
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alessandra Murabito | -- | 2078 | 2023-07-07 10:29:39 | | | |
| 2 | Wendy Huang | Meta information modification | 2078 | 2023-07-07 15:22:35 | | |
Video Upload Options
Cystic fibrosis (CF) is the rare genetic disease caused by mutations in the gene encoding the cystic fibrosis transmembrane conductance regulator (CFTR). These molecules, known as CFTR modulators, have led to unprecedented improvements in the lung function and quality of life of most CF patients. However, the efficacy of these drugs is still suboptimal, and the clinical response is highly variable even among individuals bearing the same mutation. Furthermore, not all patients carrying rare CFTR mutations are eligible for CFTR modulator therapies, indicating the need for alternative and/or add-on therapeutic approaches. Because the second messenger 3′,5′-cyclic adenosine monophosphate (cAMP) represents the primary trigger for CFTR activation and a major regulator of different steps of the life cycle of the channel, there is growing interest in devising ways to fine-tune the cAMP signaling pathway for therapeutic purposes.
1. Introduction
2. cAMP Is Recognized as the Master Regulator of CFTR Activity
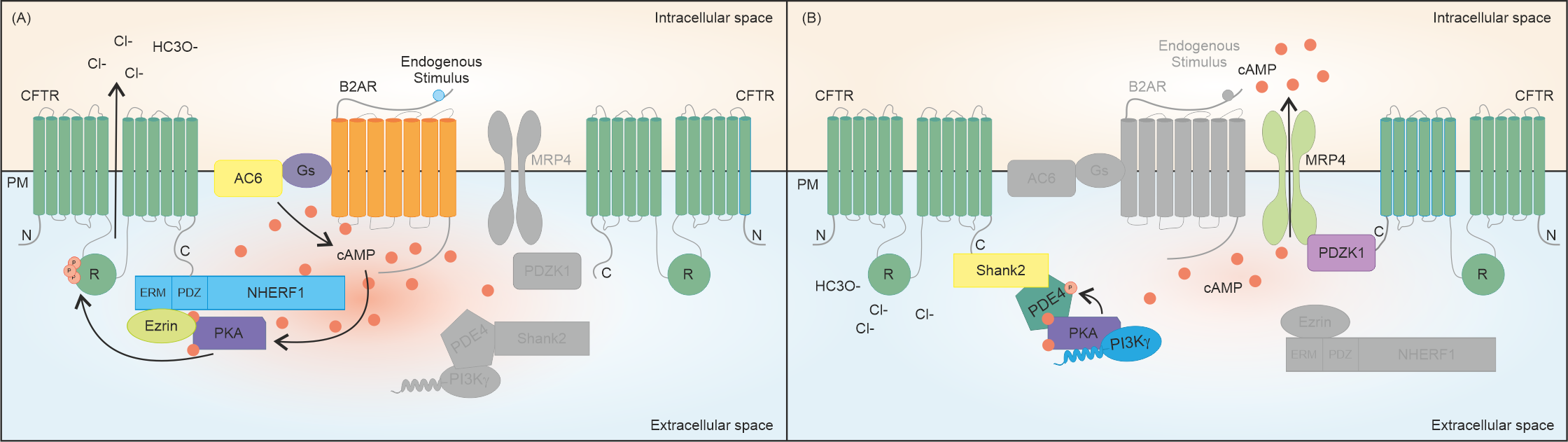
References
- Shteinberg, M.; Haq, I.J.; Polineni, D.; Davies, J.C. Cystic fibrosis. Lancet 2021, 397, 2195–2211.
- Bell, S.C.; Mall, M.A.; Gutierrez, H.; Macek, M.; Madge, S.; Davies, J.C.; Burgel, P.R.; Tullis, E.; Castanos, C.; Castellani, C.; et al. The future of cystic fibrosis care: A global perspective. Lancet Respir. Med. 2020, 8, 65–124.
- Heijerman, H.G.M.; McKone, E.F.; Downey, D.G.; Van Braeckel, E.; Rowe, S.M.; Tullis, E.; Mall, M.A.; Welter, J.J.; Ramsey, B.W.; McKee, C.M.; et al. Efficacy and safety of the elexacaftor plus tezacaftor plus ivacaftor combination regimen in people with cystic fibrosis homozygous for the F508del mutation: A double-blind, randomised, phase 3 trial. Lancet 2019, 394, 1940–1948.
- Middleton, P.G.; Mall, M.A.; Drevinek, P.; Lands, L.C.; McKone, E.F.; Polineni, D.; Ramsey, B.W.; Taylor-Cousar, J.L.; Tullis, E.; Vermeulen, F.; et al. Elexacaftor-tezacaftor-ivacaftor for cystic fibrosis with a single Phe508del allele. N. Engl. J. Med. 2019, 381, 1809–1819.
- Orenti, A.; Zolin, A.; Jung, A.; van Rens, J.; Adamoli, A.; Prasad, V.; Fox, A.; Krasnyk, M.; Lorca Mayor, S.; Naehrlich, L.; et al. ECFSPR Annual Report 2021. 2023. Available online: https://www.ecfs.eu/sites/default/files/Annual%20Report_2021_09Jun2023.pdf (accessed on 16 June 2023).
- Fajac, I.; Sermet, I. Therapeutic approaches for patients with cystic fibrosis not eligible for current CFTR modulators. Cells 2021, 10, 2793.
- Graeber, S.Y.; Vitzthum, C.; Pallenberg, S.T.; Naehrlich, L.; Stahl, M.; Rohrbach, A.; Drescher, M.; Minso, R.; Ringshausen, F.C.; Rueckes-Nilges, C.; et al. Effects of elexacaftor/tezacaftor/ivacaftor therapy on CFTR function in patients with cystic fibrosis and one or two F508del alleles. Am. J. Respir. Crit. Care Med. 2022, 205, 540–549.
- Veit, G.; Roldan, A.; Hancock, M.A.; Da Fonte, D.F.; Xu, H.; Hussein, M.; Frenkiel, S.; Matouk, E.; Velkov, T.; Lukacs, G.L. Allosteric folding correction of F508del and rare CFTR mutants by elexacaftor-tezacaftor-ivacaftor (Trikafta) combination. JCI Insight 2020, 5, e139983.
- Farinha, C.M.; Gentzsch, M. Revisiting CFTR interactions: Old partners and new players. Int. J. Mol. Sci. 2021, 22, 13196.
- Chin, S.; Hung, M.; Bear, C.E. Current insights into the role of PKA phosphorylation in CFTR channel activity and the pharmacological rescue of cystic fibrosis disease-causing mutants. Cell Mol. Life Sci. 2017, 74, 57–66.
- Murabito, A.; Cnudde, S.; Hirsch, E.; Ghigo, A. Potential therapeutic applications of AKAP disrupting peptides. Clin. Sci. 2020, 134, 3259–3282.
- Naren, A.P.; Cobb, B.; Li, C.; Roy, K.; Nelson, D.; Heda, G.D.; Liao, J.; Kirk, K.L.; Sorscher, E.J.; Hanrahan, J.; et al. A macromolecular complex of beta 2 adrenergic receptor, CFTR, and ezrin/radixin/moesin-binding phosphoprotein 50 is regulated by PKA. Proc. Natl. Acad. Sci. USA 2003, 100, 342–346.
- Trotta, T.; Guerra, L.; Piro, D.; d’Apolito, M.; Piccoli, C.; Porro, C.; Giardino, I.; Lepore, S.; Castellani, S.; Di Gioia, S.; et al. Stimulation of beta2-adrenergic receptor increases CFTR function and decreases ATP levels in murine hematopoietic stem/progenitor cells. J. Cyst. Fibros. 2015, 14, 26–33.
- Vijftigschild, L.A.; Berkers, G.; Dekkers, J.F.; Zomer-van Ommen, D.D.; Matthes, E.; Kruisselbrink, E.; Vonk, A.; Hensen, C.E.; Heida-Michel, S.; Geerdink, M.; et al. beta2-Adrenergic receptor agonists activate CFTR in intestinal organoids and subjects with cystic fibrosis. Eur. Respir. J. 2016, 48, 768–779.
- Namkung, W.; Finkbeiner, W.E.; Verkman, A.S. CFTR-adenylyl cyclase I association responsible for UTP activation of CFTR in well-differentiated primary human bronchial cell cultures. Mol. Biol. Cell 2010, 21, 2639–2648.
- Liu, F.; Zhang, Z.; Csanady, L.; Gadsby, D.C.; Chen, J. Molecular structure of the human CFTR ion channel. Cell 2017, 169, 85–95.e88.
- Mihalyi, C.; Iordanov, I.; Torocsik, B.; Csanady, L. Simple binding of protein kinase A prior to phosphorylation allows CFTR anion channels to be opened by nucleotides. Proc. Natl. Acad. Sci. USA 2020, 117, 21740–21746.
- Della Sala, A.; Prono, G.; Hirsch, E.; Ghigo, A. Role of protein kinase A-mediated phosphorylation in CFTR channel activity regulation. Front. Physiol. 2021, 12, 690247.
- Infield, D.T.; Schene, M.E.; Fazan, F.S.; Galles, G.D.; Galpin, J.D.; Ahern, C.A. Real-time observation of functional specialization among phosphorylation sites in CFTR. J. Gen. Physiol. 2023, 155, e202213216.
- Sun, F.; Hug, M.J.; Bradbury, N.A.; Frizzell, R.A. Protein kinase A associates with cystic fibrosis transmembrane conductance regulator via an interaction with ezrin. J. Biol. Chem. 2000, 275, 14360–14366.
- Li, J.; Dai, Z.; Jana, D.; Callaway, D.J.; Bu, Z. Ezrin controls the macromolecular complexes formed between an adapter protein Na+/H+ exchanger regulatory factor and the cystic fibrosis transmembrane conductance regulator. J. Biol. Chem. 2005, 280, 37634–37643.
- Guerra, L.; Fanelli, T.; Favia, M.; Riccardi, S.M.; Busco, G.; Cardone, R.A.; Carrabino, S.; Weinman, E.J.; Reshkin, S.J.; Conese, M.; et al. Na+/H+ exchanger regulatory factor isoform 1 overexpression modulates cystic fibrosis transmembrane conductance regulator (CFTR) expression and activity in human airway 16HBE14o- cells and rescues DeltaF508 CFTR functional expression in cystic fibrosis cells. J. Biol. Chem. 2005, 280, 40925–40933.
- Singh, A.K.; Riederer, B.; Krabbenhoft, A.; Rausch, B.; Bonhagen, J.; Lehmann, U.; de Jonge, H.R.; Donowitz, M.; Yun, C.; Weinman, E.J.; et al. Differential roles of NHERF1, NHERF2, and PDZK1 in regulating CFTR-mediated intestinal anion secretion in mice. J. Clin. Investig. 2009, 119, 540–550.
- Lee, J.H.; Richter, W.; Namkung, W.; Kim, K.H.; Kim, E.; Conti, M.; Lee, M.G. Dynamic regulation of cystic fibrosis transmembrane conductance regulator by competitive interactions of molecular adaptors. J. Biol. Chem. 2007, 282, 10414–10422.
- Kim, J.Y.; Han, W.; Namkung, W.; Lee, J.H.; Kim, K.H.; Shin, H.; Kim, E.; Lee, M.G. Inhibitory regulation of cystic fibrosis transmembrane conductance regulator anion-transporting activities by Shank2. J. Biol. Chem. 2004, 279, 10389–10396.
- Blanchard, E.; Zlock, L.; Lao, A.; Mika, D.; Namkung, W.; Xie, M.; Scheitrum, C.; Gruenert, D.C.; Verkman, A.S.; Finkbeiner, W.E.; et al. Anchored PDE4 regulates chloride conductance in wild-type and DeltaF508-CFTR human airway epithelia. FASEB J. 2014, 28, 791–801.
- Ghigo, A.; Murabito, A.; Sala, V.; Pisano, A.R.; Bertolini, S.; Gianotti, A.; Caci, E.; Montresor, A.; Premchandar, A.; Pirozzi, F.; et al. A PI3Kgamma mimetic peptide triggers CFTR gating, bronchodilation, and reduced inflammation in obstructive airway diseases. Sci. Transl. Med. 2022, 14, eabl6328.
- Turner, M.J.; Sato, Y.; Thomas, D.Y.; Abbott-Banner, K.; Hanrahan, J.W. Phosphodiesterase 8A regulates CFTR activity in airway epithelial cells. Cell Physiol. Biochem. 2021, 55, 784–804.
- Nguyen, J.P.; Kim, Y.; Cao, Q.; Hirota, J.A. Interactions between ABCC4/MRP4 and ABCC7/CFTR in human airway epithelial cells in lung health and disease. Int. J. Biochem. Cell Biol. 2021, 133, 105936.
- Li, C.; Krishnamurthy, P.C.; Penmatsa, H.; Marrs, K.L.; Wang, X.Q.; Zaccolo, M.; Jalink, K.; Li, M.; Nelson, D.J.; Schuetz, J.D.; et al. Spatiotemporal coupling of cAMP transporter to CFTR chloride channel function in the gut epithelia. Cell 2007, 131, 940–951.




