Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mario-Adrian Tienda-Vazquez | -- | 4090 | 2023-06-20 01:38:05 | | | |
| 2 | Dean Liu | -9 word(s) | 4081 | 2023-06-20 05:29:37 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Tienda-Vázquez, M.A.; Hanel, J.M.; Márquez-Arteaga, E.M.; Salgado-Álvarez, A.P.; Scheckhuber, C.Q.; Alanis-Gómez, J.R.; Espinoza-Silva, J.I.; Ramos-Kuri, M.; Hernández-Rosas, F.; Melchor-Martínez, E.M.; et al. Exosomes in Treatment of Skin Disorders. Encyclopedia. Available online: https://encyclopedia.pub/entry/45817 (accessed on 08 May 2026).
Tienda-Vázquez MA, Hanel JM, Márquez-Arteaga EM, Salgado-Álvarez AP, Scheckhuber CQ, Alanis-Gómez JR, et al. Exosomes in Treatment of Skin Disorders. Encyclopedia. Available at: https://encyclopedia.pub/entry/45817. Accessed May 08, 2026.
Tienda-Vázquez, Mario Adrián, Juan Manuel Hanel, Elsa Margarita Márquez-Arteaga, Ana Paola Salgado-Álvarez, Christian Quintus Scheckhuber, José Rafael Alanis-Gómez, Janette Ivone Espinoza-Silva, Manuel Ramos-Kuri, Fabiola Hernández-Rosas, Elda M. Melchor-Martínez, et al. "Exosomes in Treatment of Skin Disorders" Encyclopedia, https://encyclopedia.pub/entry/45817 (accessed May 08, 2026).
Tienda-Vázquez, M.A., Hanel, J.M., Márquez-Arteaga, E.M., Salgado-Álvarez, A.P., Scheckhuber, C.Q., Alanis-Gómez, J.R., Espinoza-Silva, J.I., Ramos-Kuri, M., Hernández-Rosas, F., Melchor-Martínez, E.M., & Parra-Saldívar, R. (2023, June 20). Exosomes in Treatment of Skin Disorders. In Encyclopedia. https://encyclopedia.pub/entry/45817
Tienda-Vázquez, Mario Adrián, et al. "Exosomes in Treatment of Skin Disorders." Encyclopedia. Web. 20 June, 2023.
Copy Citation
Exosomes are nano-sized extracellular vesicles that can be isolated and purified from different sources such as blood, urine, breast milk, saliva, urine, umbilical cord bile cells, and mesenchymal stem cells. They have bioactive compounds that, thanks to their paracrine activity, have proven to be effective as anti-inflammatory agents, inducers of macrophage polarization and accelerators of skin repair and regeneration, reducing the possible complications relating to poor wound repair, and prolonged inflammation.
exosomes
mesenchymal stem cell-derived
wound care
skin damage
1. Introduction
The skin is known as the largest organ, representing about eight percent of the body’s weight [1]. The skin covers the surface of the body, belonging to the integumentary system, and is derived from the ectoderm. Its main function is to act as a physical barrier against the external environment [2], protecting internal organs, bones, muscles, and other soft structures from pathogenic microorganisms, chemicals, loss of water, electrolytes, and thermal homeostasis has an average surface area of 1.5 m2 to 2.0 m2 and an average thickness of 0.3 cm [2][3][4]. This organ is divided into different layers, and in order from the outermost to the innermost are the epidermis, dermal-epidermal junction, dermis, and subcutaneous cell tissue, mostly known as hypodermis, which, in turn, are subdivided into the sublayers described below (Figure 1) [3][4].
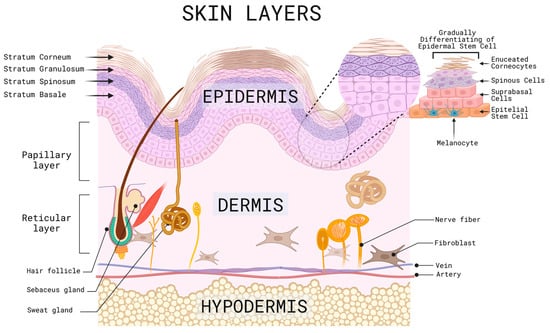
Figure 1. Representative scheme of the different layers, sublayers and cells of the skin. Created with BioRender.com (accessed on 2 June 2023).
The epidermis is the outermost layer of the skin, which makes it susceptible to the environment, with a risk of injury from factors such as UV solar radiation, trauma, ulcers, and surgical procedures, among others. It is composed of stratified squamous epithelium, melanocytes, and keratinocytes, which are in constant change and renewal and are induced to multiply by tissue injury or after a specific stimulation that requires tissue restoration, gradually differentiating from stem cells into suprabasal cells, spinous cells, cells with keratohyalin granules and finally enucleated corneocytes, as they migrate to the skin’s surface and accumulate keratin that is released daily. This process gives rise to several sublayers, including the stratum corneum, which is the outermost layer where desquamation occurs due to the detachment of these cells, followed by the stratum spinosum and the stratum granulosum, and finally, the basal layer or stratum germinativum, which is the deepest layer where the melanocyte precursor melanoblasts are located; these are in contact with keratinocytes through their dendrites, which are in charge of producing melanin, that determine the color of the skin, hair, and provides protection against UV rays [5][6][7][8].
The dermal-epidermal junction is a membrane that lies between the basal membrane of the epidermis and the papillary layer of the dermis and is composed of keratinocytes and fibroblasts, sheets and filaments of keratin, collagen IV, and proteoglycans. Its functions are to provide support, scaffolding, and adhesion to its underlying layers, in addition to participating in tissue repair processes in healing [9].
The dermis is the middle layer of the skin; it has a thickness of 2 to 4 mm, composed of connective tissue of collagen, elastin and amorphous filamentous reticular fibers, blood vessels, lymphatics, hair follicles, cutaneous nerve receptors, endothelial cells, and cells of Schwann [10][11][12]. It has two sublayers; the superficial dermis, also called the papillary layer, contains an abundant extracellular matrix made up of specialized proteins, glycosaminoglycans, and carbohydrates, while the second sublayer, the reticular layer, made up of the middle dermis and the deep or basal dermis, are cells such as fibroblasts that give firmness and hydration to the skin. In case of an injury at this level, there is no regeneration; instead, there is a repair process which will be described later [13][14]. Finally, the hypodermis or subcutaneous cell tissue is composed mainly of fat tissue and blood vessels [15].
When an injury occurs, time plays a fundamental role in the defense against pathogens, repair, and epithelial regeneration [16]. To repair the lesions, it is necessary to involve other cellular mechanisms, such as chemokines, cytokines, tumor necrosis factor (TNFα), and interleukin-1 (IL-1) as an initial step, and subsequently, growth factors, such as the fibroblast growth factor (FGF) and vascular endothelial growth (VEGF) [1]. Adult skin has two mechanisms to deal with wounds: regeneration and repair. When regeneration occurs, the tissue can replace damaged cells with new ones without leaving a noticeable scar; on the other hand, in the repair process, the damaged tissue presents an inability to produce normal cells resulting in the formation of scars (Figure 2) [15]. Moreover, an ideally healed wound is known to present a normal anatomical structure, function, and appearance after some time, while a minimally healed wound presents a normal anatomical structure but without a functional result [2].
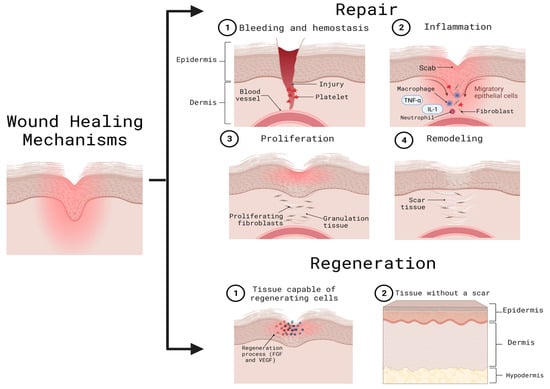
Figure 2. Wound healing mechanisms. The repair mechanism is carried out through hemostasis (an inflammatory phase) re-epithelialization or proliferation, and tissue remodeling. After this, the regeneration process is responsible for the formation of new cells without leaving an appreciable scar. Both repair and regeneration include the action of cytokines, chemokines, and growth factors. FGF: fibroblast growth factor; IL-1: interleukin-1; TNFα: tumor necrosis factor; VEGF: vascular endothelial growth. Created with BioRender.com (accessed on 2 June 2023).
The healing process starts right after an injury is made. It begins with clot formation by the platelets, better known as hemostasis, followed by an inflammatory phase involving an immune response, where neutrophils remove necrotic tissue and bacteria, and macrophages facilitate cytokine and growth factor transformation [2]. Over the following days, a proliferation phase called the re-epithelization process occurs; this consists of the proliferation, migration, differentiation, and stratification of keratinocytes to the center of the trauma creating a new epithelium. The last stage of this process, which can vary in duration between weeks and years, is called the remodeling phase, and this is characterized by the presence of fibroblasts and collagen, fibers which lead to the maturation of the wound and achieve the tissue’s complete recovery [1][2]. As previously described, the re-epithelialization process is essential when a wound occurs as cellular exhaustion is generated; this causes a stimulus to recruit hair follicle cells and produce epidermal cells [5].
Extracellular vesicles are responsible for various physiological processes such as intercellular communication and tissue repair. There is a subset of extracellular vesicles called exosomes, whose size ranges from 30 to 200 nm, which can carry different biomolecules, such as proteins, lipids, and nucleic acids (RNA and DNA). These biomolecules are specific cargoes that depend on the parental cell, that is, the cell that produced the exosomes. Exosomes act as messengers, transporting the biomolecules that can selectively be taken up by recipient cells in order to influence various cellular processes, such as gene expression, cell-to-cell signaling, immune response, and tissue repair. They have the advantage of being able to travel through body fluids such as blood, saliva, and urine, among others, allowing them to reach distant cells and tissues [17][18][19][20][21][22]. Due to their ability to transfer biological information and mediate tissue repair processes, exosomes are a promising strategy to repair, regenerate, and treat skin disorders.
2. Exosome Therapy
As previously described, there are various skin disorders that can trigger immunosuppression, DNA mutations, ROS production, and inflammation; currently seeking alternatives that can combat these disorders and avoid their consequences. Extracellular vesicles are vesicles that are secreted by cells in order to communicate and, in turn, regulate physiological processes such as tissue repair. They have the advantage of being able to easily cross biological barriers such as the blood–brain barrier and the cell membrane. Within the extracellular vesicles, researchers found a subgroup called exosomes; they are nano-sized extracellular vesicles that typically range between 30 and 200 nm [23]. Exosomes are formed (Figure 3) by a lipidic bilayer that can accept both hydrophobic and hydrophilic drugs [24]. On their surface, they contain immune regulatory molecules, membrane proteins, and membrane trafficking molecules, which can help the exosome to attach or ignore the site of interest, turning them into selective extracellular vesicles for the delivery of the biomolecules contained inside, leading to cell communication. The exosome can transport mRNA, nucleic acids, or protein chaperones if they carry substances that are instead lipids, proteins or have a cytoplasmic content; they are accepted by recipient cells and can lead to the modification of pathological or physiological functions of the target cells as a result of exosome-specific cell interactions [25]. Exosomes deposit their content to target cells through different pathways, a ligand-receptor for subsequent activation of signaling pathways, pinocytosis, phagocytosis, and fusion with the plasma membrane [26].
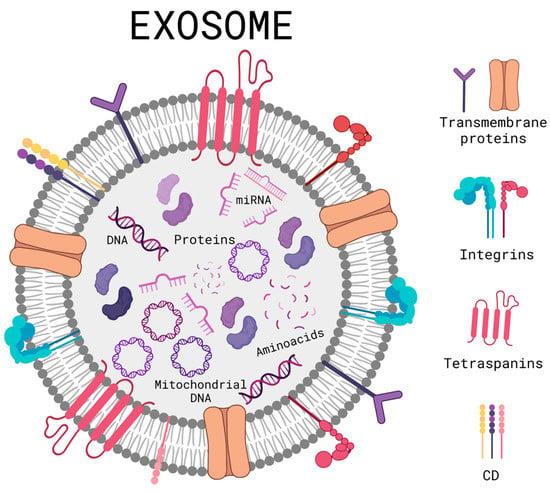
Figure 3. Schematic diagram of a general structure of an exosome. Created with BioRender.com (accessed on 2 June 2023).
The biogenesis of exosomes is based on the invagination of the surface of the cell membrane, and subsequently, primary endocytosis occurs; with the help of lipids, extracellular and membrane proteins are internalized to fuse and form early endosomes. Later they mature and become late endosomes, and, in this stage of maturation, they undergo more invaginations, achieving the specific encapsulation of certain nucleic acids and proteins, producing intraluminal vesicles and later becoming multivesicular bodies. Depending on the biomolecules encapsulated and expressed on the surface, they define the fate of these vesicles within or outside the cell; if their destination is inside the cell, they can be degraded by proceeding to the lysosomes and, on the other hand, if their destination is outside the cell, they first translocate to the membrane before being guided by RAB GTPase proteins with the dynamic support of cytoskeletal proteins. Then, they dock, and membrane fusion occurs, mediated by the N-ethylmaleimide-sensitive fusion attachment protein receptor (SNARE) family of soluble proteins, which subsequently expel them out of the cell and, when released, they are called exosomes [18][20][22]. However, multivesicular bodies do not always follow this route; they can deviate and generate hybrid molecules called “amphisomes” when fused with autophagosomes to later expel exosomes with a different biological load. This is one of the concerns and limitations of the use of exosomes as therapy, and the lack of heterogeneity can activate different undesired physiological processes [27].
Exosomes are commonly used for controlled drug delivery, biomarkers, tissue repairing agents, and even as an important part in the development of vaccines [24]. Exosomes have also been widely used as part of skin care products for anti-wrinkle, anti-aging, general hair, skincare, and wound healing. Exosomes can transport biologically active molecules such as nucleic acids, carbohydrates, proteins, antigens, and other molecules which can be used for different therapeutic treatments such as the regulation of skin pigmentation by using exosomes secreted by melanocytes or the influence of melanin production through exosomes secreted by skin cells through the mediation of paracrine, endocrine, and autocrine effects [28]. Exosomes can be harvested from several fluids and tissues of both human and animal origin, such as blood, urine, breast milk, saliva, urine, bile, and umbilical cord cells [23]. Depending on their parental cell, exosomes contain different biomolecules, such as proteins, lipids, or nucleic acids. The said cargo can also result from the exosome’s intercellular release site and the physiological condition of its cell [25].
2.1. Exosome Purification and Isolation
In order to use exosomes, they must be purified, removing unwanted molecules such as proteins, protein aggregates, and RNA granules, as these additional molecules can affect the exosome’s function, leading to unexpected effects. The technique used to separate these molecules can have an effect on the exosome’s functionality, either by damaging the exosomes themselves or by not properly eliminating certain compounds. These processes attempt to reduce the substance’s complexity due to its composition with a minimal loss of yield [29]. To isolate the exosomes, there are a plethora of methods, such as differential ultracentrifugation (DC), which are normally used to separate exosomes from biological fluids; this is based on the differences in size and density present within the exosomes and any unwanted molecules. This method has low effectiveness; the material obtained is highly heterogeneous, but its purity is notably low, and this process can also isolate unwanted molecules of a similar size and density as exosomes, affecting the compound’s function. The G-force applied alongside the centrifuge’s motor itself can also affect the result, as the process has low repeatability. Density-gradient centrifugation (DGC) is another purifying method; in addition to a centrifuge, a gradient of sucrose is used, which enables the separation of non-vesicular components. This process yields high purity while potentially isolating specific exosome subtypes. This method, however, can prove to be time-consuming, and there is a high risk of sample loss during the handling of the material. Density-cushioned ultracentrifugation (DCGC) can be used after DGC to ensure high purity in the resulting substance with large volume samples. Sedimentation velocity and buoyant density are considered to identify and eliminate unwanted molecules. Both the biological activity and physical integrity of the exosomes are maintained [30][31][32]. The affinity isolation method tags specific protein bodies on the substance, such as antibodies or peptides. Exosomes can be isolated from biofluids and cell culture media. This process yields the expected results; however, the surface proteins and the functionality of the exosomes might suffer damage. Other methods, such as polymer precipitation, have been used, and while this method is fast and suitable for large-volume samples [32], it yields a notably impure and easily contaminated solution, resulting in a less-than-ideal process [33].
2.2. Exosomes Derived from Mesenchymal Stem Cells (MSC-EXO) and Their Application in Different Skin Disorders
Mesenchymal stem cells (MSCs) can be isolated from different human tissues, such as umbilical cord blood or tissue, placenta tissue, and the spinal cord, and they are considered and called multipurpose cells [34]. This type of cells can differentiate into chondrocytes, osteocytes, and adipocytes, among others, depending on their origins, and because of this capacity and the way they can reproduce themselves, MSCs have gained the attention of researchers to start finding new treatments for different diseases [23]. MSCs are preferred for being a source of therapeutic exosomes due to their safeness and immunomodulatory properties [35]. MSCs secrete exosomes and other biomolecules that have been demonstrated to be beneficial in the repair of the attribute in paracrine signaling, which is the one that includes vesicles and exosomes [34]. These MSCs-EXOs contain different biomolecules that help them to perform specific functions such as lipids, proteins, and RNA, which play the role of maintaining their integrity and stability to enhance recognition and targeting on the surface and to improve their potential for controlling late translation and transcription receptors, respectively. MSCs-EXO present qualities similar to no other exosomes: they are small, have good biocompatibility, low immunogenicity, and have a long-circulating life. MSCs-EXO isolated from breast milk have shown stability for around 90 days [36], as opposed to other biomolecules used for drug delivery, such as neutrophils which have a much shorter life expectancy of around 16 h [37]. MSCs-EXO also penetrates easily, making them a great fit for drug delivery, immune therapies, and other therapies [23]. Due to these characteristics of exosomes, they can be used for various applications, including tissue regeneration and wound healing, as demonstrated by researchers using MSC-EXO obtained from placental umbilical cords [34]. In addition, MSC-EXO secondarily regulates the expression of growth factors, activating signaling pathways, such as phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT), the extracellular signal-regulated kinase cascade (ERK) or Wnt/β-catenin, resulting in re-epithelialization by cell proliferation and migration and angiogenesis [35].
MSCs-EXOs are considered of great interest due to their regenerative and immunomodulatory functions, leading to the development of anti-aging, wound healing, and anti-inflammatory treatments. The use of MSCs differs depending on the tissue from which they are originally extracted and can be used as regulators of immune responses through their differentiation into cells such as macrophages and natural killer cells [23]. Specific MSCs have proven their efficiency in treatments such as the use of ASC-exosomes when derived from adipose tissue in the treatment of atopic dermatitis [38] and Umbilical Cord Blood MSCs for the treatment of cutaneous wounds [39]. Therefore, it is important to consider the specific uses that different MSCs can have due to their tissue of origin.
The use of MSC-EXO has also been shown to lead to the polarization of the M2 macrophage phenotype, ensuring tissue repair, regardless of diseases such as diabetes. Ti et al. [40] found that human umbilical cord mesenchymal cells previously treated with lipopolysaccharides showed promising results through the regulation of the TLR4/NF-KB/STAT3/AKT signaling pathways and, specifically, TLR4 was downregulated while STAT3/AKT was upregulated. This model was applied to diabetic rats with cutaneous excisions, and after three days of exosome applications, the expression level of M2 macrophages ended up higher than M1 macrophages at the site of the wound [40]. Human bone marrow stromal cells have also shown great potential in the treatment of diabetic wounds through M2 macrophage polarization. Liu et al. [41] found that the inflammatory phase, or M1 macrophage phase, was inhibited by the expression of the PTEN gene, resulting in the activation of the PI3K and AKT pathways.
In the case of cutaneous injuries, several other mechanisms have been studied for their use of exosomes. Macrophage polarization has been used, as discussed previously; however, other methods were applied, such as the inhibition of white blood cells, keratinocytes, and fibroblasts, as well as the downregulation of the TLR4 pathway, which resulted in the lower production of inflammatory factors, enhancing the speed of wound healing [2][42][43].
Activating protein-1 (AP-1) refers to transcription factors, including activating transcription factor subunits: Jun and Fos dimers that bind to a specific site on DNA molecules known as the AP-1 binding site. Different factors regulate the transcription process of specific genes, leading to different physiological functions, such as keratinocyte proliferation and differentiation, cellular proliferation, apoptosis, and even cancerous transformation. The activity of AP-1 is up-regulated via the interaction of specific protein kinases such as MAPK and transcriptional coactivators. Protein interactions can also play an important role in a reduction in the transcription process. The activation of AP-1 activity can result in an extracellular stimulus or the exposure and activation of AP-1 proteins such as c-Jun, which, in turn, are regulated by the control of gene transcription. The function of activation proteins can also lead to the transcription of metalloproteinase genes, which degrade collagen and elastin, impairing the skin’s flexibility and elasticity [44].
MAPK can regulate AP-1, both during transcription processes and post-transcription, affecting the molecule’s ability to bind to DNA chains [45]. An increase in MAPK activity results in an increase in AP-1 activity, enhancing the ability of the transcription factors to bind to DNA molecules and trigger the transcription process, allowing the formation of RNA and, eventually, specific protein compounds [44]. On the other hand, molecules such as glycogen synthase kinase 3-β (GSK3-β) act as down regulators for AP-1 [46].
The MAPK signaling pathways represent pathways that activate and phosphorylate proteins, giving them the required energy to realize their function [47]. The pathway’s activation requires the binding of a ligand to a receptor known as RTK; the receptor catalyzes phosphorylation itself, then, growth factors bind to the phosphorylated RTK. Through a binding protein, a Ras molecule could be energized, exchanging its original GDP molecule with GTP and activating the protein. The activated Ras can then bind to effector proteins, such as B-Raf, which gives energy to MEK 1 and 2 via phosphorylation and, in turn, phosphorylate ERK 1 and 2. This can lead to the activation of the AP-1 factors Fos and Jun. Once activated, the factors bind to an AP-1 receptor in a chain of DNA, leading to the expression of different genes (Figure 4).
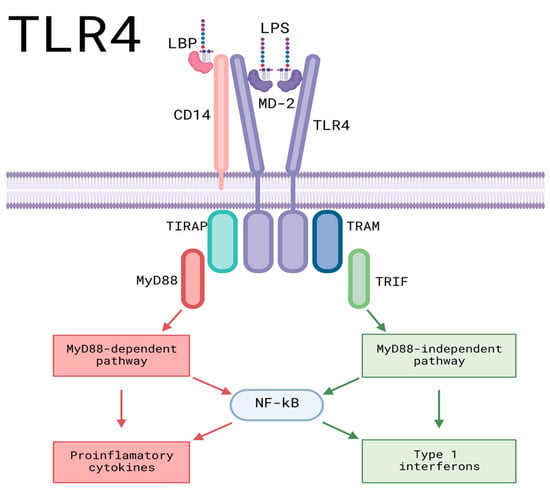
Figure 4. Pro-inflammatory signaling pathways of TLR4. LPS and LBP bind, leading the formed molecule toward CD-14, binding LPS and CD-14. It then interacts with the TLR4/MD-2 complex before being transported to the TLR4 monomers and leading to the dimerization of TLR4, activating intracellular TIR domains, which results in the activation of downstream proteins for signaling. TIRAP binds to the TIR domain, allowing the binding and activation of the myeloid differentiation molecule My-D88, leading to the My-D88-dependent pathway, which culminates in the activation of proinflammatory cytokines. TRAM can also bind to the TIR domain, resulting in the My-D88-independent pathway, culminating in the production of Type 1 interferons. AP-1: activating protein-1; CD14: cluster of differentiation 14; IKKs: IκB kinase complex; IL-1β: Interleukin-1 beta; IRAK-1: interleukin-1 receptor-associated kinase; IRAK-4: interleukin-4 receptor-associated kinase; IRF5: Interferon regulatory factor 5; IκBζ: NF-kappa-B inhibitor zeta; LBP: LPS-Binding Protein; LPS: Lipopolysaccharide; MAPK: Mitogen-activated protein kinase; MD-2: Myeloid Differentiation Factor 2; MyD88: Myeloid differentiation Factor 88; NFKB: Nuclear factor kappa B; TAK1: Transforming growth factor β-activated kinase 1; TIRAP: Toll-interleukin domain-containing adapter protein; TLR4: Toll-like receptor 4; TNF-α: Tumor Necrosis Factor-alpha; TRAF6: TNF receptor-associated factor 6; TRAM: Translocating chain-associating membrane protein; TRIF: TIR domain-containing adaptor-inducing interferon-β. Created with BioRender.com (accessed on 2 June 2023).
Gao et al. [48] reported that the levels of ERK, Jun, and Fos phosphorylation were elevated by UVB radiation, leading to increased transcription of MMPs and, specifically, MMP-1. To lower the production rate of MMPs, miR-1246-over expressing exosomes were used to suppress the activity of both the AP-1 and MAPK signaling pathways through the suppression of phosphorylated Fos and Jun production. The levels of p-ERK, p-Jun, and p-Fos were decreased by 66.5%, 70.6%, and 70.1%, respectively, greatly lowering the rate of MMP-1 transcription and the degradation of collagen and elastin through the manipulation of both the MAPK and AP-1 pathways. Li et al. [49] used a stem cell conditioned medium to facilitate organ and tissue repair through a decrease in the phosphorylation of Fos and Jun in the MAPK/AP-1 cascade, avoiding the transcription through AP-1 and the production of MMP1 along proinflammatory molecules such as IL-6.
On the other hand, considering the effects of UV radiation, MSCs-EXOs were used to counteract oxidative stress and skin aging by degrading MMPs and down-regulating the production of reactive oxygen species. Gao et al. [48] used miR-1246-overexpressing exosomes, which led to the reversal of the effects triggered by excessive UVB radiation, such as the production of reactive oxygen species, the secretion of MMPs, and the degradation of collagen. The transported miR-1246 led to the upregulation of TGF-β1, which promoted the synthesis of collagen and elastin. This molecule also leads to M2 macrophage polarization via the activation of the STAT3 signaling pathway.
Studies regarding in vitro models of keratinocytes damaged by oxidative stress with H2O2 and in vivo UV-irradiated mice, to which MSC-EXO was used, observed a decrease in ROS production and a decrease in radiation-induced DNA damage. In addition, aberrant calcium signaling and mitochondrial changes, as well as inflammation, were decreased. The authors demonstrated that MSC-EXO had antioxidant capacities, acting through the Nuclear factor erythroid 2-related factor 2 (NRF2) pathway; the size of the exosomes used could determine them as powerful nanotherapeutic agents against oxidative stress models [50].
Exosomes have been used in clinical trials for the treatment of various diseases, used as biomarkers, exosome vaccines, drug delivery systems, and exosome therapy [27]. The ClinicalTrials.gov website (https://clinicaltrials.gov/ accessed on 6 June 2023) lists different clinical trials, their current status, and the pathology for which they are used. A search was carried out with the term “exosome”, excluding conditions or diseases that were not related to the skin, and the following results were shown hair loss, alopecia, psoriasis, squamous cell carcinoma of the head and neck, melanoma, diabetic foot, wounds, and injuries. One of the clinical trials with already published results was the use of exosomes to treat patients with stage III/IV metastatic melanoma, with the inclusion criteria of HLA-DPO4+, -B35+, or HLA-A1+, stage IIIB, and IV leukocyte phenotype, and tumors expressing the MAGE3 antigen. Autologous exosomes, isolated from dendritic cells, were used to immunize the patients. Their results showed no adverse effects, in addition to tumor regression in lymph nodes and skin lesions, as well as the depigmentation of the naevi and loss of the tumor’s class I major histocompatibility complex [51].
To use exosomes in clinical trials, good manufacturing practices must be followed, which are governed by safety concerns; these practices monitor and divide the process into three major steps: the production, purification/isolation, and characterization of the exosome, always maintaining sterility and ensuring the quality and heterogeneity of the exosomes. For the production process, different types of cells can be used, such as MSCs, dendritic cells derived from monocytes, and adipose tissue-derived stem cells (ADSC), among others, in order to induce the formation and release of exosomes, the system, the environment, and the cell culture medium, as well as the dissociation enzyme, there are several methods to purify and isolate exosomes. In general, terms, what regulates the good manufacturing practices of this process is the elimination of unwanted cell debris, the concentration of the conditioning medium, and the isolation of exosomes. The last step is the characterization of the exosome, which is based on ensuring that exosomes are isolated through validation or biological and physicochemical characterization, measuring bioactivity, observing structure, and finally, measuring adsorption and protein content [52].
References
- Li, P.; Guo, X. A Review: Therapeutic Potential of Adipose-Derived Stem Cells in Cutaneous Wound Healing and Regeneration. Stem Cell Res. Ther. 2018, 9, 305.
- Tottoli, E.M.; Dorati, R.; Genta, I.; Chiesa, E.; Pisani, S.; Conti, B. Skin Wound Healing Process and New Emerging Technologies for Skin Wound Care and Regeneration. Pharmaceutics 2020, 12, 735.
- Yadav, N.; Parveen, S.; Chakravarty, S.; Banerjee, M. Skin Anatomy and Morphology; Springer: Berlin/Heidelberg, Germany, 2019; pp. 1–10. ISBN 978-981-13-2540-3.
- Kerns, M.L.; Chien, A.L.; Kang, S. Skin Aging. In Fitzpatrick’s Dermatology, 9e; Kang, S., Amagai, M., Bruckner, A.L., Enk, A.H., Margolis, D.J., McMichael, A.J., Orringer, J.S., Eds.; McGraw-Hill Education: New York, NY, USA, 2019.
- Bennion, S. Structure and Function of the Skin. In Dermatology Secrets E-Book; Elsevier: Amsterdam, The Netherlands, 2020; pp. 4690–4696.
- Jia, Q.; Nash, J.F. Pathology of Aging Skin BT—Textbook of Aging Skin; Farage, M.A., Miller, K.W., Maibach, H.I., Eds.; Springer: Berlin/Heidelberg, Germany, 2017; pp. 363–385. ISBN 978-3-662-47398-6.
- Gökçe, B.; Güngör, S. Nanocarrier-Mediated Follicular Targeting; Elsevier: Amsterdam, The Netherlands, 2020; pp. 305–326. ISBN 9780128222867.
- Freeman, S.C.; Sonthalia, S. Histology, Keratohyalin Granules; StarPerarls Publishing: Treasure Island, FL, USA, 2023.
- Roig-Rosello, E.; Rousselle, P. The Human Epidermal Basement Membrane: A Shaped and Cell Instructive Platform That Aging Slowly Alters. Biomolecules 2020, 10, 1607.
- Shpichka, A.; Butnaru, D.; Bezrukov, E.A.; Sukhanov, R.B.; Atala, A.; Burdukovskii, V.; Zhang, Y.; Timashev, P. Skin Tissue Regeneration for Burn Injury. Stem Cell Res. Ther. 2019, 10, 94.
- Brown, T.M.; Krishnamurthy, K. Histology, Hair and Follicle; StarPerarls Publishing: Treasure Island, FL, USA, 2023.
- Madden, J.; O’Mahony, C.; Thompson, M.; O’Riordan, A.; Galvin, P. Biosensing in Dermal Interstitial Fluid Using Microneedle Based Electrochemical Devices. Sens. Bio-Sens. Res. 2020, 29, 100348.
- Deniz, A.A.H.; Abdik, E.A.; Abdik, H.; Aydın, S.; Şahin, F.; Taşlı, P.N. Zooming in across the Skin: A Macro-to-Molecular Panorama. In Cell Biology and Translational Medicine; Springer: Berlin/Heidelberg, Germany, 2020; pp. 157–200.
- Stan, D.; Tanase, C.; Avram, M.; Apetrei, R.; Mincu, N.-B.; Mateescu, A.L.; Stan, D. Wound Healing Applications of Creams and “Smart” Hydrogels. Exp. Dermatol. 2021, 30, 1218–1232.
- Lopes, B.; Sousa, P.; Alvites, R.; Branquinho, M.; Sousa, A.; Mendonça, C.; Atayde, L.M.; Maurício, A.C. The Application of Mesenchymal Stem Cells on Wound Repair and Regeneration. Appl. Sci. 2021, 11, 3000.
- Takeo, M.; Lee, W.; Ito, M. Wound Healing and Skin Regeneration. Cold Spring Harb. Perspect. Med. 2015, 5, a023267.
- Zhang, Y.; Liu, Y.; Liu, H.; Tang, W.H. Exosomes: Biogenesis, Biologic Function and Clinical Potential. Cell Biosci. 2019, 9, 19.
- Jella, K.K.; Nasti, T.H.; Li, Z.; Malla, S.R.; Buchwald, Z.S.; Khan, M.K. Exosomes, Their Biogenesis and Role in Inter-Cellular Communication, Tumor Microenvironment and Cancer Immunotherapy. Vaccines 2018, 6, 69.
- Kalluri, R.; LeBleu, V. The Biology, Function, and Biomedical Applications of Exosomes. Science 2020, 367, eaau6977.
- Xu, M.; Ji, J.; Jin, D.; Wu, Y.; Wu, T.; Lin, R.; Zhu, S.; Jiang, F.; Ji, Y.; Bao, B.; et al. The Biogenesis and Secretion of Exosomes and Multivesicular Bodies (MVBs): Intercellular Shuttles and Implications in Human Diseases. Genes Dis. 2022; in press.
- Kumari, M.; Anji, A. Small but Mighty—Exosomes, Novel Intercellular Messengers. Biology 2022, 11, 413.
- Gurung, S.; Perocheau, D.; Touramanidou, L.; Baruteau, J. The Exosome Journey: From Biogenesis to Uptake and Intracellular Signalling. Cell Commun. Signal. 2021, 19, 47.
- Ha, D.H.; Kim, H.-K.; Lee, J.; Kwon, H.H.; Park, G.-H.; Yang, S.H.; Jung, J.Y.; Choi, H.; Lee, J.H.; Sung, S.; et al. Mesenchymal Stem/Stromal Cell-Derived Exosomes for Immunomodulatory Therapeutics and Skin Regeneration. Cells 2020, 9, 1157.
- Huda, M.N.; Nafiujjaman, M.; Deaguero, I.G.; Okonkwo, J.; Hill, M.L.; Kim, T.; Nurunnabi, M. Potential Use of Exosomes as Diagnostic Biomarkers and in Targeted Drug Delivery: Progress in Clinical and Preclinical Applications. ACS Biomater. Sci. Eng. 2021, 7, 2106–2149.
- McBride, J.D.; Rodriguez-Menocal, L.; Badiavas, E.V. Extracellular Vesicles as Biomarkers and Therapeutics in Dermatology: A Focus on Exosomes. J. Investig. Dermatol. 2017, 137, 1622–1629.
- Girón, J.; Maurmann, N.; Pranke, P. The Role of Stem Cell-Derived Exosomes in the Repair of Cutaneous and Bone Tissue. J. Cell Biochem. 2022, 123, 183–201.
- Rezaie, J.; Feghhi, M.; Etemadi, T. A Review on Exosomes Application in Clinical Trials: Perspective, Questions, and Challenges. Cell Commun. Signal. 2022, 20, 145.
- Xiong, M.; Zhang, Q.; Hu, W.; Zhao, C.; Lv, W.; Yi, Y.; Wang, Y.; Tang, H.; Wu, M.; Wu, Y. The Novel Mechanisms and Applications of Exosomes in Dermatology and Cutaneous Medical Aesthetics. Pharmacol. Res. 2021, 166, 105490.
- Street, J.M.; Koritzinsky, E.H.; Glispie, D.M.; Yuen, P.S.T. Urine Exosome Isolation and Characterization. Methods Mol. Biol. 2017, 1641, 413–423.
- Li, K.; Wong, D.K.; Hong, K.Y.; Raffai, R.L. Cushioned–Density Gradient Ultracentrifugation (C-DGUC): A Refined and High Performance Method for the Isolation, Characterization, and Use of Exosomes. Extracell. RNA Methods Protoc. 2018, 1740, 69–83.
- Duong, P.; Chung, A.; Bouchareychas, L.; Raffai, R.L. Cushioned-Density Gradient Ultracentrifugation (C-DGUC) Improves the Isolation Efficiency of Extracellular Vesicles. PLoS ONE 2019, 14, e0215324.
- Chen, J.; Li, P.; Zhang, T.; Xu, Z.; Huang, X.; Wang, R.; Du, L. Review on Strategies and Technologies for Exosome Isolation and Purification. Front. Bioeng. Biotechnol. 2022, 9, 811971.
- Yu, L.L.; Zhu, J.; Liu, J.X.; Jiang, F.; Ni, W.K.; Qu, L.S.; Ni, R.Z.; Lu, C.H.; Xiao, M.B. A Comparison of Traditional and Novel Methods for the Separation of Exosomes from Human Samples. BioMed Res. Int. 2018, 2018, 3634563.
- Hade, M.D.; Suire, C.N.; Suo, Z. Mesenchymal Stem Cell-Derived Exosomes: Applications in Regenerative Medicine. Cells 2021, 10, 1959.
- Sun, Y.; Liu, G.; Zhang, K.; Cao, Q.; Liu, T.; Li, J. Mesenchymal Stem Cells-Derived Exosomes for Drug Delivery. Stem Cell Res. Ther. 2021, 12, 561.
- de la Torre Gomez, C.; Goreham, R.V.; Bech Serra, J.J.; Nann, T.; Kussmann, M. “Exosomics”—A Review of Biophysics, Biology and Biochemistry of Exosomes with a Focus on Human Breast Milk. Front. Genet. 2018, 9, 92.
- Wu, Y.; Liu, Y.; Wang, T.; Jiang, Q.; Xu, F.; Liu, Z. Living Cell for Drug Delivery. Eng. Regen. 2022, 3, 131–148.
- Cho, B.S.; Kim, J.O.; Ha, D.H.; Yi, Y.W. Exosomes Derived from Human Adipose Tissue-Derived Mesenchymal Stem Cells Alleviate Atopic Dermatitis. Stem Cell Res. Ther. 2018, 9, 187.
- Qiu, X.; Liu, J.; Zheng, C.; Su, Y.; Bao, L.; Zhu, B.; Liu, S.; Wang, L.; Wang, X.; Wang, Y.; et al. Exosomes Released from Educated Mesenchymal Stem Cells Accelerate Cutaneous Wound Healing via Promoting Angiogenesis. Cell Prolif. 2020, 53, e12830.
- Ti, D.; Hao, H.; Tong, C.; Liu, J.; Dong, L.; Zheng, J.; Zhao, Y.; Liu, H.; Fu, X.; Han, W. LPS-Preconditioned Mesenchymal Stromal Cells Modify Macrophage Polarization for Resolution of Chronic Inflammation via Exosome-Shuttled Let-7b. J. Transl. Med. 2015, 13, 308.
- Liu, W.; Yu, M.; Xie, D.; Wang, L.; Ye, C.; Zhu, Q.; Liu, F.; Yang, L. Melatonin-Stimulated MSC-Derived Exosomes Improve Diabetic Wound Healing through Regulating Macrophage M1 and M2 Polarization by Targeting the PTEN/AKT Pathway. Stem Cell Res. Ther. 2020, 11, 259.
- Louiselle, A.E.; Niemiec, S.M.; Zgheib, C.; Liechty, K.W. Macrophage Polarization and Diabetic Wound Healing. Transl. Res. 2021, 236, 109–116.
- Ciesielska, A.; Matyjek, M.; Kwiatkowska, K. TLR4 and CD14 Trafficking and Its Influence on LPS-Induced pro-Inflammatory Signaling. Cell Mol. Life Sci. 2021, 78, 1233–1261.
- Eckert, R.L.; Adhikary, G.; Young, C.A.; Jans, R.; Crish, J.F.; Xu, W.; Rorke, E.A. AP1 Transcription Factors in Epidermal Differentiation and Skin Cancer. J. Skin Cancer 2013, 2013, 537028.
- Ye, N.; Ding, Y.; Wild, C.; Shen, Q.; Zhou, J. Small Molecule Inhibitors Targeting Activator Protein 1 (AP-1). J. Med. Chem. 2014, 57, 6930–6948.
- Agron, M.; Brekhman, V.; Morgenstern, D.; Lotan, T. Regulation of AP-1 by MAPK Signaling in Metal-Stressed Sea Anemone. Cell Physiol. Biochem. 2017, 42, 952–964.
- Guo, Y.; Pan, W.; Liu, S.; Shen, Z.; Xu, Y.; Hu, L. ERK/MAPK Signalling Pathway and Tumorigenesis (Review). Exp. Ther. Med. 2020, 19, 1997–2007.
- Gao, W.; Yuan, L.M.; Zhang, Y.; Huang, F.Z.; Gao, F.; Li, J.; Xu, F.; Wang, H.; Wang, Y.S. MiR-1246-Overexpressing Exosomes Suppress UVB-Induced Photoaging via Regulation of TGF-β/Smad and Attenuation of MAPK/AP-1 Pathway. Photochem. Photobiol. Sci. 2022, 22, 135–146.
- Li, L.; Ngo, H.T.T.; Hwang, E.; Wei, X.; Liu, Y.; Liu, J.; Yi, T.H. Conditioned Medium from Human Adipose-Derived Mesenchymal Stem Cell Culture Prevents Uvb-Induced Skin Aging in Human Keratinocytes and Dermal Fibroblasts. Int. J. Mol. Sci. 2020, 21, 49.
- Zhao, G.; Liu, F.; Liu, Z.; Zuo, K.; Wang, B.; Zhang, Y.; Han, X.; Lian, A.; Wang, Y.; Liu, M.; et al. MSC-Derived Exosomes Attenuate Cell Death through Suppressing AIF Nucleus Translocation and Enhance Cutaneous Wound Healing. Stem Cell Res. Ther. 2020, 11, 174.
- Escudier, B.; Dorval, T.; Chaput, N.; André, F.; Caby, M.P.; Novault, S.; Flament, C.; Leboulaire, C.; Borg, C.; Amigorena, S.; et al. Vaccination of Metastatic Melanoma Patients with Autologous Dendritic Cell (DC) Derived-Exosomes: Results of the First Phase 1 Clinical Trial. J. Transl. Med. 2005, 3, 10.
- Chen, Y.S.; Lin, E.Y.; Chiou, T.W.; Harn, H.J. Exosomes in Clinical Trial and Their Production in Compliance with Good Manufacturing Practice. Tzu Chi Med. J. 2020, 32, 113–120.
More
Information
Subjects:
Dermatology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
920
Revisions:
2 times
(View History)
Update Date:
20 Jun 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





