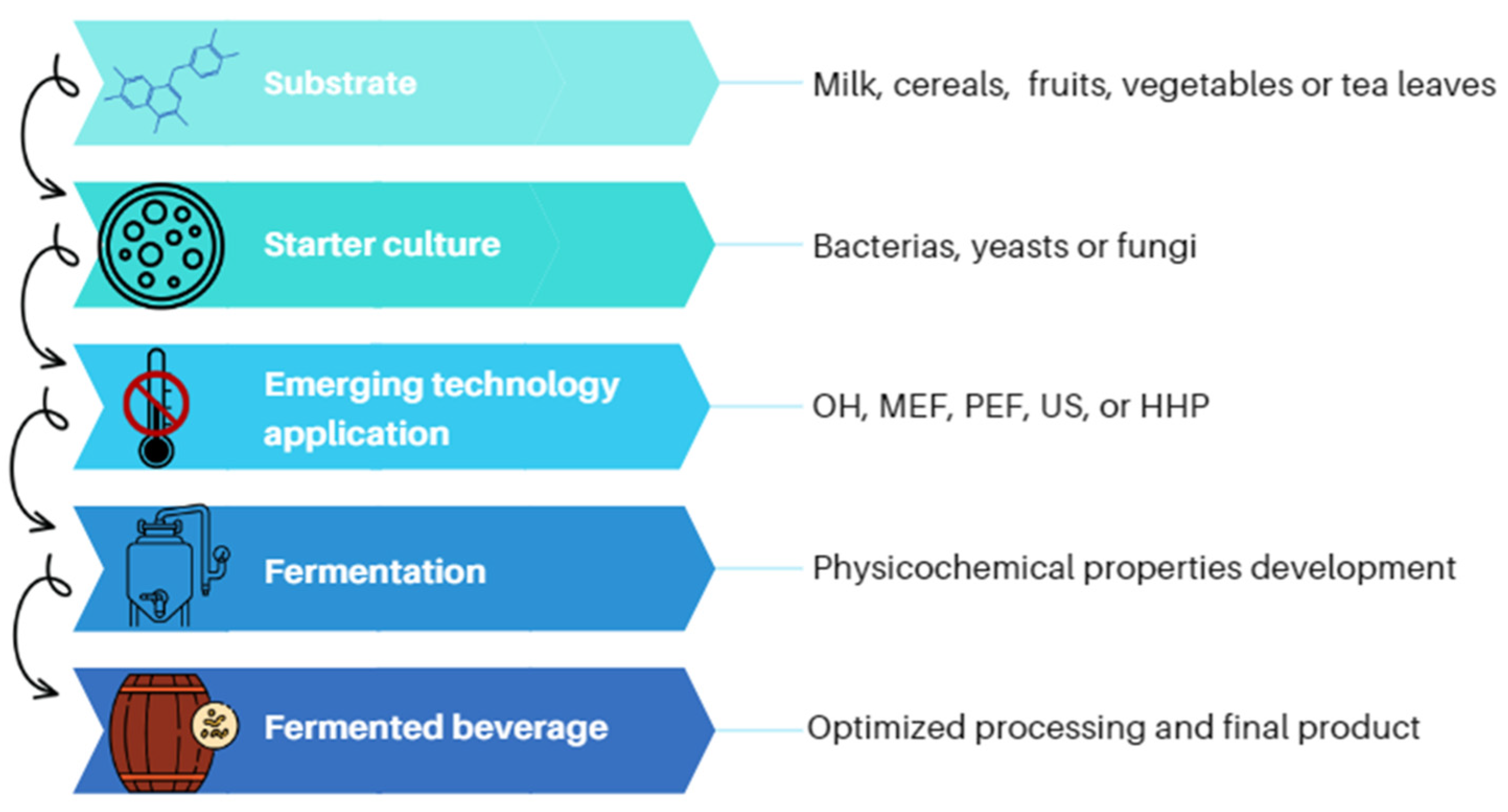
2. Fermented Beverages Development
Among the huge variety of fermented products, the beverages category represents a growing sector of the food industry since modern health-conscious consumers worldwide recognize them as refreshing, convenient, and healthy products, as well as probiotic vehicles that could improve well-being and reduce the risk of chronic and degenerative diseases
[8]. Fermented beverages can be obtained mainly from milk from different sources, cereals, fruits, vegetables, or tea leaves fermented by a variety of microorganisms. According to their alcoholic strength by volume (ABV) content, they are classified as alcoholic fermented beverages (A-FB) with >1.2% ABV, low-alcoholic fermented beverages (LA-FB) with ≤1.2% ABV, and non-alcoholic fermented beverages (NA-FB) with <0.5% ABV
[10]. A complete and interesting review of different types of traditional fermented beverages, their history, cultural aspects related to their consumption, fermentation processing, and product attributes, has been conducted by Baschali et al.
[10].
Interestingly, the prevalence of allergy to cows’ milk proteins, lactose, and gluten intolerance, as well as the popular trends towards vegan and vegetarian diets, have promoted an accelerated development of A-FB, LA-FB, and NA-FB with non-traditional ingredients as an alternative to satisfy the mentioned needs. Hence, scientific interest in the design of fermented beverages with non-conventional ingredients and health-related properties and the selection of optimal processing conditions has increased during the last decade
[10][11][12][13][14][15][16][17][18][19][20][21][22][23][24][25][26][27][28].
A-FBs are usually obtained by the action of
Saccharomyces cerevisiae yeast in different substrates, because it is the main microorganism responsible for alcoholic fermentation
[12][13]. One of the most frequently consumed A-FB is beer. It is usually produced by extracting raw materials from the malt with boiling water and then fermenting with
Saccharomyces cerevisiae yeast for a period of 7–14 days, approximately
[14][15][16]. Barley is the most used raw material in traditional beer processing; nonetheless, diverse cereals like rice, corn, sorghum, or millet are used today to produce different types of beers to satisfy the current consumers’ demand for gluten-free products
[14][15]. Likewise, wine is traditionally made from fermented grapes by different yeast species and strains which interact among them, resulting in wines of different quality characteristics
[17][18], with
S. cerevisiae being the most widespread yeast used as a starter in winemaking
[16]. Nowadays, other fruits such as berries, apricot, plum, and cherry have been used to produce new fruit wines taking advance of their high content of phenolic compounds and antioxidant potential
[19]. Recently, Maldonado et al.
[20] used tamarind (
Tamarindus indica L.) as a substrate for tamarind wine production, using
S. cerevisiae for fermentation. These authors indicated that tamarind is a potential alternative for wine production, improving the utilization of this fruit. Another example of a popular A-FB is cider, which is prepared from apple juice obtained from freshly pressed apples of different varieties and ripening stages or from concentrated apple juice fermented with
S. cerevisiae for 10–15 days
[21][22][23]. In a recent study, Hou et al.
[24] evaluated the use of non-
Saccharomyces yeasts to optimize the fermentation conditions of cider and produce volatile compounds with rose and honey scent. As a result, a cider with a different aroma profile that is highly attractive to consumers in terms of sensory properties was developed.
Otherwise, LA-FB and NA-FB are commonly produced by the action of bacteria, yeasts and fungi, or their combination, inoculated in different raw materials from plant or animal origin. For example, drinkable yogurt, considered a traditionally popular fermented beverage worldwide, is obtained by milk fermentation, specifically with
Lactobacillus bulgaricus and
Streptococcus thermophilus. It has had an important role in human diets, being a good source of proteins with high digestibility, vitamins A and B, as well as minerals such as calcium, magnesium, zinc, and phosphorus, among other important compounds
[25]. Kefir is also one of the oldest NA-FB, which has gained high popularity in recent years due to its unique sensorial attributes, nutritional value, and functional properties. It is prepared from any type of milk and the interaction of various homo- and heterofermentative LAB species such as
Lactobacillus helveticus,
Lactobacillus acidophilus,
Streptococcus thermophilus,
Bifidobacterium bifidum, and yeasts like
Kluyveromyces,
Saccharomyces,
Candida, and
Torulopsis present in “kefir grains”
[10][26]. As mentioned before, due to the high demand for dairy-alternative products, the development of plant-based yogurt- and kefir-like products has been widely investigated in recent years. Scientists and technologists have focused on finding plant-derived ingredients such as cereals, pseudocereals and legumes to be used as a substrate for fermentation with LAB, obtaining products with similar nutritional, functional, textural, and sensorial characteristics to conventional yogurt or kefir and with the ability to host LAB for long storage time
[27]. In this regard, Boek et al.
[28] reported that pulses such as beans, peas, chickpeas, cowpeas, and lentils are potential ingredients for yogurt alternatives because of their elevated protein concentration, amino acid profile and gelling behavior when fermented with LAB. In a different study, almond beverage (38.88%) and Jerusalem artichoke beverage (61.12%) were combined and fermented with
Streptococcus thermophilus,
Lactobacillus delbruckii ssp.
Bulgaricus,
Lactobacillus acidophilus, and
Bifidobacterium lactis to produce a plant-based yogurt that can be safely consumed by people who seek dairy-free products with good sensory acceptability and physicochemical characteristics
[29]. On the other hand, kombucha is considered a refreshing LA-FB with beneficial properties on human health, currently being highly consumed worldwide
[30][31]. It is commonly prepared from the fermentation of sweetened black tea with a symbiotic consortium composed of acetic acid bacteria and yeasts for 7 to 10 days at room temperature (20–30 °C), resulting in a final product with a sour and slightly sweet taste and probiotic properties
[29][32]. However, other substrates, such as green tea, oolong tea, and medicinal herbs, have also been used for kombucha fermentation
[33]. Most of the scientific efforts conducted today regarding kombucha processing rely on the isolation, enumeration, biochemical characterization, and identification of the microorganisms present in this LA-FB in order to have a better understanding of its microbial diversity and its potential probiotics benefits
[30][31][32][33].
Overall, one of the most attractive characteristics of fermented beverages is their health-promoting potential which can contribute to well-being
[11]. In this sense, the development of new products with appealing flavors, natural ingredients, and functional attributes has been a great challenge for the food industry and a wide area for research work. Furthermore, the use of sustainable technologies to reduce processing costs and environmental impact is also highly important to satisfy current consumers’ claims. In this regard, the implementation of emerging processes as assisting fermentation treatments has demonstrated potential results that could be scaled-up at industrial levels for fermented beverage elaboration, as explained in the next section.
3. Emerging Technologies as Fermentation-Assisted Processes for Fermented Beverages Development
As mentioned before, the production of fermented products has undergone remarkable improvement over the years, evolving from spontaneous fermentation up to the use of specific starter culture in a controlled environment at industrial levels and the application of novel techniques for processing optimization
[1][2][3][9][11]. During the last decades, different approaches have been conducted to better understand microbial metabolism and its functional role in fermentation by implementing tools such as next-generation sequence techniques and meta-omics technologies
[34]. At the same time, emerging processes, as well as modern and sophisticated equipment, have also been implemented to reduce operation time and energy costs, leading to competitive processes based on technological innovation, elevated yields, and high-quality, safe products
[9][35]. Among the emerging technologies used for this purpose, ohmic heating (OH), moderate electric fields (MEF), pulsed electric fields (PEF), ultrasound(US), and high hydrostatic pressures (HHP) stand out
[36][37][38].
Most of the emerging technologies are considered non-thermal processes, which have increasingly gained interest among researchers since they are safe and environment-friendly
[39]. Generally, a non-thermal process is applied at ambient temperature or lower than 40 °C during short periods of time, preserving heat-sensitive compounds mostly intact in the treated products, unlike heat-processed foods
[40]. In this regard, the application of non-thermal treatments could improve fermented beverages’ taste and nutritional-quality properties and might accelerate the rate of chemical and fermentation reactions decreasing processing time (
Figure 1)
[9][40][41][42].
Figure 1. Implementation of emerging technologies as assisted-fermentation techniques.
During the last two decades, several researchers worldwide have focused on fermentation process optimization by applying non-thermal treatments either prior to or during the fermentation stage to obtain fermented beverages with better functional properties, high-quality attributes, longer shelf life, and within shorter processing times. Generated results have indicated that PEF, US, and HHP have shown promising results in achieving these objectives, as reported in Table 1. However, the evaluation of each treatment for a specific substrate and microorganism is highly relevant in order to define the best processing conditions to achieve desired results.
Table 1. Effects of emerging technologies applied as assisted-fermentation techniques for the development of fermented beverages.
3.1. Ohmic Heating, Moderate Electric Fields and Pulsed Electric Fields
OH, MEF, and PEF processes involve the application of electrical energy at different intensities and short periods of time (μs-min) to food placed between two electrodes. The main difference among these technologies is the temperature achieved during processing: while OH is considered a thermal treatment in which the product acts as an electrical resistor, being rapidly heated by the dissipation of electrical energy, MEF and PEF are described as non-thermal processes
[52]. Nonetheless, the main mechanism of action in OH, MEF and PEF is electroporation, which depends on the electric field strength, and it could be irreversible or reversible, causing different effects on the treated product
[25].
Few studies have been conducted combining OH or MEF with fermentation, reporting that the electroporation phenomenon improves the fermentation process by increasing the metabolic rate and growth of microorganisms. Nonetheless, depending on the electric field strength applied and the microorganism’s characteristics, the metabolic effects could be different
[35][52][53][54]. Gally et al.
[55] and Knirsch et al.
[56] concluded that OH and MEF induce sublethal temperatures with a constant distribution in batch processing, enhancing the fermentation technique and causing positive effects on microorganisms’ activity. Interesting results from the application of OH and MEF as assisted fermentation techniques have been reviewed previously by Mota et al.
[51] and Gavahian et al.
[35]. Despite the fact that reported results showed a great potential to improve fermentation processes in terms of processing time reduction, further research must be conducted to fully understand occurring mechanisms and optimize processing parameters in order to obtain the best results.
OH technology has also been applied to fermented beverages for preservation purposes. Namely, Alcántara-Zavala et al.
[57] applied OH (65 °C/5 and 7 min; 70 °C/3 and 5 min) to pulque (a traditional Mexican probiotic A-FB) to extend the product’s shelf life. These authors highlighted that OH is a potential alternative to increase the shelf life of pulque up to 22 days, with no negative impact on physicochemical and sensory properties, and preserving significant counts of LAB, such as
Lactobacillus acidophilus,
Lactobacillus kefiri, and
Saccharomyces cerevisiae. Likewise, probiotic-fermented milk was treated with OH (4, 6 and 8 V/cm, 90–95 °C/5 min), and a Weibull predicted model was applied to evaluate the survival kinetics of
Listeria monocytogenes as post-fermentation contamination
[58]. Results from this study indicated that OH diminished the viability of
Listeria monocytogenes, and OH-treated beverages had suitable
Lactobacillus acidophilus counts. Furthermore, the application of OH enhanced the extraction of bioactive compounds and sensory properties of treated fermented beverages.
Regarding PEF technology, it has been applied for the development of high-value fermented beverages demonstrating promising results in terms of shelf life extension and beverage stability, fermentation time reduction, production of secondary metabolites, and improvement of microbial metabolism favoring their growth and increasing their population
[9]. On the one hand, high-intensity PEFs (>15 kV/cm) have been used for preservation purposes by the inactivation of pathogens or deteriorative microorganisms. In a recent study, Rios-Corpio et al.
[59] compared microbial stability, physicochemical parameters, bioactive compounds content, and sensory characteristics of a pomegranate (Punica granatum) fermented beverage treated by PEF (6 ms-bipolar pulses at 18 kV/cm and 200 Hz) or thermal pasteurization (63 °C, 30 min and 72 °C, 15 s) and stored for 56 days at refrigerated conditions (4 °C). Authors reported that microbial loads of
Brettanomyces ssp. were reduced by approximately four log cycles in the PEF-treated beverage. Also, the antioxidant compounds concentration in heat-treated beverages was lower than in the PEF-treated ones. Both treated beverages were stable during 56 days of storage; nevertheless, those beverages processed by PEF showed better sensory acceptability. Likewise, it has been reported that the PEF process at a high intensity (37–53 kV/cm) is a potential alternative to preserve kombucha beverages with minimal changes in their physicochemical attributes, antioxidant activity, and bioactive compounds content
[47]. Similarly, PEF technology can be used for microbial decontamination during wine production, achieving up to 4.0 Log
10 cycles reduction of
S. cerevisiae and
O. oeni in red wine after alcoholic and malolactic fermentation with no detrimental effects in its quality characteristics in terms of oenological parameters and sensory attributes
[60]. One of the most important advantages of high-intensity PEF is the short processing time, avoiding the increase of temperature and causing irreversible electroporation. As a result, microbial cells could be inactivated, and thermolabile compounds, such as antioxidants and volatile substances, are retained, obtaining safe, shelf life stable, and high-quality products
[9][55][56].
On the other hand, low intensity-PEF (<1 kV/cm) has been used to assist the fermentation of a
Hanseniapora sp. strain and control the sugar/ethanol conversion rate to produce low-alcohol apple cider
[61]. PEF treatment at 0.29 kV/cm and 10.7 s applied to the pre-culture during 6 h showed the highest alcohol reduction by 1.6% (v,v) with a significant increase of biomass yield and yeast concentration. Interestingly, authors reported that the sensitivity of
Hanseniaspora sp. yeast to PEF was more noticeable during the lag phase than in the log phase in terms of fermentation time decrease and ethanol content reduction. In a different study, El Darra et al.
[62] compared the effects of PEF (0.8 kV/cm-100 ms; 5 kV/cm-1 ms), US (24 kHz-5, 10 and 15 min), and mild pasteurization (50 °C-15 min) as pre-treatments of Cabernet Franc grapes for alcoholic fermentation. It was observed that all pre-treatments enhanced phenolics extraction, color intensity, and antioxidant activity of the grapes during fermentation; nonetheless, PEF at 0.8 kV/cm and 5 kV/cm caused the highest phenolic, anthocyanin, and tannin content in the obtained wine. Similarly, Saldaña et al.
[63] applied PEF treatments to induce permeabilization in the skin cells of different grape varieties to improve the vinification process in terms of polyphenol content enhancement or reduction of maceration time. The authors reported that depending on processing conditions and the grape variety, PEF could reduce the maceration time to obtain the highest phenolic concentration in the wine for two days. Furthermore, it was observed that the longer the pulse width, the higher the efficacy in electroporating cells from the grape skin. Ricci et al.
[64] gathered valuable information from 2007 to 2017 about PEF technology implemented in winemaking and concluded that PEF is a low-cost process that could improve red wines’ color quality and polyphenol profile, significantly reducing the maceration time. However, according to the authors, more research should be conducted to better understand the possible mechanisms involved in the reactions during fermentation and maceration, as well as to evaluate the potential electrochemical contamination induced by the PEF chamber electrodes during processing.
Recently, some authors have explored the effects of low-intensity PEF in fermented dairy beverage development. Chanos et al.
[65] reported that the PEF process at 1 kV/cm for 3 cycles of 50 pulses and 4 Hz applied to a mixed culture of
Streptococcus thermophilus DIL 5218 and
Lactobacillus delbrueckii subsp.
bulgaricus DSMZ 20081T inoculated in reconstituted skim milk medium successfully reduced yogurt fermentation time by 12 min. It was stated that PEF could cause cell stress and accelerate the metabolism of LAB, improving their performance during fermentation. Overall, it could be stated that low/moderate intensity PEF might induce reversible electroporation in the cell membrane or cell wall, which accelerates microbial activity or bioactive compounds extraction obtaining fermented beverages in a shorter processing time with improved quality. Understanding the effects caused by PEF processing parameters, such as pulse electric strength, pulse width, pulse frequency, pulse polarity or pulse shape on yogurt starter microorganisms as well as their connection to the development of the unique yogurt sensory characteristics, could open a new field of study for the optimization of dairy products fermentation process, as well as other fermented beverages. In addition, a comparison of energy consumption from PEF technology and conventional processes for fermented beverages elaboration is of high importance to validate the viability of this emerging technologies implementation at the industrial level.
3.2. Ultrasound
US processing refers to the propagation of sound waves of frequencies above the human hearing (20–40 kHz) through a liquid medium generating shear fields, agitation, turbulence, vibration, pressure, and acoustic streaming
[66][67][68]. Depending on the frequency applied during US processing, the liquid medium can be exposed to transient or stable cavitation, which is the main mechanism of action. The application of high-intensity US induces desirable physical and chemical effects that usually enhance process efficiency in the food industry towards improving mass transfer
[68][69]. Umego et al.
[70] gathered complete and concise information on US technology applied to increase fermentation efficiency.
Different research works have explored the use of US to control or stimulate microorganisms’ activity in fermented beverages. Ojha et al.
[71] reported that US enhances the growth and metabolic activity of different microorganisms. Likewise, Nguyen et al.
[72] evaluated the effects of US at 20 kHz on the fermentative activity of different strains of
Bifidobacterium in milk and observed that US could reduce the fermentation time required to reach a pH of 4.7 up to 30 min. Authors attribute this positive effect to the improvement in lactose hydrolysis and transgalactosylation by the enzyme β-galactosidase released from the intracellular content of bifidobacteria during processing. Similarly, Barukĉić et al.
[45] demonstrated the activation of a mixed culture of
Streptococcus thermophilus and
Lactobacillus delbrueckii subsp.
Bulgaricus inoculated in reconstituted sweet whey and treated with power US of 84 W over 150 s, resulting in a decreased fermentation time of up to 30 min and higher viable cell counts than the untreated fermented whey. Liu et al.
[73] applied US to LAB previous to their use for soymilk fermentation and observed a stress response due to the higher β-glucosidase activity and higher aglycone content in the fermented soymilk. US effects have been mainly associated with the formation of reversible pores in microbial cell membranes caused by cavitation, increasing their permeability for the internalization of essential nutrients and subsequently promoting their growth during fermentation. In addition, the generation of pressure fluctuation during US might induce cell growth, proliferation, and changes in the metabolic processes; as well, the cell membrane rupture causes the release of vitamins, nucleotides, amino acids, and enzymes which are growth-promoting factors that may stimulate the growth of the intact cells
[74].
Different authors have analyzed the changes in gel structure formation caused by US when applied as an assisted-fermentation process of yogurt production. It is well known that this characteristic is an important quality attribute of yogurt
[75]. It has been reported that US applied as pre-treatment or during fermentation affects the gel formation and can positively or negatively modify the final product texture
[76]. For example, the application of US at 45 kHz for 5 min during yogurt fermentation led to the formation of large colloidal particles that are not desirable in this kind of product, affecting its rheological properties
[77]. Conversely, as reported by Carrillo-López et al.
[75], US processing of milk at 24 kHz before fermentation results in higher yogurt firmness than when sonication is applied during fermentation.
Considering the production of A-FB, US has been used in wineries to improve wine taste, color, aroma, phenolic profile, and extraction yields from grape to must
[42][78]. US has shown great potential for accelerating the wine aging process by positively modifying the chemical reactions occurring during this time
[79]. Recently, US was applied to crushed grapes using winery scale equipment to optimize the maceration process
[80]. Obtained results from this study indicated that US altered the physical characteristics of grape skin, improving phenolic extraction and wine color with minimal impact on the physicochemical attributes of the finished wines. In addition, US processing may increase the efficiency of traditional winery processes by allowing a reduction of more than 50% of maceration time. As reviewed by Umego et al.
[70], sonication of
S. cerevisiae increased its growth rate, cell membrane permeability, and alcohol tolerance, improving its fermentation performance.
Recently, NA-FB with probiotic characteristics and high levels of bioactive and antioxidant compounds have been developed by applying US as an assisted fermentation process. Different whey–oat beverage formulations were prepared and sonicated (40 kHz) at 0, 3, and 10 min before fermentation with
L. casei 431, having microbial population as a response variable. Results indicated that fermented beverage with a whey–oat ratio of 50:50 and US-treated for 3 min presented the highest growth of
L. casei (7–8.85 Log CFU/mL), high antioxidant activity and good sensory acceptance, considering it as a probiotic beverage with potential health benefits
[81]. It could be possible that US induced the acceleration of microbial metabolisms because of the cavitation effects improving the mass transfer of substrates into the cell.
3.3. High Hydostratic Pressure
HHP treatment is a non-thermal technique based on applying elevated pressures (100–800 MPa) to solid or liquid foods for a short time (3–15 min). The pressure is transmitted uniformly and quasi-instantaneously to the product through a non-compressible pressure-transmitting medium, usually water, at low or ambient temperature, avoiding the loss of biological components
[82][83][84][85]. The HHP process has been successfully applied over the years to deliver safe and nutritious foods with functional properties and fresh taste
[86].
Unlike PEF and US, HHP has been mainly used as a preservation process of fermented beverages, demonstrating the inactivation of bacteria and yeasts, keeping their quality attributes and nutritional composition. The effectiveness of HHP on microbial inactivation mainly depends on the pressure level applied, the come of time (CUT) and processing time, microorganisms characteristics and food matrix composition
[82][83][84][85]. Rios-Corpio et al.
[87] compared the effect of the HHP (500, 550, 600 MPa, 5–10 min) with thermal pasteurization (63 °C/min; 72 °C/15 s) on the microbiological, physicochemical, antioxidant, and sensory characteristics of a fermented pomegranate beverage during 42 d of storage at 4 °C. HHP- or heat-treated beverages were microbiologically stable along the storage; nonetheless, HHP-processed beverages had higher levels of antioxidant compounds immediately after processing (399.22 mg GA/100 mL of total phenolic compounds and 121.54 mg of Q/100 mL of total flavonoids). Likewise, HHP (500 MPa, 2 min) process applied to a fermented litchi juice with
Lactobacillus casei did not affect its color and flavor characteristics and retained more phenolic compounds and antioxidant capacity, presenting higher quality attributes than the juice conventionally treated
[88].
Other studies have focused on evaluating the microbial viability of fermented beverages after HHP, which in some cases is highly desirable due to their probiotic properties. Pega et al.
[89] assessed the impact of HHP at 200 and 400 MPa for 10 min and 1 min, respectively, on the evolution of the starter LAB used to prepare a sweet whey-fermented beverage. The authors observed that fermented beverages retained flavor and texture attributes immediately after processing and during 45 d of storage. Furthermore, it was reported that HHP at 200 MPa for 10 min kept an optimal concentration of total starter microorganisms, resulting in a fermented beverage with potential health benefits due to the viability of lactic acid bacteria. As can be seen, it is important to find the optimal pressure level to be applied as sublethal conditions for each specific microorganism. Hence, more research is needed in this area to optimize processing parameters and obtain fermented beverages with probiotic properties.
Some authors also investigated the use of HHP as a pre-treatment in milk as raw material to produce low-fat yogurt with a similar texture and consistency to the full-fat product. HHP processes (100–300 MPa or 600–700 MPa) of milk used for yogurt elaboration induced an increase in viscosity values, amino acid content and better texture profile of the obtained yogurt compared to the yogurt prepared from the untreated milk
[90][91]. It is well known that when milk is subjected to HHP, the casein micelle fragments into smaller subunits with better aggregating properties
[90], resulting in a product with an improved texture profile. Therefore, milk pre-treatment with HHP could be a potential alternative to produce yogurts with low-fat content and high-quality attributes, similar to those prepared with conventional ingredients.