Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Chung-Sung Lee | -- | 1675 | 2023-08-07 04:03:31 | | | |
| 2 | Wendy Huang | Meta information modification | 1675 | 2023-08-07 07:27:36 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Hwang, H.S.; Lee, C. Design and Formulation of Hyaluronic-Acid-Based Hydrogels. Encyclopedia. Available online: https://encyclopedia.pub/entry/47710 (accessed on 28 March 2026).
Hwang HS, Lee C. Design and Formulation of Hyaluronic-Acid-Based Hydrogels. Encyclopedia. Available at: https://encyclopedia.pub/entry/47710. Accessed March 28, 2026.
Hwang, Hee Sook, Chung-Sung Lee. "Design and Formulation of Hyaluronic-Acid-Based Hydrogels" Encyclopedia, https://encyclopedia.pub/entry/47710 (accessed March 28, 2026).
Hwang, H.S., & Lee, C. (2023, August 07). Design and Formulation of Hyaluronic-Acid-Based Hydrogels. In Encyclopedia. https://encyclopedia.pub/entry/47710
Hwang, Hee Sook and Chung-Sung Lee. "Design and Formulation of Hyaluronic-Acid-Based Hydrogels." Encyclopedia. Web. 07 August, 2023.
Copy Citation
Hyaluronic acid (HA) is a natural polymer that is widely distributed in the human body and plays a significant role in numerous physiological processes such as cell migration, tissue hydration, and wound healing. Hydrogels based on HA and its derivatives have gained popularity as potential treatments for bone-related diseases. HA-based hydrogels have been extensively studied for their ability to mimic the natural extracellular matrix of bone tissue and provide a suitable microenvironment for cell support and tissue regeneration. The physical and chemical properties of HA can be modified to improve its mechanical strength, biocompatibility, and osteogenic potential.
hyaluronic acid
hydrogel
scaffold
bone tissue engineering
bone regeneration
1. Introduction
Bone tissue engineering is an emerging field that aims to regenerate damaged or diseased bone tissue using biomaterials, cells, and growth factors to restore normal skeletal function [1][2]. The field has grown rapidly in recent years, driven by the increasing prevalence of bone disorders and injuries such as osteoporosis, bone fractures, and bone defects caused by trauma or disease [3]. The current treatments for these conditions, such as bone grafting and implantation of metal or ceramic implants, have limitations and drawbacks, including limited availability of donor tissue, risk of infection, and poor integration with surrounding tissues.
Hydrogels have emerged as promising biomaterials for bone tissue engineering applications due to their ability to mimic the extracellular matrix (ECM) of bone tissue and their tunable physical and chemical properties [4]. Hydrogels are three-dimensional networks of hydrophilic polymer chains that can absorb large amounts of water while maintaining their structural integrity [5]. Hydrogels can be designed and formulated to provide a suitable microenvironment for bone cells and promote bone tissue regeneration.
Hyaluronic acid (HA) is a naturally occurring polysaccharide that has emerged as a promising biomaterial for bone regeneration due to its biocompatibility, biodegradability, and ability to interact with the cells and growth factors involved in bone formation [6][7]. HA is a linear molecule consisting of repeating disaccharide units of glucuronic acid and N-acetylglucosamine (Figure 1) [8]. The molecular weight of HA can vary widely, from a few hundred to millions of Daltons. One of the key features of HA that make it an attractive biomaterial for bone regeneration is its ability to interact with the cells and growth factors involved in bone formation. HA can bind to cell surface receptors, such as CD44 and the receptor for hyaluronan-mediated motility (RHAMM), that are expressed on osteoblasts and osteoclasts, as well as on mesenchymal stem cells (MSCs), and that can differentiate into bone-forming cells [9]. HA can also interact with growth factors, such as bone morphogenetic proteins (BMPs) and transforming growth factor-beta (TGF-β), which play important roles in bone formation and regeneration [10].
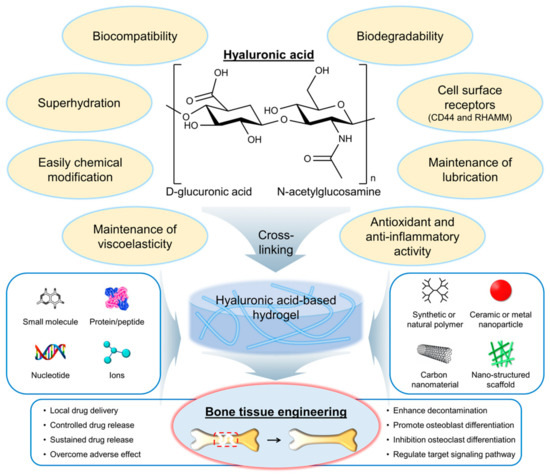
Figure 1. Chemical structure and properties of hyaluronic acid (HA), as well as its application in the development of hydrogels for bone tissue engineering. Red dashed box indicates the bone defect and injury area. RHAMM, a receptor for hyaluronan-mediated motility.
HA has several properties and characteristics that make it a promising biomaterial for bone tissue engineering applications (Figure 1) [11][12]. First, HA is biocompatible, meaning it does not have adverse effects on living tissues and cells. The biocompatibility of HA is due to its natural occurrence in the body and its nonimmunogenic nature [13]. This means that it is unlikely to cause an immune response in the body, reducing the risk of rejection. Second, HA is biodegradable, meaning it can be broken down and metabolized by the body over time. The biodegradability of HA is due to the presence of specific enzymes in the body, such as hyaluronidases, that can cleave the glycosidic bonds between the disaccharide units of HA [14]. The biodegradation of HA can be controlled by modifying the molecular weight and degree of cross-linking of the polymer chains. Third, HA can interact with the cells and growth factors involved in bone formation. As mentioned earlier, HA can bind to cell surface receptors, such as CD44 and RHAMM, which are expressed on osteoblasts and osteoclasts, as well as on MSCs, that can differentiate into bone-forming cells [9]. The interaction of HA with cells and growth factors can be modulated by modifying the physical and chemical properties of the HA. Fourth, HA is a highly hydrated molecule, allowing it to absorb large amounts of water and create a hydrophilic environment that is favorable for cell attachment and proliferation [15]. This makes HA-based hydrogels particularly well suited for tissue engineering applications because they can provide a suitable microenvironment for cell growth and differentiation. Finally, HA can be easily modified to introduce functional groups that can be used for cross-linking and the incorporation of bioactive molecules [16]. This allows for the creation of HA-based hydrogels with tailored physical and chemical properties such as mechanical strength, degradation rate, and bioactivity. Furthermore, HA is also known as a lubricant or support for other lubricants, such as lipids, in synovial joints [17]. Antioxidant activity and anti-inflammatory activity of HA have also been reported [18][19][20][21][22].
HA-based hydrogels have several advantages over other biomaterials for bone tissue engineering applications. The hydrophilic nature of HA-based hydrogels promotes the adsorption of BMPs and other growth factors that are involved in bone formation and regeneration [23]. The presence of these growth factors can enhance the osteogenic differentiation of MSCs and promote the formation of new bone tissue. In addition, the mechanical properties of HA-based hydrogels can be easily tuned by adjusting the degree of cross-linking and the molecular weight of the HA polymer chains [24]. This allows for the creation of hydrogels with a wide range of mechanical properties that are suitable for different applications. Hydrogels with higher mechanical strength may be used for load-bearing applications, whereas hydrogels with lower mechanical strength may be used for applications where flexibility and conformability are important. Furthermore, HA-based hydrogels can be easily modified to incorporate bioactive molecules, such as growth factors and ECM components, that can further enhance the osteogenic differentiation of MSCs and promote the formation of new bone tissue [25]. The incorporation of these bioactive molecules can also improve the integration of the hydrogel with surrounding tissues. Finally, HA-based hydrogels are biocompatible and biodegradable, meaning they are unlikely to cause adverse effects in the body and can be broken down and metabolized over time [26]. This reduces the risk of inflammation and other complications associated with the use of synthetic biomaterials.
2. The Selection of HA-Based Hydrogel Formulations for Bone Regeneration Applications
The selection and optimization of HA-based hydrogel formulations for bone regeneration applications are critical for achieving the desired mechanical and biological properties. The choice of cross-linking agent, cross-linking density, and HA concentration can have a significant impact on the properties of the resulting hydrogel.
Several cross-linking agents and strategies have been used to cross-link HA-based hydrogels, including physical and chemical cross-linking agents [26][27][28]. Physical cross-linking includes methods such as thermal, pH-sensitive, and photomediated cross-linking, which rely on the reversible formation of physical cross-links between the HA chains. Chemical cross-linking agents such as glutaraldehyde, genipin, and carbodiimide form covalent bonds between the HA chains. The choice of cross-linking agent can have a significant impact on the properties of the resulting hydrogel. Physical cross-linking agents generally tend to result in hydrogels with lower mechanical strength but higher swelling capacity, whereas chemical cross-linking agents tend to result in hydrogels with higher mechanical strength but lower swelling capacity. Figure 2 shows the chemical structure of HA modified with selected chemical cross-linking points for gelation. Chemical modifications are mainly performed by targeting the carboxylic acid of the glucuronic acid residue or the C-6 hydroxyl group of the N-acetylglucosamine sugar of the HA backbone. In particular, approaches of chemical modification through addition, disulfide, enzyme, click reaction, and hydrazide are mainly used to form HA-based hydrogels without additional initiators [29].
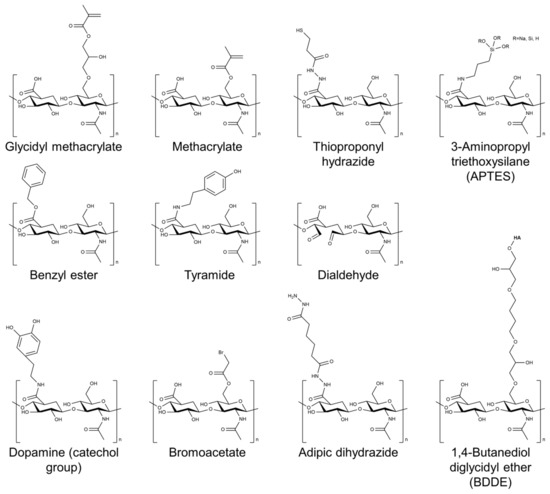
Figure 2. Chemical structure of HA modified with selected chemical cross-linking points for gelation of HA.
In addition to the cross-linking agent, the concentration of HA in the hydrogel formulation can also affect the properties of the resulting hydrogel [30]. Higher concentrations of HA typically result in hydrogels with higher mechanical strength but lower swelling capacity. In addition, modifications to the cross-linking agent or the HA molecule itself can also be used to improve the properties of the hydrogel. The introduction of methacrylate or aldehyde groups onto the HA molecule has also been shown to enhance the mechanical properties of the resulting hydrogel [31][32][33].
To enhance the mechanical and biological properties of HA-based hydrogels for bone regeneration applications, several strategies have been investigated. One approach to improve the mechanical properties of HA-based hydrogels is to incorporate reinforcing agents into the hydrogel matrix. Reinforcing agents can provide additional strength and stiffness to the hydrogel, allowing it to withstand mechanical loading better. Examples of reinforcing agents that have been used in combination with HA-based hydrogels include various types of nanoparticles, such as carbon nanotubes, graphene oxide, and hydroxyapatite nanoparticles, as well as micro- and nanofibers made of biodegradable polymers such as poly(lactic-co-glycolic acid) (PLGA) and polycaprolactone (PCL) [34][35][36][37]. The incorporation of these reinforcing agents can significantly improve the mechanical properties of HA-based hydrogels, as demonstrated by studies showing increased compressive and tensile strength, modulus, and toughness [35]. In particular, the addition of bioceramics, such as hydroxyapatite or tricalcium phosphate, can improve the mechanical properties of the hydrogel and enhance its osteoconductive properties [38][39].
In addition to enhancing the mechanical properties of HA-based hydrogels, the incorporation of growth factors and other bioactive molecules can also improve their biological properties, such as their bioactivity and osteoinductivity. Growth factors such as BMPs, TGF-β, and platelet-derived growth factor (PDGF) have been shown to enhance bone regeneration when incorporated into HA-based hydrogels [39]. Similarly, other bioactive molecules such as ECM proteins, such as collagen and fibronectin, as well as small-molecule drugs such as dexamethasone and simvastatin, have been shown to improve the biological properties of HA-based hydrogels [16][40]. The incorporation of bone morphogenetic protein-2 (BMP-2) and stromal cell-derived factor-1α (SDF-1α) into an HA-based hydrogel was shown to promote bone formation and accelerate bone regeneration in vivo [41].
HA-based hydrogels can be formed into various shapes, such as particles, films, and porous scaffolds, using different fabrication techniques [38]. Advanced fabrication techniques, such as 3D printing, have been explored to create complex and functional HA-based scaffolds for bone regeneration [29][42].
References
- Marew, T.; Birhanu, G. Three dimensional printed nanostructure biomaterials for bone tissue engineering. Regen. Ther. 2021, 18, 102–111.
- Kang, M.; Lee, C.-S.; Lee, M. Bioactive scaffolds integrated with liposomal or extracellular vesicles for bone regeneration. Bioengineering 2021, 8, 137.
- Dimitriou, R.; Jones, E.; McGonagle, D.; Giannoudis, P.V. Bone regeneration: Current concepts and future directions. BMC Med. 2011, 9, 66.
- Gough, J.E.; Saiani, A.; Miller, A.F. Peptide hydrogels: Mimicking the extracellular matrix. Bioinspired Biomim. Nanobiomater. 2012, 1, 4–12.
- Rao, T.; Chvs, P. Hydrogels the three dimensional networks: A review. Int. J. Curr. Pharm. Res. 2021, 13, 12–17.
- Dahiya, P.; Kamal, R. Hyaluronic acid: A boon in periodontal therapy. N. Am. J. Med. Sci. 2013, 5, 309.
- Lee, C.-S.; Na, K. Photochemically triggered cytosolic drug delivery using pH-responsive hyaluronic acid nanoparticles for light-induced cancer therapy. Biomacromolecules 2014, 15, 4228–4238.
- Cantor, J.O.; Nadkarni, P.P. Hyaluronan: The Jekyll and Hyde molecule. Inflamm. Allergy Drug Targets 2006, 5, 257–260.
- Leng, Y.; Abdullah, A.; Wendt, M.K.; Calve, S. Hyaluronic acid, CD44 and RHAMM regulate myoblast behavior during embryogenesis. Matrix Biol. 2019, 78, 236–254.
- Kawano, M.; Ariyoshi, W.; Iwanaga, K.; Okinaga, T.; Habu, M.; Yoshioka, I.; Tominaga, K.; Nishihara, T. Mechanism involved in enhancement of osteoblast differentiation by hyaluronic acid. Biochem. Biophys. Res. Commun. 2011, 405, 575–580.
- Gupta, R.C.; Lall, R.; Srivastava, A.; Sinha, A. Hyaluronic acid: Molecular mechanisms and therapeutic trajectory. Front. Vet. Sci. 2019, 6, 192.
- Li, H.; Qi, Z.; Zheng, S.; Chang, Y.; Kong, W.; Fu, C.; Yu, Z.; Yang, X.; Pan, S. The application of hyaluronic acid-based hydrogels in bone and cartilage tissue engineering. Adv. Mater. Sci. Eng. 2019, 2019, 3027303.
- Lee, S.Y.; Kang, M.S.; Jeong, W.Y.; Han, D.-W.; Kim, K.S. Hyaluronic acid-based theranostic nanomedicines for targeted cancer therapy. Cancers 2020, 12, 940.
- Snetkov, P.; Zakharova, K.; Morozkina, S.; Olekhnovich, R.; Uspenskaya, M. Hyaluronic acid: The influence of molecular weight on structural, physical, physico-chemical, and degradable properties of biopolymer. Polymers 2020, 12, 1800.
- He, Y.; Hou, Z.; Wang, J.; Wang, Z.; Li, X.; Liu, J.; Liang, Q.; Zhao, J. Assessment of biological properties of recombinant collagen-hyaluronic acid composite scaffolds. Int. J. Biol. Macromol. 2020, 149, 1275–1284.
- Zhai, P.; Peng, X.; Li, B.; Liu, Y.; Sun, H.; Li, X. The application of hyaluronic acid in bone regeneration. Int. J. Biol. Macromol. 2020, 151, 1224–1239.
- Lin, W.; Liu, Z.; Kampf, N.; Klein, J. The role of hyaluronic acid in cartilage boundary lubrication. Cells 2020, 9, 1606.
- Ke, C.; Sun, L.; Qiao, D.; Wang, D.; Zeng, X. Antioxidant acitivity of low molecular weight hyaluronic acid. Food Chem. Toxicol. 2011, 49, 2670–2675.
- De Paiva, W.K.V.; de Medeiros, W.R.D.B.; de Assis, C.F.; dos Santos, E.S.; de Sousa Júnior, F.C. Physicochemical characterization and in vitro antioxidant activity of hyaluronic acid produced by Streptococcus zooepidemicus CCT 7546. Prep. Biochem. Biotechnol. 2022, 52, 234–243.
- Mohammed, A.A.; Niamah, A.K. Identification and antioxidant activity of hyaluronic acid extracted from local isolates of Streptococcus thermophilus. Mater. Today Proc. 2022, 60, 1523–1529.
- Han, W.; Lv, Y.; Sun, Y.; Wang, Y.; Zhao, Z.; Shi, C.; Chen, X.; Wang, L.; Zhang, M.; Wei, B. The anti-inflammatory activity of specific-sized hyaluronic acid oligosaccharides. Carbohydr. Polym. 2022, 276, 118699.
- Aznabaev, M.; Imaeva, A.; Bashkatov, S.; Gabdrakhmanova, A. Anti-inflammatory activity of hyaluronic acid. Eksperimental’naia Klin. Farmakol. 2003, 66, 28–29.
- Kim, J.; Kim, I.S.; Cho, T.H.; Kim, H.C.; Yoon, S.J.; Choi, J.; Park, Y.; Sun, K.; Hwang, S.J. In vivo evaluation of MMP sensitive high-molecular weight HA-based hydrogels for bone tissue engineering. J. Biomed. Mater. Res. Part A 2010, 95, 673–681.
- Xue, Y.; Chen, H.; Xu, C.; Yu, D.; Xu, H.; Hu, Y. Synthesis of hyaluronic acid hydrogels by crosslinking the mixture of high-molecular-weight hyaluronic acid and low-molecular-weight hyaluronic acid with 1, 4-butanediol diglycidyl ether. RSC Adv. 2020, 10, 7206–7213.
- Frith, J.E.; Menzies, D.J.; Cameron, A.R.; Ghosh, P.; Whitehead, D.L.; Gronthos, S.; Zannettino, A.C.; Cooper-White, J.J. Effects of bound versus soluble pentosan polysulphate in PEG/HA-based hydrogels tailored for intervertebral disc regeneration. Biomaterials 2014, 35, 1150–1162.
- Ding, Y.-W.; Wang, Z.-Y.; Ren, Z.-W.; Zhang, X.-W.; Wei, D.-X. Advances in modified hyaluronic acid-based hydrogels for skin wound healing. Biomater. Sci. 2022, 10, 3393–3409.
- Khunmanee, S.; Jeong, Y.; Park, H. Crosslinking method of hyaluronic-based hydrogel for biomedical applications. J. Tissue Eng. 2017, 8, 2041731417726464.
- Patrick Micheels, M.; Didier Sarazin, M.; Christian Tran, M. Effect of different crosslinking technologies on hyaluronic acid behavior: A visual and microscopic study of seven hyaluronic acid gels. J. Drugs Dermatol. 2016, 15, 600–606.
- Burdick, J.A.; Prestwich, G.D. Hyaluronic acid hydrogels for biomedical applications. Adv. Mater. 2011, 23, H41–H56.
- Ibrahim, S.; Kang, Q.K.; Ramamurthi, A. The impact of hyaluronic acid oligomer content on physical, mechanical, and biologic properties of divinyl sulfone-crosslinked hyaluronic acid hydrogels. J. Biomed. Mater. Res. Part A 2010, 94, 355–370.
- Tan, H.; Li, H.; Rubin, J.P.; Marra, K.G. Controlled gelation and degradation rates of injectable hyaluronic acid-based hydrogels through a double crosslinking strategy. J. Tissue Eng. Regen. Med. 2011, 5, 790–797.
- Zhang, M.; Chen, X.; Yang, K.; Dong, Q.; Yang, H.; Gu, S.; Xu, W.; Zhou, Y. Dual-crosslinked hyaluronic acid hydrogel with self-healing capacity and enhanced mechanical properties. Carbohydr. Polym. 2023, 301, 120372.
- Spearman, B.S.; Agrawal, N.K.; Rubiano, A.; Simmons, C.S.; Mobini, S.; Schmidt, C.E. Tunable methacrylated hyaluronic acid-based hydrogels as scaffolds for soft tissue engineering applications. J. Biomed. Mater. Res. Part A 2020, 108, 279–291.
- Vashist, A.; Kaushik, A.; Vashist, A.; Sagar, V.; Ghosal, A.; Gupta, Y.; Ahmad, S.; Nair, M. Advances in carbon nanotubes–hydrogel hybrids in nanomedicine for therapeutics. Adv. Healthc. Mater. 2018, 7, 1701213.
- Li, X.; Yang, Z.; Fang, L.; Ma, C.; Zhao, Y.; Liu, H.; Che, S.; Zvyagin, A.V.; Yang, B.; Lin, Q. Hydrogel composites with different dimensional nanoparticles for bone regeneration. Macromol. Rapid Commun. 2021, 42, 2100362.
- Amiryaghoubi, N.; Fathi, M.; Barzegari, A.; Barar, J.; Omidian, H.; Omidi, Y. Recent advances in polymeric scaffolds containing carbon nanotube and graphene oxide for cartilage and bone regeneration. Mater. Today Commun. 2021, 26, 102097.
- Patel, M.; Koh, W.-G. Composite hydrogel of methacrylated hyaluronic acid and fragmented polycaprolactone nanofiber for osteogenic differentiation of adipose-derived stem cells. Pharmaceutics 2020, 12, 902.
- Sikkema, R.; Keohan, B.; Zhitomirsky, I. Hyaluronic-Acid-Based Organic-Inorganic Composites for Biomedical Applications. Materials 2021, 14, 4982.
- Zheng, Z.; Patel, M.; Patel, R. Hyaluronic acid-based materials for bone regeneration: A review. React. Funct. Polym. 2022, 171, 105151.
- Lee, C.-S.; Singh, R.K.; Hwang, H.S.; Lee, N.-H.; Kurian, A.G.; Lee, J.-H.; Kim, H.S.; Lee, M.; Kim, H.-W. Materials-based nanotherapeutics for injured and diseased bone. Prog. Mater. Sci. 2023, 135, 101087.
- Holloway, J.L.; Ma, H.; Rai, R.; Hankenson, K.D.; Burdick, J.A. Synergistic Effects of SDF-1α and BMP-2 Delivery from Proteolytically Degradable Hyaluronic Acid Hydrogels for Bone Repair. Macromol. Biosci. 2015, 15, 1218–1223.
- Xu, X.; Jha, A.K.; Harrington, D.A.; Farach-Carson, M.C.; Jia, X. Hyaluronic acid-based hydrogels: From a natural polysaccharide to complex networks. Soft Matter 2012, 8, 3280–3294.
More
Information
Subjects:
Engineering, Biomedical
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
627
Revisions:
2 times
(View History)
Update Date:
07 Aug 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





