Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Dana Antonia Tapoi | -- | 3391 | 2023-07-11 14:49:45 | | | |
| 2 | Rita Xu | Meta information modification | 3391 | 2023-07-12 03:46:40 | | | | |
| 3 | Rita Xu | Meta information modification | 3391 | 2023-07-12 03:47:21 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Dumitru, A.V.; Țăpoi, D.A.; Halcu, G.; Munteanu, O.; Dumitrascu, D.; Ceaușu, M.C.; Gheorghișan-Gălățeanu, A. CD30 in Disease. Encyclopedia. Available online: https://encyclopedia.pub/entry/46649 (accessed on 04 June 2026).
Dumitru AV, Țăpoi DA, Halcu G, Munteanu O, Dumitrascu D, Ceaușu MC, et al. CD30 in Disease. Encyclopedia. Available at: https://encyclopedia.pub/entry/46649. Accessed June 04, 2026.
Dumitru, Adrian Vasile, Dana Antonia Țăpoi, Georgian Halcu, Octavian Munteanu, David-Ioan Dumitrascu, Mihail Constantin Ceaușu, Ancuța-Augustina Gheorghișan-Gălățeanu. "CD30 in Disease" Encyclopedia, https://encyclopedia.pub/entry/46649 (accessed June 04, 2026).
Dumitru, A.V., Țăpoi, D.A., Halcu, G., Munteanu, O., Dumitrascu, D., Ceaușu, M.C., & Gheorghișan-Gălățeanu, A. (2023, July 11). CD30 in Disease. In Encyclopedia. https://encyclopedia.pub/entry/46649
Dumitru, Adrian Vasile, et al. "CD30 in Disease." Encyclopedia. Web. 11 July, 2023.
Copy Citation
CD30, also known as TNFRSF8 (tumor necrosis factor receptor superfamily member 8), is a protein receptor that is heavily glycosylated inside the Golgi apparatus, as well as a tumor marker that is found on the surface of specific cells in the body, including certain immune cells and cancer ones.
CD30
anaplastic large cell lymphoma
embryonal carcinoma
1. Introduction
CD30 (TNFRSF8) is a protein receptor located on the surface of cells and belongs to the tumor necrosis factor receptor superfamily. It can be found on in multiple cell types, including activated T cells, B cells, natural killer (NK) cells, and lymphoid cells. CD30 engages in various biological processes by interacting with its ligand CD30L (also known as CD153).
CD30 is a transmembrane protein with an approximate molecular weight of 120 kDa and is classified as a type I protein. Its extracellular domain features six cysteine-rich pseudo repeat motifs; within its cytoplasmic tail, multiple sequences exist that are capable of binding to TNF receptor-associated factors (TRAFs). These TRAF-binding sequences facilitate the activation of various signaling pathways, including nuclear factor kappa B (NF-kB) and extracellular signal-regulated kinase (ERK) pathways. Notably, overexpression of CD30 has been found to cause self-aggregation, which recruits TRAFs for recruitment and activation of NF-kB regardless of CD30L expression levels. When present naturally in individuals without pathologic conditions, CD30 expression is typically limited to activated B and T lymphocytes [1]. CD30L, commonly known as CD153, is a type II transmembrane protein belonging to the TNF superfamily 8 (TNFSF8). The human CD30L gene can be found on chromosome 9q33, and CD30L is expressed in both resting and activated B cells, activated T cells, natural killer (NK) cells, eosinophils granulocytes, monocytes, and mast cells. In addition, CD30L can be found in epithelial cells and Hassall’s corpuscles within the thymus medulla, as well as both protein and mRNA forms of CD30L that are expressed in malignant hematopoietic cells, such as chronic lymphocytic leukemia, follicular B-cell lymphoma, hairy cell leukemia, T-cell lymphoblastic lymphoma, and adult T-cell leukemia lymphoma. However, its exact role within healthy individuals remains ill-understood since no human diseases have been associated with defects in either gene [2][3].
Initial identification of CD30 as an antigen found only on Reed–Sternberg cells associated with Hodgkin’s disease led to further investigations, showing that it is also present on activated lymphoid cells and anaplastic large cell lymphomas. This antigen can now be found in tissues such as mesothelium, soft tissue tumors, decidua, and activated macrophages. Studies conducted in vitro indicate that CD30 may play a key role in lymphoid cell signaling pathways related to cell proliferation, apoptosis, and cytotoxicity. Its most prominent non-lymphoid expression can be seen in embryonal carcinoma. Studies have consistently demonstrated the strong expression of CD30 at both protein and mRNA levels in embryonal carcinoma tumors. Lower expression was also detected in seminomas and yolk sac tumors. However, a more recent investigation contradicted these results by finding no CD30 positivity among 27 mixed tumor cases that did not contain embryonal carcinoma elements. Due to an increasing rate of testicular tumors with embryonal elements, such as embryonal carcinomas, it has become more important than ever before to detect embryonal antibodies and confirm their presence within nonseminomatous germ cell tumors. Antibodies, such as anti-phospholipid, are also extremely helpful when trying to determine which tumor cells contain embryonal elements and distinguishing embryonal carcinoma from other elements within nonseminomatous germ cell tumors [4][5][6].
Even though CD30 has long been recognized as an essential marker in various lymphomas and activation molecule in B and T cells, its biological role remains unexplored. Researchers wanted to explore the effects of CD30 signaling, so they employed the C10 antibody, an agonist of human CD30, to stimulate CD30 on YT (yolk tumor) cells. Subsequent gene array analysis demonstrated the induction and suppression of approximately 750 gene products and 90 gene products by CD30 signals, showing more than two-fold changes. CD30 signaling involves both TRAF2-dependent and independent pathways. These signals have the ability to reduce the activity of effector cells by modulating gene expression associated with their cytotoxic functions in natural killer (NK) and T cells. At YT, a large granular lymphoma cell line, CD30 signals interfere with expression of mRNA-encoding cytotoxic effector molecules, such as Fas ligand, Perforin, and Granzyme B, which leads to their loss and, consequently, reduces cytotoxicity. CD30 completely inhibits Cellula myc (cmyc), a regulator of proliferation and an upstream regulator of Fas ligand. Furthermore, CD30 induces and upregulates CCR7 expression, suggesting its involvement in lymphocyte trafficking to lymph nodes. CD30 upregulates Fas (TNFRSF6), death receptor 3 (TNFRSF25), and TNF-related apoptosis-inducing ligand (TNFSF10), which indicates increased susceptibility to apoptotic signals. Conversely, upregulation of TNFR-associated factor 1 and cellular inhibitor of apoptosis 2 protects cells against specific forms of apoptosis. These studies demonstrate how CD30 signaling may hinder lymphocyte effector function and proliferation while simultaneously driving them towards lymph nodes and increasing susceptibility to specific apoptotic signals. They offer one potential mechanism behind CD30’s observed suppression of CD8 cytotoxic T lymphocyte (CTL) activity in diabetes models in vivo [5][7][8][9]. The effects of CD30 in oncogenesis are presented in Figure 1.
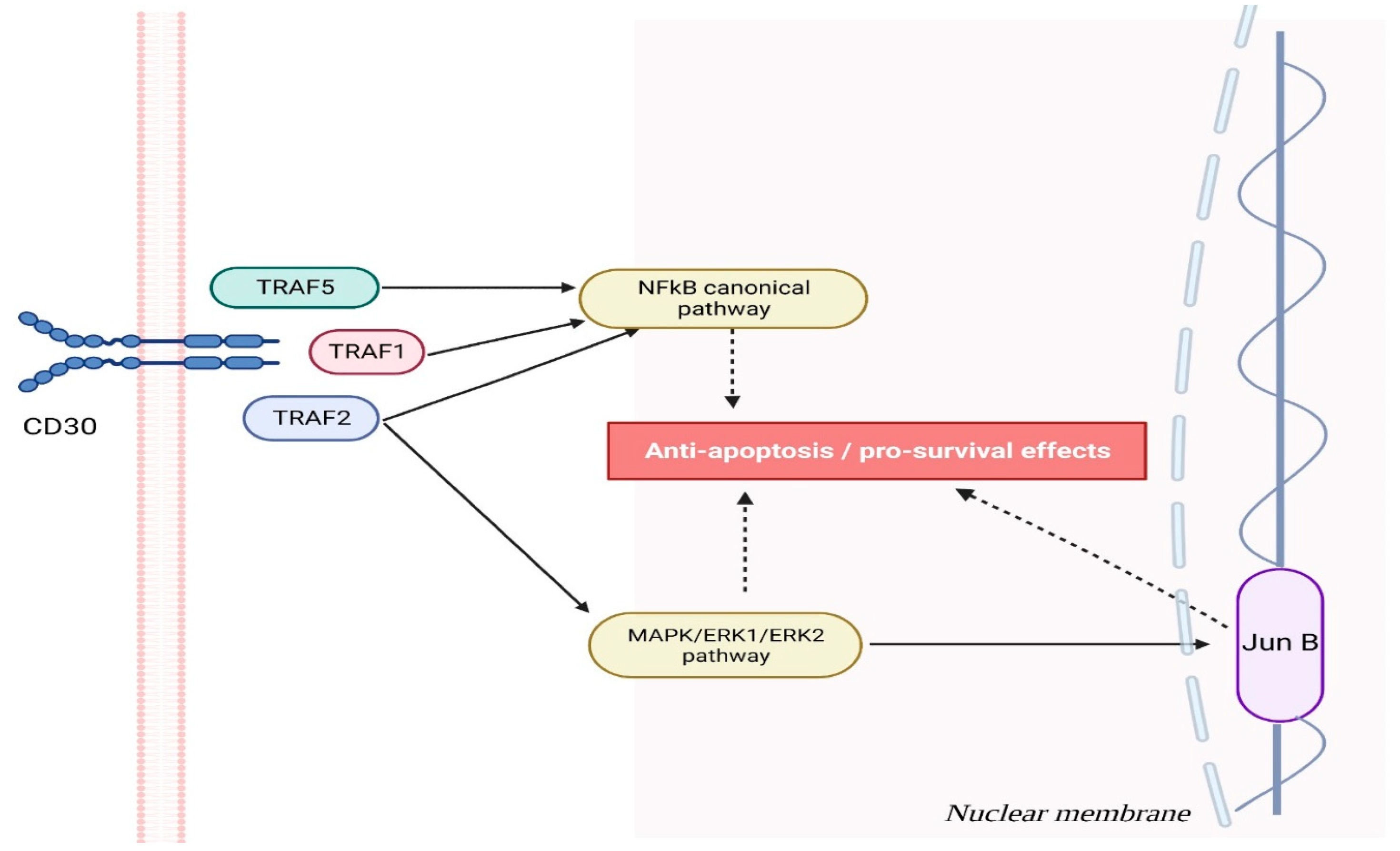
Figure 1. The CD30 molecule plays a critical role in cell survival by engaging various signaling pathways that confer an advantage to cells with higher CD30 levels. When activated, CD30 forms trimers and sends out signals through tumor necrosis factor receptor-associated proteins (TRAF), specifically TRAF2, TRAF1, and TRAF5, to stimulate nuclear factor-kappa B (NFkB) pathways and thus increase survival chances. CD30 binding initiates signaling through mitogen-activated protein kinase (MAPK) pathways, including ERK1 and ERK2, that activate mitogen-activated protein kinase kinase kinase. This provides cancerous cells with various anti-apoptotic and pro-survival benefits, with JunB acting as a nuclear transcription factor to further support survival while upregulating CD30 expression.
Understanding CD30 and its functions within various biological processes continues to advance, with research providing new insights and potential therapeutic implications. CD30 has been identified as playing an essential role in the activation and survival of lymphocytes and in modulating immune responses. Researchers are exploring CD30’s role in organizing and maintaining lymphoid tissues, as well as uncovering its significance for life-threatening conditions, such as Hodgkin lymphoma, embryonal carcinoma, anaplastic large cell lymphoma, and cutaneous T-cell lymphoma. Their investigations aim to gain more insight into its mechanisms as well as potential implications for disease management.
2. CD30 in Disease
2.1. Hodgkin Lymphoma and CD30
Hodgkin lymphoma, characterized by Reed–Sternberg cells, has long been linked with CD30 expression on their surface cells. CD30 plays an integral part in the Hodgkin lymphoma pathology by supporting Reed–Sternberg survival and proliferation; activation via signaling pathway by CD30 triggers the activation of nuclear factor-kappa B (NF-kB), an activator transcription factor responsible for gene expression which promotes survival resistance to apoptosis and ultimately contributes to sustained growth and survival of Reed–Sternberg cells over time.
Lymphomas that affect the thymus can be divided into T-cell and B-cell lineages and Hodgkin lymphomas. Lymphomas involving this organ account for approximately 25% of mediastinal tumors; about 13% are Hodgkin lymphomas (HL), while 13% are non-Hodgkin lymphomas (NHL). Only around 3% and 6%, respectively, arise as primary mediastinal malignancies. Approximately 50% or 20%, respectively, have systemic involvement; mediastinal and hilar lymph nodes are affected in cases with generalized involvement. Currently, two histological subtypes that most frequently affect localized mediastinal involvement are PMBL (primary mediastinal B-cell lymphoma) l and T-lymphoblastic lymphoma (T-Lb). Both may arise from thymic tissue. In addition, age may influence which subtype is diagnosed. Subtype incidence has been discussed extensively [10].
Below, researchers will examine some of the more prevalent mediastinal/primary thymic lymphomas and their key immunohistochemical markers, notable morphological characteristics, and diagnostic challenges. Furthermore, researchers will describe rare, tumor-like lesions of particular interest due to their complex pathogenesis, clinical features, and status as specific or uncommon biological entities, such as Castleman disease, in both mediastinum as well as thymus and IgG4-related diseases [11].
Lymphomas of the thymus may encompass numerous subtypes, from immature precursor cells (both T and B cells) to mature peripheral T and B cells. Hodgkin lymphoma of the classical type (cHL), most frequently observed in both pediatric patients and adults, is the most prevalent lymphoma. The nodular sclerosis subtype of Hodgkin lymphoma is especially prevalent here; T-cell precursor type lymphomas tend to be more predominant among young patients compared to lymphomas originating in adults; primary cHL cases that specifically target the thymus are relatively rare in both age groups [12][13].
Thymic lymphomas typically arise from B cells. Thymic B cells tend to reside mainly within the medulla or perivascular spaces and exhibit distinct phenotypic characteristics compared to other subsets of B cells, although their exact relationship remains poorly understood. Thymic B cells may contribute to B-cell thymic lymphoma or classical Hodgkin lymphoma (cHL) of the thymus, among other cancers. Due to neoplastic growth’s destructive nature, biopsies or surgical specimens of lymphoid masses may make it challenging to identify remnants and features indicative of their thymic origin. If these remnants are identified, however, they should be accurately acknowledged as such [14].
The nodular sclerosis (NS) variant of classical Hodgkin lymphomas (cHLs) in the mediastinum are the most prevalent subtype, making up 50–70% of primary mediastinal lymphomas. Thymus-NS lymphoma, in particular, affects young individuals more often than older ones and women more often than men. Under conditions of sclerosis with a polymorphous inflammatory cell population, it is beneficial to identify CD30+ cells, even though they may be sparse in fibrous backgrounds that will aid diagnosis. Reed–Sternberg cells (RS) can be identified by their large size, abundant eosinophilic cytoplasm, two or more nuclei, and an abundance of eosinophilic nucleoli. Lacunar cells (LCs) typically feature small hyperlobulated nuclei with small nucleoli and clear, retractile cytoplasm, and they often represent the NS subtype of chronic herpetic lymphoma (cHL). Furthermore, this subtype can often lead to reactive epithelial cell (EC) proliferation and cystic changes, which can easily resemble a thymoma. An extensive sampling of mediastinal cystic lesions is necessary to identify possible focal sites of chronic lymphocytic leukemia within their walls. Misdiagnosis of chronic lymphocytic leukemia of the thymus as primary mediastinal B-cell lymphoma (PMBL) can be common as both can induce sclerotic reactions and may display RS-like cells that have similar characteristics. Both disorders share B-cell origins and display similar morphologies and clinical presentations [15].
CD30 expression on Reed–Sternberg cells makes it an attractive target for diagnostic and therapeutic uses, including Hodgkin lymphoma treatment. Antibody–drug conjugates specifically targeting CD30, such as brentuximab vedotin, have demonstrated positive outcomes in treatment modalities. These novel therapeutic interventions offer significant promise in managing this disease.
As T-lymphoblastic lymphoma and B1 thymoma share many morphological similarities, it can be difficult to differentiate them using lymphoblasts alone. In addition, both tumors often exhibit frequent mitosis and extensive necrosis, which makes diagnosis even more challenging. Therefore, when dealing with necrotic tumors or small biopsies that cannot provide sufficient tissue samples for diagnosis, relying heavily on specific immunohistochemical markers that target lymphoblastic precursors like LMO2 can often help. Previous research has established that CDK6 staining is limited to T-lymphoblastic lymphoma cells, and subcapsular lymphoblasts present in normal or hyperplastic thymuses. Furthermore, T-lymphoblastic lymphomas tend to exhibit rare remnants and epithelial cell (EC) networks due to the highly destructive nature of tumor growth. In contrast, B1 thymomas may exhibit partial presence of EC networks even within necrotic tumors, which can be detected using appropriate immunohistochemical stains (e.g., cytokeratins). Also, rarely, mature, peripheral T-cell lymphomas have been reported in thymus or mediastinal lymph nodes [16][17].
Almost all the CD30 positive neoplasms are composite tumors in which the CD30 component is at least a minimal part of corresponding tumors. A composite tumor consists of different cell types or components. In the context of CD30-positive neoplasms, this means that the tumors are not solely composed of CD30-positive cells, but also contain other types of cells. For example, in Hodgkin lymphoma, the CD30 component is only a part of the overall tumor, and it may be present in minimal quantities compared to other components of the tumor [18].
Therefore, any diagnostic or predictive speculation based solely on the identification and quantification of CD30-positive cells cannot be reliable. In other words, the presence or number of CD30-positive cells alone cannot provide a definitive diagnosis or predict the behavior of the tumor. Additional morphological (structural) features and further investigations are necessary for accurate diagnosis and prediction.
In addition to conventional histological sections, CD30 positive cells can be identified on various alternative technical supports. These alternative methods allow for the detection and characterization of CD30 positive cells in different formats, expanding the diagnostic capabilities beyond traditional histology.
For example, in the diagnostic process of Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL), the cytopathological analysis of the effusions associated with a late breast implant plays a pivotal role [19]. It involves the examination of cellular components within the collected fluid sample, which is typically obtained through procedures such as fine-needle aspiration or seroma fluid analysis. These characteristics may include atypical lymphoid cells, large cell size, abnormal cell shape, and distinct cell membrane markers, such as CD30 expression.
Furthermore, in the context of fine-needle aspiration cytology (FNAC), the utilization of specific markers such as CD15, CD30, and PAX5 has proven to be valuable for the identification of Hodgkin lymphoma (HL) and non-Hodgkin large B-cell lymphoma, specifically diffuse large B-cell lymphoma (DLBCL). These markers aid in distinguishing between these subtypes, and their differential expression patterns provide insights into the diagnostic utility of FNAC [20].
2.2. Embryonal Carcinoma and CD30
Embryonal carcinoma, a malignant germ cell tumor found primarily in the testes and ovaries, is linked to the expression of CD30. CD30 serves as an invaluable diagnostic marker to detect embryonal carcinoma from other types of germ cell tumors; additionally, its signaling could play a part in embryonal carcinoma cell survival and proliferation similar to its role in other forms of cancer; therefore, more research must be conducted into how CD30 influences embryonal carcinoma cells’ behavior [21].
Immunohistochemistry plays an essential role in accurately classifying and distinguishing malignant germ cell neoplasms from other gynecological and nongynecological tumors, thus having a profound impact on their clinical management. Nuclear markers like SALL4, OCT4, NANOG, and SOX2 are often employed; SOX2 can also be found in the cytoplasm. In addition, membranous markers like CD117, D2-40 CD30 PLAP GP-3 may also be useful; Gp-3 and PLAP proteins have been observed within cells. By combining these markers with non-germ cell tumors markers, such as PLAP and SALL4, it possible to differentiate germ cell tumors from epithelial and gonadal stromal neoplasms in most cases. SALL4 has proven highly specific for germ cell tumors, showing only weak staining on some ovarian clear cell tumors and metastatic gastrointestinal adenocarcinomas, without reacting with any granulosa cell, theca cell, or ovarian stromal cell tumors [22][23].
OCT3/4, NANOG, SOX2, GP-3, and CD30 markers can help reliably differentiate yolk sac tumors and embryonal carcinomas. Positive staining for CD30, OCT3/4, NANOG, SOX2 GP-3, or CD30 tends to occur more commonly among embryonal carcinomas while negative staining occurs more commonly among yolk sac tumors, serving as reliable indicators between them both neoplasms. Furthermore, GP-3 staining is positive in yolk sac tumors but negative in embryonal carcinomas, allowing for further differentiation between them [24][25].
CD30 plays an intricate role in embryonal cancer development and requires further study to understand its specific mechanisms and therapeutic applications within embryonal carcinoma.
2.3. Anaplastic Large Cell Lymphoma (ALCL) and CD30
ALCL is an aggressive form of non-Hodgkin lymphoma characterized by large lymphoma cells expressing CD30. CD30 plays a crucial role in its development as it contributes to cell survival, proliferation, and resistance against apoptosis. Furthermore, abnormal activation of CD30 signaling in ALCL often stems from genetic translocations affecting anaplastic lymphoma kinase (ALK) gene translocations that result in hybrid ALK fusion proteins, which further intensify CD30 signaling [26]. Moreover, ALCL ALK+ are more prevalent in children and young adults, with better outcomes, whereas ALCL AKL− occur mostly in adults and elders, with the worst outcome [27].
ALK-Positive (ALK+) Anaplastic Large Cell Lymphoma (ALCL)
An ALK+ anaplastic large cell lymphoma (ALCL) is a subtype of T-cell lymphoma that features large lymphoid cells characterized by their unique, horseshoe-shaped nuclei and ample cytoplasm. ALK+ ALCL can be distinguished from other cancer types by a chromosomal translocation involving ALK, leading to the expression of both ALK protein and CD30 proteins [27]. Noteworthy is another category of ALCL called ALK-negative ALCL, which exhibits similar morphological and phenotypic features but lacks ALK rearrangement and protein expression. Therefore, differentiation diagnosis must also be conducted between ALK+ ALCL and other subtypes of T-cell or B-cell lymphomas that display anaplastic features or CD30 expression.
ALK+ ALCL typically involves both lymph nodes and extranodal sites. Extranodal involvement includes skin, bone, soft tissue, lungs, and liver, and only rarely affecting the gastrointestinal tract or central nervous system. Contrary to classic Hodgkin lymphoma, mediastinal disease is less frequently present with ALK+ ALCL. When investigating bone marrow involvement based on H&E staining, it is estimated to occur in approximately 10% of the cases. Immunohistochemical stains reveal a higher incidence (around 30%) as ALK+ ALCL may involve bone marrow less significantly. Additionally, the small cell variant of ALK+ ALCL may present as leukemic leukemia involving peripheral blood [26][27][28].
ALK+ ALCLs exhibit a diverse array of morphological characteristics. However, all cases contain cells with eccentric, horseshoe-shaped, or kidney-shaped nuclei that often exhibit an eosinophilic region near their nucleus. These cells, commonly called hallmark cells, can be found across all morphologies. While hallmark cells typically feature larger cell sizes with similar cytological features, smaller variants with the same characteristics can also be observed, which aids in accurate diagnosis. Certain cells may appear to contain nuclear inclusions; however, these are invaginations of the nuclear membrane rather than true inclusions and thus they are known as doughnut cells [29].
Morphologically, ALK+ ALCLs range from small cell neoplasms to those dominated by large cells. Multiple patterns have been identified, with the common pattern representing roughly 60% of cases. Tumor cells that display this pattern feature abundant cytoplasms that may appear clear, basophilic, or eosinophilic. Multiple nuclei arranged in an arc-shaped configuration give rise to cells resembling Reed–Sternberg cells. Nuclear chromatin tends to be finely clumped or dispersed, with multiple small, basophilic nucleoli. When composed of larger cells, nucleoli may become more prominent; however, eosinophilic or inclusion-like nucleoli are rare. When lymph node architecture remains partially preserved, tumor growth typically occurs within its sinuses, resembling metastatic tumor growth [30].
The lymphohistiocytic pattern, found in approximately 10% of cases, is characterized by tumor cells mixed with an abundance of reactive histiocytes that may obscure any malignant cells present, leading to misdiagnosis as reactive lesions. Neoplastic cells found here tend to be smaller than in the common pattern but tend to cluster around blood vessels; CD30 and ALK antibodies may help identify and highlight them more readily than others can; histiocytes may even exhibit evidence of red blood cell engulfment (erythrophagocytosis) [31].
The small-cell pattern, accounting for 5–10% of cases, typically feature small to medium-sized neoplastic cells with irregular nuclei, and pale cytoplasm and centrally located nuclei that have become cancerous over time. This is the origin of the cells’ “fried egg cells” nickname. Rarely, signet ring-like cells may also be observed. Hallmark cells, characteristic of ALCL, are always present and often cluster around blood vessels. This variant can often be misdiagnosed during the traditional examination as peripheral T-cell lymphoma not otherwise specified (NOS). Smear preparations from peripheral blood samples may reveal small, atypical cells with folded nuclei that resemble flower petals and occasional large cells with vacuolated, blue cytoplasm. The Hodgkin-like pattern, found in approximately 3% of cases, displays nodular sclerosis typical for Hodgkin lymphoma. A total of 15% of lymph node biopsies may show multiple patterns simultaneously—known as composite patterns—on one biopsy sample; it is important to remember that relapses may reveal different characteristics than the initial presentation. For example, ALCL tumors display cells characterized by monomorphic, round nuclei either predominantly or mixed with more pleomorphic nuclei, either alone or mixed together with more pleomorphic ones. Some cases may include high proportions of multinucleated giant cells or display sarcomatous features. Occasionally, the tumor may exhibit a hypocellular appearance characterized by myxoid or edematous background. Myxoid tumors can sometimes present themselves, with spindle cells mimicking sarcoma while occasional rare cases containing clusters of malignant cells within an otherwise reactive lymph node are present; capsular fibrosis combined with tumor nodules can also be seen, suggesting metastatic non-lymphoid malignancies [31][32][33][34].
References
- Younes, A.; Carbone, A. Clinicopathologic and Molecular Features of Hodgkin Lymphoma. Cancer Biol. 2003, 2, 500–507.
- Fonatsch, C.; Latza, U.; Dürkop, H.; Rieder, H.; Stein, H. Assignment of the human CD30 (Ki-1) gene to 1p36. Genomics 1992, 14, 825–826. Available online: https://www.sciencedirect.com/science/article/pii/S0888754305802034 (accessed on 15 May 2023).
- Smith, C.A.; Gruss, H.J.; Davis, T.; Anderson, D.; Farrah, T.; Baker, E.; Sutherland, G.R.; Brannan, C.I.; Copeland, N.G.; Jenkins, N.A. CD30 antigen, a marker for Hodgkin’s lymphoma, is a receptor whose ligand defines an emerging family of cytokines with homology to TNF. Cell 1993, 73, 1349–1360.
- Berney, D.M.; Shamash, J.; Pieroni, K.; Oliver, R.T.D. Loss of CD30 expression in metastatic embryonal carcinoma: The effects of chemotherapy? Histopathology 2001, 39, 382–385.
- Bowen, M.A.; Olsen, K.J.; Cheng, L.; Avila, D.; Podack, E.R. Functional effects of CD30 on a large granular lymphoma cell line, YT. Inhibition of cytotoxicity, regulation of CD28 and IL-2R, and induction of homotypic aggregation. J. Immunol. 1993, 151, 5896–5906.
- Ranjitha, V.N.; Khemani, R.; Rao, B.V.; Fonseca, D.; Murthy, S.S.; Giridhar, A.; Jayakarthik, Y.; Sharma, R.; Raju, K.V.V.N.; Rao, T.S.; et al. The core four—A panel of immunohistochemistry markers to diagnose and subtype testicular germ cell tumors. Urol Ann. 2022, 14, 21–26.
- Buchan, S.L.; Al-Shamkhani, A. Distinct Motifs in the Intracellular Domain of Human CD30 Differentially Activate Canonical and Alternative Transcription Factor NF-κB Signaling. PLoS ONE 2012, 7, e45244.
- Muta, H.; Boise, L.H.; Fang, L.; Podack, E.R. CD30 Signals Integrate Expression of Cytotoxic Effector Molecules, Lymphocyte Trafficking Signals, and Signals for Proliferation and Apoptosis1. J. Immunol. 2000, 165, 5105–5111.
- Telford, W.G.; Nam, S.Y.; Podack, E.R.; Miller, R.A. CD30-Regulated Apoptosis in Murine CD8 T Cells after Cessation of TCR Signals. Cell Immunol. 1997, 182, 125–136. Available online: https://www.sciencedirect.com/science/article/pii/S0008874997912284 (accessed on 18 May 2023).
- Chen, L.; Wang, M.; Fan, H.; Hu, F.; Liu, T. Comparison of pediatric and adult lymphomas involving the mediastinum characterized by distinctive clinicopathological and radiological features. Sci. Rep. 2017, 7, 2577.
- Simonetti, S.; Muñoz, N.P.; Vivancos, J.L.; Sitjes, L.S.; Pérez, J.C.H.; Bohorquez, N.L.; Maestre Alcacer, J.A.; de García, I.K.; Carrasco García, M.Á. IgG4-related Disease in Thymus. A Very Rare Case of Chronic Fibrosis Mimicking Sarcoidosis. Tumori J. 2017, 103 (Suppl. S1), S19–S24.
- Johnson, P.W.M., IV. Masses in the mediastinum: Primary mediastinal lymphoma and intermediate types. Hematol. Oncol. 2015, 33, 29–32.
- Maeshima, A.M.; Taniguchi, H.; Suzuki, T.; Yuda, S.; Toyoda, K.; Yamauchi, N.; Makita, S.; Fukuhara, S.; Munakata, W.; Maruyama, D.; et al. Distribution of malignant lymphomas in the anterior mediastinum: A single-institution study of 76 cases in Japan, 1997–2016. Int. J. Hematol. 2017, 106, 675–680.
- Duwe, B.V.; Sterman, D.H.; Musani, A.I. Tumors of the Mediastinum. Chest 2005, 128, 2893–2909.
- Marchevsky, A.; Marx, A.; Ströbel, P.; Suster, S.; Venuta, F.; Marino, M.; Yousem, S.; Zakowski, M. Policies and Reporting Guidelines for Small Biopsy Specimens of Mediastinal Masses. J. Thorac. Oncol. 2011, 6, S1724–S1729.
- Jevremovic, D.; Roden, A.C.; Ketterling, R.P.; Kurtin, P.J.; McPhail, E.D. LMO2 Is a Specific Marker of T-Lymphoblastic Leukemia/Lymphoma. Am. J. Clin. Pathol. 2016, 145, 180–190.
- Menter, T.; Gasser, A.; Juskevicius, D.; Dirnhofer, S.; Tzankov, A. Diagnostic Utility of the Germinal Center–associated Markers GCET1, HGAL, and LMO2 in Hematolymphoid Neoplasms. Appl. Immunohistochem. Mol. Morphol. 2015, 23, 491–498. Available online: https://journals.lww.com/appliedimmunohist/Fulltext/2015/08000/Diagnostic_Utility_of_the_Germinal.3.aspx (accessed on 19 May 2023).
- Alduaij, W.; Hilton, L.K.; Al Moosawi, M.; Ben-Neriah, S.; Meissner, B.; Boyle, M.; Mekwunye, K.; Scott, D.W.; Leitch, H.A.; Craig, J.W. Concurrent Composite Lymphomas Collectively Bearing Three Diagnostic Entities of Shared Clonal Origin. Hemasphere 2022, 6, e705.
- Julien, L.; Michel, R.P.; Auger, M. Breast implant–associated anaplastic large cell lymphoma and effusions: A review with emphasis on the role of cytopathology. Cancer Cytopathol. 2020, 128, 440–451.
- Cozzolino, I.; Vitagliano, G.; Caputo, A.; Montella, M.; Franco, R.; Ciancia, G.; Selleri, C.; Zeppa, P. CD15, CD30, and PAX5 evaluation in Hodgkin’s lymphoma on fine-needle aspiration cytology samples. Diagn Cytopathol. 2020, 48, 211–216.
- Takeda, S.; Miyoshi, S.; Ohta, M.; Minami, M.; Masaoka, A.; Matsuda, H. Primary germ cell tumors in the mediastinum. Cancer 2003, 97, 367–376.
- Low, J.J.H.; Ilancheran, A.; Ng, J.S. Malignant ovarian germ-cell tumours. Best Pract. Res. Clin. Obs. Gynaecol. 2012, 26, 347–355.
- Pectasides, D.; Pectasides, E.; Kassanos, D. Germ cell tumors of the ovary. Cancer Treat. Rev. 2008, 34, 427–441.
- Hoei-Hansen, C.E.; Kraggerud, S.M.; Abeler, V.M.; Kærn, J.; Rajpert-De Meyts, E.; Lothe, R.A. Ovarian dysgerminomas are characterised by frequent KIT mutations and abundant expression of pluripotency markers. Mol. Cancer 2007, 6, 12.
- Lifschitz-Mercer, B.; Walt, H.; Kushnir, I.; Jacob, N.; Diener, P.A.; Moll, R.; Czernobilsky, B. Differentiation potential of ovarian dysgerminoma: An immunohistochemical study of 15 cases. Hum. Pathol. 1995, 26, 62–66. Available online: https://www.sciencedirect.com/science/article/pii/0046817795901159 (accessed on 19 May 2023).
- Bayle, C.; Charpentier, A.; Duchayne, E.; Manel, A.M.; Pages, M.P.; Robert, A.; Lamant, L.; Dastugue, N.; Bertrand, Y.; Dijoud, F.; et al. Leukaemic presentation of small cell variant anaplastic large cell lymphoma: Report of four cases. Br. J. Haematol. 1999, 104, 680–688.
- Falini, B.; Pileri, S.; Zinzani, P.L.; Carbone, A.; Zagonel, V.; Wolf-Peeters, C.; Verhoef, G.; Menestrina, F.; Todeschini, G.; Paulli, M.; et al. ALK+ Lymphoma: Clinico-Pathological Findings and Outcome. Blood 1999, 93, 2697–2706. Available online: https://www.sciencedirect.com/science/article/pii/S0006497120489566 (accessed on 19 May 2023).
- Stein, H.; Foss, H.D.; Dürkop, H.; Marafioti, T.; Delsol, G.; Pulford, K.; Pileri, S.; Falini, B. CD30+ anaplastic large cell lymphoma: A review of its histopathologic, genetic, and clinical features. Blood 2000, 96, 3681–3695. Available online: https://www.sciencedirect.com/science/article/pii/S0006497120482060 (accessed on 20 May 2023).
- Falini, B.; Bigerna, B.; Fizzotti, M.; Pulford, K.; Pileri, S.A.; Delsol, G.; Carbone, A.; Paulli, M.; Magrini, U.; Menestrina, F.; et al. ALK Expression Defines a Distinct Group of T/Null Lymphomas (‘ALK Lymphomas’) with a Wide Morphological Spectrum. Am. J. Pathol. 1998, 153, 875–886.
- Kumar, S.; Dispenzieri, A.; Lacy, M.Q.; Hayman, S.R.; Buadi, F.K.; Colby, C.; Laumann, K.; Zeldenrust, S.R.; Leung, N.; Dingli, D.; et al. Revised Prognostic Staging System for Light Chain Amyloidosis Incorporating Cardiac Biomarkers and Serum Free Light Chain Measurements. J. Clin. Oncol. 2012, 30, 989–995.
- Pileri, S.A.; Pulford, K.; Mori, S.; Mason, D.Y.; Sabattini, E.; Roncador, G.; Piccioli, M.; Ceccarelli, C.; Piccaluga, P.P.; Santini, D.; et al. Frequent expression of the NPM-ALK chimeric fusion protein in anaplastic large-cell lymphoma, lympho-histiocytic type. Am. J. Pathol. 1997, 150, 1207.
- Agur, A.; Amir, G.; Paltiel, O.; Klein, M.; Dann, E.J.; Goldschmidt, H.; Goldschmidt, N. CD68 staining correlates with the size of residual mass but not with survival in classical Hodgkin lymphoma. Leuk. Lymphoma 2015, 56, 1315–1319.
- Isaacson, P.G.; Du, M.Q. MALT lymphoma: From morphology to molecules. Nat. Rev. Cancer 2004, 4, 644–653.
- Hodges, K.B.; Collins, R.D.; Greer, J.P.; Kadin, M.E.; Kinney, M.C. Transformation of the small cell variant Ki-1+ lymphoma to anaplastic large cell lymphoma: Pathologic and clinical features. Am. J. Surg. Pathol. 1999, 23, 49–58.
More
Information
Subjects:
Hematology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
876
Revisions:
3 times
(View History)
Update Date:
12 Jul 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





