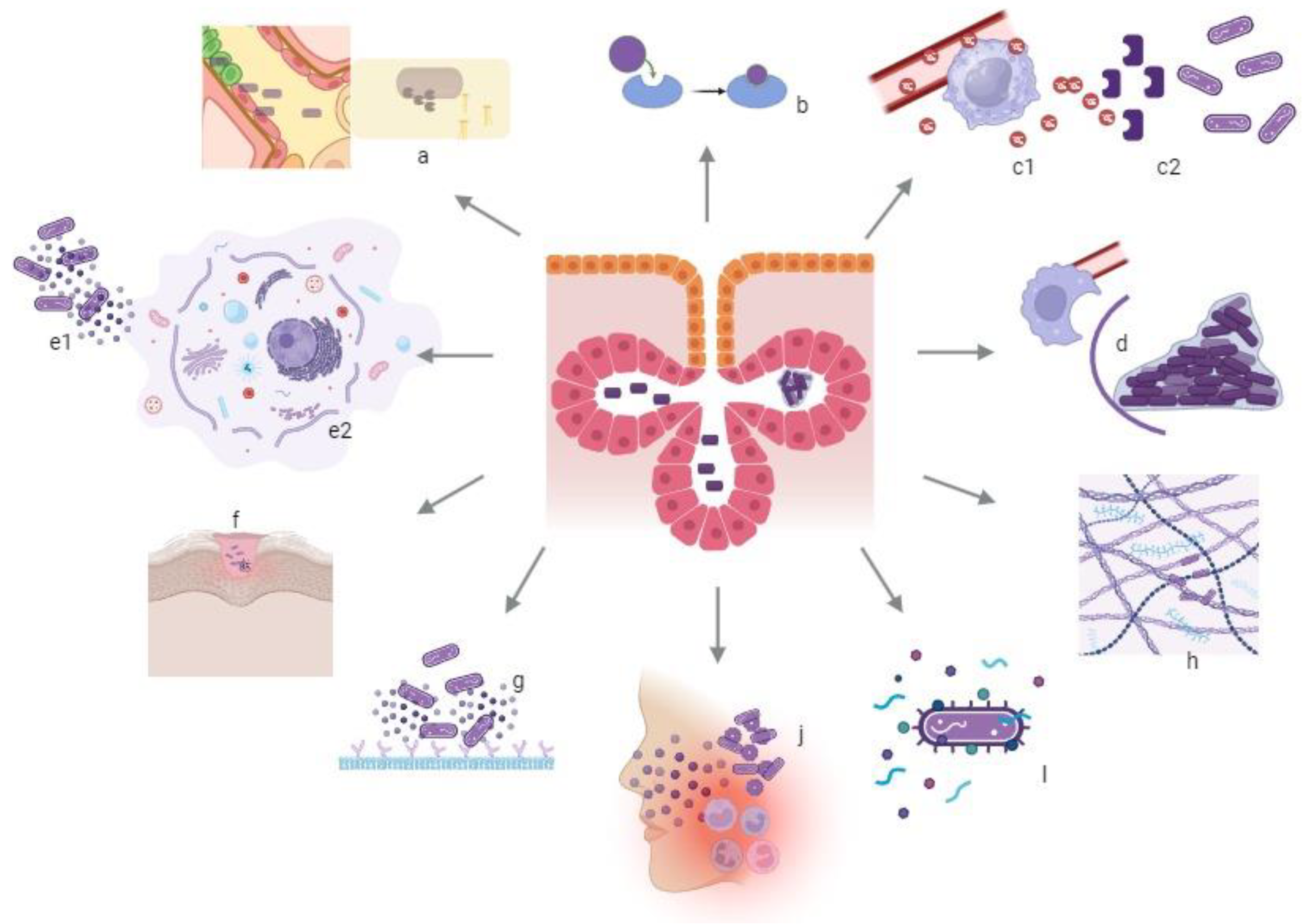
2. C. acnes Infection and Virulence Factors
|
Virulence Factors |
Function |
|---|---|
|
Lipases |
Enzymes involved in the metabolization of sebum and free fatty acid release and triglycerides. |
|
Polyunsaturated Fatty Acid Isomerase |
Catalyzation and isomerization of linoleic acid. |
|
Hyaluronate Lyase |
Promotes the degradation of hyaluronic acid andolaolablablaother glycosaminoglycans, such as chondroitin-4-sulfate, chondroitin-6-sulfate, and dermatan sulfate, of the extracellular matrix in the epidermis and dermis. |
|
Glycosidase |
Disruption of carbohydrate and glycan structures that constitute the eukaryotic host glycolipids and glycoproteins. |
|
Sialidase |
Discard the sialic acid from sialoglycoconjugates. |
|
Radical oxygenase |
Reduction of the oxygen free radicals. |
|
Sortase F |
Capacity to covalently attach to various proteins, including adhesion factors. |
|
Porphyrin |
Fluorescent molecules that can stimulate inflammatory host reactions. |
|
Biofilm |
Matrix that provides bacterial resistance to adverse compounds, such as antibiotics |
|
Adhesin dermatan-sulfate protein |
Molecular surface components that recognize adhesive molecules of the matrix. |
|
Christie–Atkins–Munch–Petersen Factors |
Promote the formation of pores in host cells membranes. |
3. C. acnes Clinical Relevance
Acne Vulgaris (AV)
|
Score |
Acne Severity Denomination |
Type of Lesions Observed |
|---|---|---|
|
0 |
Clear skin |
No lesions observed. |
|
1 |
The skin is almost unchanged |
Few comedones and less, or 1, small inflammatory lesion. |
|
2 |
Mild severity |
12 comedones and less or equal severe inflammatory lesions. |
|
3 |
Moderate severity |
Many comedones and more several inflammatory lesions and less, or 1, nodule. |
|
4 |
Severe severity |
Many comedones and inflammatory lesions, less or equal several nodules and cysts. |
|
Global Score |
Lesions Count |
|
None: 0 Mild: 1 to 18 Moderate: 31 to 38 Very severe: >39 |
|
|
Local score = Factor × Grade (0–4) |
|
|
Factor (1–3) |
Grade (0–4) |
|
Nose/chin: 1 Forehead/right cheek/left cheek: 2 Chest and upper back: 3 |
No lesions: 0 One or more comedone: 1 One or more papule: 2 One or more pustule: 3 One or more nodule: 4 |
4. C. acnes Antibiotic Treatment
|
Compound |
Antibacterial Mechanism |
Positive Factors |
Negative Factors |
|---|---|---|---|
|
Benzoyl peroxide (BPO) |
The discharge of reactive oxygen intermediates oxidizes the proteins in the bacterial cell membrane. |
No bacterial resistance to BPO has emerged despite decades of use. Keratolytic and anti-inflammatory properties are an additional component of BPO. |
BPO is expensive and is a skin irritant, especially in darker skin types. |
|
Clindamycin |
Inhibits the bacterial 50S ribosome-mediated protein production. |
Has a synergetic effect when used with BPO. Fox–Fordyce illness, folliculitis, periorificial face dermatitis, and rosacea have all been treated successfully with topical clindamycin, according to reports. |
C. acnes isolates was shown to be resistant to clindamycin. Topical clindamycin side effects generally take the shape of dryness, stinging, burning, and erythema. |
|
Micozanole Nitrate (MN) |
Antifungal drug that affects the integrity of fungal cell membranes. |
Annihilates Malassezia furfur, a fungus that provides an optimal environment for the growth of C. acnes. |
May provoke allergic reactions, skin irritation such as erythema, pruritus, and occasionally exudation. |
|
Hydrogen Peroxide (HP) |
Is known by its powerful antiseptic activity against the vast known microorganisms in the skin. It can be used in the concentrations of 3 to 6% of (v/v). Even though the precise mechanism of action of hydrogen peroxide is unknown, it is widely thought that it is connected to its oxidizing activity. |
No cases of acquired bacterial resistance to HP have been reported. PVP–I and HP interact positively. |
HP concentrated solutions (20–30% or more) are extremely irritating to the skin and mucous membranes and should be handled carefully. |
|
Chlorhexidine (CHX) |
CHX has an antibacterial activity by affecting the integrity of cell membranes. |
Being a highly safe topical medication, chlorhexidine is also commonly found in wound dressings and central line catheters. Chlorhexidine has a broad spectrum of activity and persistent residual effects. |
Associated with poor efficacy, chlorhexidine side effects are uncommon but include minor skin irritation and, less often, allergic responses such as severe anaphylaxis. |
|
Povidone-iodine (PVP-I) |
It is hypothesized that PVP–I mechanisms include the inhibition of the electron transportation and cellular and inhibiting protein synthesis. |
It is considered, among the antiseptics, the one with the broadest spectrum of activity against viruses, bacteria, molds, fungi, yeasts, and protozoa. |
Low solubility, poor chemical stability, and shows local toxicity if not used in a soluble polymer matrix. PVP–I should not be used in patients with thyroid diseases and applicated iodine radiotherapy and it is also contraindicated to pregnant women or during lactation, and to newborns, and to young children. |
|
Isopropanol |
It is hypothesized that alcohols promote the protein denaturation or inhibition of mRNA and protein synthesis. |
Rapid bacterial activity and broad spectrum of activity (vegetative bacteria, including mycobacteria, viruses, fungi, but not against bacterial spores). No reported allergic reactions. |
Alcohols’ antimicrobial properties are brief, so they are commonly combined with compounds such as chlorhexidine, which keep working after the alcohol has evaporated. |
5. New Therapeutic Strategies
To overcome bacterial resistance concerns, there are strategies being explored, not only envisioning therapy efficacy but also a personalized medical approach [28]. Improvements in the treatment of C. acnes rely on the development of novel therapies that maximize efficacy while reducing side effects, as well as issues in public health, such as antibiotics prescriptions.
Since a healthy skin flora can be influenced by exogenous and endogenous factors causing pathological conditions for the host, one of the strategies is the use of bacterial extracts and compounds. This strategy aims to help recover the healthy state of the skin in different pathological conditions such as AV [29]. Ho et al. studied the effects of fermented postbiotics (TYCA06, AP-32 and CP-9 and collagen gel) in the growth inhibition of C. acnes, as well as an in vivo assay on the skin of patients with oily skin and severe AV. The postbiotics displayed a good growth inhibition for C. acnes, and in clinical trials no adverse effects was observed, showing significant reduction in redness, inflammation, and accumulation of porphyrins in skin brown spots. Moreover, there was a significant improvement in the skin hydration and in the AV lesions (in only one week of treatment), even though there was no significant reduction in the sebum skin [30]. Han et al. studied the effects of the E. faecalis CBT SL-5 extract, isolated from healthy Korean human fecal samples in patients with mild to moderate AV. The treatment significantly reduced the phylogenetic diversity in the patient’s skin and was well tolerated by the patients. However, no significant difference was observed in the treatment and in the vehicle lotion in the improvement of AV [31]. Tsai et al. applied a base cream including heat-killed L. plantarum-GMNL6 on one side of the face in 15 females. The treatment significantly reduced the amount of C. acnes and reduced, inclusively, the red areas and the porphyrin [32]. Karoglan et al. performed a microbiome transplantation of beneficial strains of C. acnes to the patient’s skin. The results were not statistically significant but clinical improvements in the noninflammatory lesions were observed. The major limitation of the study was the absence of a control or placebo group and the small participant sample, highlighting the need for further studies with larger groups to correlate the microbiome transplantation with beneficial outcome in AV patients [33].
More recently, phage therapy has been increasingly studied as an innovative approach in bacterial infection treatments.
Phage therapy, short for bacteriophage therapy, is a type of treatment that uses bacteriophages to target and kill specific bacteria. Bacteriophages, often referred to as phages, are viruses that infect and replicate within bacteria, ultimately leading to the bacterial cell’s death [34]. This way, phage therapy constitutes a potential approach for treating bacterial infections, including those caused by bacteria like C. acnes, and involves identifying and isolating specific bacteriophages that can target and infect C. acnes bacteria. Once suitable phages are isolated, they can be purified and used to treat the infection [35]. The phages attach to the surface of C. acnes bacteria, inject their genetic material, and then replicate inside the bacterial cell. This replication ultimately leads to the lysis (bursting) of the bacterial cell, killing it, and releasing more phages to attack other bacteria [35].
Phage therapy entails several advantages, such as (a) specificity, as phages are highly specific to the target bacteria, reducing the risk of harming beneficial bacteria in the body; (b) reduced antibiotic resistance, since phages can be effective against bacteria with multiresistant profile and help overcome virulence mechanisms such as the production of biofilms; and (c) potential for personalized treatment, since it constitutes a therapy that can be tailored to a specific strain that is causing infection [28][34]. This therapy has been considered an optimistic possibility to provide specific treatment in infections with pathogenic bacteria and in dysbiosis conditions when compared to the use of antibiotics [28]. As such, several studies have been conducted, using phages as the vehicle to treat C. acnes bacterial infection.
An important therapeutic aspect is phages’ temperate nature, allowing coexistence with their host and enabling gene transfer, including antibiotic resistance genes. The safety of phage therapy relies on understanding these dynamics. A study conducted with antibiotic treated mice was found to increase phage integration into bacterial genomes, enriching the phage metagenome with stress-specific functions, shaping the phage–bacterial network. This enrichment also included functions related to host metabolism, such as a broader carbohydrate pathway in ampicillin-treated mice [36]. Such strategies could provide a protective effect in the gut microflora during antibiotic treatments.
Despite all the advantages in this innovative approach, phage therapy also entails some challenges that make its applicability not straight forward.
Identifying and isolating appropriate phages can be challenging and time-consuming, since the affinity towards a specific strain is essential to an effective treatment. Another challenge is the regulatory approval. In many countries, phage therapy is considered experimental, failing to check all the necessary bureaucracies for clinical application and, therefore, not reaching global population. One more challenge, that somewhat correlates with the former, is the lack of clinical data. This limitation constricts the flexibility in terms of phage therapy as a personalized treatment, due to its regulatory limitations that, in the end, result in few clinical trials.
References
- Corvec, S.; Dagnelie, M.-A.; Khammari, A.; Dréno, B. Taxonomy and phylogeny of Cutibacterium (formerly Propionibacterium) acnes in inflammatory skin diseases. Ann. Dermatol. Venereol. 2019, 146, 26–30.
- Dekio, I.; Asahina, A.; Shah, H.N. Unravelling the eco-specificity and pathophysiological properties of Cutibacterium species in the light of recent taxonomic changes. Anaerobe 2021, 71, 102411.
- Mayslich, C.; Grange, P.A. Cutibacterium acnes as an Opportunistic Pathogen: An Update of Its Virulence-Associated Factors. Microorganisms 2021, 9, 303.
- Yang, Y.; Qu, L.; Mijakovic, I.; Wei, Y. Advances in the human skin microbiota and its roles in cutaneous diseases. Microb. Cell Fact. 2022, 21, 176.
- Falconer, T.M.; Baba, M.; Kruse, L.M.; Dorrestijn, O.; Donaldson, M.J.; Smith, M.M.; Figtree, M.C.; Hudson, B.J.; Cass, B.; Young, A.A. Contamination of the Surgical Field with Propionibacterium acnes in Primary Shoulder Arthroplasty. J. Bone Jt. Surg. Am. 2016, 98, 1722–1728.
- Khatoon, Z.; McTiernan, C.D.; Suuronen, E.J.; Mah, T.-F.; Alarcon, E.I. Bacterial biofilm formation on implantable devices and approaches to its treatment and prevention. Heliyon 2018, 4, e01067.
- Siddiqui, R.; Makhlouf, Z.; Ahmed, N. The increasing importance of the gut microbiome in acne vulgaris. Folia Microbiol. 2022, 67, 825–835.
- Elston, M.J.; Dupaix, J.P.; Opanova, M.I.; Atkinson, R.E. Cutibacterium acnes (formerly Proprionibacterium acnes) and Shoulder Surgery. Hawai’i J. Health Soc. Welf. 2019, 78 (Suppl. S2), 3–5.
- Martin, D.R.; Witten, J.C.; Tan, C.D.; Rodriguez, E.R.; Blackstone, E.H.; Pettersson, G.B.; Seifert, D.E.; Willard, B.B.; Apte, S.S. Proteomics identifies a convergent innate response to infective endocarditis and extensive proteolysis in vegetation components. JCI Insight 2020, 5, e135317.
- Boman, J.; Nilson, B.; Sunnerhagen, T.; Rasmussen, M. True infection or contamination in patients with positive Cutibacterium blood cultures—A retrospective cohort study. Eur. J. Clin. Microbiol. Infect. Dis. 2022, 41, 1029–1037.
- Kraaijvanger, R.; Veltkamp, M. The Role of Cutibacterium acnes in Sarcoidosis: From Antigen to Treatable Trait? Microorganisms 2022, 10, 1649.
- Ugge, H.; Carlsson, J.; Söderquist, B.; Fall, K.; Andén, O.; Davidsson, S. The influence of prostatic Cutibacterium acnes infection on serum levels of IL6 and CXCL8 in prostate cancer patients. Infect. Agents Cancer 2018, 13, 7.
- Radej, S.; Szewc, M.; Maciejewski, R. Prostate Infiltration by Treg and Th17 Cells as an Immune Response to Propionibacterium acnes Infection in the Course of Benign Prostatic Hyperplasia and Prostate Cancer. Int. J. Mol. Sci. 2022, 23, 8849.
- Coenye, T.; Spittaels, K.-J.; Achermann, Y. The role of biofilm formation in the pathogenesis and antimicrobial susceptibility of Cutibacterium acnes. Biofilm 2022, 4, 100063.
- Xu, H.; Li, H. Acne, the Skin Microbiome, and Antibiotic Treatment. Am. J. Clin. Dermatol. 2019, 20, 335–344.
- Zohra, F.T.; Sultana, T.; Islam, S.; Nasreen, A. Evaluation of Severity in Patients of Acne Vulgaris by Global Acne Grading System in Bangladesh. Clin. Pathol. Res. J. 2017, 1, 000105.
- Szymańska, A.; Budzisz, E.; Erkiert-Polguj, A. The Anti-Acne Effect of Near-Infrared Low-Level Laser Therapy. Clin. Cosmet. Investig. Dermatol. 2021, 14, 1045–1051.
- Mawardi, P.; Ardiani, I.; Primisawitri, P.P.; Nareswari, A. Dual role of Cutibacterium acnes in acne vulgaris pathophysiology. Bali Med. J. 2021, 10, 486–490.
- Sheffer-Levi, S.; Rimon, A.; Lerer, V.; Shlomov, T.; Coppenhagen-Glazer, S.; Rakov, C.; Zeiter, T.; Nir-Paz, R.; Hazan, R.; Molho-Pessach, V. Antibiotic Susceptibility of Cutibacterium acnes Strains Isolated from Israeli Acne Patients. Acta Derm. Venereol. 2020, 100, adv00295.
- Kuriyama, T.; Karasawa, T.; Williams, D.W. Chapter Thirteen—Antimicrobial Chemotherapy: Significance to Healthcare. In Biofilms in Infection Prevention and Control; Percival, S.L., Williams, D.W., Randle, J., Cooper, T., Eds.; Academic Press: Boston, MA, USA, 2014; pp. 209–244.
- Peckman, B.; Kharel, M.K. Erythromycin. In Reference Module in Biomedical Sciences; Elsevier: Amsterdam, The Netherlands, 2022.
- Alkhawaja, E.; Hammadi, S.; Abdelmalek, M.; Mahasneh, N.; Alkhawaja, B.; Abdelmalek, S.M. Antibiotic resistant Cutibacterium acnes among acne patients in Jordan: A cross sectional study. BMC Dermatol. 2020, 20, 17.
- Legiawati, L.; Halim, P.A.; Fitriani, M.; Hikmahrachim, H.G.; Lim, H.W. Microbiomes in Acne Vulgaris and Their Susceptibility to Antibiotics in Indonesia: A Systematic Review and Meta-Analysis. Antibiotics 2023, 12, 145.
- Bandyopadhyay, D. Topical Antibacterials in Dermatology. Indian J. Dermatol. 2021, 66, 117–125.
- Letzelter, J.; Hill, J.B.; Hacquebord, J. An Overview of Skin Antiseptics Used in Orthopaedic Surgery Procedures. J. Am. Acad. Orthop. Surg. 2019, 27, 599–606.
- Fatemi, F.; Najafian, J.; Savabi Nasab, S.; Nilforoushzadeh, M.A. Treatment of Acne Vulgaris Using the Combination of Topical Erythromycin and Miconazole. J. Ski. Stem Cell 2014, 1, e23330.
- National Center for Biotechnology Information. PubChem Compound Summary for CID 68553, Miconazole Nitrate. 2023. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Miconazole-nitrate (accessed on 18 September 2023).
- Kim, S.; Song, H.; Jin, J.S.; Lee, W.J.; Kim, J. Genomic and Phenotypic Characterization of Cutibacterium acnes Bacteriophages Isolated from Acne Patients. Antibiotics 2022, 11, 1041.
- Condrò, G.; Guerini, M.; Castello, M.; Perugini, P. Acne Vulgaris, Atopic Dermatitis and Rosacea: The Role of the Skin Microbiota-A Review. Biomedicines 2022, 10, 2523.
- Ho, H.-H.; Chen, C.-W.; Yi, T.-H.; Huang, Y.-F.; Kuo, Y.-W.; Lin, J.-H.; Chen, J.-F.; Tsai, S.-Y.; Chan, L.-P.; Liang, C.-H. Novel application of a Co-Fermented postbiotics of TYCA06/AP-32/CP-9/collagen in the improvement of acne vulgaris-A randomized clinical study of efficacy evaluation. J. Cosmet. Dermatol. 2022, 21, 6249–6260.
- Han, H.S.; Shin, S.H.; Choi, B.-Y.; Koo, N.; Lim, S.; Son, D.; Chung, M.J.; Park, K.Y.; Sul, W.J. A split face study on the effect of an anti-acne product containing fermentation products of Enterococcus faecalis CBT SL-5 on skin microbiome modification and acne improvement. J. Microbiol. 2022, 60, 488–495.
- Tsai, W.; Chou, C.; Chiang, Y.; Lin, C.; Lee, C. Regulatory effects of Lactobacillus plantarum-GMNL6 on human skin health by improving skin microbiome. Int. J. Med. Sci. 2021, 18, 1114–1120.
- Karoglan, A.; Paetzold, B.; Pereira de Lima, J.; Brüggemann, H.; Tüting, T.; Schanze, D.; Güell, M.; Gollnick, H. Safety and Efficacy of Topically Applied Selected Cutibacterium acnes Strains over Five Weeks in Patients with Acne Vulgaris: An Open-label, Pilot Study. Acta Derm. Venereol. 2019, 99, 1253–1257.
- Xuan, G.; Wang, Y.; Wang, Y.; Lin, H.; Wang, C.; Wang, J. Characterization of the newly isolated phage Y3Z against multi-drug resistant Cutibacterium acnes. Microb. Pathog. 2023, 180, 106111.
- Han, M.-H.; Khan, S.A.; Moon, G.-S. Cutibacterium acnes KCTC 3314 Growth Reduction with the Combined Use of Bacteriophage PAP 1-1 and Nisin. Antibiotics 2023, 12, 1035.
- Modi, S.R.; Lee, H.H.; Spina, C.S.; Collins, J.J. Antibiotic treatment expands the resistance reservoir and ecological network of the phage metagenome. Nature 2013, 499, 219–222.





