
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mohamed O. Radwan | -- | 1895 | 2023-08-06 10:02:32 | | | |
| 2 | Lindsay Dong | + 29 word(s) | 1924 | 2023-08-08 02:42:57 | | | | |
| 3 | Lindsay Dong | Meta information modification | 1924 | 2023-08-08 02:46:51 | | |
Video Upload Options
Gypsogenin possesses a versatile and unique aldehyde group that can be utilized to create covalent interactions with undruggable targets. Gypsogenin carboxamides have demonstrated high cytotoxic activity against breast and lung cancer. The bisamides of gypsogenic acid possess prominent activity as well; however, their anti-leukemic activity is yet to be explored.
1. Introduction
Found in higher plants, pentacyclic triterpenes (PTs) are bio-nutrient phytochemicals endowed with a diverse range of bioactivities such as hepatoprotective [1][2][3], anti-inflammatory [4][5][6], anti-hypertensive [3][7][8][9], anti-atherosclerotic [7][8], anti-viral [10][11][12], anti-fibrosis [13][14][15], and anti-ulcer effects [4][16][17]. In particular, PTs have ubiquitous applications in terms of anti-cancer drug discovery [18][19][20][21][22][23][24][25][26].
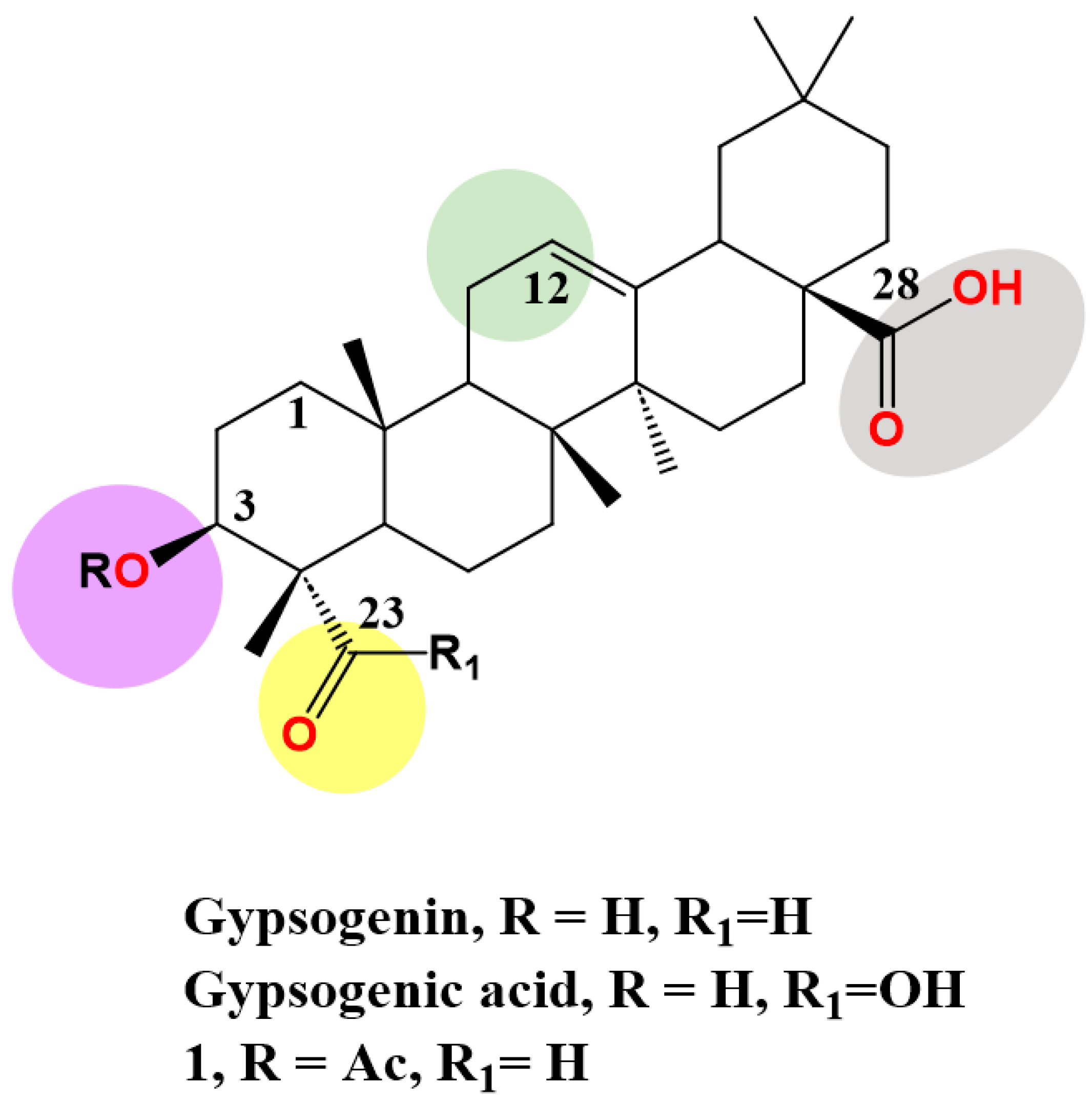
2. Anti-Cancer Effect of Gypsogenin, Gypsogenic Acid, and Their Semisynthetic Derivatives
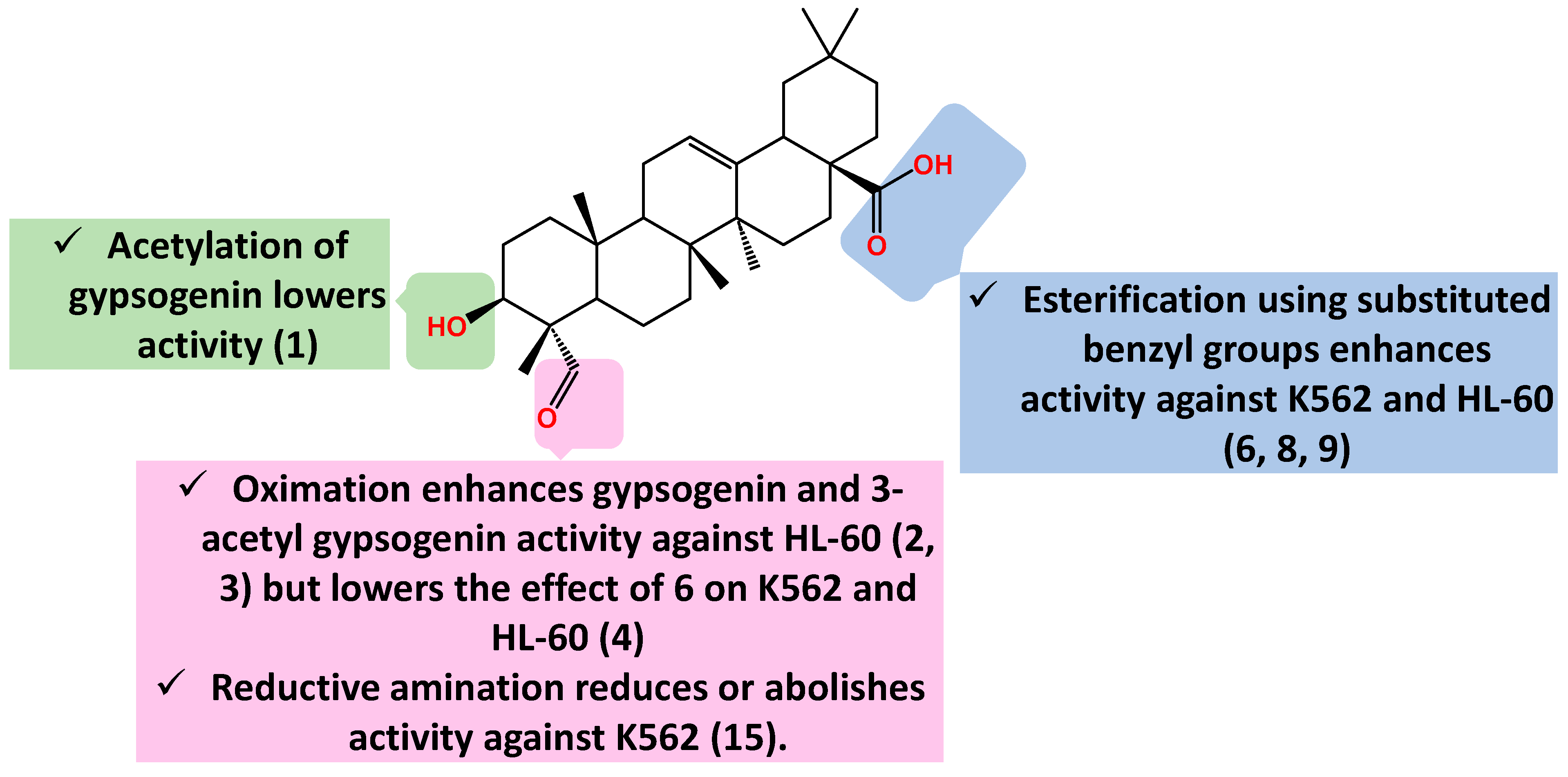
2.1. Anti-Leukemic Activity

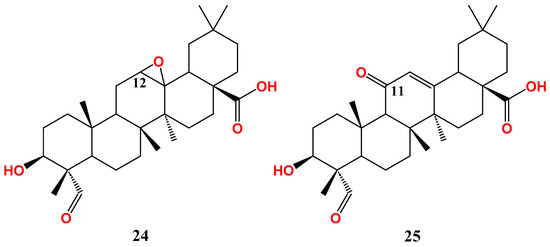
2.2. Anti-Breast Cancer Activity

2.3. Anti-Lung Cancer Activity
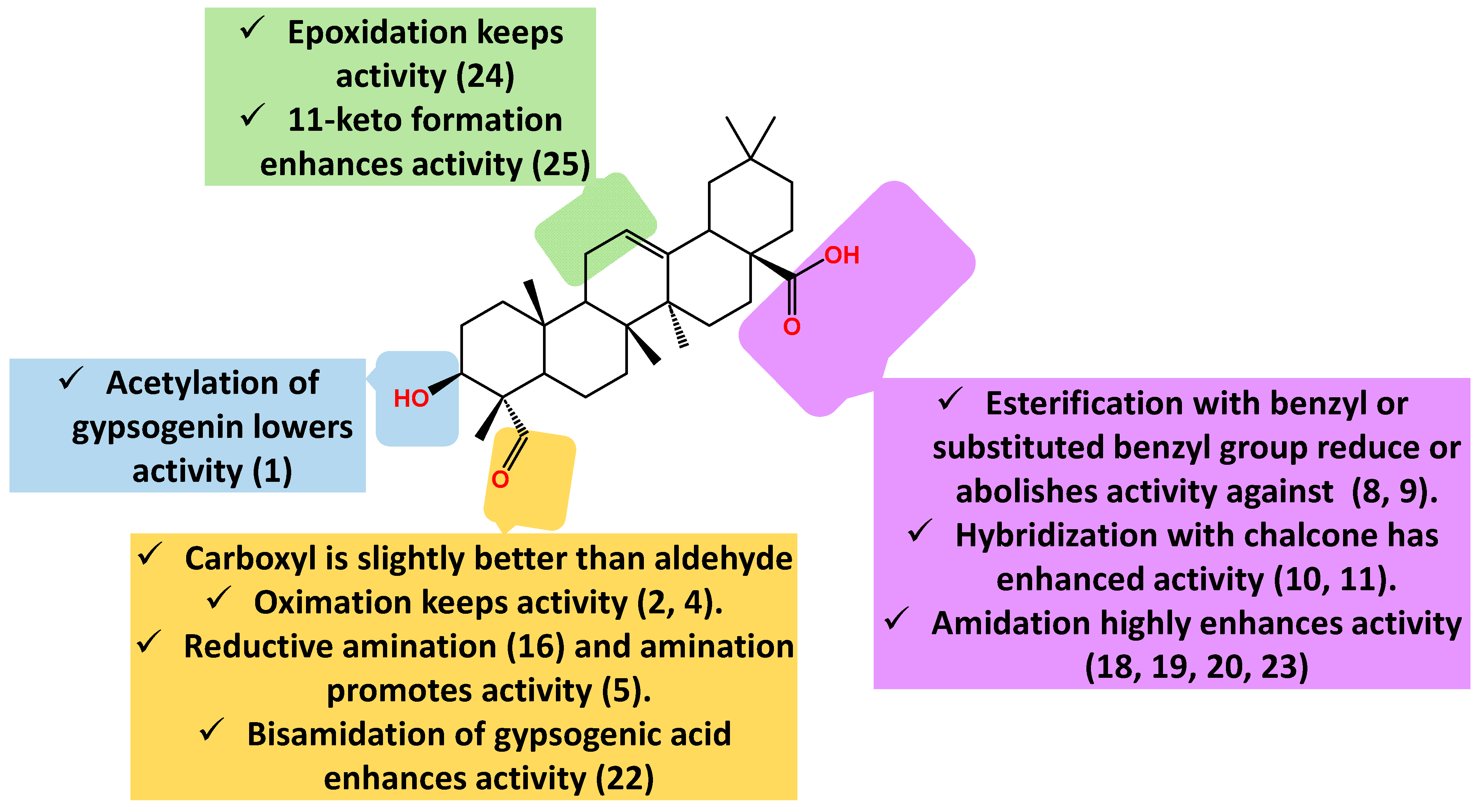
2.4. Other Anti-Cancer Activities
References
- Gutiérrez-Rebolledo, G.A.; Siordia-Reyes, A.G.; Meckes-Fischer, M.; Jiménez-Arellanes, A. Hepatoprotective Properties of Oleanolic and Ursolic Acids in Antitubercular Drug-Induced Liver Damage. Asian Pac. J. Trop. Med. 2016, 9, 644–651.
- Xu, G.B.; Xiao, Y.H.; Zhang, Q.Y.; Zhou, M.; Liao, S.G. Hepatoprotective Natural Triterpenoids. Eur. J. Med. Chem. 2018, 145, 691–716.
- Ayeleso, T.B.; Matumba, M.G.; Mukwevho, E. Oleanolic Acid and Its Derivatives: Biological Activities and Therapeutic Potential in Chronic Diseases. Molecules 2017, 22, 1915.
- Aly, A.M.; Al-Alousi, L.; Salem, H.A. Licorice: A Possible Anti-Inflammatory and Anti-Ulcer Drug. AAPS PharmSciTech 2005, 6, E74–E82.
- Tsai, S.J.; Yin, M.C. Antioxidative and Anti-Inflammatory Protection of Oleanolic Acid and Ursolic Acid in PC12 Cells. J. Food Sci. 2008, 73, H174–H178.
- Radwan, M.O.; Ismail, M.A.H.; El-Mekkawy, S.; Ismail, N.S.M.; Hanna, A.G. Synthesis and Biological Activity of New 18β-Glycyrrhetinic Acid Derivatives. Arab. J. Chem. 2016, 9, 390–399.
- Somova, L.I.; Shode, F.O.; Ramnanan, P.; Nadar, A. Antihypertensive, Antiatherosclerotic and Antioxidant Activity of Triterpenoids Isolated from Olea Europaea, Subspecies Africana Leaves. J. Ethnopharmacol. 2003, 84, 299–305.
- Somova, L.O.; Nadar, A.; Rammanan, P.; Shode, F.O. Cardiovascular, Antihyperlipidemic and Antioxidant Effects of Oleanolic and Ursolic Acids in Experimental Hypertension. Phytomedicine 2003, 10, 115–121.
- Zhang, S.; Liu, Y.; Wang, X.; Tian, Z.; Qi, D.; Li, Y.; Jiang, H. Antihypertensive Activity of Oleanolic Acid Is Mediated via Downregulation of Secretory Phospholipase A2 and Fatty Acid Synthase in Spontaneously Hypertensive Rats. Int. J. Mol. Med. 2020, 46, 2019–2034.
- Pompei, R.; Laconi, S.; Ingianni, A. Antiviral Properties of Glycyrrhizic Acid and Its Semisynthetic Derivatives. Mini-Rev. Med. Chem. 2012, 9, 996–1001.
- Sun, Z.-G.; Zhao, T.-T.; Lu, N.; Yang, Y.-A.; Zhu, H.-L. Research Progress of Glycyrrhizic Acid on Antiviral Activity. Mini-Rev. Med. Chem. 2019, 19, 826–832.
- Tohmé, M.J.; Giménez, M.C.; Peralta, A.; Colombo, M.I.; Delgui, L.R. Ursolic Acid: A Novel Antiviral Compound Inhibiting Rotavirus Infection in Vitro. Int. J. Antimicrob. Agents 2019, 54, 601–609.
- Yang, Y.; Huang, Y.; Huang, C.; Lv, X.; Liu, L.; Wang, Y.; Li, J. Antifibrosis Effects of Triterpene Acids of Eriobotrya Japonica (Thunb.) Lindl. Leaf in a Rat Model of Bleomycin-Induced Pulmonary Fibrosis. J. Pharm. Pharmacol. 2012, 64, 1751–1760.
- Lee, M.K.; Lee, K.Y.; Jeon, H.Y.; Sung, S.H.; Kim, Y.C. Antifibrotic Activity of Triterpenoids from the Aerial Parts of Euscaphis Japonica on Hepatic Stellate Cells. J. Enzyme Inhib. Med. Chem. 2009, 24, 1276–1279.
- Xiang, H.; Han, Y.; Zhang, Y.; Yan, W.; Xu, B.; Chu, F.; Xie, T.; Jia, M.; Yan, M.; Zhao, R.; et al. A New Oleanolic Acid Derivative against CCl4-Induced Hepatic Fibrosis in Rats. Int. J. Mol. Sci. 2017, 18, 553.
- Farina, C.; Pinza, M.; Pifferi, G. Synthesis and Anti-Ulcer Activity of New Derivatives of Glycyrrhetic, Oleanolic and Ursolic Acids. Il Farm. 1998, 53, 22–32.
- Somensi, L.B.; Costa, P.; Boeing, T.; Mariano, L.N.B.; Longo, B.; Magalhães, C.G.; Duarte, L.P.; Maciel e Silva, A.T.; de Souza, P.; de Andrade, S.F.; et al. Gastroprotective Properties of Lupeol-Derived Ester: Pre-Clinical Evidences of Lupeol-Stearate as a Potent Antiulcer Agent. Chem. Biol. Interact. 2020, 321, 108964.
- Chudzik, M.; Korzonek-Szlacheta, I.; Król, W. Triterpenes as Potentially Cytotoxic Compounds. Molecules 2015, 20, 1610–1625.
- Tang, Z.-Y.; Li, Y.; Tang, Y.-T.; Ma, X.-D.; Tang, Z.-Y. Anticancer Activity of Oleanolic Acid and Its Derivatives: Recent Advances in Evidence, Target Profiling and Mechanisms of Action. Biomed. Pharmacother. 2022, 145, 112397.
- Salvador, J.A.R.; Leal, A.S.; Valdeira, A.S.; Gonçalves, B.M.F.; Alho, D.P.S.; Figueiredo, S.A.C.; Silvestre, S.M.; Mendes, V.I.S. Oleanane-, Ursane-, and Quinone Methide Friedelane-Type Triterpenoid Derivatives: Recent Advances in Cancer Treatment. Eur. J. Med. Chem. 2017, 142, 95–130.
- Laszczyk, M.N. Pentacyclic Triterpenes of the Lupane, Oleanane and Ursane Group as Tools in Cancer Therapy. Planta Med. 2009, 75, 1549–1560.
- Ghante, M.H.; Jamkhande, P.G. Role of Pentacyclic Triterpenoids in Chemoprevention and Anticancer Treatment: An Overview on Targets and Underling Mechanisms. J. Pharmacopunct. 2019, 22, 55–67.
- Shaheen, U.; Ragab, E.A.; Abdalla, A.N.; Bader, A. Triterpenoidal Saponins from the Fruits of Gleditsia Caspica with Proapoptotic Properties. Phytochemistry 2018, 145, 168–178.
- Kumar, A.; Gupta, K.B.; Dhiman, M.; Arora, S.; Jaitak, V. New Pentacyclic Triterpene from Potentilla Atrosanguinea Lodd. as Anticancer Agent for Breast Cancer Targeting Estrogen Receptor-α. Nat. Prod. Res. 2022, 36, 4352–4357.
- Ghosh, A.; Panda, C.K. Role of Pentacyclic Triterpenoid Acids in the Treatment of Bladder Cancer. Mini Rev. Med. Chem. 2022, 22, 1331–1340.
- Liese, J.; Abhari, B.A.; Fulda, S. Smac Mimetic and Oleanolic Acid Synergize to Induce Cell Death in Human Hepatocellular Carcinoma Cells. Cancer Lett. 2015, 365, 47–56.
- Wang, X.; Bai, H.; Zhang, X.; Liu, J.; Cao, P.; Liao, N.; Zhang, W.; Wang, Z.; Hai, C. Inhibitory Effect of Oleanolic Acid on Hepatocellular Carcinoma via ERK-P53-Mediated Cell Cycle Arrest and Mitochondrial-Dependent Apoptosis. Carcinogenesis 2013, 34, 1323–1330.
- Mu, D.-W.; Guo, H.-Q.; Zhou, G.-B.; Li, J.-Y.; Su, B. Oleanolic Acid Suppresses the Proliferation of Human Bladder Cancer by Akt/MTOR/S6K and ERK1/2 Signaling. Int. J. Clin. Exp. Pathol. 2015, 8, 13864–13870.
- Amara, S.; Zheng, M.; Tiriveedhi, V. Oleanolic Acid Inhibits High Salt-Induced Exaggeration of Warburg-like Metabolism in Breast Cancer Cells. Cell Biochem. Biophys. 2016, 74, 427–434.
- Chakravarti, B.; Maurya, R.; Siddiqui, J.A.; Bid, H.K.; Rajendran, S.M.; Yadav, P.P.; Konwar, R. In Vitro Anti-Breast Cancer Activity of Ethanolic Extract of Wrightia Tomentosa: Role of pro-Apoptotic Effects of Oleanolic Acid and Urosolic Acid. J. Ethnopharmacol. 2012, 142, 72–79.
- Zhao, X.; Liu, M.; Li, D. Oleanolic Acid Suppresses the Proliferation of Lung Carcinoma Cells by MiR-122/Cyclin G1/MEF2D Axis. Mol. Cell. Biochem. 2015, 400, 1–7.
- Furtado, R.A.; Rodrigues, E.P.; Araújo, F.R.R.; Oliveira, W.L.; Furtado, M.A.; Castro, M.B.; Cunha, W.R.; Tavares, D.C. Ursolic Acid and Oleanolic Acid Suppress Preneoplastic Lesions Induced by 1,2-Dimethylhydrazine in Rat Colon. Toxicol. Pathol. 2008, 36, 576–580.
- Janakiram, N.B.; Indranie, C.; Malisetty, S.V.; Jagan, P.; Steele, V.E.; Rao, C.V. Chemoprevention of Colon Carcinogenesis by Oleanolic Acid and Its Analog in Male F344 Rats and Modulation of COX-2 and Apoptosis in Human Colon HT-29 Cancer Cells. Pharm. Res. 2008, 25, 2151–2157.
- Žiberna, L.; Šamec, D.; Mocan, A.; Nabavi, S.F.; Bishayee, A.; Farooqi, A.A.; Sureda, A.; Nabavi, S.M. Oleanolic Acid Alters Multiple Cell Signaling Pathways: Implication in Cancer Prevention and Therapy. Int. J. Mol. Sci. 2017, 18, 643.
- Yadav, V.R.; Prasad, S.; Sung, B.; Kannappan, R.; Aggarwal, B.B. Targeting Inflammatory Pathways by Triterpenoids for Prevention and Treatment of Cancer. Toxins 2010, 2, 2428–2466.
- Borella, R.; Forti, L.; Gibellini, L.; De Gaetano, A.; De Biasi, S.; Nasi, M.; Cossarizza, A.; Pinti, M. Synthesis and Anticancer Activity of CDDO and CDDO-Me, Two Derivatives of Natural Triterpenoids. Molecules 2019, 24, 4097.
- Hsu, Y.-C.; Hsieh, W.-C.; Chen, S.-H.; Li, Y.-Z.; Liao, H.-F.; Lin, M.-Y.; Sheu, S.-M. 18β-Glycyrrhetinic Acid Modulated Autophagy Is Cytotoxic to Breast Cancer Cells. Int. J. Med. Sci. 2023, 20, 444–454.
- Chen, J.; Zhang, Z.-Q.; Song, J.; Liu, Q.-M.; Wang, C.; Huang, Z.; Chu, L.; Liang, H.-F.; Zhang, B.-X.; Chen, X.-P. 18β-Glycyrrhetinic-Acid-Mediated Unfolded Protein Response Induces Autophagy and Apoptosis in Hepatocellular Carcinoma. Sci. Rep. 2018, 8, 9365.
- Sun, Y.; Dai, C.; Yin, M.; Lu, J.; Hu, H.; Chen, D. Hepatocellular Carcinoma-Targeted Effect of Configurations and Groups of Glycyrrhetinic Acid by Evaluation of Its Derivative-Modified Liposomes. Int. J. Nanomed. 2018, 13, 1621–1632.
- Lee, C.S.; Kim, Y.J.; Lee, M.S.; Han, E.S.; Lee, S.J. 18beta-Glycyrrhetinic Acid Induces Apoptotic Cell Death in SiHa Cells and Exhibits a Synergistic Effect against Antibiotic Anti-Cancer Drug Toxicity. Life Sci. 2008, 83, 481–489.
- Yamaguchi, H.; Noshita, T.; Yu, T.; Kidachi, Y.; Kamiie, K.; Umetsu, H.; Ryoyama, K. Novel Effects of Glycyrrhetinic Acid on the Central Nervous System Tumorigenic Progenitor Cells: Induction of Actin Disruption and Tumor Cell-Selective Toxicity. Eur. J. Med. Chem. 2010, 45, 2943–2948.
- Roohbakhsh, A.; Iranshahy, M.; Iranshahi, M. Glycyrrhetinic Acid and Its Derivatives: Anti-Cancer and Cancer Chemopreventive Properties, Mechanisms of Action and Structure- Cytotoxic Activity Relationship. Curr. Med. Chem. 2016, 23, 498–517.
- Zafar, S.; Khan, K.; Hafeez, A.; Irfan, M.; Armaghan, M.; ur Rahman, A.; Gürer, E.S.; Sharifi-Rad, J.; Butnariu, M.; Bagiu, I.-C.; et al. Ursolic Acid: A Natural Modulator of Signaling Networks in Different Cancers. Cancer Cell Int. 2022, 22, 399.
- Raphael, T.J.; Kuttan, G. Effect of Naturally Occurring Triterpenoids Ursolic Acid and Glycyrrhizic Acid on the Cell-Mediated Immune Responses of Metastatic Tumor-Bearing Animals. Immunopharmacol. Immunotoxicol. 2008, 30, 243–255.
- Kim, D.-K.; Baek, J.H.; Kang, C.-M.; Yoo, M.-A.; Sung, J.-W.; Kim, D.-K.; Chung, H.-Y.; Kim, N.D.; Choi, Y.H.; Lee, S.-H.; et al. Apoptotic Activity of Ursolic Acid May Correlate with the Inhibition of Initiation of DNA Replication. Int. J. Cancer 2000, 87, 629–636.
- Liu, X.-S.; Jiang, J. Induction of Apoptosis and Regulation of the MAPK Pathway by Ursolic Acid in Human Leukemia K562 Cells. Planta Med. 2007, 73, 1192–1194.
- Pisha, E.; Chai, H.; Lee, I.S.; Chagwedera, T.E.; Farnsworth, N.R.; Cordell, G.A.; Beecher, C.W.; Fong, H.H.; Kinghorn, A.D.; Brown, D.M. Discovery of Betulinic Acid as a Selective Inhibitor of Human Melanoma That Functions by Induction of Apoptosis. Nat. Med. 1995, 1, 1046–1051.
- Hordyjewska, A.; Ostapiuk, A.; Horecka, A.; Kurzepa, J. Betulin and Betulinic Acid: Triterpenoids Derivatives with a Powerful Biological Potential. Phytochem. Rev. 2019, 18, 929–951.
- Fulda, S. Betulinic Acid: A Natural Product with Anticancer Activity. Mol. Nutr. Food Res. 2009, 53, 140–146.
- Selzer, E.; Pimentel, E.; Wacheck, V.; Schlegel, W.; Pehamberger, H.; Jansen, B.; Kodym, R. Effects of Betulinic Acid Alone and in Combination with Irradiation in Human Melanoma Cells. J. Investig. Dermatol. 2000, 114, 935–940.
- Fulda, S.; Debatin, K.-M. Sensitization for Anticancer Drug-Induced Apoptosis by Betulinic Acid. Neoplasia 2005, 7, 162–170.
- Li, X.; Jiang, W.; Li, W.; Dong, S.; Du, Y.; Zhang, H.; Zhou, W. Betulinic Acid-Mediating MiRNA-365 Inhibited the Progression of Pancreatic Cancer. Oncol. Res. 2023, 31, 505–514.
- Lim, H.Y.; Ong, P.S.; Wang, L.; Goel, A.; Ding, L.; Li-Ann Wong, A.; Ho, P.C.; Sethi, G.; Xiang, X.; Goh, B.C. Celastrol in Cancer Therapy: Recent Developments, Challenges and Prospects. Cancer Lett. 2021, 521, 252–267.
- Yang, H.; Chen, D.; Cui, Q.C.; Yuan, X.; Dou, Q.P. Celastrol, a Triterpene Extracted from the Chinese “Thunder of God Vine,” Is a Potent Proteasome Inhibitor and Suppresses Human Prostate Cancer Growth in Nude Mice. Cancer Res. 2006, 66, 4758–4765.
- Nagase, M.; Oto, J.; Sugiyama, S.; Yube, K.; Takaishi, Y.; Sakato, N. Apoptosis Induction in HL-60 Cells and Inhibition of Topoisomerase II by Triterpene Celastrol. Biosci. Biotechnol. Biochem. 2003, 67, 1883–1887.
- Kannaiyan, R.; Manu, K.A.; Chen, L.; Li, F.; Rajendran, P.; Subramaniam, A.; Lam, P.; Kumar, A.P.; Sethi, G. Celastrol Inhibits Tumor Cell Proliferation and Promotes Apoptosis through the Activation of C-Jun N-Terminal Kinase and Suppression of PI3 K/Akt Signaling Pathways. Apoptosis Int. J. Program. Cell Death 2011, 16, 1028–1041.
- Zhang, H.; Mu, Y.; Wang, F.; Song, L.; Sun, J.; Liu, Y.; Sun, J. Synthesis of Gypsogenin Derivatives with Capabilities to Arrest Cell Cycle and Induce Apoptosis in Human Cancer Cells. R. Soc. Open Sci. 2018, 5, 171510.
- Lee, I.; Yoo, J.K.; Na, M.; Min, B.S.; Lee, J.; Yun, B.S.; Jin, W.; Kim, H.; Youn, U.; Chen, Q.C.; et al. Cytotoxicity of Triterpenes Isolated from Aceriphyllum Rossii. Chem. Pharm. Bull. 2007, 55, 1376–1378.
- Ciftci, H.I.; Radwan, M.O.; Ozturk, S.E.; Ulusoy, N.G.; Sozer, E.; Ellakwa, D.E.; Ocak, Z.; Can, M.; Ali, T.F.S.; Abd-Alla, H.I.; et al. Design, Synthesis and Biological Evaluation of Pentacyclic Triterpene Derivatives: Optimization of Anti-ABL Kinase Activity. Molecules 2019, 24, 3535.
- Emirdağ-Öztürk, S.; Babahan, İ.; Özmen, A. Synthesis, Characterization and In Vitro Anti-Neoplastic Activity of Gypsogenin Derivatives. Bioorganic Chem. 2014, 53, 15–23.
- Emirdağ-Öztürk, S.; Karayıldırım, T.; Çapcı-Karagöz, A.; Alankuş-Çalışkan, Ö.; Özmen, A.; Poyrazoğlu-Çoban, E. Synthesis, Antimicrobial and Cytotoxic Activities, and Structure-Activity Relationships of Gypsogenin Derivatives against Human Cancer Cells. Eur. J. Med. Chem. 2014, 82, 565–573.
- Ciftci, H.I.; Ozturk, S.E.; Ali, T.F.S.; Radwan, M.O.; Tateishi, H.; Koga, R.; Ocak, Z.; Can, M.; Otsuka, M.; Fujita, M. The First Pentacyclic Triterpenoid Gypsogenin Derivative Exhibiting Anti-ABL1 Kinase and Anti-Chronic Myelogenous Leukemia Activities. Biol. Pharm. Bull. 2018, 41, 570–574.
- Ulusoy, N.G.; Emirdağ, S.; Sözer, E.; Radwan, M.O.; Çiftçi, H.; Aksel, M.; Bölükbaşı, S.Ş.; Özmen, A.; Yaylı, N.; Karayıldırım, T.; et al. Design, Semi-Synthesis and Examination of New Gypsogenin Derivatives against Leukemia via Abl Tyrosine Kinase Inhibition and Apoptosis Induction. Int. J. Biol. Macromol. 2022, 222, 1487–1499.
- Gypsogenin Türevi Ile Kalkon Bileşiklerinin Yeni Yari Sentezi ve bu Türevlerinin Insan Kanser Hücre Hatlari Üzerindeki Çalişmalari—TR201922043A2|PatentGuru. Available online: https://www.patentguru.com/TR201922043A2 (accessed on 11 July 2023).
- Almansour, N.M. Triple-Negative Breast Cancer: A Brief Review About Epidemiology, Risk Factors, Signaling Pathways, Treatment and Role of Artificial Intelligence. Front. Mol. Biosci. 2022, 9, 836417.
- Wu, G.; Chu, H.; Wang, J.; Mu, Y.; Sun, J. Synthesis of Gypsogenin and Gypsogenic Acid Derivatives with Antitumor Activity by Damaging Cell Membranes. New J. Chem. 2019, 43, 18898–18914.
- Sun, K.-P.; Zhao, T.-T.; Liu, L.; Mu, X.-D.; Sun, J.-Y. Anticancer Structure-Activity Relationships and Potential Target Exploration of the Natural Product Gypsogenin. ChemistrySelect 2023, 8, e202300072.
- Tian, G.; Zhou, L.; Zhong, Y.; Xu, W.; Bai, H.; Liu, L.; Cui, S. Experimental Studies of the Therapeutic Effect of Gypsophila oldhamiana Gypsogenin on Lewis Lung Cancer in Mice. Chin. J. Clin. Oncol. 2008, 5, 206–210.
- Sun, J.; Zhang, H.; Mou, Y.; Sun, J.; Wang, F.; Wu, Z.; Wang, Y.; Song, L. Gypsogenin Derivatives. CN107236017A. 10 October 2017. Available online: https://patents.google.com/patent/CN107236017A/en?oq=CN+107236017A (accessed on 11 July 2023).
- Liu, Y.; Li, X.; Jiang, S.; Ge, Q. Inhibitory effect of Gypsophila oldhamiana gypsogenin on NCI-N87 gastric cancer cell line. Eur. J. Inflamm. 2018, 16.





