
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Hao Yang | -- | 3043 | 2023-07-20 15:12:52 | | | |
| 2 | Lindsay Dong | Meta information modification | 3043 | 2023-07-24 03:21:26 | | |
Video Upload Options
The presence of oily wastewater poses a significant threat to both the ecological environment and public health. In order to solve this problem, the design and preparation of an efficient oil–water separation membrane is very important. Metal–organic frameworks (MOFs) are currently a promising material for oil–water separation due to their tunable wettability, adjustable pore size and also low density, high porosity, and high surface area. Therefore, MOFs-based membranes show great potential in the field of oil–water separation.
1. Introduction
2. The Mechanism of Oil–Water Separation
2.1. Basic Theory of Wettability
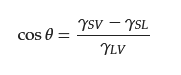
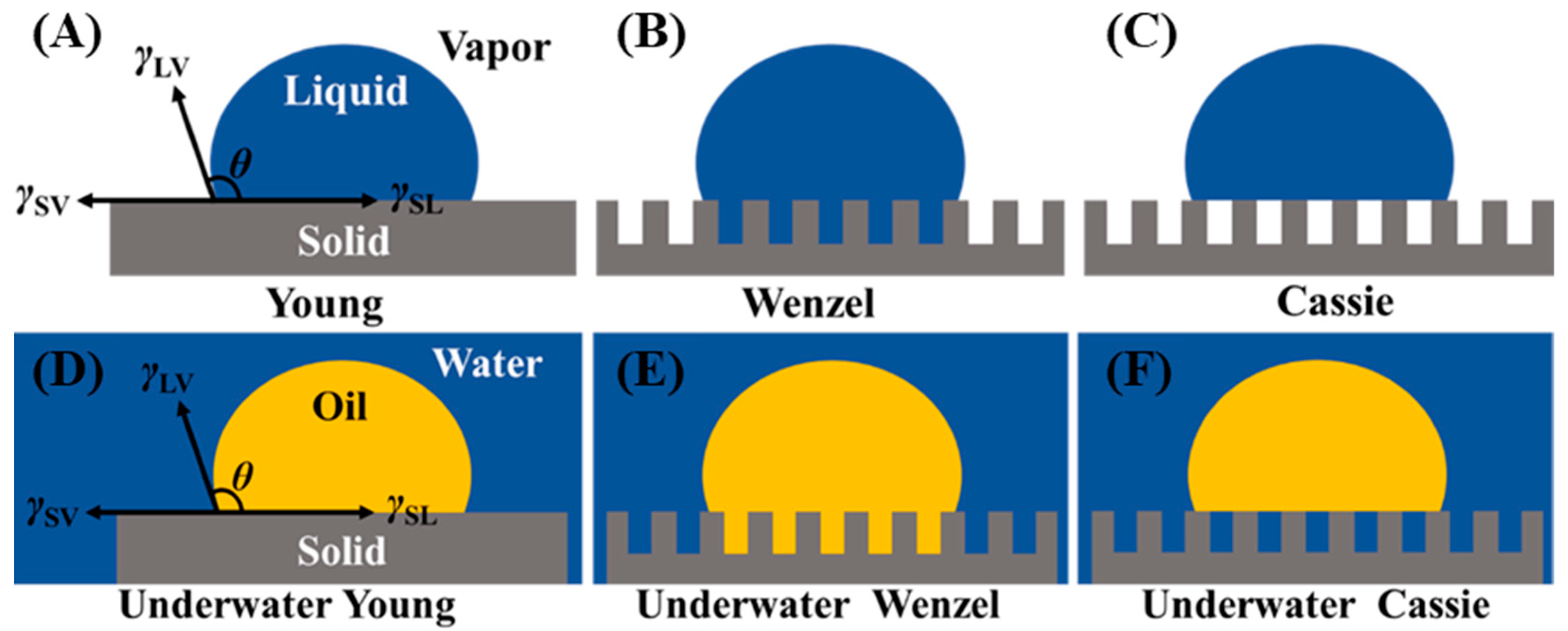
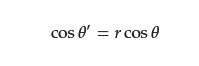
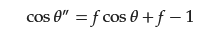
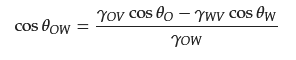
2.2. Separation of Free Oil–Water Mixture
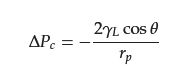
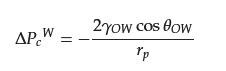
2.3. Separation of Oil–Water Emulsion
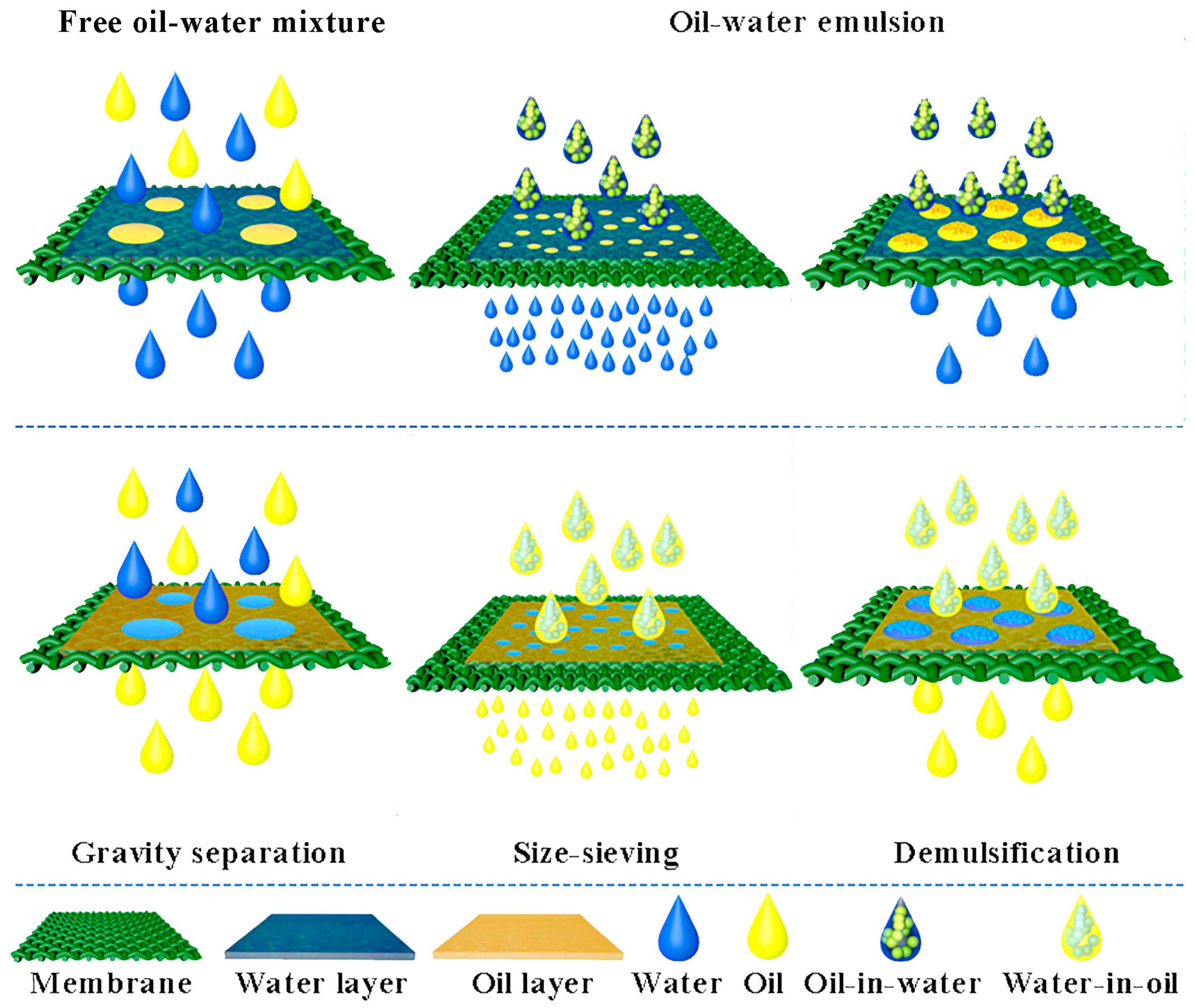
3. Methods to Prepare MOFs-Based Materials for Oil–Water Separation
3.1. In Situ Growth Method
3.1.1. Substrate Pre-Modification Method
3.1.2. Precursor Sacrifice Method
3.1.3. Crystal Seed Growth
3.2. Deposition Method
3.2.1. Direct Deposition
3.2.2. Electrochemical Deposition
3.2.3. Layer-by-Layer Self-Assembly
3.2.4. Filtration Deposition
3.2.5. Spin-Coating
3.3. Blending Membrane Method
3.3.1. Electrospinning
3.3.2. Phase Inversion
4. The Classification of MOFs Used for Oil–Water Separation
4.1. ZIF Series
4.2. UiO Series
4.3. MIL Series
4.4. Other MOFs
5. Conclusions
Most studies using MOF-based oil–water separation membranes are somewhat flawed, and the following efforts are still needed to solve the environmental problems of wastewater: (1) The long-term performance of MOFs-based membranes has been neglected, especially in continuous oil–water separation processes. Researchers can therefore focus on ways to improve the long-term durability of the membranes; (2) Most MOFs materials are unstable in wet environments due to weak coordination between their own metals and organic ligands. How to improve the stability of MOFs without affecting their structural properties remains to be investigated. (3) The specific wastewater environment is very complex and may contain organic molecules, heavy metals, microorganisms, etc. MOFs-based membranes need to be more stable and fouling resistant in the complex components. Based on this, there is a need for researchers to create materials with better stability and self-cleaning properties. (4) MOFs-based membranes should be used in a wider range of applications, and they must also be able to treat actual wastewater, from municipal wastewater to industrial wastewater, broadening their practical applications. (5) In addition, there is still a lack of longitudinal comparisons of data on the separation efficiency, mechanical and chemical stability, and pollution resistance of MOF-based membranes based on the separation of complex oil–water mixtures.
References
- Beyer, J.; Trannum, H.C.; Bakke, T.; Hodson, P.V.; Collier, T.K. Environmental effects of the deepwater horizon oil spill: A review. Mar. Pollut. Bull. 2016, 110, 28–51.
- Adetunji, A.I.; Olaniran, A.O. Treatment of industrial oily wastewater by advanced technologies: A review. Appl. Water Sci. 2021, 11, 98.
- Monteiro, C.B.; Oleinik, P.H.; Leal, T.F.; Marques, W.C.; Nicolodi, J.L.; Lopes, B. Integrated environmental vulnerability to oil spills in sensitive areas. Environ. Pollut. 2020, 267, 115238.
- Murawski, S.A.; Kilborn, J.P.; Bejarano, A.C.; Chagaris, D.; Donaldson, D.; Hernandez, F.J.; MacDonald, T.C.; Newton, C.; Peebles, E.; Robinson, K.L. A synthesis of deepwater horizon impacts on coastal and nearshore living marine resources. Front. Mar. Sci. 2021, 7, 594862.
- Owusu, B.A.; Lim, A.; Intawong, C.; Rheanpumikankit, S.; Suksri, S.; Ingviya, T. Haematological, renal, and hepatic function changes among Rayong oil spill clean-up workers: A longitudinal study. Int. Arch. Occup. Environ. Health 2022, 95, 1481–1489.
- Ingviya, T.; Intawong, C.; Abubaker, S.; Strickland, P.T. Exposure assessment of rayong oil spill cleanup workers. Expo. Health 2020, 12, 617–628.
- Padikkal, S.; Sumam, K.S.; Sajikumar, N. Sustainability indicators of water sharing compacts. Environ. Dev. Sustain. 2018, 20, 2027–2042.
- Zhang, W.F.; Liu, N.; Cao, Y.Z.; Lin, X.; Liu, Y.N.; Feng, L. Superwetting porous materials for wastewater treatment: From immiscible oil/water mixture to emulsion separation. Adv. Mater. Interfaces 2017, 4, 1700029.
- Guha, I.F.; Varanasi, K.K. Separating nanoscale emulsions: Progress and challenges to date. Curr. Opin. Colloid Interface Sci. 2018, 36, 110–117.
- Putatunda, S.; Bhattacharya, S.; Sen, D.; Bhattacharjee, C. A review on the application of different treatment processes for emulsified oily wastewater. Int. J. Environ. Sci. Technol. 2019, 16, 2525–2536.
- Abuhasel, K.; Kchaou, M.; Alquraish, M.; Munusamy, Y.; Jeng, Y.T. Oily wastewater treatment: Overview of conventional and modern methods, challenges, and future opportunities. Water 2021, 13, 980.
- Etkin, D.S.; Nedwed, T.J. Effectiveness of mechanical recovery for large offshore oil spills. Mar. Pollut. Bull. 2021, 163, 111848.
- Zhu, Y.Z.; Wang, D.; Jiang, L.; Jin, J. Recent progress in developing advanced membranes for emulsified oil/water separation. NPG Asia Mater. 2014, 6, e101.
- Zhang, R.; Sun, Y.H.; Guo, Z.G.; Liu, W.M. Janus membranes with asymmetric wettability applied in oil/water emulsion separations. Adv. Sustain. Syst. 2021, 5, 2000253.
- Cai, Y.H.; Shi, S.Q.; Fang, Z.; Li, J.Z. Design, development, and outlook of superwettability membranes in oil/water emulsions separation. Adv. Mater. Interfaces 2021, 8, 2100799.
- Kitao, T.; Zhang, Y.Y.; Kitagawa, S.; Wang, B.; Uemura, T. Hybridization of MOFs and polymers. Chem. Soc. Rev. 2017, 46, 3108–3133.
- Wang, C.H.; Liu, X.L.; Demir, N.K.; Chen, J.P.; Li, K. Applications of water stable metal-organic frameworks. Chem. Soc. Rev. 2016, 45, 5107–5134.
- Ryu, U.; Jee, S.; Rao, P.C.; Shin, J.; Ko, C.; Yoon, M.; Park, K.S.; Choi, K.M. Recent advances in process engineering and upcoming applications of metal-organic frameworks. Coord. Chem. Rev. 2021, 426, 213544.
- Venkatesan, N.; Yuvaraj, P.; Fathima, N.N. Fabrication of non-fluorinated superhydrophobic and flame retardant porous material for efficient oil/water separation. Mater. Chem. Phys. 2022, 286, 126190.
- Borazjani, A.R.; Akhlaghi, B.; Abbasi, M.; Osfouri, S. Investigation of petroleum products dehydration using natural zeolite and activated carbon. Pet. Sci. Technol. 2023, 1–20.
- Chen, X.P.; Li, Y.M.; Yang, Y.S.; Zhang, D.; Guan, Y.H.; Bao, M.T.; Wang, Z.N. A super-hydrophobic and antibiofouling membrane constructed from carbon sphere-welded MnO2 nanowires for ultra-fast separation of emulsion. J. Membr. Sci. 2022, 653, 120514.
- Rego, R.M.; Kuriya, G.; Kurkuri, M.D.; Kigga, M. MOF based engineered materials in water remediation: Recent trends. J. Hazard. Mater. 2021, 403, 123605.
- Mukherjee, S.; Sharma, S.; Ghosh, S.K. Hydrophobic metal-organic frameworks: Potential toward emerging applications. APL Mater. 2019, 7, 050701.
- Bhuyan, A.; Ahmaruzzaman, M. Metal-organic frameworks: A new generation potential material for aqueous environmental remediation. Inorg. Chem. Commun. 2022, 140, 109436.
- Beydaghdari, M.; Saboor, F.H.; Babapoor, A.; Karve, V.V.; Asgari, M. Recent Advances in MOF-Based Adsorbents for Dye Removal from the Aquatic Environment. Energies 2022, 15, 34.
- Zheng, M.Y.; Chen, J.Y.; Zhang, L.; Cheng, Y.; Lu, C.Y.; Liu, Y.F.; Singh, A.; Trivedi, M.; Kumar, A.; Liu, J.Q. Metal organic frameworks as efficient adsorbents for drugs from wastewater. Mater. Today Commun. 2022, 31, 103514.
- Dong, X.Y.; Li, Y.Y.; Li, D.Q.C.; Liao, D.H.; Qin, T.R.; Prakash, O.; Kumar, A.; Liu, J.Q. A new 3D 8-connected Cd(II) MOF as a potent photocatalyst for oxytetracycline antibiotic degradation. CrystEngComm 2022, 24, 6933–6943.
- Li, L.T.; Zou, J.F.; Han, Y.T.; Liao, Z.H.; Lu, P.F.; Nezamzadeh-Ejhieh, A.; Liu, J.Q.; Peng, Y.Q. Recent advances in Al(iii)/In(iii)-based MOFs for the detection of pollutants. N. J. Chem. 2022, 46, 19577–19592.
- Ke, F.; Pan, A.; Liu, J.Q.; Liu, X.X.; Yuan, T.; Zhang, C.Y.; Fu, G.N.; Peng, C.Y.; Zhu, J.F.; Wan, X.C. Hierarchical camellia-like metal-organic frameworks via a bimetal competitive coordination combined with alkaline-assisted strategy for boosting selective fluoride removal from brick tea. J. Colloid Interface Sci. 2023, 642, 61–68.
- Ge, B.; Yang, H.; Xu, X.C.; Ren, G.N.; Zhao, X.C.; Pu, X.P.; Li, W.Z. Facile synthesis of superhydrophobic ZIF-8/bismuth oxybromide photocatalyst aerogel for oil/water separation and hazardous pollutant degradation. Appl. Nanosci. 2020, 10, 1409–1419.
- Ahmad, N.; Samavati, A.; Nordin, N.; Jaafar, J.; Ismail, A.F.; Malek, N. Enhanced performance and antibacterial properties of amine-functionalized ZIF-8-decorated GO for ultrafiltration membrane. Sep. Purif. Technol. 2020, 239, 116554.
- Miao, W.N.; Tian, Y.; Jiang, L. Bioinspired superspreading surface: From essential mechanism to application. Acc. Chem. Res. 2022, 55, 1467–1479.
- Zhao, W.J. Bio-inspired superwettable materials: An interview with Lei Jiang. Natl. Sci. Rev. 2017, 4, 781–784.
- Gore, P.M.; Naebe, M.; Wang, X.G.; Kandasubramanian, B. Nano-fluoro dispersion functionalized superhydrophobic degummed & waste silk fabric for sustained recovery of petroleum oils & organic solvents from wastewater. J. Hazard. Mater. 2022, 426, 127822.
- Wang, Y.; Zhao, W.N.; Han, M.; Guan, L.; Han, L.; Hemraj, A.; Tam, K.C. Sustainable superhydrophobic surface with tunable nanoscale hydrophilicity for water harvesting applications. Angew. Chem. Int. Ed. 2022, 61, e202115238.
- Zheng, L.Z.; Li, H.Q.; Lai, X.J.; Huang, W.; Lin, Z.Y.; Zeng, X.R. Superwettable Janus nylon membrane for multifunctional emulsion separation. J. Membr. Sci. 2022, 642, 119995.
- Yang, J.; Li, H.N.; Chen, Z.X.; He, A.; Zhong, Q.Z.; Xu, Z.K. Janus membranes with controllable asymmetric configurations for highly efficient separation of oil-in-water emulsions. J. Mater. Chem. A 2019, 7, 7907–7917.
- Li, L.; Xu, Z.Z.; Sun, W.; Chen, J.; Dai, C.L.; Yan, B.; Zeng, H.B. Bio-inspired membrane with adaptable wettability for smart oil/water separation. J. Membr. Sci. 2020, 598, 117661.
- Song, P.; Lu, Q.Y. Porous clusters of metal-organic framework coated stainless steel mesh for highly efficient oil/water separation. Sep. Purif. Technol. 2020, 238, 116454.
- Gao, X.; Ma, Q.; Jin, Z.W.; Nian, P.; Wang, Z. Switchable superlyophobic zeolitic imidazolate framework-8 film-coated stainless-steel meshes for selective oil-water emulsion separation with high flux. N. J. Chem. 2020, 44, 13534–13541.
- Yang, X.H.; Li, S.P.; Yao, Y.Q.; Zhao, J.; Zhu, Z.G.; Chai, C.P. Preparation and characterization of polypropylene non-woven fabric/ZIF-8 composite film for efficient oil/water separation. Polym. Test 2021, 100, 107263.
- Zhou, P.Z.; Cheng, J.; Yan, Y.Y.; Xu, S.P.; Zhou, C.L. Ultrafast preparation of hydrophobic ZIF-67/copper mesh via electrodeposition and hydrophobization for oil/water separation and dyes adsorption. Sep. Purif. Technol. 2021, 272, 118871.
- Gao, J.; Wei, W.; Yin, Y.; Liu, M.H.; Zheng, C.B.; Zhang, Y.F.; Deng, P.Y. Continuous ultrathin UiO-66-NH2 coatings on a polymeric substrate synthesized by a layer-by-layer method: A kind of promising membrane for oil-water separation. Nanoscale 2020, 12, 6658–6663.
- Zhu, X.M.; Yu, Z.X.; Zeng, H.J.; Feng, X.F.; Liu, Y.C.; Cao, K.Y.; Li, X.Y.; Long, R.X. Using a simple method to prepare UiO-66-NH2/chitosan composite membranes for oil-water separation. J. Appl. Polym. Sci. 2021, 138, e50765.
- Li, H.Y.; Mu, P.; Li, J.; Wang, Q.T. Inverse desert beetle-like ZIF-8/PAN composite nanofibrous membrane for highly efficient separation of oil-in-water emulsions. J. Mater. Chem. A 2021, 9, 4167–4175.





