Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Umesh K. Reddy | -- | 2217 | 2023-09-26 16:41:01 | | | |
| 2 | Lindsay Dong | Meta information modification | 2217 | 2023-09-28 03:06:06 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Lopez-Ortiz, C.; Gracia-Rodriguez, C.; Belcher, S.; Flores-Iga, G.; Das, A.; Nimmakayala, P.; Balagurusamy, N.; Reddy, U.K. Drosophila melanogaster as Phytochemicals Translational Model System. Encyclopedia. Available online: https://encyclopedia.pub/entry/49679 (accessed on 24 May 2026).
Lopez-Ortiz C, Gracia-Rodriguez C, Belcher S, Flores-Iga G, Das A, Nimmakayala P, et al. Drosophila melanogaster as Phytochemicals Translational Model System. Encyclopedia. Available at: https://encyclopedia.pub/entry/49679. Accessed May 24, 2026.
Lopez-Ortiz, Carlos, Celeste Gracia-Rodriguez, Samantha Belcher, Gerardo Flores-Iga, Amartya Das, Padma Nimmakayala, Nagamani Balagurusamy, Umesh K. Reddy. "Drosophila melanogaster as Phytochemicals Translational Model System" Encyclopedia, https://encyclopedia.pub/entry/49679 (accessed May 24, 2026).
Lopez-Ortiz, C., Gracia-Rodriguez, C., Belcher, S., Flores-Iga, G., Das, A., Nimmakayala, P., Balagurusamy, N., & Reddy, U.K. (2023, September 26). Drosophila melanogaster as Phytochemicals Translational Model System. In Encyclopedia. https://encyclopedia.pub/entry/49679
Lopez-Ortiz, Carlos, et al. "Drosophila melanogaster as Phytochemicals Translational Model System." Encyclopedia. Web. 26 September, 2023.
Copy Citation
Drosophila melanogaster, an invertebrate model with its extensively studied genome, has more than 70% gene homology to humans and has been used as a model system in biological studies for a long time. The notable advantages of Drosophila as a model system, such as their low maintenance cost, high reproductive rate, short generation time and lifespan, and the high similarity of metabolic pathways between Drosophila and mammals, have encouraged the use of Drosophila in the context of screening and evaluating the impact of phytochemicals present in the diet.
Drosophila
phytochemical
human health
metabolism
disease
1. Introduction
Phytochemicals are specialized metabolites with biological properties stored in plant tissues, such as roots, stems, leaves, flowers, and fruits [1]. Phytochemicals include a wide range of compounds such as polyphenols, carotenoids, flavonoids, coumarins, terpenoids, glucosinolates, saponins, and capsaicinoids, which are often associated with the vibrant colors of fruits and vegetables [2]. Although phytochemicals are not essential nutrients in plants, they are responsible for many health benefits associated with a plant-based diet [3][4]. Potential phytochemical health benefits include antioxidant, anti-inflammatory, anti-cancer, and anti-microbial properties [5]. Currently, numerous phytochemicals are being studied for their possible use in developing novel drugs and dietary supplements [6].
Phytochemicals exhibit their beneficial and harmful effects by interaction with multiple cell signaling molecules [7]. Nevertheless, the molecular mechanisms underlying the effectiveness of phytochemicals continue to grow. In this context, employing cost-effective, rapid, reliable, and efficient in vitro and in vivo assays will facilitate analyzing these compounds’ metabolic processes, dosage response, and pharmacological and toxicological profiles [8][9][10]. D. melanogaster, known as the fruit fly, has emerged as an alternate animal model. Due to its short lifespan, small size, and well-understood genome, it has been widely used to examine the efficacy and safety of phytochemicals on various physiological processes, including metabolism, aging, and immunity [11][12].
2. Phytochemicals and Their Potential Therapeutic Benefits
Regular consumption of fruits, vegetables, and grains has been associated with a reduced risk of certain chronic diseases due to phytochemicals with antioxidant and anti-inflammatory properties [13]. Phytochemicals regulate oxidative stress, which has been recognized as a significant factor in the pathogenesis of metabolic disorders and cancer [14]. Phytochemical therapeutic benefits are divided mainly into five categories (i) enhancers of the body’s immune system; (ii) preventers of diabetes and heart diseases; (iii) hypocholesterolemic agents; (iv) promoters of digestion and absorption; and (v) retardants of aging [15][16]. The major classes of phytochemicals with disease-preventing functions are dietary fiber, antioxidants, detoxifying agents, immunity-potentiating, and neuropharmacological agents [17].
For instance, polyphenols such as resveratrol have been associated with decreased risk of myocardial infarction, stroke, and diabetes [18][19]. Polyphenols in diet also help to improve lipid profiles, blood pressure, insulin resistance, and systemic inflammation [20]. Furthermore, vitamin C and carotenoids may benefit immune function, thereby reducing cancer risk by enhancing the tumor surveillance of the immune system [21]. Capsaicinoids, including capsaicin and dihydrocapsaicin found in peppers, have been found to have beneficial roles in humans, including managing pain inflammation during rheumatoid arthritis, anti-cancer agent by generating reactive oxygen species, and in the prevention or treatment of neurodegenerative diseases such as Alzheimer’s disease due to its antioxidant activity [22][23][24].
Other spices, such as turmeric obtained from the roots of Curcuma domestica, contain a yellow coloring principle, curcumin, a powerful antioxidant that can offer protection against cancer, inhibiting lipid peroxide-induced DNA damage [25]. Flavonoid consumption, such as quercetin and kaemferol, through vegetables and fruits, reduces the risk of death from coronary heart disease [26].
3. Advantages of Using Drosophila as a Translational Model for Testing Phytochemicals
For decades, D. melanogaster has been widely used as an excellent animal model to study genetics, evolution, and developmental biology [27]. It is a cost-effective option due to its high reproductive rate (30–50 eggs/day), short generation time (approximately ten days at 25 °C), and low maintenance cost. In addition, Drosophila short lifespan (average three months at 25 °C) and easy generation of large populations allow for performing longevity and lifespan assays in only a few months [28]. Furthermore, it offers powerful molecular and genetic tools that permit gene overexpression or knock-down studies [29].
Although Drosophila is evolutionarily distant from humans, fly development, physiological, biological, and metabolic processes are equivalent to many of those found in mammals. Recently, similarities between humans and fruit flies in terms of metabolic regulation, including the role of insulin signaling, nutrient sensing, and energy homeostasis in metabolic disorders, such as diabetes and obesity [30][31][32][33]. Moreover, ingesting complex foods rich in phytochemicals by an organism leads to the degradation of nutrients that directly affect the gastrointestinal microbiome because the host and microbiome share the same food source [34]. These microbiome changes influence the organism’s phenotype and behavior by altering the genome, transcriptome, epigenome, proteome, and metabolome [35].
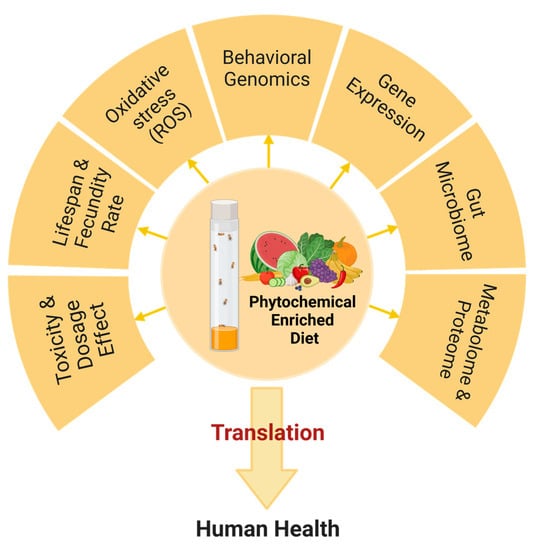
4. Methods for Testing the Efficacy of Phytochemicals in Drosophila
Phytochemical ingestion effects are observed in general physiology, including metabolism, behavior, stress resistance, reproductive capacity, nervous system, and immune capacity in both Drosophila and humans, and aspects of these physiological changes can be used as parameters to determine the phytochemical effects and toxicity [36]. Among them, lifespan and survival rate are simple and efficient longitudinal assays to determine the effects after administration of the candidate plant-based compounds [37]. In addition, various strains with different lifespan characteristics and transgenic flies with symptoms similar to human diseases are available to evaluate plant compounds’ effects on mortality rate.
Oxidative stress mitigation in Drosophila is also an essential parameter in assessing the efficacy of phytochemicals. Levels of reactive oxygen species (ROS), activities of antioxidant enzymes, such as superoxide dismutase and catalase, and lipid peroxidation in treated fly cells provide insight into the potential antioxidative activities of phytochemicals (Figure 1) [38]. Moreover, Drosophila has a relatively simple nervous system, making it an ideal model to study neurodevelopment and neurodegeneration using automated tracking systems, locomotor activity, ring assay, gustatory, social, and circadian rhythm patterns, providing insight into potential impacts of phytochemicals on the nervous system function and behavior [39].

Figure 1. Approaches for evaluating the effects of phytochemicals in Drosophila melanogaster model.
Although administration of compounds via feeding is the most typical method of phytochemical delivery to Drosophila [36], it is important to consider several concerns about feeding-related artifacts, such as reduced feeding related to the preference of flies for each compound, uncertainty about the amount of food consumed by each individual and of the actual phytochemical concentration achieved in the tissues of the flies.
5. Studies Evaluating the Effect of Phytochemicals by Using Drosophila
5.1. Phytochemical Effect on Aging
Anti-aging research has gained significant attention due to the growing aging population, which experiences a natural decline in the body’s capacity to repair molecular, biochemical, and organ damage, resulting in increased susceptibility to age-related diseases [40]. The primary cause of radical damage to macromolecules in aging is the progressive decline of the endogenous antioxidant system, triggered by reactive oxygen species (ROS). To restore the ROS balance, natural plant-derived antioxidants can be utilized to reinforce the endogenous antioxidant system [41].
Plant-derived compounds with anti-aging properties have been investigated. Moringa oleifera has been identified as one of the plants with the highest levels of bioactive molecules, such as polyphenols, flavonoids, and tannins, which elicit an antioxidant response [42].
Withanolides are widely found in Solanaceae species and have been investigated for their potential to enhance resilience against age-related stress [43]. Withania somnifera extracts with high withanolides concentration have been shown to ameliorate behavioral deficits in an in vivo D. melanogaster model of oxidative stress, reducing the effects of aging in locomotion and cognition [44].
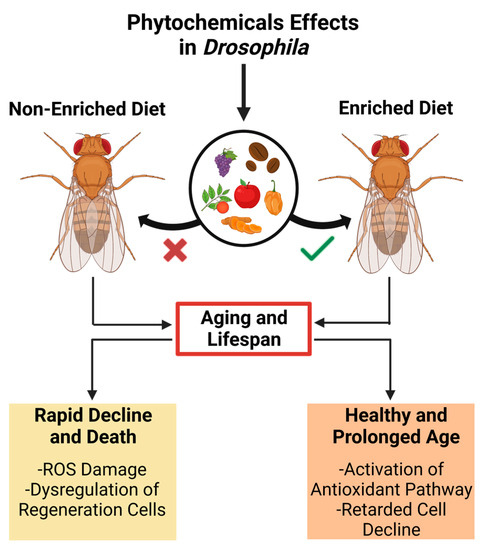
5.2. Development and Lifespan
Age-related dysregulation of development is closely associated with functional organ and tissue decline, affecting lifespan and age-related disease development [40]. A modern lifestyle characterized by high caloric intake and minimal physical activity in humans results in high lipid storage levels that reduce overall lifespan [45]. Nevertheless, several studies have shown that several phytochemicals can expand the lifespan using Drosophila as a model (Figure 2) [46].

Figure 2. The impact of a phytochemical-enriched diet on the lifespan of Drosophila melanogaster suggests potential applications for promoting healthy aging and longevity in humans.
5.3. Metabolism
Recent evidence suggests that the metabolic state of an organism is closely tied to its diet, with an important factor being that the dietary habits of parents can impact the metabolic states of their offspring [47]. Controlled dietary conditions are crucial for studying metabolism and organism physiology [48]. Drosophila possesses notable metabolic systems that share many conserved functions with vertebrates, including insulin, insulin-like growth factor, the target of rapamycin signaling pathways, and energy regulation [49][50].
Obesity is a prevalent metabolic syndrome in humans, leading to various metabolic complications, such as impaired glucose tolerance, insulin resistance, dyslipidemia, hypertension, type 2 diabetes, and premature heart disease [51]. Heinrichsen et al. [52] evaluated the metabolic response of Drosophila fed a high-fat diet, which resulted in increased levels of triglyceride and glucose, decreased stress tolerance and lifespan, and activation of pathways associated with fat metabolism, insulin signaling, cardiac fat accumulation, and dysfunction.
Moreover, the effects of resveratrol were also analyzed in Drosophila metabolism. It was observed that resveratrol supplementation improved metabolic parameters, such as enhanced glucose tolerance and reduced lipid accumulation. Nevertheless, the implementation of target-specific therapy might be beneficial in mitigating any negative consequences arising from the pro-oxidant activity associated with high dosages [53]. Meanwhile, quercetin supplementation, a flavonoid in various fruits and vegetables, improved glucose homeostasis, reduced oxidative stress, and enhanced mitochondrial function in flies [54].
5.4. Microbiome
The gut microbiome in the human intestines plays a critical role in nutrient absorption, lysis, and storage [55]. Additionally, they are essential for various physiological processes, including metabolism, digestion, circadian rhythms, and vitamin synthesis in humans [56]. Imbalances in the gut microbiota (dysbiosis) due to diet, antibiotic use, age, and stress contribute to disease development, including diabetes, obesity, colon cancer, inflammatory bowel disease, inflammation, and neurodegeneration [57].
The Drosophila gut microbiome is extracellular and encompasses three regions: the foregut, midgut, and hindgut, each creating distinct conditions for microbial cells [58]. Although the Drosophila gut microbiome has been well-documented, it is important to note that fruit flies have a limited number of microorganisms, about 30 species, compared to mammals with more than 500 species [59]. The gut microbial population is profoundly influenced by the dietary habits associated with consuming different types and amounts of phytochemicals (99). It also varies depending on the host genotype, age, sex, and habitat [60][61].
Moreover, Jimenez-Padilla et al. [62] found that Drosophila flies fed diets with strawberries and blueberries had an increased abundance of Acetobacter in their microbiome. Acetobacter, particularly Acetobacter pomorum, has probiotic properties and produces acetic acid, which promotes insulin signaling, reducing lipid and sugar levels in adult flies [63]. The blueberry diet also led to higher levels of Actinobacteria compared to the control diet [62]. Actinobacteria, including the genus Bifidobacterium, metabolize anthocyanins into small compounds with probiotic effects for obesity and gastrointestinal and systemic diseases [64][65].
5.5. Neurodegenerative Diseases
D. melanogaster has been widely used in drug screening studies to identify high-quality hits that exhibit crucial characteristics, including metabolic stability, oral or transdermal availability, and, most notably, low toxicity, providing a valuable resource for drug development [28]. Phytochemicals have been extensively studied for their potential in preventing and controlling the proliferation and development of tumor cells [66]. Black beans, specifically Phaseolus vulgaris, contain phenolic compounds, including cyanidin-3-O-glucoside (C3G), recognized as an anti-cancer compound [67].
Likewise, phytochemicals can offer neuroprotection, which is crucial for developing new treatments for neurodegenerative diseases, such as Alzheimer’s. In Alzheimer’s disease, memory loss is associated with the forming beta-amyloid plaques, which give rise to oligomers that generate reactive oxygen species (ROS) and promote Tau protein aggregation, ultimately leading to neuronal cell death [68].
Moreover, epilepsy is a neurological disorder characterized by sensory-motor deficits and convulsions. It can have various causes, including gene mutations that encode ion channels in brain cells responsible for transmitting signals between neurons [69]. To alleviate epilepsy symptoms, researchers have investigated using Imperata cylindrica root extracts in a mutant Drosophila “para” gene model. The treated flies exhibited an inhibitory effect on voltage-gated sodium ion channels, which reduced inflammation and increased tissue repair in brain cells, confirming the extract’s efficacy in treating epilepsy [69].
6. Gene Regulation Induced by Phytochemicals in Drosophila
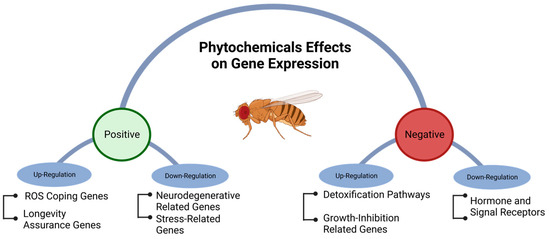
Phytochemical intake can modulate gene expression, influenced by factors, such as cell type, life stage, and growing conditions. Different mechanisms of gene regulation, including cis-regulatory elements, repressor proteins, non-coding RNA, and epigenetic processes, such as methylation, may be affected by phytochemicals [70][71][72][73]. High-throughput technologies, such as RNA-seq, are commonly used to study gene regulation and metabolic pathways on a genome-wide scale [73][74]. Phytochemicals have the potential to modulate metabolic pathways and coping mechanisms that counteract age-related neurodegenerative diseases [75]. Although phytochemicals can positively influence the health of D. melanogaster by upregulating genes associated with longer lifespans and down-regulating genes related to diseases and stress, they can also have negative effects by upregulating genes associated with reduced growth rates. Additionally, they may trigger mechanisms related to detoxification and reduce the expression of genes involved in coping with reactive oxygen species (ROS) and hormone-signaling receptors (Figure 3).

Figure 4. Positive and negative genetic regulation of metabolic pathways in Drosophila melanogaster exposed to phytochemicals.
7. Conclusions
Plant extracts have long been employed for their therapeutic and preventive properties in addressing various disorders. These extracts encompass a wide array of bioactive compounds, including polyphenols, carotenoids, flavonoids, curcuminoids, terpenoids, and capsaicinoids, contributing to their potential beneficial effects. Drosophila is an excellent model organism with extensive use in studying diverse biological processes. Leveraging the vast range of powerful genetic and molecular biology tools available, the Drosophila model offers a valuable and cheap alternative for investigating the effects of plant extracts and their derived compounds on large populations in a short period.
References
- Oladipo, A.; Enwemiwe, V.; Ejeromedoghene, O.; Adebayo, A.; Ogunyemi, O.; Fu, F. Production and functionalities of specialized metabolites from different organic sources. Metabolites 2022, 12, 534.
- Kumar, A.; Kumar, M.; Jose, A.; Tomer, V.; Oz, E.; Proestos, C.; Zeng, M.; Elobeid, T.; Oz, F. Major Phytochemicals: Recent Advances in Health Benefits and Extraction Method. Molecules 2023, 28, 887.
- Tyagi, S.; Singh, G.; Sharma, A.; Aggarwal, G. Phytochemicals as candidate therapeutics: An overview. Int. J. Pharm. Sci. Rev. Res. 2010, 3, 53–55.
- Mullins, A.P.; Arjmandi, B.H. Health benefits of plant-based nutrition: Focus on beans in cardiometabolic diseases. Nutrients 2021, 13, 519.
- Zhang, Y.-J.; Gan, R.-Y.; Li, S.; Zhou, Y.; Li, A.-N.; Xu, D.-P.; Li, H.-B. Antioxidant phytochemicals for the prevention and treatment of chronic diseases. Molecules 2015, 20, 21138–21156.
- Alam, S.; Sarker, M.M.R.; Sultana, T.N.; Chowdhury, M.N.R.; Rashid, M.A.; Chaity, N.I.; Zhao, C.; Xiao, J.; Hafez, E.E.; Khan, S.A. Antidiabetic phytochemicals from medicinal plants: Prospective candidates for new drug discovery and development. Front. Endocrinol. 2022, 13, 800714.
- Pham, D.-C.; Shibu, M.; Mahalakshmi, B.; Velmurugan, B.K. Effects of phytochemicals on cellular signaling: Reviewing their recent usage approaches. Crit. Rev. Food Sci. Nutr. 2020, 60, 3522–3546.
- Kim, S.-I.; Jung, J.-W.; Ahn, Y.-J.; Restifo, L.L.; Kwon, H.-W. Drosophila as a model system for studying lifespan and neuroprotective activities of plant-derived compounds. J. Asia-Pac. Entomol. 2011, 14, 509–517.
- Sharma, R.; Kumar, S.; Kumar, V.; Thakur, A. Comprehensive review on nutraceutical significance of phytochemicals as functional food ingredients for human health management. J. Pharmacogn. Phytochem. 2019, 8, 385–395.
- Sharma, A.; D’Souza, C.; Rai, V.; Gupta, S.C. Evaluation of Safety and Efficacy of Nutraceuticals Using Drosophila as an in vivo Tool. In Nutraceuticals in Veterinary Medicine; Springer: Berlin/Heidelberg, Germany, 2019; pp. 685–692.
- Moraes, K.C.; Montagne, J. Drosophila melanogaster: A powerful tiny animal model for the study of metabolic hepatic diseases. Front. Physiol. 2021, 12, 728407.
- Pratomo, A.R.; Salim, E.; Hori, A.; Kuraishi, T. Drosophila as an Animal Model for Testing Plant-Based Immunomodulators. Int. J. Mol. Sci. 2022, 23, 14801.
- Samtiya, M.; Aluko, R.E.; Dhewa, T.; Moreno-Rojas, J.M. Potential health benefits of plant food-derived bioactive components: An overview. Foods 2021, 10, 839.
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative stress: An essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol. Rev. 2014, 94, 329–354.
- Rao, B.N. Bioactive phytochemicals in Indian foods and their potential in health promotion and disease prevention. Asia Pac. J. Clin. Nutr. 2003, 12, 9–22.
- AlAli, M.; Alqubaisy, M.; Aljaafari, M.N.; AlAli, A.O.; Baqais, L.; Molouki, A.; Abushelaibi, A.; Lai, K.-S.; Lim, S.-H.E. Nutraceuticals: Transformation of conventional foods into health promoters/disease preventers and safety considerations. Molecules 2021, 26, 2540.
- Saxena, M.; Saxena, J.; Nema, R.; Singh, D.; Gupta, A. Phytochemistry of medicinal plants. J. Pharmacogn. Phytochem. 2013, 1, 168–182.
- Fraga, C.G.; Croft, K.D.; Kennedy, D.O.; Tomás-Barberán, F.A. The effects of polyphenols and other bioactives on human health. Food Funct. 2019, 10, 514–528.
- Rana, A.; Samtiya, M.; Dhewa, T.; Mishra, V.; Aluko, R.E. Health benefits of polyphenols: A concise review. J. Food Biochem. 2022, 46, e14264.
- Amiot, M.; Riva, C.; Vinet, A. Effects of dietary polyphenols on metabolic syndrome features in humans: A systematic review. Obes. Rev. 2016, 17, 573–586.
- Gombart, A.F.; Pierre, A.; Maggini, S. A review of micronutrients and the immune system–working in harmony to reduce the risk of infection. Nutrients 2020, 12, 236.
- Basith, S.; Cui, M.; Hong, S.; Choi, S. Harnessing the therapeutic potential of capsaicin and its analogues in pain and other diseases. Molecules 2016, 21, 966.
- Batiha, G.E.-S.; Alqahtani, A.; Ojo, O.A.; Shaheen, H.M.; Wasef, L.; Elzeiny, M.; Ismail, M.; Shalaby, M.; Murata, T.; Zaragoza-Bastida, A. Biological properties, bioactive constituents, and pharmacokinetics of some Capsicum spp. and capsaicinoids. Int. J. Mol. Sci. 2020, 21, 5179.
- Lu, M.; Chen, C.; Lan, Y.; Xiao, J.; Li, R.; Huang, J.; Huang, Q.; Cao, Y.; Ho, C.-T. Capsaicin—The major bioactive ingredient of chili peppers: Bio-efficacy and delivery systems. Food Funct. 2020, 11, 2848–2860.
- Chanda, S.; Ramachandra, T. Phytochemical and pharmacological importance of turmeric (Curcuma longa): A review. Res. Rev. A J. Pharmacol. 2019, 9, 16–23.
- Micek, A.; Godos, J.; Del Rio, D.; Galvano, F.; Grosso, G. Dietary flavonoids and cardiovascular disease: A comprehensive dose–response meta-analysis. Mol. Nutr. Food Res. 2021, 65, 2001019.
- Tolwinski, N.S. Introduction: Drosophila—A model system for developmental biology. J. Dev. Biol. 2017, 5, 9.
- Pandey, U.B.; Nichols, C.D. Human disease models in Drosophila melanogaster and the role of the fly in therapeutic drug discovery. Pharmacol. Rev. 2011, 63, 411–436.
- Jeibmann, A.; Paulus, W. Drosophila melanogaster as a model organism of brain diseases. Int. J. Mol. Sci. 2009, 10, 407–440.
- Torres-Fuentes, C.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. A natural solution for obesity: Bioactives for the prevention and treatment of weight gain. A review. Nutr. Neurosci. 2015, 18, 49–65.
- Garofalo, R.S. Genetic analysis of insulin signaling in Drosophila. Trends Endocrinol. Metab. 2002, 13, 156–162.
- Owusu-Ansah, E.; Perrimon, N. Modeling metabolic homeostasis and nutrient sensing in Drosophila: Implications for aging and metabolic diseases. Dis. Models Mech. 2014, 7, 343–350.
- Chatterjee, N.; Perrimon, N. What fuels the fly: Energy metabolism in Drosophila and its application to the study of obesity and diabetes. Sci. Adv. 2021, 7, eabg4336.
- Catalkaya, G.; Venema, K.; Lucini, L.; Rocchetti, G.; Delmas, D.; Daglia, M.; De Filippis, A.; Xiao, H.; Quiles, J.L.; Xiao, J. Interaction of dietary polyphenols and gut microbiota: Microbial metabolism of polyphenols, influence on the gut microbiota, and implications on host health. Food Front. 2020, 1, 109–133.
- Baenas, N.; Wagner, A.E. Drosophila melanogaster as an alternative model organism in nutrigenomics. Genes Nutr. 2019, 14, 14.
- Lee, S.-H.; Min, K.-J. Drosophila melanogaster as a model system in the study of pharmacological interventions in aging. Transl. Med. Aging 2019, 3, 98–103.
- Campisi, J.; Kapahi, P.; Lithgow, G.J.; Melov, S.; Newman, J.C.; Verdin, E. From discoveries in ageing research to therapeutics for healthy ageing. Nature 2019, 571, 183–192.
- Nagpal, I.; Abraham, S.K. Ameliorative effects of gallic acid, quercetin and limonene on urethane-induced genotoxicity and oxidative stress in Drosophila melanogaster. Toxicol. Mech. Methods 2017, 27, 286–292.
- Tello, J.A.; Williams, H.E.; Eppler, R.M.; Steinhilb, M.L.; Khanna, M. Animal models of neurodegenerative disease: Recent advances in fly highlight innovative approaches to drug discovery. Front. Mol. Neurosci. 2022, 15, 883358.
- Guo, J.; Huang, X.; Dou, L.; Yan, M.; Shen, T.; Tang, W.; Li, J. Aging and aging-related diseases: From molecular mechanisms to interventions and treatments. Signal Transduct. Target. Ther. 2022, 7, 391.
- Forni, C.; Facchiano, F.; Bartoli, M.; Pieretti, S.; Facchiano, A.; D’Arcangelo, D.; Norelli, S.; Valle, G.; Nisini, R.; Beninati, S. Beneficial role of phytochemicals on oxidative stress and age-related diseases. BioMed Res. Int. 2019, 2019, 8748253.
- Abd Rani, N.Z.; Husain, K.; Kumolosasi, E. Moringa genus: A review of phytochemistry and pharmacology. Front. Pharmacol. 2018, 9, 108.
- Singh, A.; Duggal, S.; Singh, H.; Singh, J.; Katekhaye, S. Withanolides: Phytoconstituents with significant pharmacological activities. Int. J. Green Pharm. 2010, 4, 229.
- Cabey, K.; Long, D.M.; Law, A.; Gray, N.E.; McClure, C.; Caruso, M.; Lak, P.; Wright, K.M.; Stevens, J.F.; Maier, C.S. Withania somnifera and Centella asiatica Extracts Ameliorate Behavioral Deficits in an In vivo Drosophila melanogaster Model of Oxidative Stress. Antioxidants 2022, 11, 121.
- Jebb, S. Obesity: Causes and consequences. Women’s Health Med. 2004, 1, 38–41.
- Chen, J.-C.; Wang, R.; Wei, C.-C. Anti-aging effects of dietary phytochemicals: From Caenorhabditis elegans, Drosophila melanogaster, rodents to clinical studies. Crit. Rev. Food Sci. Nutr. 2022, 1–26, Online ahead of print.
- Buescher, J.L.; Musselman, L.P.; Wilson, C.A.; Lang, T.; Keleher, M.; Baranski, T.J.; Duncan, J.G. Evidence for transgenerational metabolic programming in Drosophila. Dis. Models Mech. 2013, 6, 1123–1132.
- Colombani, J.; Andersen, D.S. The Drosophila gut: A gatekeeper and coordinator of organism fitness and physiology. Wiley Interdiscip. Rev. Dev. Biol. 2020, 9, e378.
- Baker, K.D.; Thummel, C.S. Diabetic larvae and obese flies—Emerging studies of metabolism in Drosophila. Cell Metab. 2007, 6, 257–266.
- Schlegel, A.; Stainier, D.Y.R. Lessons from “lower” organisms: What worms, flies, and zebrafish can teach us about human energy metabolism. PLoS Genet. 2007, 3, e199.
- Singla, P.; Bardoloi, A.; Parkash, A.A. Metabolic effects of obesity: A review. World J. Diabetes 2010, 1, 76.
- Heinrichsen, E.T.; Zhang, H.; Robinson, J.E.; Ngo, J.; Diop, S.; Bodmer, R.; Joiner, W.J.; Metallo, C.M.; Haddad, G.G. Metabolic and transcriptional response to a high-fat diet in Drosophila melanogaster. Mol. Metab. 2014, 3, 42–54.
- Chung, J.H.; Manganiello, V.; Dyck, J.R. Resveratrol as a calorie restriction mimetic: Therapeutic implications. Trends Cell Biol. 2012, 22, 546–554.
- Bayliak, M.M.; Shmihel, H.V.; Lylyk, M.P.; Vytvytska, O.M.; Storey, J.M.; Storey, K.B.; Lushchak, V.I. Alpha-ketoglutarate attenuates toxic effects of sodium nitroprusside and hydrogen peroxide in Drosophila melanogaster. Environ. Toxicol. Pharmacol. 2015, 40, 650–659.
- De Filippis, F.; Paparo, L.; Nocerino, R.; Della Gatta, G.; Carucci, L.; Russo, R.; Pasolli, E.; Ercolini, D.; Berni Canani, R. Specific gut microbiome signatures and the associated pro-inflamatory functions are linked to pediatric allergy and acquisition of immune tolerance. Nat. Commun. 2021, 12, 5958.
- Kumar, V.; Singh, D.D.; Lakhawat, S.S.; Yasmeen, N.; Pandey, A.; Singla, R.K. Biogenic phytochemicals modulating obesity: From molecular mechanism to preventive and therapeutic approaches. Evid.-Based Complement. Altern. Med. 2022, 2022, 1–20.
- Vamanu, E.; Rai, S.N. The link between obesity, microbiota dysbiosis, and neurodegenerative pathogenesis. Diseases 2021, 9, 45.
- Douglas, A.E. The Drosophila model for microbiome research. Lab Anim. 2018, 47, 157–164.
- Jehrke, L.; Stewart, F.A.; Droste, A.; Beller, M. The impact of genome variation and diet on the metabolic phenotype and microbiome composition of Drosophila melanogaster. Sci. Rep. 2018, 8, 6215.
- Wong, A.C.-N.; Dobson, A.J.; Douglas, A.E. Gut microbiota dictates the metabolic response of Drosophila to diet. J. Exp. Biol. 2014, 217, 1894–1901.
- Xu, Z.; Knight, R. Dietary effects on human gut microbiome diversity. Br. J. Nutr. 2015, 113, S1–S5.
- Jiménez-Padilla, Y.; Esan, E.O.; Floate, K.D.; Sinclair, B.J. Persistence of diet effects on the microbiota of Drosophila suzukii (Diptera: Drosophilidae). Can. Entomol. 2020, 152, 516–531.
- Newell, P.D.; Douglas, A.E. Interspecies interactions determine the impact of the gut microbiota on nutrient allocation in Drosophila melanogaster. Appl. Environ. Microbiol. 2014, 80, 788–796.
- Binda, C.; Lopetuso, L.R.; Rizzatti, G.; Gibiino, G.; Cennamo, V.; Gasbarrini, A. Actinobacteria: A relevant minority for the maintenance of gut homeostasis. Dig. Liver Dis. 2018, 50, 421–428.
- Jayarathne, S.; Stull, A.J.; Park, O.H.; Kim, J.H.; Thompson, L.; Moustaid-Moussa, N. Protective effects of anthocyanins in obesity-associated inflammation and changes in gut microbiome. Mol. Nutr. Food Res. 2019, 63, 1900149.
- Islam, M.S.; Akhtar, M.M.; Ciavattini, A.; Giannubilo, S.R.; Protic, O.; Janjusevic, M.; Procopio, A.D.; Segars, J.H.; Castellucci, M.; Ciarmela, P. Use of dietary phytochemicals to target inflammation, fibrosis, proliferation, and angiogenesis in uterine tissues: Promising options for prevention and treatment of uterine fibroids? Mol. Nutr. Food Res. 2014, 58, 1667–1684.
- Fonseca-Hernández, D.; Lugo-Cervantes, E.D.C.; Escobedo-Reyes, A.; Mojica, L. Black bean (Phaseolus vulgaris L.) polyphenolic extract exerts antioxidant and antiaging potential. Molecules 2021, 26, 6716.
- Shin, W.S.; Di, J.; Cao, Q.; Li, B.; Seidler, P.M.; Murray, K.A.; Bitan, G.; Jiang, L. Amyloid β-protein oligomers promote the uptake of tau fibril seeds potentiating intracellular tau aggregation. Alzheimer’s Res. Ther. 2019, 11, 86.
- Ssempijja, F.; Dare, S.S.; Bukenya, E.E.; Kasozi, K.I.; Kenganzi, R.; Fernandez, E.M.; Vicente-Crespo, M. Attenuation of Seizures, Cognitive Deficits, and Brain Histopathology by Phytochemicals of Imperata cylindrica (L.) P. Beauv (Poaceae) in Acute and Chronic Mutant Drosophila melanogaster Epilepsy Models. J. Evid. -Based Integr. Med. 2023, 28, 2515690X231160191.
- Saliu, J.A.; Olajuyin, A.M.; Akinnubi, A. Modulatory effect of Artocarpus camansi on ILP-2, InR, and Imp-L2 genes of sucrose–induced diabetes mellitus in Drosophila melanogaster. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2021, 246, 109041.
- Ing-Simmons, E.; Vaid, R.; Bing, X.Y.; Levine, M.; Mannervik, M.; Vaquerizas, J.M. Independence of chromatin conformation and gene regulation during Drosophila dorsoventral patterning. Nat. Genet. 2021, 53, 487–499.
- Rivera, J.; Keränen, S.V.E.; Gallo, S.M.; Halfon, M.S. REDfly: The transcriptional regulatory element database for Drosophila. Nucleic Acids Res. 2019, 47, D828–D834.
- Fear, J.M.; León-Novelo, L.G.; Morse, A.M.; Gerken, A.R.; Van Lehmann, K.; Tower, J.; Nuzhdin, S.V.; McIntyre, L.M. Buffering of genetic regulatory networks in Drosophila melanogaster. Genetics 2016, 203, 1177–1190.
- Zhang, Z.-G.; Niu, X.-Y.; Lu, A.-P.; Xiao, G.G. Effect of curcumin on aged Drosophila Melanogaster: A pathway prediction analysis. Chin. J. Integr. Med. 2015, 21, 115–122.
- Park, J.H.; Jung, J.W.; Ahn, Y.-J.; Kwon, H.W. Neuroprotective properties of phytochemicals against paraquat-induced oxidative stress and neurotoxicity in Drosophila melanogaster. Pestic. Biochem. Physiol. 2012, 104, 118–125.
More
Information
Subjects:
Food Science & Technology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
950
Revisions:
2 times
(View History)
Update Date:
28 Sep 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





