
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Shu-Lin Liu | -- | 2655 | 2023-07-26 11:06:36 | | | |
| 2 | Peter Tang | Meta information modification | 2655 | 2023-07-26 11:26:12 | | |
Video Upload Options
Sphingomyelin (SM) and its metabolites are crucial regulators of tumor cell growth, differentiation, senescence, and programmed cell death. With the rise in lipid-based nanomaterials, engineered lipidic nanomaterials inspired by SM metabolism, corresponding lipid targeting, and signaling activation have made fascinating advances in cancer therapeutic processes.
1. Introduction
2. Synthesis and Metabolism of SM
2.1. Structure and Distribution of SM
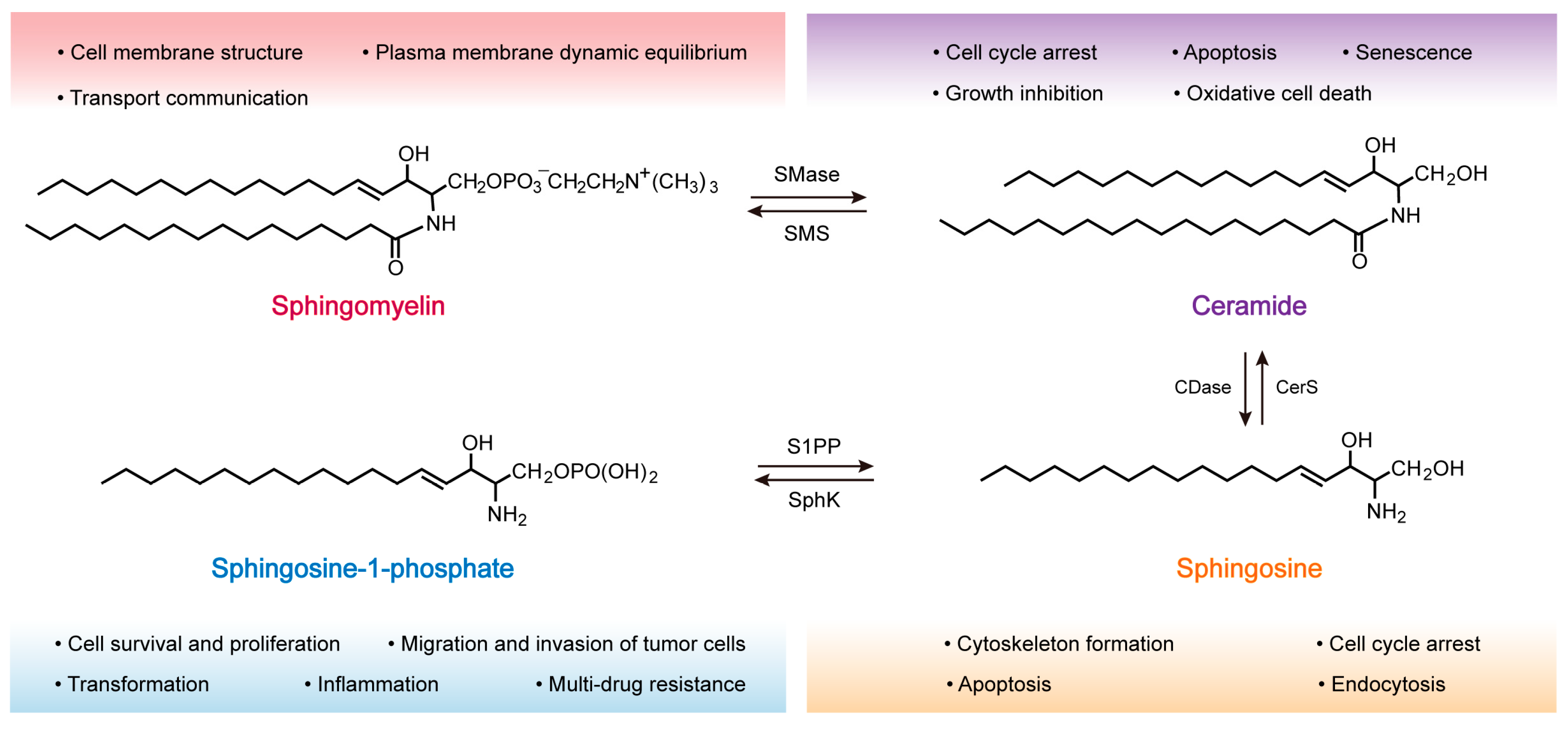
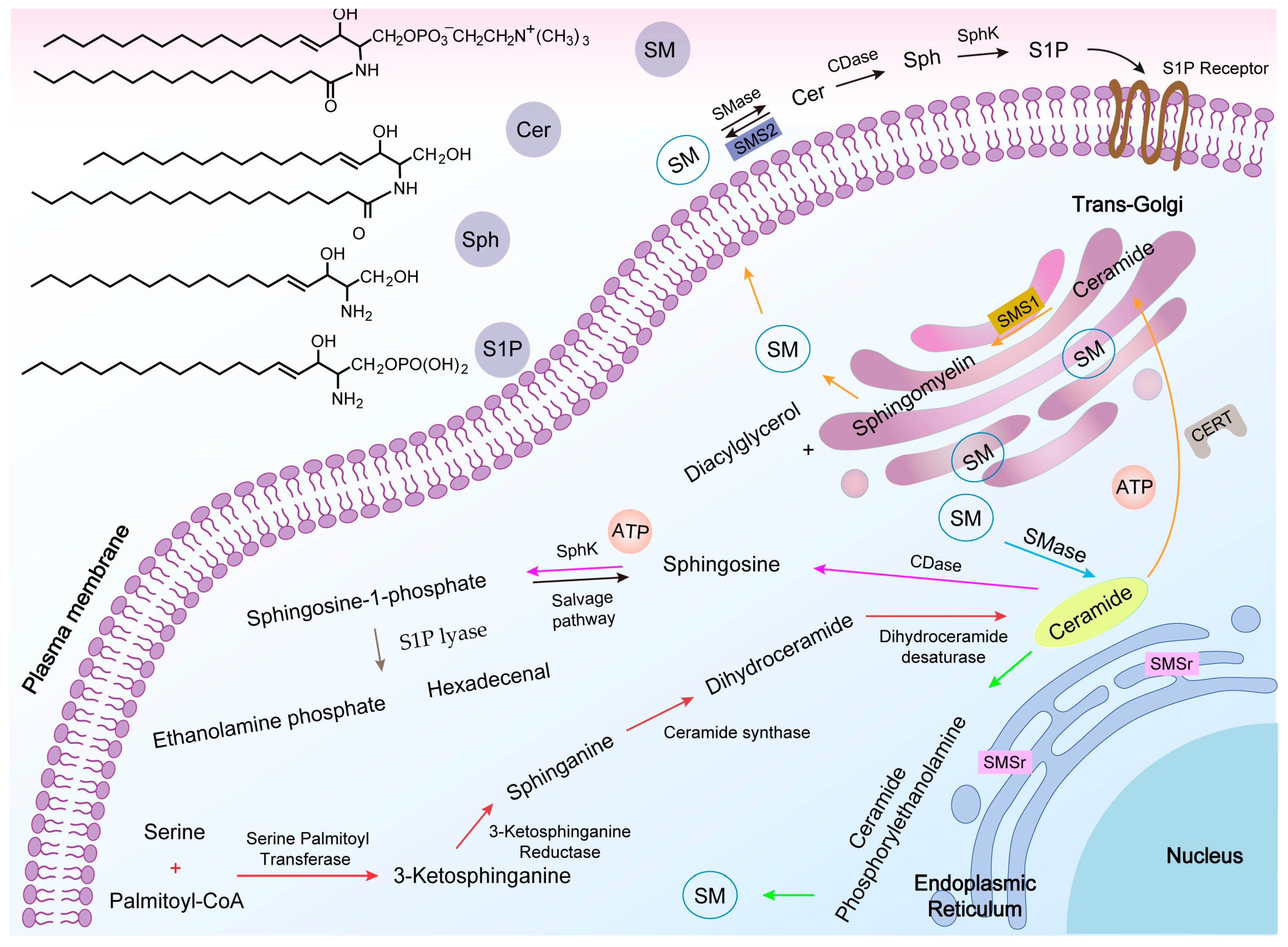
2.2. SM Synthesis
2.3. SM Metabolism
3. Role of SM in the Development of Tumors
|
Active Molecule |
Pathway |
Function or Activity |
Cancer |
Refs. |
|---|---|---|---|---|
|
Sphingosine kinase |
Akt/NF-κB |
Cancer progression and chemoresistance |
Colon |
[42] |
|
S1P/S1PR1 |
Inflammation and angiogenesis |
Breast |
[43] |
|
|
PI3k/Akt/FOXO3a |
Apoptosis resistance |
Breast |
[44] |
|
|
S1P/Stat3/AKT |
Proliferation |
Colon |
[45] |
|
|
E-cad |
Tumorigenesis and metastasis |
Breast |
[46] |
|
|
S1P/ERK/CD44 |
Chemoresistance |
Colon |
[47] |
|
|
S1P/AKT/GSK-3β |
HIF-1α stabilization |
Glioblastoma |
[48] |
|
|
Sphingomyelinase |
↑ Sensitivity |
Glioblastoma |
[49] |
|
|
↑ Resistance |
Melanoma |
[50] |
||
|
Induce apoptosis |
Colon |
[51] |
||
|
Induce apoptosis and resistance |
Ovarian |
[52] |
||
|
Acid ceramidase |
↑ Proliferative ↓ Sensitivity |
Melanoma |
||
|
↓ Sensitivity |
Prostate |
[55] |
||
|
↑ Radioresistant |
Glioblastoma |
[55] |
||
|
Sphingosine kinase 2 |
Mcl-1 |
↑ Cell survival |
Leukemia |
[56] |
|
Sphingosine-1-phosphate lyase |
p53 and p38 |
↑ Apoptosis |
Colon |
[57] |
|
Sphingomyelin synthase |
TGF- b1 |
↑ Migratory, invasion |
Breast |
[58] |
|
Overexpression of SMS1 |
↓ Cell death |
Lymphatic |
[59] |
|
|
Sphingomyelinase |
CD95 |
↑ Apoptosis |
Lymphatic |
[60] |
|
p53 |
↑ Apoptosis |
Lung |
[61] |
|
|
Sphingosine |
Cdk4 |
↓ Cell proliferation |
Intestinal adenoma |
[62] |
|
Ceramide |
CerS6/C16-ceramide activated |
↑ Apoptosis |
Lung |
[63] |
|
High cytotoxicity in p53 |
↑ Apoptosis |
Breast |
[64] |
↑, increase; ↓, decrease.
4. SM Metabolism-Based Lipidic Nanomaterials for Cancer Therapy
4.1. SM-Based Lipidic Nanomaterials for Cancer Therapy
4.2. Cer-Based Lipidic Nanomaterials for Cancer Therapy
4.3. S1P-Based Lipidic Nanomaterials for Cancer Therapy
|
Materials |
Size (d. nm) |
Therapeutics |
Cancer Cell Type |
Refs. |
|---|---|---|---|---|
|
SM-CSS-CPT |
93.1 ± 7.63 |
Intravenous injection |
CT26, b16, mc38 |
[67] |
|
UroGm-SNs |
131 ± 12 |
SW620 |
[68] |
|
|
DOTAP (DSN) |
142 ± 2 |
HCT-116 |
[71] |
|
|
PEGylated Cu (DDC)2 liposomes |
121.5 ± 0.57 |
Intravenous injection |
4T1 |
[90] |
|
SNs–ST |
131 ± 8 |
SW480 |
[72] |
|
|
SNs_PEG |
77 ± 3 |
PANC-1, |
[91] |
|
|
Lipid–porphyrin conjugates |
180 ± 10 |
Kyse-30 |
[92] |
|
|
SMLs@PDA |
229.5 ± 26.3 |
[93] |
||
|
C6-NBD-SM Liposomes |
71 ± 3 |
CCRF-CEM |
[94] |
|
|
CPNPs |
20 |
MCF-7 |
[78] |
|
|
C6 ceramide |
~80 |
B16, WM-115 |
[95] |
|
|
TRI-Gel |
Subcutaneous injection |
Lewis lung carcinoma |
[96] |
|
|
Celastrol |
Subcutaneous injection |
CT26, HCT-8, HCT-116, DLD-1 |
[97] |
|
|
LipC6 |
90 |
Intra-splenic injections |
liver tumors |
[80] |
|
Thirty molar percent C6-ceramide in a twelve molar percent pegylated |
80 |
Intraperitoneal injection |
SKOV3, TOV112D, A2780, A2780CP, PE01, PE04 |
[98] |
|
S1P/JS-K/Lipo |
189 |
Intravenous injection |
U87MG |
[81] |
|
PP2A |
A549 |
[82] |
||
|
3-[4-(5-aryl-1, 2, 4-oxadiazol-3-yl)-1H-indol-1-yl]propanoic acid series |
Intravenous injection |
peripheral lymphocyte |
[84] |
References
- Miller, K.D.; Siegel, R.L.; Lin, C.C.; Mariotto, A.B.; Kramer, J.L.; Rowland, J.H.; Stein, K.D.; Alteri, R.; Jemal, A. Cancer treatment and survivorship statistics, 2016. CA Cancer J. Clin. 2016, 66, 271–289.
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Parkin, D.M.; Pineros, M.; Znaor, A.; Bray, F. Cancer statistics for the year 2020: An overview. Int. J. Cancer 2021, 149, 778–789.
- Riehemann, K.; Schneider, S.W.; Luger, T.A.; Godin, B.; Ferrari, M.; Fuchs, H. Nanomedicine--challenge and perspectives. Angew. Chem. Int. Ed. 2009, 48, 872–897.
- Zhou, Q.; Zhang, L.; Wu, H. Nanomaterials for cancer therapies. Nanotechnol. Rev. 2017, 6, 473–496.
- Sun, T.; Zhang, Y.S.; Pang, B.; Hyun, D.C.; Yang, M.; Xia, Y. Engineered nanoparticles for drug delivery in cancer therapy. Angew. Chem. Int. Ed. 2014, 53, 12320–12364.
- Dadfar, S.M.; Camozzi, D.; Darguzyte, M.; Roemhild, K.; Varvara, P.; Metselaar, J.; Banala, S.; Straub, M.; Guvener, N.; Engelmann, U.; et al. Size-isolation of superparamagnetic iron oxide nanoparticles improves MRI, MPI and hyperthermia performance. J. Nanobiotechnol. 2020, 18, 22.
- Li, M.; Huang, Y.; Wu, J.; Li, S.; Mei, M.; Chen, H.; Wang, N.; Wu, W.; Zhou, B.; Tan, X.; et al. A PEG-lipid-free COVID-19 mRNA vaccine triggers robust immune responses in mice. Mater. Horiz. 2023, 10, 466–472.
- Chaudhari, V.S.; Murty, U.S.; Banerjee, S. Lipidic nanomaterials to deliver natural compounds against cancer: A review. Environ. Chem. Lett. 2020, 18, 1803–1812.
- Limongi, T.; Susa, F.; Marini, M.; Allione, M.; Torre, B.; Pisano, R.; di Fabrizio, E. Lipid-based nanovesicular drug delivery systems. Nanomaterials 2021, 11, 3391.
- Porter, C.J.; Trevaskis, N.L.; Charman, W.N. Lipids and lipid-based formulations: Optimizing the oral delivery of lipophilic drugs. Nat. Rev. Drug Discov. 2007, 6, 231–248.
- Quehenberger, O.; Armando, A.M.; Brown, A.H.; Milne, S.B.; Myers, D.S.; Merrill, A.H.; Bandyopadhyay, S.; Jones, K.N.; Kelly, S.; Shaner, R.L.; et al. Lipidomics reveals a remarkable diversity of lipids in human plasma. J. Lipid Res. 2010, 51, 3299–3305.
- Wolrab, D.; Jirasko, R.; Cifkova, E.; Horing, M.; Mei, D.; Chocholouskova, M.; Peterka, O.; Idkowiak, J.; Hrnciarova, T.; Kuchar, L.; et al. Lipidomic profiling of human serum enables detection of pancreatic cancer. Nat. Commun. 2022, 13, 124.
- Hu, Y.; Zhang, R.Q.; Wang, Z.G.; Liu, S.L. In situ quantification of lipids in live cells by using lipid-binding domain-based biosensors. Bioconjug. Chem. 2022, 33, 2076–2087.
- Radin, N.S. Cancer progression in the kidney and prostate: Vital roles of sphingolipids in chemotherapy. Urology 2002, 60, 562–568.
- Vykoukal, J.; Fahrmann, J.F.; Gregg, J.R.; Tang, Z.; Basourakos, S.; Irajizad, E.; Park, S.; Yang, G.; Creighton, C.J.; Fleury, A.; et al. Caveolin-1-mediated sphingolipid oncometabolism underlies a metabolic vulnerability of prostate cancer. Nat. Commun. 2020, 11, 4279.
- D’Angelo, G.; Moorthi, S.; Luberto, C. Role and function of sphingomyelin biosynthesis in the development of cancer. Adv. Cancer Res. 2018, 140, 61–96.
- Bienias, K.; Fiedorowicz, A.; Sadowska, A.; Prokopiuk, S.; Car, H. Regulation of sphingomyelin metabolism. Pharmacol. Rep. 2016, 68, 570–581.
- Ogretmen, B. Sphingolipid metabolism in cancer signalling and therapy. Nat. Rev. Cancer 2018, 18, 33–50.
- Ogretmen, B.; Hannun, Y.A. Biologically active sphingolipids in cancer pathogenesis and treatment. Nat. Rev. Cancer 2004, 4, 604–616.
- Modrak, D.E.; Gold, D.V.; Goldenberg, D.M. Sphingolipid targets in cancer therapy. Mol. Cancer Ther. 2006, 5, 200–208.
- Newton, J.; Lima, S.; Maceyka, M.; Spiegel, S. Revisiting the sphingolipid rheostat: Evolving concepts in cancer therapy. Exp. Cell Res. 2015, 333, 195–200.
- Taniguchi, M.; Okazaki, T. Role of ceramide/sphingomyelin (SM) balance regulated through “SM cycle” in cancer. Cell. Signal. 2021, 87, 110119.
- Cartier, A.; Hla, T. Sphingosine 1-phosphate: Lipid signaling in pathology and therapy. Science 2019, 366, eaar5551.
- Taha, T.A.; Mullen, T.D.; Obeid, L.M. A house divided: Ceramide, sphingosine, and sphingosine-1-phosphate in programmed cell death. Biochim. Biophys. Acta 2006, 1758, 2027–2036.
- Kolesnick, R. The therapeutic potential of modulating the ceramide/sphingomyelin pathway. J. Clin. Investig. 2002, 110, 3–8.
- Huwiler, A.; Pfeilschifter, J. Altering the sphingosine-1-phosphate/ceramide balance: A promising approach for tumor therapy. Curr. Pharm. Des. 2006, 12, 4625–4635.
- Merrill, A.H., Jr.; Schmelz, E.M.; Dillehay, D.L.; Spiegel, S.; Shayman, J.A.; Schroeder, J.J.; Riley, R.T.; Voss, K.A.; Wang, E. Sphingolipids--the enigmatic lipid class: Biochemistry, physiology, and pathophysiology. Toxicol. Appl. Pharmacol. 1997, 142, 208–225.
- Filippov, A.; Oradd, G.; Lindblom, G. Sphingomyelin structure influences the lateral diffusion and raft formation in lipid bilayers. Biophys. J. 2006, 90, 2086–2092.
- Christie; William, W. Lipids: Their structures and occurrence. In Lipid Analysis, 4th ed.; Elsevier: Amsterdam, The Netherlands, 2012; pp. 3–19.
- Furland, N.E.; Zanetti, S.R.; Oresti, G.M.; Maldonado, E.N.; Aveldano, M.I. Ceramides and sphingomyelins with high proportions of very long-chain polyunsaturated fatty acids in mammalian germ cells. J. Biol. Chem. 2007, 282, 18141–18150.
- Slotte, J.P. Biological functions of sphingomyelins. Prog. Lipid Res. 2013, 52, 424–437.
- Allan, D.; Quinn, P. Resynthesis of sphingomyelin from plasma-membrane phosphatidylcholine in BHK cells treated with staphylococcus aureus sphingomyelinase. Biochem. J. 1988, 254, 765–771.
- Gault, C.R.; Obeid, L.M.; Hannun, Y.A. An overview of sphingolipid metabolism: From synthesis to breakdown. Adv. Exp. Med. Biol. 2010, 688, 1–23.
- Futerman, A.H.; Stieger, B.; Hubbard, A.L.; Pagano, R.E. Sphingomyelin synthesis in rat liver occurs predominantly at the cis and medial cisternae of the Golgi apparatus. J. Biol. Chem. 1990, 265, 8650–8657.
- Huitema, K.; van den Dikkenberg, J.; Brouwers, J.F.; Holthuis, J.C. Identification of a family of animal sphingomyelin synthases. EMBO J. 2004, 23, 33–44.
- Hanada, K.; Kumagai, K.; Yasuda, S.; Miura, Y.; Kawano, M.; Fukasawa, M.; Nishijima, M. Molecular machinery for non-vesicular trafficking of ceramide. Nature 2003, 426, 803–809.
- Hanada, K. Regulation of CERT-mediated trafficking of ceramide. Chem. Phys. Lipids 2007, 149, S7.
- Mitsutake, S.; Zama, K.; Yokota, H.; Yoshida, T.; Tanaka, M.; Mitsui, M.; Ikawa, M.; Okabe, M.; Tanaka, Y.; Yamashita, T.; et al. Dynamic modification of sphingomyelin in lipid microdomains controls development of obesity, fatty liver, and type 2 diabetes. J. Biol. Chem. 2011, 286, 28544–28555.
- Vacaru, A.M.; Tafesse, F.G.; Ternes, P.; Kondylis, V.; Hermansson, M.; Brouwers, J.F.; Somerharju, P.; Rabouille, C.; Holthuis, J.C. Sphingomyelin synthase-related protein SMSr controls ceramide homeostasis in the ER. J. Cell Biol. 2009, 185, 1013–1027.
- Saddoughi, S.A.; Song, P.; Ogretmen, B. Roles of bioactive sphingolipids in cancer biology and therapeutics. Subcell. Biochem. 2008, 49, 413–440.
- Haddadi, N.; Lin, Y.; Simpson, A.M.; Nassif, N.T.; McGowan, E.M. “Dicing and splicing” sphingosine kinase and relevance to cancer. Int. J. Mol. Sci. 2017, 18, 1891.
- Shamekhi, S.; Abdolalizadeh, J.; Ostadrahimi, A.; Mohammadi, S.A.; Barzegari, A.; Lotfi, H.; Bonabi, E.; Zarghami, N. Apoptotic effect of saccharomyces cerevisiae on human colon cancer SW480 cells by regulation of Akt/NF-kB signaling pathway. Probiotics Antimicrob. Proteins 2020, 12, 311–319.
- Nagahashi, M.; Yamada, A.; Katsuta, E.; Aoyagi, T.; Huang, W.C.; Terracina, K.P.; Hait, N.C.; Allegood, J.C.; Tsuchida, J.; Yuza, K.; et al. Targeting the SphK1/S1P/S1PR1 axis that links obesity, chronic inflammation, and breast cancer metastasis. Cancer Res. 2018, 78, 1713–1725.
- Zhang, X.; Zhuang, T.; Liang, Z.; Li, L.; Xue, M.; Liu, J.; Liang, H. Breast cancer suppression by aplysin is associated with inhibition of PI3K/AKT/FOXO3a pathway. Oncotarget 2017, 8, 63923–63934.
- Bao, Y.; Li, K.; Guo, Y.; Wang, Q.; Li, Z.; Yang, Y.; Chen, Z.; Wang, J.; Zhao, W.; Zhang, H.; et al. Tumor suppressor PRSS8 targets Sphk1/S1P/Stat3/Akt signaling in colorectal cancer. Oncotarget 2016, 7, 26780–26792.
- Zheng, X.D.; Zhang, Y.; Qi, X.W.; Wang, M.H.; Sun, P.; Zhang, Y.; Jiang, J. Role of Sphk1 in the malignant transformation of breast epithelial cells and breast cancer progression. Indian J. Cancer 2014, 51, 524–529.
- Kawahara, S.; Otsuji, Y.; Nakamura, M.; Murakami, M.; Murate, T.; Matsunaga, T.; Kanoh, H.; Seishima, M.; Banno, Y.; Hara, A. Sphingosine kinase 1 plays a role in the upregulation of CD44 expression through extracellular signal-regulated kinase signaling in human colon cancer cells. Anticancer Drugs 2013, 24, 473–483.
- Ader, I.; Brizuela, L.; Bouquerel, P.; Malavaud, B.; Cuvillier, O. Sphingosine kinase 1: A new modulator of hypoxia inducible factor 1alpha during hypoxia in human cancer cells. Cancer Res. 2008, 68, 8635–8642.
- Pchejetski, D.; Golzio, M.; Bonhoure, E.; Calvet, C.; Doumerc, N.; Garcia, V.; Mazerolles, C.; Rischmann, P.; Teissie, J.; Malavaud, B.; et al. Sphingosine kinase-1 as a chemotherapy sensor in prostate adenocarcinoma cell and mouse models. Cancer Res. 2005, 65, 11667–11675.
- Alshaker, H.; Wang, Q.; Kawano, Y.; Arafat, T.; Bohler, T.; Winkler, M.; Cooper, C.; Pchejetski, D. Everolimus (RAD001) sensitizes prostate cancer cells to docetaxel by down-regulation of HIF-1alpha and sphingosine kinase 1. Oncotarget 2016, 7, 80943–80956.
- Sauer, L.; Nunes, J.; Salunkhe, V.; Skalska, L.; Kohama, T.; Cuvillier, O.; Waxman, J.; Pchejetski, D. Sphingosine kinase 1 inhibition sensitizes hormone-resistant prostate cancer to docetaxel. Int. J. Cancer 2009, 125, 2728–2736.
- Sukocheva, O.; Wang, L.; Verrier, E.; Vadas, M.A.; Xia, P. Restoring endocrine response in breast cancer cells by inhibition of the sphingosine kinase-1 signaling pathway. Endocrinology 2009, 150, 4484–4492.
- Giussani, P.; Bassi, R.; Anelli, V.; Brioschi, L.; De Zen, F.; Riccitelli, E.; Caroli, M.; Campanella, R.; Gaini, S.M.; Viani, P.; et al. Glucosylceramide synthase protects glioblastoma cells against autophagic and apoptotic death induced by temozolomide and Paclitaxel. Cancer Investig. 2012, 30, 27–37.
- Liu, Y.Y.; Han, T.Y.; Giuliano, A.E.; Cabot, M.C. Expression of glucosylceramide synthase, converting ceramide to glucosylceramide, confers adriamycin resistance in human breast cancer cells. J. Biol. Chem. 1999, 274, 1140–1146.
- Liu, Y.Y.; Patwardhan, G.A.; Xie, P.; Gu, X.; Giuliano, A.E.; Cabot, M.C. Glucosylceramide synthase, a factor in modulating drug resistance, is overexpressed in metastatic breast carcinoma. Int. J. Oncol. 2011, 39, 425–431.
- LeBlanc, F.R.; Pearson, J.M.; Tan, S.F.; Cheon, H.; Xing, J.C.; Dunton, W.; Feith, D.J.; Loughran, T.P., Jr. Sphingosine kinase-2 is overexpressed in large granular lymphocyte leukaemia and promotes survival through Mcl-1. Br. J. Heaematol. 2020, 190, 405–417.
- Oskouian, B.; Sooriyakumaran, P.; Borowsky, A.D.; Crans, A.; Dillard-Telm, L.; Tam, Y.Y.; Bandhuvula, P.; Saba, J.D. Sphingosine-1-phosphate lyase potentiates apoptosis via p53- and p38-dependent pathways and is down-regulated in colon cancer. Proc. Natl. Acad. Sci. USA 2006, 103, 17384–17389.
- Zheng, K.; Chen, Z.; Feng, H.; Chen, Y.; Zhang, C.; Yu, J.; Luo, Y.; Zhao, L.; Jiang, X.; Shi, F. Sphingomyelin synthase 2 promotes an aggressive breast cancer phenotype by disrupting the homoeostasis of ceramide and sphingomyelin. Cell Death Dis. 2019, 10, 157.
- Lafont, E.; Milhas, D.; Carpentier, S.; Garcia, V.; Jin, Z.X.; Umehara, H.; Okazaki, T.; Schulze-Osthoff, K.; Levade, T.; Benoist, H.; et al. Caspase-mediated inhibition of sphingomyelin synthesis is involved in FasL-triggered cell death. Cell Death Differ. 2010, 17, 642–654.
- Grassme, H.; Schwarz, H.; Gulbins, E. Molecular mechanisms of ceramide-mediated CD95 clustering. Biochem. Biophys. Res. Commun. 2001, 284, 1016–1030.
- Eroica, S.; Susan, C.E.; Cynthia, C.; Elroy, F. Characterizing the sphingomyelinase pathway triggered by PRIMA-1 derivatives in lung cancer cells with differing p53 status. Anticancer Res. 2014, 34, 3271.
- Kohno, M.; Momoi, M.; Oo, M.L.; Paik, J.H.; Lee, Y.M.; Venkataraman, K.; Ai, Y.; Ristimaki, A.P.; Fyrst, H.; Sano, H.; et al. Intracellular role for sphingosine kinase 1 in intestinal adenoma cell proliferation. Mol. Cell. Biol. 2006, 26, 7211–7223.
- Senkal, C.E.; Ponnusamy, S.; Manevich, Y.; Meyers-Needham, M.; Saddoughi, S.A.; Mukhopadyay, A.; Dent, P.; Bielawski, J.; Ogretmen, B. Alteration of ceramide synthase 6/C16-ceramide induces activating transcription factor 6-mediated endoplasmic reticulum (ER) stress and apoptosis via perturbation of cellular Ca2+ and ER/Golgi membrane network. J. Biol. Chem. 2011, 286, 42446–42458.
- Chang, W.T.; Wu, C.Y.; Lin, Y.C.; Wu, M.T.; Su, K.L.; Yuan, S.S.; Wang, H.D.; Fong, Y.; Lin, Y.H.; Chiu, C.C. C(2)-ceramide-induced rb-dominant senescence-like phenotype leads to human breast cancer Mcf-7 escape from p53-dependent cell death. Int. J. Mol. Sci. 2019, 20, 4292.
- Zhong, L.; Li, Y.; Xiong, L.; Wang, W.; Wu, M.; Yuan, T.; Yang, W.; Tian, C.; Miao, Z.; Wang, T.; et al. Small molecules in targeted cancer therapy: Advances, challenges, and future perspectives. Signal Transduct. Target. Ther. 2021, 6, 201.
- Phillips, M.A.; Gran, M.L.; Peppas, N.A. Targeted nanodelivery of drugs and diagnostics. Nano Today 2010, 5, 143–159.
- Wang, Z.; Little, N.; Chen, J.; Lambesis, K.T.; Le, K.T.; Han, W.; Scott, A.J.; Lu, J. Immunogenic camptothesome nanovesicles comprising sphingomyelin-derived camptothecin bilayers for safe and synergistic cancer immunochemotherapy. Nat. Nanotechnol. 2021, 16, 1130–1140.
- Bouzo, B.L.; Lores, S.; Jatal, R.; Alijas, S.; Alonso, M.J.; Conejos-Sanchez, I.; de la Fuente, M. Sphingomyelin nanosystems loaded with uroguanylin and etoposide for treating metastatic colorectal cancer. Sci. Rep. 2021, 11, 17213.
- Medina, O.P.; Tower, R.J.; Medina, T.P.; Ashkenani, F.; Appold, L.; Bötcher, M.; Huber, L.; Will, O.; Ling, Q.; Hauser, C.; et al. Multimodal targeted nanoparticle-based delivery system for pancreatic tumor imaging in cellular and animal models. Curr. Pharm. Des. 2020, 28, 313–323.
- Penate Medina, T.; Gerle, M.; Humbert, J.; Chu, H.; Kopnick, A.L.; Barkmann, R.; Garamus, V.M.; Sanz, B.; Purcz, N.; Will, O.; et al. Lipid-iron nanoparticle with a cell stress release mechanism combined with a local alternating magnetic field enables site-activated drug release. Cancers 2020, 12, 3767.
- Masoumi, F.; Saraiva, S.M.; Bouzo, B.L.; Lopez-Lopez, R.; Esteller, M.; Diaz-Lagares, A.; de la Fuente, M. Modulation of colorectal tumor behavior via lncrna tp53tg1-lipidic nanosystem. Pharmaceutics 2021, 13, 1507.
- Nagachinta, S.; Bouzo, B.L.; Vazquez-Rios, A.J.; Lopez, R.; Fuente, M. Sphingomyelin-based nanosystems (sns) for the development of anticancer miRNA therapeutics. Pharmaceutics 2020, 12, 189.
- Morad, S.A.; Messner, M.C.; Levin, J.C.; Abdelmageed, N.; Park, H.; Merrill, A.H., Jr.; Cabot, M.C. Potential role of acid ceramidase in conversion of cytostatic to cytotoxic end-point in pancreatic cancer cells. Cancer Chemoth. Pharm. 2013, 71, 635–645.
- Flowers, M.; Fabrias, G.; Delgado, A.; Casas, J.; Abad, J.L.; Cabot, M.C. C6-ceramide and targeted inhibition of acid ceramidase induce synergistic decreases in breast cancer cell growth. Breast Cancer Res. Treat. 2012, 133, 447–458.
- Huang, H.; Zhang, Y.; Liu, X.; Li, Z.; Xu, W.; He, S.; Huang, Y.; Zhang, H. Acid sphingomyelinase contributes to evodiamine-induced apoptosis in human gastric cancer SGC-7901 cells. DNA Cell Biol. 2011, 30, 407–412.
- Kester, M.; Heakal, Y.; Fox, T.; Sharma, A.; Robertson, G.P.; Morgan, T.T.; Altinoglu, E.I.; Tabakovic, A.; Parette, M.R.; Rouse, S.M.; et al. Calcium phosphate nanocomposite particles for in vitro imaging and encapsulated chemotherapeutic drug delivery to cancer cells. Nano Lett. 2008, 8, 4116–4121.
- Ganta, S.; Singh, A.; Patel, N.R.; Cacaccio, J.; Rawal, Y.H.; Davis, B.J.; Amiji, M.M.; Coleman, T.P. Development of EGFR-targeted nanoemulsion for imaging and novel platinum therapy of ovarian cancer. Pharm. Res. 2014, 31, 2490–2502.
- Stover, T.; Kester, M. Liposomal delivery enhances short-chain ceramide-induced apoptosis of breast cancer cells. J. Pharmacol. Exp. Ther. 2003, 307, 468–475.
- Chen, L.; Alrbyawi, H.; Poudel, I.; Arnold, R.D.; Babu, R.J. Co-delivery of doxorubicin and ceramide in a liposomal formulation enhances cytotoxicity in murine B16Bl6 melanoma cell lines. AAPS PharmSciTech 2019, 20, 99.
- Li, G.; Liu, D.; Kimchi, E.T.; Kaifi, J.T.; Qi, X.; Manjunath, Y.; Liu, X.; Deering, T.; Avella, D.M.; Fox, T.; et al. Nanoliposome C6-ceramide increases the anti-tumor immune response and slows growth of liver tumors in mice. Gastroenterology 2018, 154, 1024–1036.
- Liu, Y.; Wang, X.; Li, J.; Tang, J.; Li, B.; Zhang, Y.; Gu, N.; Yang, F. Sphingosine 1-phosphate liposomes for targeted nitric oxide delivery to mediate anticancer effects against brain glioma tumors. Adv. Mater. 2021, 33, e2101701.
- Saddoughi, S.A.; Gencer, S.; Peterson, Y.K.; Ward, K.E.; Mukhopadhyay, A.; Oaks, J.; Bielawski, J.; Szulc, Z.M.; Thomas, R.J.; Selvam, S.P.; et al. Sphingosine analogue drug FTY720 targets I2PP2A/SET and mediates lung tumour suppression via activation of PP2A-RIPK1-dependent necroptosis. EMBO Mol. Med. 2013, 5, 105–121.
- Companioni, O.; Mir, C.; Garcia-Mayea, Y.; ME, L.L. Targeting Sphingolipids for Cancer Therapy. Front. Oncol. 2021, 11, 745092.
- Meng, Q.; Zhao, B.; Xu, Q.; Xu, X.; Deng, G.; Li, C.; Luan, L.; Ren, F.; Wang, H.; Xu, H. Indole-propionic acid derivatives as potent, S1P3-sparing and EAE efficacious sphingosine-1-phosphate 1 (S1P1) receptor agonists. Bioorg. Med. Chem. Lett. 2012, 22, 2794–2797.
- Luo, D.; Guo, Z.; Zhao, X.; Wu, L.; Liu, X.; Zhang, Y.; Zhang, Y.; Deng, Z.; Qu, X.; Cui, S.; et al. Novel 5-fluorouracil sensitizers for colorectal cancer therapy: Design and synthesis of S1P receptor 2 (S1PR2) antagonists. Eur. J. Med. Chem. 2021, 227, 113923.
- Beljanski, V.; Knaak, C.; Smith, C.D. A novel sphingosine kinase inhibitor induces autophagy in tumor cells. J. Pharmacol. Exp. Ther. 2010, 333, 454–464.
- Huang, X.; Taeb, S.; Jahangiri, S.; Emmenegger, U.; Tran, E.; Bruce, J.; Mesci, A.; Korpela, E.; Vesprini, D.; Wong, C.S.; et al. miRNA-95 mediates radioresistance in tumors by targeting the sphingolipid phosphatase SGPP1. Cancer Res. 2013, 73, 6972–6986.
- Billich, A.; Bornancin, F.; Mechtcheriakova, D.; Natt, F.; Huesken, D.; Baumruker, T. Basal and induced sphingosine kinase 1 activity in A549 carcinoma cells: Function in cell survival and IL-1beta and TNF-alpha induced production of inflammatory mediators. Cell. Signal. 2005, 17, 1203–1217.
- Leroux, M.E.; Auzenne, E.; Evans, R.; Hail, N., Jr.; Spohn, W.; Ghosh, S.C.; Farquhar, D.; McDonnell, T.; Klostergaard, J. Sphingolipids and the sphingosine kinase inhibitor, SKI II, induce BCL-2-independent apoptosis in human prostatic adenocarcinoma cells. Prostate 2007, 67, 1699–1717.
- Liu, H.; Kong, Y.; Liu, Z.; Guo, X.; Yang, B.; Yin, T.; He, H.; Gou, J.; Zhang, Y.; Tang, X. Sphingomyelin-based PEGylation Cu (DDC)(2) liposomes prepared via the dual function of Cu(2+) for cancer therapy: Facilitating DDC loading and exerting synergistic antitumor effects. Int. J. Pharm. 2022, 621, 121788.
- Bidan, N.; Lores, S.; Vanhecke, A.; Nicolas, V.; Domenichini, S.; Lopez, R.; de la Fuente, M.; Mura, S. Before in vivo studies: In vitro screening of sphingomyelin nanosystems using a relevant 3D multicellular pancreatic tumor spheroid model. Int. J. Pharm. 2022, 617, 121577.
- Massiot, J.; Rosilio, V.; Ibrahim, N.; Yamamoto, A.; Nicolas, V.; Konovalov, O.; Tanaka, M.; Makky, A. Newly synthesized lipid-porphyrin conjugates: Evaluation of their self-assembling properties, their miscibility with phospholipids and their photodynamic activity in vitro. Chemistry 2018, 24, 19179–19194.
- Lim, E.B.; Haam, S.; Lee, S.W. Sphingomyelin-based liposomes with different cholesterol contents and polydopamine coating as a controlled delivery system. Colloid Surface A 2021, 618, 126447.
- Zembruski, N.C.; Nguyen, C.D.; Theile, D.; Ali, R.M.; Herzog, M.; Hofhaus, G.; Heintz, U.; Burhenne, J.; Haefeli, W.E.; Weiss, J. Liposomal sphingomyelin influences the cellular lipid profile of human lymphoblastic leukemia cells without effect on P-glycoprotein activity. Mol. Pharm. 2013, 10, 1020–1034.
- Teng, Y.; Li, J.; Hui, S. C6 ceramide potentiates curcumin-induced cell death and apoptosis in melanoma cell lines in vitro. Cancer Chemother. Pharmacol. 2010, 66, 999–1003.
- Pal, S.; Medatwal, N.; Kumar, S.; Kar, A.; Komalla, V.; Yavvari, P.S.; Mishra, D.; Rizvi, Z.A.; Nandan, S.; Malakar, D.; et al. A localized chimeric hydrogel therapy combats tumor progression through alteration of sphingolipid metabolism. ACS Cent. Sci. 2019, 5, 1648–1662.
- Medatwal, N.; Ansari, M.N.; Kumar, S.; Pal, S.; Jha, S.K.; Verma, P.; Rana, K.; Dasgupta, U.; Bajaj, A. Hydrogel-mediated delivery of celastrol and doxorubicin induces a synergistic effect on tumor regression via upregulation of ceramides. Nanoscale 2020, 12, 18463–18475.
- Zhang, X.; Kitatani, K.; Toyoshima, M.; Ishibashi, M.; Usui, T.; Minato, J.; Egiz, M.; Shigeta, S.; Fox, T.; Deering, T.; et al. Ceramide nanoliposomes as a MLKL-dependent, necroptosis-inducing, chemotherapeutic reagent in ovarian cancer. Mol. Cancer Ther. 2018, 17, 50–59.





