Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Vasile Valeriu Lupu | -- | 3471 | 2023-07-25 08:49:16 | | | |
| 2 | Camila Xu | Meta information modification | 3471 | 2023-07-25 08:54:52 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Fotea, S.; Ghiciuc, C.M.; Stefanescu, G.; Cianga, A.L.; Mihai, C.M.; Lupu, A.; Butnariu, L.I.; Starcea, I.M.; Salaru, D.L.; Mocanu, A.; et al. Diabetes Mellitus and COVID-19 in Children. Encyclopedia. Available online: https://encyclopedia.pub/entry/47206 (accessed on 27 March 2026).
Fotea S, Ghiciuc CM, Stefanescu G, Cianga AL, Mihai CM, Lupu A, et al. Diabetes Mellitus and COVID-19 in Children. Encyclopedia. Available at: https://encyclopedia.pub/entry/47206. Accessed March 27, 2026.
Fotea, Silvia, Cristina Mihaela Ghiciuc, Gabriela Stefanescu, Anca Lavinia Cianga, Cristina Maria Mihai, Ancuta Lupu, Lacramioara Ionela Butnariu, Iuliana Magdalena Starcea, Delia Lidia Salaru, Adriana Mocanu, et al. "Diabetes Mellitus and COVID-19 in Children" Encyclopedia, https://encyclopedia.pub/entry/47206 (accessed March 27, 2026).
Fotea, S., Ghiciuc, C.M., Stefanescu, G., Cianga, A.L., Mihai, C.M., Lupu, A., Butnariu, L.I., Starcea, I.M., Salaru, D.L., Mocanu, A., Chisnoiu, T., Thet, A.A., Miron, L., & Lupu, V.V. (2023, July 25). Diabetes Mellitus and COVID-19 in Children. In Encyclopedia. https://encyclopedia.pub/entry/47206
Fotea, Silvia, et al. "Diabetes Mellitus and COVID-19 in Children." Encyclopedia. Web. 25 July, 2023.
Copy Citation
Coronavirus disease 2019 (COVID-19) is a complex infectious disease caused by the SARS-CoV-2 virus, and it currently represents a worldwide public health emergency. The pediatric population is less prone to develop severe COVID-19 infection, but children presenting underlying medical conditions, such as diabetes mellitus, are thought to be at increased risk of developing more severe forms of COVID-19.
children
COVID-19
diabetes mellitus
SARS-CoV-2
1. Introduction
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was first isolated and identified, in Wuhan, China, in 2019 [1]. Many studies suggested that viruses, like enteroviruses (Coxsackie virus B), rotavirus, mumps virus and cytomegalovirus, are potential triggers of Type 1 diabetes mellitus (T1DM) in children and young adults [2]. Currently, SARS-CoV-2 infection also appears to be a potential trigger for the development of diabetes mellitus type 1 and 2 in children, which represents the most frequent chronic metabolic disorder within the pediatric population. In their study, Graff et al. [3] found that among 454 patients identified with SARS-CoV-2 infection, those with diabetes were more prone for admission (aOR, 6.6; p = 0.04), and, moreover, data also showed that diabetes mellitus and other cardiovascular comorbidities were identified as major risk factors regarding outcome and mortality in patients with COVID-19. ACE2 is a receptor expressed in various organs, including both exocrine and endocrine tissues of the pancreas. In a manner similar to SARS-CoV, the virus responsible for the 2003 pandemic, SARS-CoV-2 binds to ACE2 receptors through its spike protein. Since the emergence of the SARS-CoV-2 pandemic, diabetes has been acknowledged as a risk factor associated with increased morbidity and mortality in patients with COVID-19. Moreover, recent evidence indicated that COVID-19 may result in poorer outcomes among individuals with pre-existing diabetes, potentially leading to the development of diabetic ketoacidosis (DKA) [4].
Clinical manifestations of COVID-19 are correlated with age according to the data available in the literature [5]. Pediatric patients with no important medical history are susceptible to COVID-19 but usually have a milder course compared to adults [6]. COVID-19 can exhibit a pronounced clinical progression involving acute respiratory distress syndrome (ARDS), accompanied by a localized and systemic surge of cytokines, which can potentially lead to rapid clinical deterioration and failure of multiple organs. Compared to adults who develop respiratory symptoms that can evolve into ARDS, most children do not have respiratory diseases but can develop a life-threatening multisystem inflammatory syndrome (MIS-C). MIS-C is a multi-system inflammatory syndrome that affects individuals aged <21 years with a history of SARS-CoV-2 infection within 4–6 weeks prior to the onset of symptoms presenting with fever, laboratory evidence of inflammation and multisystem (>2) organ involvement, with cardiac and gastrointestinal being the most frequent issues and no alternative plausible diagnoses [7]. In children, MIS-C exhibits similarities to Kawasaki disease, such as febrile illness with inflammation of the blood vessels and possible subsequent consequences of coronary artery aneurysm, conjunctivitis, rash and congestion of the oropharynx [8]. Neurological manifestations, such as headache, meningism, skin hyperesthesia and altered consciousness, were also described in MIS-C [9]. Nevertheless, these findings are non-specific and are found in a vast area of other pediatric infectious diseases [10].
The defects in insulin action and/or secretion are present in diabetes mellitus, which sums up a group of chronic metabolic diseases that imply elevated blood glucose values, mentioning that insulin action and/or secretion disorder may coexist in the same patient [11]. Diabetes can be classified as type 1 diabetes (insulin-dependent diabetes mellitus) and type 2 diabetes (non-insulin-dependent).
1.1. T1DM
Type 1 diabetes mellitus (T1DM) occurs when the beta cells in Langerhans pancreatic islets are gradually destroyed by the immune system, leading to a decrease in the body’s insulin production capacity, eventually resulting in no insulin production [12]. If insulin is lacking, the production of hepatic glucose increases and the use of insulin decreases. This leads to a significant breakdown of adipose tissue, ketonemia and successive diabetic ketoacidosis that can be life threatening if not treated [13].
The diagnosis of type 1 diabetes is established based on the association between clinical manifestations, such as polydipsia, polyuria, weight loss, the laboratory findings and the autoantibody positivity, and other clinical signs can include abdominal pain, vomiting, shortness of breath and an altered general status. Diabetes can sometimes be misdiagnosed as asthma (because of Kussmaul respiration) or as an acute abdomen, but these errors are becoming rare with the help of laboratory testing [14]. In the absence of important comorbidities, T1DM and its treatment are known to have multiple complications, the two major ones being diabetic ketoacidosis and hypoglycemia.
Acute hyperglycemia leads to diabetic ketoacidosis, which is an acute life-threatening metabolic complication of diabetes with a mortality rate of 0.5 percent and with almost 40 percent of DKA cases at presentation. Cerebral edema represents the most frequent cause of death among the diabetic population. For the already known cases of diabetes, the rate of this complication is 1 to 8 percent per year. DKA is managed with immediate hospitalization, insulin replacement and rehydration [15][16].
On the other hand, hypoglycemia is a T1DM complication of insulin treatment. Its symptoms are the result of a fall in blood glucose, leading to a range of neurogenic and neuroglycopenic symptoms, including emotional instability and tremor. In critical cases, seizures and unconsciousness may be present, and there are suspicions regarding permanent cognitive sequelae caused by repeated hypoglycemic episodes, which are to be confirmed [17][18]. This condition was reported to exist in approximately 19–37 percent of children and adolescents with the tendency of reduction in the prevalence over time [19]. When severe hypoglycemia occurs, urgent treatment is required and can be effective with the administration of glucagon intravenously, intramuscularly or subcutaneously [20].
Studies describe that SARS-CoV-2 infects [21] pancreatic cells, leading to replication and alteration of the β-cell function precisely and via impairment of insulin secretion. Moreover, SARS-CoV-2 triggers autoimmunity [22], meaning that both autoantibody-positive insulin-dependent and autoantibody-negative diabetes can develop during an infection with the novel coronavirus [23].
1.2. T2DM
Type 2 diabetes mellitus (T2DM) is a complex, chronic metabolic disease that consists of hyperglycemia as a result of insulin resistance and a variable grade of impairment in insulin secretion due to β-cell dysfunction (lipotoxicity, inborn genetic defect or acquired from glucose toxicity or other mechanisms) [24].
The frequency of type 2 diabetes has increased significantly around the world over the past 20 years [25]. In the pediatric populations, T2DM accounts for less than 50% of all diabetes cases, and there are significant ethnic differences in prevalence all around the world. After adjustment for other demographic characteristics, Pham et al. demonstrated, in their study, that the probability of having type 2 diabetes was double among the Asian population, 65% more likely among Black people, and 17% more likely among people of mixed/other ethnicities [26][27]. The constantly increasing incidence of type 2 diabetes mellitus among children and adolescents is also becoming a source of concern to all those involved in the care of diabetic children [28].
Usually, the debut of T2DM occurs during puberty in high-risk obese adolescents having insulin resistance, knowing that the data regarding the prevalence of T2DM among children under 10 years of age are very limited [29]. Clinically, it presents characteristics of metabolic syndrome, such as arterial hypertension, hyperlipidemia, acanthosis nigricans, fatty liver disease and polycystic ovary disease [26]. The complications of T2DM are of great importance. Arterial hypertension and imbalance of lipids have an impact on the early development of retinopathy, nephropathy and increase the risk of future cardiovascular disease, which is a major cause of morbidity and mortality in adults with T2DM. These comorbidities occur early and progress at an alarming rate in children and adolescents, even in those with efficient metabolic control. Unfortunately, in comparison to the adult population, treatment options for children are limited. Needless to mention, an early initiation of treatment with metformin alongside lifestyle modifications and physical activity for all youth with T2D remains imperative and leads to better compliance [30][31].
In addition, measures, like social distancing, online schooling, increased intake of high-calorie foods, isolation with consequent reduction in physical exercises, leading to a worsening of preexistent obesogenic factors in a population, and health disparities with temporization of seeking medical care, may have all led towards an increased incidence of the disease as the pandemic developed [19].
During the COVID-19 pandemic, diabetic children encountered difficulties regarding dietary habits, physical activity, insulin dose adjustment and, more rarely, access to insulin supply due to the challenging lifestyle changes [32]. Moreover, a significant increase in severe ketoacidosis was discovered in diabetic pediatric cases during the same interval of time [33].
2. Diabetes Mellitus and COVID-19 in Children
2.1. T1DM and COVID-19
From the beginning of the pandemic, it was quickly discovered that pre-existing T1DM represented one of the high risk factors for developing severe COVID-19 and related complications. On the other hand, SARS-CoV-2 has been suggested as a potential inducer of new-onset pediatric T1DM [34]. Data available in the literature at this moment suggest that severe acute respiratory syndrome coronaviruses (SARS-CoV-2, for instance) can enter in islet cells via angiotensin-converting enzyme-2 (ACE-2) receptors and cause reversible β-cell damage and transient hyperglycemia. Also, the discovery of SARS-CoV-2 in pancreatic tissue samples taken from deceased individuals, along with the evidence of decreased pancreatic function in both the pediatric and adult population with COVID-19, implies that this virus might harm the β cells of the pancreas and trigger the onset of T1DM through direct infection, inflammatory response and interactions with the renin-angiotensin system (Figure 1). It was proposed that several pathways may be involved, and β cells can be infected through several transmembrane receptors beyond ACE-2, such as neuropilin 1 (NRP-1), transferrin receptor (TFRC) and FES Upstream Region (FURIN). Also, immunofluorescent studies demonstrated a stronger expression of NRP-1 in β cells compared to α cells, and inhibition of this receptor with a specific antagonist reduced the uptake of SARS-CoV-2, suggesting its important role in viral entry. Downstream effects appear to involve the activation of key signaling pathways, such as c-Jun N-terminal kinase (JNK) and p21-activated kinase (PAK), that lead to apoptosis and impaired insulin excretion. Moreover, according to Tang et al., SARS-CoV-2 can lead to a transdifferentiation of β cells into α and acinar cells via the signaling pathway, involving the phosphorylation of protein kinase R (PKR) and eukaryotic translation initiation factor 2 (eIF2), which, in turn, determines integrated stress responses and cellular conversion [35][36][37][38].
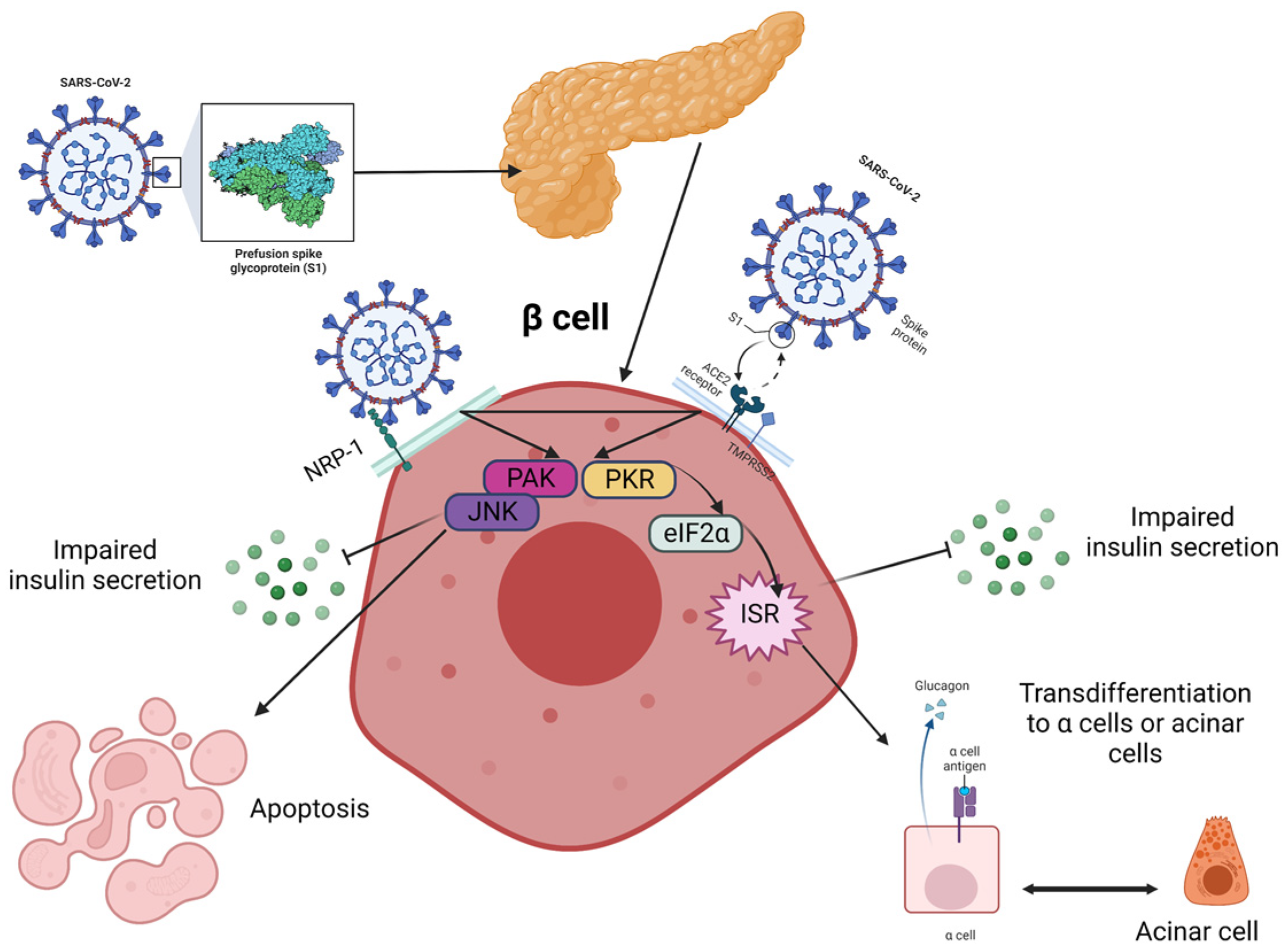
Figure 1. Proposed pathways for the fate of β cells post-COVID-19 are currently being investigated, although it remains unclear how the virus is transmitted to the pancreas and islets. Neurophilin-1 (NRP-1) has higher expression in β cells compared to angiotensin-converting enzyme 2 (ACE2) and could play a critical role in the infection. The virus stimulated β cells via p21-activated kinase (PAK) and c-Jun N-terminal kinase (JNK) and triggered the transformation of β cells into glucagon-producing α-cells or trypsin-producing acinar cells, leading to decreased insulin secretion. This transformation is facilitated through the PKR-eIF2a-mediated integrated stress response (IKR), and these two pathways may interact with each other. The eIF2 is the eukaryotic translation initiation factor 2, and PKR stands for protein kinase R. Created with Biorender.com, adapted after [38].
From a different perspective, the importance of psychological stress is also not to be neglected as it is known for decreasing insulin sensitivity and increasing insulin resistance and, therefore, may be important in the development or the onset of T1DM. The pandemic and the subsequent lockdown have had biological, economic, psychological and social consequences, and they may have increased the risk of type 1 diabetes during this period [39].
Usually, most pediatric patients with COVID-19 present mild clinical manifestations and have good prognosis. They can either have fever and mild upper respiratory symptoms or no symptoms at all. Also, they may exhibit gastrointestinal symptoms or manifestations related to DKA and may present polyuria, polydipsia, extreme fatigue, elevated temperature, drowsiness, tachypnea, deep breathing, abdominal pain, nausea, vomiting and somnolence with a clinical course that can even progress to coma and life-threatening status [40].
Diabetes and COVID-19 can have immunosuppressive effects, which lead to an increased risk for fungal disease, especially in those patients who receive corticosteroid therapy. Brothers et al. described the clinical findings of a fungal infection with Candida glabrata, a known pathogen in diabetic patients that can give rise to fulminant septic shock due to its increased resistance to antifungal agents in a child with type 1 diabetes [41][42].
A case report also described an association between new-onset diabetes and MIS-C with overlapping symptoms from both diseases with typical signs of DKA and associated bilateral conjunctivitis with limbic sparing swollen, bright red with dry mucous membranes, scattered cervical lymphadenopathy, stage 1 acanthosis nigricans and impaired mental status that were identified in the evolution of a patient. The simultaneous presence of both conditions may suggest that COVID-19 may have an impact on both beta cell function and beta cell death by accelerating its course [42].
Data published in the literature prove that there is a positive association between COVID-19 and thrombotic disorders, with evidence also suggesting that macroangiopathic processes and complement-mediated inflammation may be involved [43]. For instance, Grigore et al. described the case of cerebral venous thrombosis secondary to SARS-CoV-2 infection in a female teenager [44], whereas Alizadeh et al. presented a case of new-onset diabetes and atypical hemolytic-uremic syndrome, raising the suspicion that SARS-CoV-2 infection may represent an infectious trigger for the patient’s condition [45].
In a cross-sectional survey conducted in Italy in November 2020, Rabbone et al. [46] described how the COVID-19 pandemic might have reduced diabetes presentation and the severity of diabetic ketoacidosis by comparing data across two consecutive years (2019 and 2020). Surprisingly, compared to the same period in 2019, the results in 2020 showed a reduction of approximately 20% in new diabetes cases. Similar situations were noticed both in India and the UK [47][48]. A possible explanation would be that along with the lockdown, children were less exposed to seasonal viruses, which are known to be triggers for new-onset type 1 diabetes cases [49][50]. On the other hand, children presenting with DKA had a more aggressive form of DKA in 2020 compared to 2019 (44.3% vs. 35%) [46], and a significant rise in DKA at follow-up was observed during the first wave in many countries with high COVID-19 mortality [51]. A summary of the studies regarding the impact of the COVID-19 pandemic on the presence of DKA at the moment of diabetes mellitus diagnosis is represented in Figure 2.
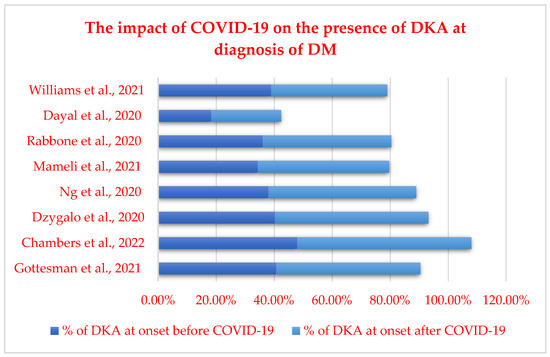
A study from Germany suggested an increase in the number of cases of diabetic ketoacidosis alongside more severe cases of DKA at diagnosis among the pediatric population during the COVID-19 pandemic, with an incidence of DKA that almost doubled in 2020 compared to the previous year [34]. Additionally, a study conducted in the UK demonstrated an increase of 80% regarding the number of cases of T1DM in children compared to 2019 and 2018, along with a high rate of severe DKA but without delayed presentation [35]. Ho et al. agreed with these facts in their study, which proved that not only the frequency of DKA at the debut of T1DM was more increased during the pandemic (68.2% vs. 45.6%; p < 0.001) but the incidence of severe cases of DKA was also more important (27.1% in 2020 vs. 13.2% in 2019; p = 0.01) [55].
Moreover, there are also results that show that children presenting HbA1c measurement was higher in those presenting during COVID-19 than one year earlier (3.0 ± 1.7 vs. 10.4 ± 3.2%; 119 ± 19 vs. 90 ± 35 mmol/mL; p = 0.008) [56]. Alonso et al. even found that higher HbA1c was significantly associated with hospitalization and considered HbA1c as a predictor for hospitalization with COVID-19 [57].
In another study, Trieu et al. showed an important increased incidence of 16.3% of new-onset T1DM in 2020 compared to the same period in 2019, which becomes even more relevant when compared to the same period in the previous two years (2018 and 2019), during which only a 6.5% decrease was observed. These results also apply to patients with T1DM who presented with DKA during 2020, representing 64.3% compared to 56.9% in 2019 and 47.1% in 2018 [58]. Another interesting aspect of the relationship between COVID-19 and children with T1DM is the seasonality, which was evaluated by Kostopoulou et al. and shows an increasing tendency from spring to winter (spring: 9.5% vs. 23.5%, autumn: 23.8% vs. 29.4%, summer: 19% vs. 11.8%, winter: 47.6% vs. 35.3%) [59].
2.2. T2DM and COVID-19
Statistically, the prevalence of T2D among the pediatric population has significantly risen in recent years [60][61]. The World Health Organization (WHO) reports that in America, Europe and the Eastern Mediterranean, roughly 50% of the population is classified as overweight or obese, with lower rates in Africa and Asia [62]. The COVID-19 pandemic and its consequent circumstances, such as the movement restrictions and the repeated lockdown measures, could have played a substantial role in increasing the number of these patients in the world [63]. The mechanisms that may explain this phenomenon are described in Figure 3.
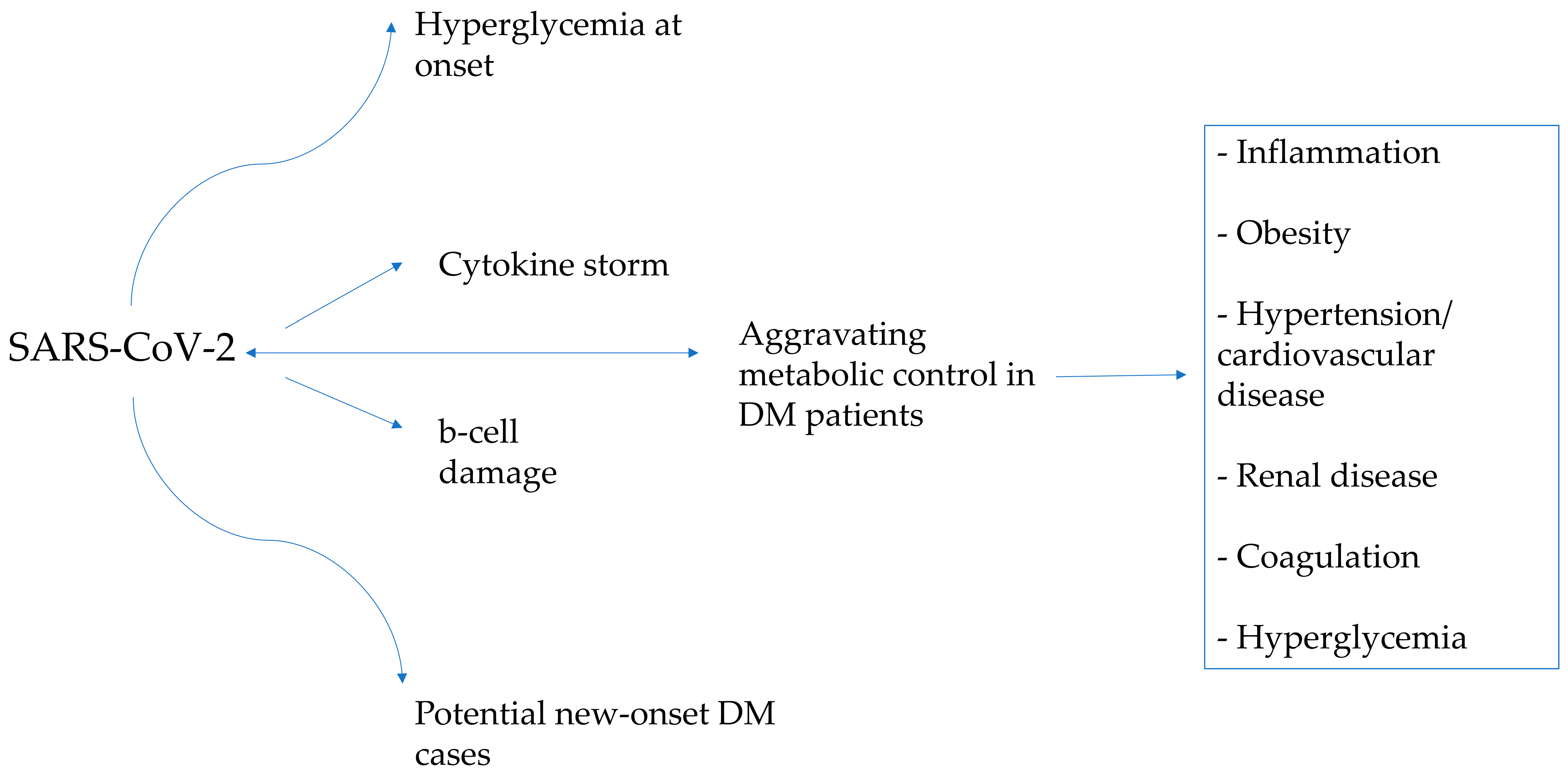
Figure 3. The interplay between diabetes and COVID-19 and how they affect each other.
The pathways leading to hyperglycemia at the initiation of T1DM/T2DM and hyperglycemia during infection-related exacerbation of metabolic control in children with diabetes may present both convergent and distinct mechanisms. During the onset of diabetes, hyperglycemia results as a consequence of pancreatic beta cell loss/dysfunction with impaired insulin production, whereas during infection-related exacerbation of metabolic control in individuals with diabetes, factors, such as stress hormones, cytokines and the consequent inflammatory response, are responsible for the compromise of the insulin action. Clearly, while some shared pathways may be implicated in hyperglycemia at the debut of diabetes and hyperglycemia during SARS-CoV-2 infection aggravating metabolic control in DM patients, the underlying mechanisms can also exhibit divergence, contingent upon the specific circumstances and individual characteristics.
Multiple studies have indicated that children and adolescents experienced an abnormal increase in weight during the COVID-19 pandemic, exceeding the expected weight gain for their age [64][65][66][67][68]. In this regard, implementing an integrated approach that promotes physical activity, lifestyle counseling and psychological support is deemed critical [69][70]. Additionally, obesity is considered a risk factor for COVID-19 infection, and patients with obesity who become infected are more susceptible to experiencing severe forms of the disease [71][72][73]. During the pandemic, there were various risk factors, such as online school activities, a more sedentary lifestyle, chronic stress and increased caloric intake, reduced availability and access to sports and physical exercise, increased social isolation and food insecurity, that led to an increased body mass index (BMI) during this period. Recently, the Centers for Disease Control showed in a data analysis that BMI doubled its values during the pandemic compared to a pre-pandemic period among the pediatric population [74]. This is explained by the increase in the number of snacks and a reduced rate of physical exercise during lockdown [75].
Anderson et al. proved, in their systematic review and meta-analysis, that in the initial year of the COVID-19 pandemic, there were consequential rises in weight gain, BMI and a higher incidence of obesity in both adults and children [76].
The same affirmation was stated by Sasidharan Pillai et al. in their retrospective study conducted on new-onset T2DM patients, who found that BMI increased during the COVID-19 pandemic compared to previous years (129% of 95th percentile vs. 141%, p = 0.02). Bond et al. discovered that not only did the BMI percentile peak after COVID-19 restrictions, but over the course of the following 21 months, it returned to pre-pandemic levels (β = −0.04 (95% CI −0.13, 0.04)) [77][78].
During the period from April to November 2020, Trieu et al. noticed a 16.3% increase in the occurrence of newly developed T1DM and a 205.3% increase in the occurrence of newly developed T2DM when compared to the same time frame in 2019. In 2019 and 2018, among children who experienced new-onset T1DM, 56.9% and 47.1%, respectively, presented with DKA, whereas in 2020, this percentage rose to 64.3%, a value which exceeded the national average. Within this period, a total of 28 children were diagnosed with both COVID-19 and diabetes. Notably, two cases exhibited significant complications due to COVID-19 and DKA, necessitating the administration of high doses of intravenous insulin over an extended duration [58]. Like many other viral infections, COVID-19 may worsen the already dysregulated glucose metabolism, leading to an increased insulin requirement [79].
Hospitalizations for new-onset type 2 diabetes in children also increased in 2020 compared with the same interval of time in 2019, according to findings presented at the American Diabetes Association Scientific Sessions.
The data obtained from a hospital in Louisiana indicated a rise in the number of admissions for new-onset T2DM in 2020 (8 cases out of 2964 hospitalizations) accompanied by DKA compared to 2019 (17 cases out of 2729). Additionally, two young individuals met the criteria for hyperosmolar hyperglycemic syndrome in 2020, whereas no cases were observed in 2019. Furthermore, increases were observed in the average levels of HbA1c, glucose and serum osmolality upon admission from 2019 to 2020 [80].
On the other hand, despite various studies indicating an increased occurrence of new-onset T2DM and DKA, Lee et al. did not observe similar findings in Korea. The data collected between 2018 and 2020 indicated that the annual incidence of DKA in T2DM patients did not differ notably, nor did the total number of newly diagnosed T2DM patients (24 in 2018, 24 in 2019 and 33 in 2020). Furthermore, they reported no notable changes in BMI and weight parameters between the pre-pandemic and pandemic intervals in patients with T2DM [81]. This contrasting result raises questions and suggests potential regional- or population-specific variations in the relationship between the pandemic and diabetes.
References
- Cardona-Hernandez, R.; Cherubini, V.; Iafusco, D.; Schiaffini, R.; Luo, X.; Maahs, D.M. Children and youth with diabetes are not at increased risk for hospitalization due to COVID-19. Pediatr. Diabetes 2021, 22, 202–206.
- Filippi, C.M.; von Herrath, M.G. Viral trigger for type 1 diabetes: Pros and cons. Diabetes 2008, 57, 2863–2871.
- Graff, K.; Smith, C.; Silveira, L.; Jung, S.; Curran-Hays, S.; Jarjour, J.; Carpenter, L.; Pickard, K.; Mattiucci, M.; Fresia, J.; et al. Risk Factors for Severe COVID-19 in Children. Pediatr. Infect. Dis. J. 2021, 40, e137–e145.
- Nielsen-Saines, K.; Li, E.; Olivera, A.M.; Martin-Blais, R.; Bulut, Y. Case Report: Insulin-Dependent Diabetes Mellitus and Diabetic Keto-Acidosis in a Child with COVID-19. Front. Pediatr. 2021, 9, 628810.
- CDC COVID-19 Response Team. Coronavirus Disease 2019 in Children-United States, February 12–April 2, 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 69, 422–426.
- Weisberg, S.P.; Connors, T.J.; Zhu, Y.; Baldwin, M.R.; Lin, W.H.; Wontakal, S.; Szabo, P.A.; Wells, S.B.; Dogra, P.; Gray, J.; et al. Distinct antibody responses to SARS-CoV-2 in children and adults across the COVID-19 clinical spectrum. Nat. Immunol. 2021, 22, 25–31.
- Vogel, T.P.; Top, K.A.; Karatzios, C.; Hilmers, D.C.; Tapia, L.I.; Moceri, P.; Giovannini-Chami, L.; Wood, N.; Chandler, R.E.; Klein, N.P.; et al. Multisystem inflammatory syndrome in children and adults (MIS-C/A): Case definition & guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine 2021, 39, 3037–3049.
- Matic, K.M. SARS-CoV-2 and Multisystem Inflammatory Syndrome in Children (MIS-C). Curr. Probl. Pediatr. Adolesc. Health Care 2021, 51, 101000.
- Mihai, C.M.; Chisnoiu, T.; Cambrea, C.S.; Frecus, C.E.; Mihai, L.; Balasa, A.L.; Stroe, A.Z.; Gogu, A.E.; Docu Axelerad, A. Neurological manifestations found in children with multisystem inflammatory syndrome. Exp. Ther. Med. 2022, 23, 261.
- Lu, X.; Zhang, L.; Du, H.; Zhang, J.; Li, Y.Y.; Qu, J.; Zhang, W.; Wang, Y.; Bao, S.; Li, Y.; et al. SARS-CoV-2 Infection in Children. N. Engl. J. Med. 2020, 382, 1663–1665.
- American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2018. Diabetes Care 2018, 41 (Suppl. 1), S13–S27.
- Roep, B.O.; Thomaidou, S.; van Tienhoven, R.; Zaldumbide, A. Type 1 diabetes mellitus as a disease of the β-cell (do not blame the immune system?). Nat. Rev. Endocrinol. 2021, 17, 150–161.
- Crawford, J.M. Liver, biliary tract, and pancreas. In Pocket Companion to Robbins Pathologic Basis of Disease; Robbins, S.L., Cotran, R.S., Kumar, V., Eds.; WB Saunders: Philadelphia, PA, USA, 1991; pp. 313–317.
- Ziegler, R.; Neu, A. Diabetes in Childhood and Adolescence. Dtsch. Arztebl. Int. 2018, 115, 146–156.
- Wolfsdorf, J.I.; Allgrove, J.; Craig, M.E.; Edge, J.; Glaser, N.; Jain, V.; Lee, W.W.; Mungai, L.N.; Rosenbloom, A.L.; Sperling, M.A.; et al. ISPAD Clinical Practice Consensus Guidelines 2014. Diabetic ketoacidosis and hyperglycemic hyperosmolar state. Pediatr. Diabetes 2014, 15, 154–179.
- Couper, J.J.; Haller, M.J.; Greenbaum, C.J.; Ziegler, A.G.; Wherrett, D.K.; Knip, M.; Craig, M.E. ISPAD Clinical Practice Consensus Guidelines 2018: Stages of type 1 diabetes in children and adolescents. Pediatr. Diabetes 2018, 19 (Suppl. 27), 20–27.
- Couch, R.; Jetha, M.; Dryden, D.M.; Hooton, N.; Liang, Y.; Durec, T.; Sumamo, E.; Spooner, C.; Milne, A.; O’Gorman, K.; et al. Diabetes Education for Children with Type 1 Diabetes Mellitus and Their Families; (Evidence Reports/Technology Assessments, No. 166.) 1, Introduction; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2008.
- UK Hypoglycaemia Study Group. Risk of hypoglycaemia in types 1 and 2 diabetes: Effects of treatment modalities and their duration. Diabetologia 2007, 50, 1140–1147.
- Rundle, A.G.; Park, Y.; Herbstman, J.B.; Kinsey, E.W.; Wang, Y.C. COVID-19 related school closings and risk of weight gain among children. Obesity 2020, 28, 1008.
- Urakami, T. Severe Hypoglycemia: Is It Still a Threat for Children and Adolescents with Type 1 Diabetes? Front. Endocrinol. 2020, 11, 609.
- Mine, K.; Nagafuchi, S.; Mori, H.; Takahashi, H.; Anzai, K. SARS-CoV-2 Infection and Pancreatic β Cell Failure. Biology 2022, 11, 22.
- Vlad, A.; Serban, V.; Timar, R.; Sima, A.; Botea, V.; Albai, O.; Timar, B.; Vlad, M. Increased Incidence of Type 1 Diabetes during the COVID-19 Pandemic in Romanian Children. Medicina 2021, 57, 973.
- Hollstein, T.; Schulte, D.M.; Schulz, J.; Glück, A.; Ziegler, A.G.; Bonifacio, E.; Wendorff, M.; Franke, A.; Schreiber, S.; Bornstein, S.R.; et al. Autoantibody-negative insulin-dependent diabetes mellitus after SARS-CoV-2 infection: A case report. Nat. Metab. 2020, 2, 1021–1024.
- Galicia-Garcia, U.; Benito-Vicente, A.; Jebari, S.; Larrea-Sebal, A.; Siddiqi, H.; Uribe, K.B.; Ostolaza, H.; Martín, C. Pathophysiology of Type 2 Diabetes Mellitus. Int. J. Mol. Sci. 2020, 21, 6275.
- Reinehr, T. Type 2 diabetes mellitus in children and adolescents. World J. Diabetes 2013, 4, 270–281.
- Mayer-Davis, E.J.; Kahkoska, A.R.; Jefferies, C.; Dabelea, D.; Balde, N.; Gong, C.X.; Aschner, P.; Craig, M.E. ISPAD Clinical Practice Consensus Guidelines 2018: Definition, epidemiology, and classification of diabetes in children and adolescents. Pediatr. Diabetes 2018, 19 (Suppl. 27), 7–19.
- Pham, T.M.; Carpenter, J.R.; Morris, T.P.; Sharma, M.; Petersen, I. Ethnic Differences in the Prevalence of Type 2 Diabetes Diagnoses in the UK: Cross-Sectional Analysis of the Health Improvement Network Primary Care Database. Clin. Epidemiol. 2019, 11, 1081–1088.
- Tamborlane, W.V.; Haymond, M.W.; Dunger, D.; Shankar, R.; Gubitosi-Klug, R.; Bethin, K.; Karres, J.; Tomasi, P.; Libman, I.; Hale, P.H.; et al. Expanding treatment options for youth with type 2 diabetes: Current problems and proposed solutions: A white paper from the NICHD diabetes working group. Diabetes Care 2016, 393, 323–329.
- Hutchins, J.; Barajas, R.A.; Hale, D.; Escaname, E.; Lynch, J. Type 2 diabetes in a 5-year-old and single center experience of type 2 diabetes in youth under 10. Pediatr. Diabetes 2017, 18, 674–677.
- Nambam, B.; Silverstein, J.; Cheng, P.; Ruedy, K.J.; Beck, R.W.; Paul Wadwa, R.; Klingensmith, G.; Willi, S.M.; Wood, J.R.; Bacha, F.; et al. A cross-sectional view of the current state of treatment of youth with type 2 diabetes in the USA: Enrollment data from the Pediatric Diabetes Consortium Type 2 Diabetes Registry: PDC Type 2 Diabetes Registry. Pediatr. Diabetes 2017, 18, 222–229.
- Scaramuzza, A.E.; Rabbone, I.; Maffeis, C.; Schiaffini, R.; Diabetes Study Group of the Italian Society for Pediatric Endocrinology, Diabetes. Seasonal flu and COVID-19 recommendations for children, adolescents and young adults with diabetes. Diabet. Med. 2021, 38, e14427.
- Güemes, M.; Storch-de-Gracia, P.; Enriquez, S.V.; Martín-Rivada, Á.; Brabin, A.G.; Argente, J. Severity in pediatric type 1 diabetes mellitus debut during the COVID-19 pandemic. J. Pediatr. Endocrinol. Metab. 2020, 33, 1601–1603.
- Kamrath, C.; Mönkemöller, K.; Biester, T.; Rohrer, T.R.; Warncke, K.; Hammersen, J.; Holl, R.W. Ketoacidosis in Children and Adolescents with Newly Diagnosed Type 1 Diabetes During the COVID-19 Pandemic in Germany. JAMA 2020, 324, 801–804.
- Unsworth, R.; Wallace, S.; Oliver, N.S.; Yeung, S.; Kshirsagar, A.; Naidu, H.; Kwong, R.M.W.; Kumar, P.; Logan, K.M. New-onset type 1 diabetes in children during COVID-19: Multicenter regional findings in the U.K. Diabetes Care 2020, 43, e170–e171.
- Tang, X.; Uhl, S.; Zhang, T.; Xue, D.; Li, B.; Vandana, J.J.; Acklin, J.A.; Bonnycastle, L.L.; Narisu, N.; Erdos, M.R.; et al. SARS-CoV-2 infection induces beta cell transdifferentiation. Cell Metab. 2021, 33, 1577–1591.e7.
- Hamming, I.; Timens, W.; Bulthuis, M.L.; Lely, A.T.; Navis, G.V.; van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. A J. Pathol. Soc. Great Br. Irel. 2004, 203, 631–637.
- Wu, C.T.; Lidsky, P.V.; Xiao, Y.; Lee, I.T.; Cheng, R.; Nakayama, T.; Jiang, S.; Demeter, J.; Bevacqua, R.J.; Chang, C.A.; et al. SARS-CoV-2 infects human pancreatic β cells and elicits β cell impairment. Cell Metab. 2021, 33, 1565–1576.e5.
- Shirakawa, J. Pancreatic β-cell fate in subjects with COVID-19. J. Diabetes Investig. 2021, 12, 2126–2128.
- Tittel, S.R.; Rosenbauer, J.; Kamrath, C.; Ziegler, J.; Reschke, F.; Hammersen, J.; Mönkemöller, K.; Pappa, A.; Kapellen, T.; Holl, R.W. Did the COVID-19 Lockdown Affect the Incidence of Pediatric Type 1 Diabetes in Germany? Diabetes Care 2020, 43, e172–e173.
- Vasconez, W.A.; Bustamante Escobar, C.L.; Agarwal, N.; Solano, J.P.; Sanchez, J.E. Severe Diabetic Ketoacidosis in a Child with Type-1 Diabetes, Asthma, and COVID-19. J. Pediatr. Intensive Care 2021, 10, 232–234.
- Ordooei, M.; Behniafard, N.; Soheilipour, F.; Akbarian, E. New onset of diabetes in a child infected with COVID-19: A case report. J. Diabetes Metab. Disord. 2021, 20, 2129–2132.
- Brothers, E.M.; Lidsky, K.; Simmons, J.; Nakagawa, T. A Child With COVID-19, Type 1 Diabetes, and Candida glabrata: A Case Report and Literature Review. Clin. Pediatr. 2021, 60, 554–558.
- Naguib, M.N.; Raymond, J.K.; Vidmar, A.P. New onset diabetes with diabetic ketoacidosis in a child with multisystem inflammatory syndrome due to COVID-19. J. Pediatr. Endocrinol. Metab. 2020, 34, 147–150.
- Grigore, I.; Miron, I.; Gavrilovici, C.; Lupu, V.V.; Antal, D.C.; Schreiner, T.G.; Prazaru, C.; Lupu, A.; Dragan, F.; Grigore, E. SARS-CoV-2 Possible Etiology of Cerebral Venous Thrombosis in a Teenager: Case Report and Review of Literature. Viruses 2023, 15, 405.
- Alizadeh, F.; O’Halloran, A.; Alghamdi, A.; Chen, C.; Trissal, M.; Traum, A.; DeCourcey, D. Toddler with New Onset Diabetes and Atypical Hemolytic-Uremic Syndrome in the Setting of COVID-19. Pediatrics 2021, 147, e2020016774.
- Rabbone, I.; Schiaffini, R.; Cherubini, V.; Maffeis, C.; Scaramuzza, A.; Diabetes Study Group of the Italian Society for Pediatric Endocrinology and Diabetes. Has COVID-19 Delayed the Diagnosis and Worsened the Presentation of Type 1 Diabetes in Children? Diabetes Care 2020, 43, 2870–2872.
- Dayal, D.; Gupta, S.; Raithatha, D.; Jayashree, M. Missing during COVID-19 lockdown: Children with onset of type 1 diabetes. Acta Paediatr. 2020, 109, 2144–2146.
- Williams, G.; McLean, R.; Liu, J.F.; Ritzmann, T.A.; Dandapani, M.; Shanmugavadivel, D.; Sachdev, P.; Brougham, M.; Mitchell, R.T.; Conway, N.T.; et al. Multicentre service evaluation of presentation of newly diagnosed cancers and type 1 diabetes in children in the UK during the COVID-19 pandemic. BMJ Paediatr. Open 2021, 5, e001078.
- Zhu, N.; Zhang, D.; Wang, W.; Li, X.; Yang, B.; Song, J.; Zhao, X.; Huang, B.; Shi, W.; Lu, R.; et al. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020, 382, 727–733.
- Gottesman, B.L.; Yu, J.; Tanaka, C.; Longhurst, C.A.; Kim, J.J. Incidence of New-Onset Type 1 Diabetes among US Children during the COVID-19 Global Pandemic. JAMA Pediatr. 2022, 176, 414–415.
- Chambers, M.A.; Mecham, C.; Arreola, E.V.; Sinha, M. Increase in the number of pediatric new-onset diabetes and diabetic ketoacidosis cases during the COVID-19 pandemic. Endocr. Pract. 2022, 28, 479–485.
- Dżygało, K.; Nowaczyk, J.; Szwilling, A.; Kowalska, A. Increased frequency of severe diabetic ketoacidosis at type 1 diabetes onset among children during COVID-19 pandemic lockdown: An observational cohort study. Pediatr. Endocrinol. Diabetes Metab. 2020, 26, 167–175.
- Ng, S.M.; Woodger, K.; Regan, F.; Soni, A.; Wright, N.; Agwu, J.C.; Williams, E.; Timmis, A.; Kershaw, M.; Moudiotis, C.; et al. Presentation of newly diagnosed type 1 diabetes in children and young people during COVID-19: A national UK survey. BMJ Paediatr. Open 2020, 4, e000884.
- Mameli, C.; Scaramuzza, A.; Macedoni, M.; Marano, G.; Frontino, G.; Luconi, E.; Pelliccia, C.; Felappi, B.; Guerraggio, L.P.; Spiri, D.; et al. Type 1 diabetes onset in Lombardy region, Italy, during the COVID-19 pandemic: The doublewave occurrence. EClinicalMedicine 2021, 39, 101067.
- Ho, J.; Rosolowsky, E.; Pacaud, D.; Huang, C.; Lemay, J.A.; Brockman, N.; Rath, M.; Doulla, M. Diabetic ketoacidosis at type 1 diabetes diagnosis in children during the COVID-19 pandemic. Pediatr. Diabetes 2021, 22, 552–557.
- McGlacken-Byrne, S.M.; Drew, S.E.V.; Turner, K.; Peters, C.; Amin, R. The SARS-CoV-2 pandemic is associated with increased severity of presentation of childhood onset type 1 diabetes mellitus: A multi-centre study of the first COVID-19 wave. Diabet. Med. 2021, 38, e14640.
- Alonso, G.T.; Ebekozien, O.; Gallagher, M.P.; Rompicherla, S.; Lyons, S.K.; Choudhary, A.; Majidi, S.; Pinnaro, C.T.; Balachandar, S.; Gangat, M.; et al. Diabetic ketoacidosis drives COVID-19 related hospitalizations in children with type 1 diabetes. J. Diabetes 2021, 13, 681–687.
- Trieu, C.; Sunil, B.; Ashraf, A.P.; Cooper, J.; Yarbrough, A.; Pinninti, S.; Boppana, S. SARS-CoV-2 infection in hospitalized children with type 1 and type 2 diabetes. J. Clin. Transl. Endocrinol. 2021, 26, 100271.
- Kostopoulou, E.; Eliopoulou, M.I.; Rojas Gil, A.P.; Chrysis, D. Impact of COVID-19 on new-onset type 1 diabetes mellitus—A one-year prospective study. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 5928–5935.
- Pulungan, A.B.; Afifa, I.T.; Annisa, D. Type 2 diabetes mellitus in children and adolescent: An Indonesian perspective. Ann. Pediatr. Endocrinol. Metab. 2018, 23, 119.
- Prosperi, S.; Chiarelli, F. COVID-19 and diabetes in children. Ann. Pediatr. Endocrinol. Metab. 2022, 27, 157–168.
- Boutari, C.; Mantzoros, C.S. A 2022 update on the epidemiology of obesity and a call to action: As its twin COVID-19 pandemic appears to be receding, the obesity and dysmetabolism pandemic continues to rage on. Metabolism 2022, 133, 155217.
- Adams, E.L.; Caccavale, L.J.; Smith, D.; Bean, M.K. Food Insecurity, the Home Food Environment, and Parent Feeding Practices in the Era of COVID-19. Obesity 2020, 28, 2056–2063.
- Dutta, M. COVID-19 and Impact of School Closures on the Children of the United States; a Point of View with an Empirical Analysis. SSRN J. 2020, 3596096.
- Fernandez-Rio, J.; Cecchini, J.A.; Mendez-Gimenez, A.; Carriedo, A. Weight changes during the COVID-19 home confinement. Effects on psychosocial variables. Obes. Res. Clin. Pract. 2020, 14, 383–385.
- Sidor, A.; Rzymski, P. Dietary Choices and Habits during COVID-19 Lockdown: Experience from Poland. Nutrients 2020, 12, 1657.
- Vandoni, M.; Codella, R.; Pippi, R.; Carnevale Pellino, V.; Lovecchio, N.; Marin, L.; Silvestri, D.; Gatti, A.; Magenes, V.C.; Regalbuto, C.; et al. Combatting Sedentary Behaviors by Delivering Remote Physical Exercise in Children and Adolescents with Obesity in the COVID-19 Era: A Narrative Review. Nutrients 2021, 13, 4459.
- Al Heialy, S.; Hachim, M.Y.; Senok, A.; Gaudet, M.; Abou Tayoun, A.; Hamoudi, R.; Alsheikh-Ali, A.; Hamid, Q. Regulation of Angiotensin- Converting Enzyme 2 in Obesity: Implications for COVID-19. Front. Physiol. 2020, 11, 555039.
- Dicker, D.; Bettini, S.; Farpour-Lambert, N.; Frühbeck, G.; Golan, R.; Goossens, G.; Halford, J.; O’Malley, G.; Mullerova, D.; Ramos Salas, X.; et al. Obesity and COVID-19: The Two Sides of the Coin. Obes. Facts 2020, 13, 430–438.
- Nogueira-de-Almeida, C.A.; Del Ciampo, L.A.; Ferraz, I.S.; Del Ciampo, I.R.L.; Contini, A.A.; Ued, F.D.V. COVID-19 and obesity in childhood and adolescence: A clinical review. J. Pediatr. 2020, 96, 546–558.
- Tsankov, B.K.; Allaire, J.M.; Irvine, M.A.; Lopez, A.A.; Sauvé, L.J.; Vallance, B.A.; Jacobson, K. Severe COVID-19 Infection and Pediatric Comorbidities: A Systematic Review and Meta-Analysis. Int. J. Infect. Dis. 2021, 103, 246–256.
- Neshteruk, C.D.; Zizzi, A.; Suarez, L.; Erickson, E.; Kraus, W.E.; Li, J.S.; Skinner, A.C.; Story, M.; Zucker, N.; Armstrong, S.C. Weight-Related Behaviors of Children with Obesity during the COVID-19 Pandemic. Child. Obes. 2021, 17, 371–378.
- Storz, M.A. The COVID-19 pandemic: An unprecedented tragedy in the battle against childhood obesity. Clin. Exp. Pediatr. 2020, 63, 477–482.
- Lange, S.J.; Kompaniyets, L.; Freedman, D.S.; Kraus, E.M.; Porter, R.; Blanck, H.M.; Goodman, A.B. Longitudinal trends in body mass index before and during the COVID-19 pandemic among persons aged 2–19 years—United States, 2018–2020. Morb. Mortal. Wkly. Rep. 2021, 70, 1278.
- Wu, X.; Luo, S.; Zheng, X.; Ding, Y.; Wang, S.; Ling, P.; Yue, T.; Xu, W.; Yan, J.; Weng, J. Glycemic control in children and teenagers with type 1 diabetes around lockdown for COVID-19: A continuous glucose monitoring-based observational study. J Diabetes Investig. 2021, 12, 1708–1717.
- Anderson, L.N.; Yoshida-Montezuma, Y.; Dewart, N.; Jalil, E.; Khattar, J.; De Rubeis, V.; Carsley, S.; Griffith, L.E.; Mbuagbaw, L. Obesity and weight change during the COVID-19 pandemic in children and adults: A systematic review and meta-analysis. Obes. Rev. 2023, 24, e13550.
- Sasidharan Pillai, S.; Has, P.; Quintos, J.B.; Serrano Gonzalez, M.; Kasper, V.L.; Topor, L.S.; Fredette, M.E. Incidence, Severity, and Presentation of Type 2 Diabetes in Youth During the First and Second Year of the COVID-19 Pandemic. Diabetes Care 2023, 46, dc221702.
- Bond, D.M.; Seimon, R.; Schneuer, F.J.; Baur, L.A.; Craig, M.; Alexander, S.; Garnett, S.P.; Henderson, J.; Nassar, N. Impact and recovery of the COVID-19 pandemic on weight status of children and adolescents. Clin. Obes. 2023, 13, e12579.
- Rubino, F.; Amiel, S.A.; Zimmet, P.; Alberti, G.; Bornstein, S.; Eckel, R.H.; Mingrone, G.; Boehm, B.; Cooper, M.E.; Chai, Z.; et al. New-onset diabetes in COVID-19. N. Engl. J. Med. 2020, 383, 789–790.
- Hsia, D.S.; Lim, M.; Beyl, R.A.; Hasan, H.A.; Gardner, J. Initial Presentation of Children with Type 2 Diabetes during the COVID-19 Pandemic. Diabetes 2021, 70 (Suppl. 1), 153-LB.
- Lee, Y.; Kim, M.; Oh, K.; Kang, E.; Rhie, Y.J.; Lee, J.; Hong, Y.H.; Shin, Y.L.; Kim, J.H. Comparison of Initial Presentation of Pediatric Diabetes Before and During the Coronavirus Disease 2019 Pandemic Era. J. Korean Med. Sci. 2022, 37, e176.
More
Information
Subjects:
Pediatrics
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
655
Revisions:
2 times
(View History)
Update Date:
25 Jul 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





