Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ourania Manta | -- | 1283 | 2023-06-30 11:10:22 | | | |
| 2 | Sirius Huang | Meta information modification | 1283 | 2023-07-03 02:57:50 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Manta, O.; Sarafidis, M.; Schlee, W.; Mazurek, B.; Matsopoulos, G.K.; Koutsouris, D.D. Tinnitus-Related Distress Classification Using Auditory Evoked Potential Signals. Encyclopedia. Available online: https://encyclopedia.pub/entry/46266 (accessed on 26 March 2026).
Manta O, Sarafidis M, Schlee W, Mazurek B, Matsopoulos GK, Koutsouris DD. Tinnitus-Related Distress Classification Using Auditory Evoked Potential Signals. Encyclopedia. Available at: https://encyclopedia.pub/entry/46266. Accessed March 26, 2026.
Manta, Ourania, Michail Sarafidis, Winfried Schlee, Birgit Mazurek, George K. Matsopoulos, Dimitrios D. Koutsouris. "Tinnitus-Related Distress Classification Using Auditory Evoked Potential Signals" Encyclopedia, https://encyclopedia.pub/entry/46266 (accessed March 26, 2026).
Manta, O., Sarafidis, M., Schlee, W., Mazurek, B., Matsopoulos, G.K., & Koutsouris, D.D. (2023, June 30). Tinnitus-Related Distress Classification Using Auditory Evoked Potential Signals. In Encyclopedia. https://encyclopedia.pub/entry/46266
Manta, Ourania, et al. "Tinnitus-Related Distress Classification Using Auditory Evoked Potential Signals." Encyclopedia. Web. 30 June, 2023.
Copy Citation
Tinnitus is a highly prevalent condition, affecting more than 1 in 7 adults in the EU and causing negative effects on sufferers’ quality of life. The objective identification and detection of bothersome tinnitus is a critical step in the proper management and administration of appropriate interventions or the combination of interventions for the patients. A detailed audiological evaluation, including auditory brainstem responses and auditory middle latency responses analysis, could constitute an objective method for reflecting the functions of the cochlear or auditory nerve to auditory cortex.
tinnitus
auditory evoked potential
auditory brainstem response
auditory middle latency response
tinnitus-related distress
1. Tinnitus
Tinnitus is defined as the perception of a phantom sound and the patient’s reaction to it [1][2]. Most experts distinguish between subjective and objective tinnitus [2][3]. Tinnitus constitutes a common auditory symptom which can lead to severe impairment, especially when comorbidities are present [4]. In most sufferers, tinnitus is not due to medical causes, and there is no treatment available [5][6]. In many aspects, the presence of tinnitus is a heterogeneous and complex condition [7], and it can occur at any age with varying frequencies, intensities, and duration scales.
According to large, independent epidemiological studies, tinnitus affects more than 10% of the general population, whereas 1% of the population considers tinnitus to be their most serious health problem. The prevalence estimates in Europe are expected to double by 2050 [8][9][10]. Although significant scientific progress has been made in recent years [11][12][13][14][15], many patients with tinnitus remain either untreated or receive incomplete treatment. This situation contributes to increased complaints, prolonged suffering, social disengagement, excessive utilisation of healthcare services, and a complex network of referral pathways. Consequently, these result in substantial psychological and financial burdens at both national and global levels [1]. Despite its extremely high prevalence and socioeconomic burden, tinnitus remains a scientific and clinical mystery [16][17].
It is worth mentioning that the degree of tinnitus-related distress experienced by patients ranges from complete absence of discomfort to suicidal tendencies [18], resulting in a spectrum of different conditions that require distinct management and therapeutic approaches [19][20]. The various testing methods are unable to represent the degree to which tinnitus is troublesome on a case-by-case basis, although they are very useful in the diagnosis and planning of therapeutic interventions. For instance, patients with identical audiograms may have varying degrees of tinnitus perception in terms of intensity, severity, and induced disability [21][22]. This weakness in clinical and paraclinical examinations is well compensated by the use of tinnitus self-assessment questionnaires, which have been widely used in the clinical evaluation of tinnitus sufferers [7]. The use of these tests to objectively classify severity is considered to be an extremely useful tool in the hands of health professionals. Although the use of self-reported measures is considered a good practice, it is important to remember that self-assessment involves bias [1], which influences judgments and responses [19][23].
Several theories have been proposed to explain the mechanisms underlying tinnitus. Over the last decades, it has become apparent that tinnitus is closely related to hearing loss, but their degree of severity cannot be correlated. Moreover, the causal relevance of tinnitus is not limited to the cochlea, but probably involves many levels of the central auditory pathway, and even the central nervous system [24]. There is growing evidence that tinnitus etiopathogenetic mechanisms may be related to dysfunction or damage in parts of the auditory pathway [22][24]. This clinical hypothesis motivated auditory pathway testing through the use of auditory evoked potentials (AEPs) of tinnitus sufferers to assess and evaluate the severity of their tinnitus. In other words, given that the auditory pathway includes several stations involved in the conduction of sound, it is hypothesised that each of them could be associated with the occurrence of tinnitus; or, to state it simply, in order for an individual to hear tinnitus, one or more of the above stations or the connections between them must be affected.
2. Auditory Evoked Potentials (AEPs)
An AEP is an electrical signal produced by the brain in response to the presentation of a time-locked auditory stimulus [25][26]. The final AEP signal is composed of the average responses to thousands of stimulus repeats [27]. AEPs are a form of non-invasive and non-behavioural test whose main advantages are their simplicity, objectivity, reproducibility, and cost-effectiveness [22]. Based on a subject’s AEP response, audiologists are able to investigate potential obstacles along the neural pathways that lead to the brain. In addition, these signals may be useful for ruling out or confirming hearing impairments, particularly in neonates, and for medico-legal purposes to rule out benign tumours of the acoustic nerves, such as acoustic neuromas [28].
AEPs are classified as early (auditory brainstem responses—ABRs), middle (auditory middle latency responses—AMLRs), or late (auditory late latency responses—ALLRs) based on their occurrence time after the triggering stimulus [29].
In more detail, ABR is a sequence of acoustically stimulated signals that indicates synchronised neuronal activity along the neural pathways leading to the brain. It has a lengthy history of application and is regarded as one of the most reliable electrophysiological methods [30][31]. Within 10 milliseconds after the commencement of a moderately intense click stimulus, the derived ABR consists of five peaks coming from the auditory nerve and brainstem, annotated using Roman numerals (I through V) in capital letters (Figure 1). Wave I of the ABR reflects the activity of spiral ganglion cells in the distal eighth auditory nerve; wave II originates from the globular cells in the cochlear nucleus; wave III is generated by the cochlear nucleus’ spherical cells and globular cells; and waves IV and V are generated by the medial superior olive and its projections to the nuclei in the lateral lemniscus and the inferior colliculus [32][33]. Typically, these electrophysiological responses have an amplitude of less than one microvolt (μV) [28][34].
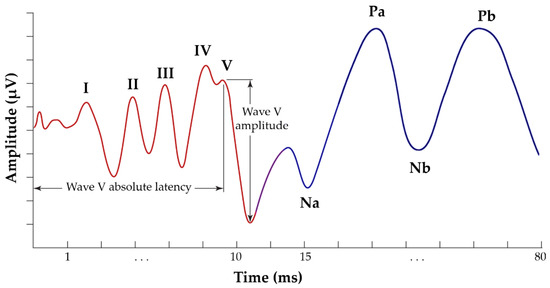
Figure 1. Typical annotated ABR signal, presenting the five waves of interest, from I to V (red waveform), and AMLR signal, presenting the four waves of interest: Na, Pa, Nb, and Pb (blue waveform).
AMLR is typically recorded in a time window of 80 to 100 milliseconds and occurs around 12 to 60 milliseconds following the external stimulation. It is hypothesised that the thalamus and the auditory cortex are responsible for generating this response. AMLR is a waveform with four waves of interest: two troughs (Na and Nb) and two peaks (Pa and Pb) (Figure 1). The AMLR signal is sensitive to low frequencies, and there is typically a discrepancy of approximately 10 dB between the auditory thresholds measured behaviourally and electrophysiologically [35][36]. The shape of these waveforms varies considerably even among healthy individuals, with the Nb and Pb components appearing inconsistently [28].
ALLR is produced by non-primary cortical areas and is utilised to evaluate the integrity of the auditory system beyond the level of AMLR. It typically occurs 60 to 800 milliseconds after the external stimulus [37].
In brief, AEPs have predictable patterns and consist of discrete waves (peaks and troughs), which are the signal’s primary waves of interest and which are generated by specific stations along the auditory pathway [22]. The major metrics of an AEP are the latencies (the time between the initial auditory stimulus and the peak or trough of a waveform [31]), and absolute amplitudes related to the signal’s waves of interest [38]. Clinicians contemplate these measurements as metrics when interpreting these waveforms.
The objective identification and detection of bothersome tinnitus is a critical step in the proper management and administration of appropriate interventions or the combination of interventions for the patients. A detailed audiological evaluation, including ABR and AMLR analysis, could constitute an objective method for reflecting the functions of the cochlear or auditory nerve to auditory cortex. These electrophysiological methods are not currently included in routine clinical approaches, and have not been clearly correlated with the pathophysiology of tinnitus [39]. However, the utilisation of more objective data could have a strong influence on the way otolaryngologists investigate and understand tinnitus, focused towards an evidence-based approach.
References
- Cima, R.F.F.; Mazurek, B.; Haider, H.; Kikidis, D.; Lapira, A.; Noreña, A.; Hoare, D.J. A Multidisciplinary European Guideline for Tinnitus: Diagnostics, Assessment, and Treatment. HNO 2019, 67, 10–42.
- Kreuzer, P.M.; Vielsmeier, V.; Langguth, B. Chronic Tinnitus: An Interdisciplinary Challenge. Dtsch. Arztebl. Int. 2013, 110, 278.
- Gopinath, B.; McMahon, C.M.; Rochtchina, E.; Karpa, M.J.; Mitchell, P. Incidence, Persistence, and Progression of Tinnitus Symptoms in Older Adults: The Blue Mountains Hearing Study. Ear Hear. 2010, 31, 407–412.
- Cima, R.F.F.; Kikidis, D.; Mazurek, B.; Haider, H.; Cederroth, C.R.; Norena, A.; Lapira, A.; Bibas, A.; Hoare, D.J. Tinnitus Healthcare: A Survey Revealing Extensive Variation in Opinion and Practices across Europe. BMJ Open 2020, 10, e029346.
- Baguley, D.; McFerran, D.; Hall, D. Tinnitus. Lancet 2013, 382, 1600–1607.
- Langguth, B.; Elgoyhen, A.B. Current Pharmacological Treatments for Tinnitus. Expert Opin. Pharmacother. 2012, 13, 2495–2509.
- Person, O.C.; Veiga, F.; Junior, A.; Altoé, J.; Portes, L.M.; Lopes, P.R.; Dos, M.E.; Puga, S.; Clayton, O.; Brasileiros De Ciências Da, A.; et al. O Que Revisões Sistemáticas Cochrane Dizem Sobre Terapêutica Para Zumbido? ABCS Heal. Sci. 2022, 47, e022301.
- Gallus, S.; Lugo, A.; Garavello, W.; Bosetti, C.; Santoro, E.; Colombo, P.; Perin, P.; La Vecchia, C.; Langguth, B. Prevalence and Determinants of Tinnitus in the Italian Adult Population. Neuroepidemiology 2015, 45, 12–19.
- Nondahl, D.M.; Cruickshanks, K.J.; Huang, G.H.; Klein, B.E.K.; Klein, R.; Tweed, T.S.; Zhan, W. Generational Differences in the Reporting of Tinnitus. Ear Hear. 2012, 33, 640.
- Hasson, D.; Theorell, T.; Westerlund, H.; Canlon, B. Prevalence and Characteristics of Hearing Problems in a Working and Non-Working Swedish Population. J. Epidemiol. Community Health 2010, 64, 453–460.
- McFerran, D.; Hoare, D.J.; Carr, S.; Ray, J.; Stockdale, D. Tinnitus Services in the United Kingdom: A Survey of Patient Experiences. BMC Health Serv. Res. 2018, 18, 110.
- Hoare, D.J.; Gander, P.E.; Collins, L.; Smith, S.; Hall, D.A. Management of Tinnitus in English NHS Audiology Departments: An Evaluation of Current Practice. J. Eval. Clin. Pract. 2012, 18, 326–334.
- Hall, D.A.; Láinez, M.J.A.; Newman, C.W.; Sanchez, T.; Egler, M.; Tennigkeit, F.; Koch, M.; Langguth, B. Treatment Options for Subjective Tinnitus: Self Reports from a Sample of General Practitioners and ENT Physicians within Europe and the USA. BMC Health Serv. Res. 2011, 11, 302.
- El-Shunnar, S.K.; Hoare, D.J.; Smith, S.; Gander, P.E.; Kang, S.; Fackrell, K.; Hall, D.A. Primary Care for Tinnitus: Practice and Opinion among GPs in England. J. Eval. Clin. Pract. 2011, 17, 684–692.
- Cima: TINNET COST Action BM1306-Clinical WG1: Establishme—Google Scholar. Available online: https://scholar.google.com/scholar_lookup?title=Establishment of a standard for Tinnitus%3B patient assessment%2C characterization%2C and treatment options&publication_year=2016&author=Cima%2CRFF&author=Haider%2CH&author=Mazurek%2CB&author=Cederroth%2CCR&author=Lapira%2CA&author=Kikidis%2CD&author=Noreña%2CA (accessed on 15 October 2022).
- Elgoyhen, A.B.; Langguth, B.; De Ridder, D.; Vanneste, S. Tinnitus: Perspectives from Human Neuroimaging. Nat. Rev. Neurosci. 2015, 16, 632–642.
- Sarafidis, M.; Manta, O.; Kouris, I.; Schlee, W.; Kikidis, D.; Vellidou, E.; Koutsouris, D. Why a Clinical Decision Support System Is Needed for Tinnitus. In Proceedings of the 2021 43rd Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Online, 1–5 November 2021; pp. 2075–2078.
- Schlee, W.; Schoisswohl, S.; Staudinger, S.; Schiller, A.; Lehner, A.; Langguth, B.; Schecklmann, M.; Simoes, J.; Neff, P.; Marcrum, S.C.; et al. Towards a unification of treatments and interventions for tinnitus patients: The EU research and innovation action UNITI. In Progress in Brain Research; Elsevier B.V.: Amsterdam, The Netherlands, 2021; Volume 260, pp. 441–451. ISBN 9780128215869.
- Husain, F.T.; Gander, P.E.; Jansen, J.N.; Shen, S. Expectations for Tinnitus Treatment and Outcomes: A Survey Study of Audiologists and Patients. J. Am. Acad. Audiol. 2018, 29, 313–336.
- Cima, R.F.F. Stress-related tinnitus treatment protocols. In Tinnitus and Stress: An Interdisciplinary Companion for Healthcare Professionals; Springer: Berlin/Heidelberg, Germany, 2017; pp. 139–172.
- Humes, L.E.; Rogers, S.E.; Quigley, T.M.; Main, A.K.; Kinney, D.L.; Herring, C. The Effects of Service-Delivery Model and Purchase Price on Hearing-Aid Outcomes in Older Adults: A Randomized Double-Blind Placebo-Controlled Clinical Trial. Am. J. Audiol. 2017, 26, 53–79.
- Manta, O.; Sarafidis, M.; Schlee, W.; Consoulas, C.; Kikidis, D.; Koutsouris, D. Electrophysiological Differences in Distinct Hearing Threshold Level Individuals with and without Tinnitus Distress. In Proceedings of the 2022 44th Annual International Conference of the IEEE Engineering in Medicine & Biology Society (EMBC), Glasgow, UK, 11–15 July 2022; Volume 2022, pp. 1630–1633.
- Searchfield, G.D.; Zhang, J. The Behavioral Neuroscience of Tinnitus; Springer: Berlin/Heidelberg, Germany, 2021; Volume 51.
- Zhang, L.; Wu, C.; Martel, D.T.; West, M.; Sutton, M.A.; Shore, S.E. Noise Exposure Alters Glutamatergic and GABAergic Synaptic Connectivity in the Hippocampus and Its Relevance to Tinnitus. Neural Plast. 2021, 2021, 8833087.
- Picton, T.W.; John, M.S.; Purcell, D.W.; Plourde, G. Human Auditory Steady-State Responses: The Effects of Recording Technique and State of Arousal. Anesth. Analg. 2003, 97, 1396–1402.
- Paulraj, M.P.; Subramaniam, K.; Bin Yaccob, S.; Bin Adom, A.H.; Hema, C.R. Auditory Evoked Potential Response and Hearing Loss: A Review. Open Biomed. Eng. J. 2015, 9, 17.
- Polonenko, M.J.; Maddox, R.K. The Parallel Auditory Brainstem Response. Trends Hear. 2019, 23, 17.
- Hall, J.W. (Ed.) Handbook of Auditory Evoked Responses; Pearson Education, Inc.: New York, NY, USA, 2015; ISBN 0205135668.
- Sörnmo, L.; Laguna, P. Evoked Potentials. In Bioelectrical Signal Processing in Cardiac and Neurological Applications; Academic Press: Cambridge, MA, USA, 2005; pp. 181–336.
- Winkler, I.; Denham, S.; Escera, C. Auditory Event-Related Potentials. In Encyclopedia of Computational Neuroscience; Springer: New York, NY, USA, 2013; pp. 1–29.
- Young, A.; Cornejo, J.; Spinner, A. Auditory Brainstem Response; StatPearls Publishing: Treasure Island, FL, USA, 2022.
- Milloy, V.; Fournier, P.; Benoit, D.; Noreña, A.; Koravand, A. Auditory Brainstem Responses in Tinnitus: A Review of Who, How, and What? Front. Aging Neurosci. 2017, 9, 237.
- Melcher, J.R.; Kiang, N.Y.S. Generators of the Brainstem Auditory Evoked Potential in Cat. III: Identified Cell Populations. Hear. Res. 1996, 93, 52–71.
- Chalak, S.; Kale, A.; Deshpande, V.K.; Biswas, D.A. Establishment of Normative Data for Monaural Recordings of Auditory Brainstem Response and Its Application in Screening Patients with Hearing Loss: A Cohort Study. J. Clin. Diagn. Res. 2013, 7, 2677–2679.
- Schoisswohl, S.; Langguth, B.; Schecklmann, M.; Bernal-Robledano, A.; Boecking, B.; Cederroth, C.R.; Chalanouli, D.; Cima, R.; Denys, S.; Dettling-Papargyris, J.; et al. Unification of Treatments and Interventions for Tinnitus Patients (UNITI): A Study Protocol for a Multi-Center Randomized Clinical Trial. Trials 2021, 22, 875.
- Watson, D.R. The Effects of Cochlear Hearing Loss, Age and Sex on the Auditory Brainstem Response. Int. J. Audiol. 2007, 35, 246–258.
- Konadath, S.; Manjula, P. Auditory Brainstem Response and Late Latency Response in Individuals with Tinnitus Having Normal Hearing. Intractable Rare Dis. Res. 2016, 5, 262–268.
- Eggermont, J.J. Auditory Brainstem Response. Handb. Clin. Neurol. 2019, 160, 451–464.
- McFadden, D.; Champlin, C.A.; Pho, M.H.; Pasanen, E.G.; Malone, M.M.; Leshikar, E.M. Auditory Evoked Potentials: Differences by Sex, Race, and Menstrual Cycle and Correlations with Common Psychoacoustical Tasks. PLoS ONE 2021, 16, e0251363.
More
Information
Subjects:
Others
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
689
Revisions:
2 times
(View History)
Update Date:
03 Jul 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





