
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jacques Christian Mbongue | -- | 2345 | 2023-06-19 16:43:10 | | | |
| 2 | Sirius Huang | Meta information modification | 2345 | 2023-06-20 03:28:15 | | | | |
| 3 | Jacques Christian Mbongue | + 1602 word(s) | 3947 | 2023-07-18 03:30:18 | | |
Video Upload Options
Oral delivery of tissue-specific autoantigens may be helpful for the clinical prevention of spontaneous autoimmune diabetes. However, the therapeutic potential has been restricted by the need for recurrent delivery of large amounts of autoantigen, and tolerance is often less successful in already sensitized hosts. These limitations were overcome by transporting chemically conjugated autoantigens for the induction of oral tolerance utilizing the nontoxic B subunit of cholera toxin (CTB).
1. Introduction
2. CTB as an Adjuvant
| Antigen. | Fused to CTB | Animal Model, Pathogen or Route | Response | Reference |
|---|---|---|---|---|
| Human Proinsulin | C-terminal | Monocyte-derived Dendritic cells (moDCs) Human, in vitro | Upregulation of IDO1, Tolerogenic effect on DCs | [28][29] |
| Human Glutamic Acid Decarboxylase 55 (GAD55) | C-terminal, Recombinant vaccinia virus | NOD mouse, i.p., +/− CFA | Comparing the same vaccine without CFA to the same vaccine, diabetes was reduced by 50–20%. | [30][31][32] |
| Insulin | C-terminal, +/− GFP | NOD Mouse | 50 % reduction in T1D incidence | [33] |
| Myelin Basic Protein (MBP) | C-terminal | 3X TgAD (Alzheimer’s Disease) Mice, Oral | 70% less amyloid buildup in the hippocampus and cerebral cortex | [34] |
3. CTB in Transgenic Plants
-
How the fusion protein of cholera toxin B subunit and insulin, made from potatoes, prevents the onset of autoimmune diabetes.
It has been demonstrated that oral administration of disease-specific autoantigens can halt or postpone the development of autoimmune disease symptoms[11, 12, 23, 43, 48, 49]. Arakawa et al. have generated transgenic potato plants synthesize human insulin, which is a major insulin-dependent diabetes mellitus autoantigen, at levels up to 0.05% of total soluble protein[2, 3, 29]. In this study, insulin was coupled to the C-terminus of the cholera toxin B subunit (CTB) to facilitate the distribution of plant-produced insulin to the lymphoid tissues associated with the gut. Transgenic potato tubers produced pentameric CTB-insulin fusion, which conserved the native antigenicity of both CTB and insulin, as well as 0.1 percent of the total soluble protein. Nonobese diabetic mice given CTB-insulin fusion protein-infused altered potato tuber tissues showed markedly reduced pancreatic islet inflammation (insulitis) and a postponed onset of clinical diabetes. When insulin- or CTB-producing transgenic potato tissues were fed on their own, neither insulitis nor diabetic symptoms were noticeably lessened. The results of the experiments suggest that food plants can be used as both a source and a vehicle for immune-tolerization against this T cell-mediated autoimmune illness.
The pentameric shape of the CTB fusion protein increases the molar concentration of the antigen per CTB pentamer molecule in addition to enabling site-specific delivery and presentation of conjugated proteins to the GALT. The traditional necessity for high levels of antigen biosynthesis in the food plant for effective vaccine manufacture may be greatly decreased as a result of the enhanced antigen concentration in the GALT. The proinsulin was coupled with CTB via the flexible hinge peptide to minimize steric hindrance between the CTB and insulin moieties and enhance CTB subunit production in plant cells[50].
On the basis of the NOD mice's induction of both systemic and intestinal anti-CTB antibodies after being fed altered potato tubers that contain the CTB-INS fusion protein, it is possible that the fusion protein was successfully transported to the intestine immune system. The discovery of an elevated serum anti-insulin IgG antibody titer provided more evidence in favor of this assertion. The humoral reaction mounted against the CTB and insulin proteins may be a secondary outcome of oral tolerance's stimulation of Th cells [12, 39, 43, 51, 52].
Serum anti-insulin IgG1 antibodies were more frequently induced when mice were fed transgenic potato tissues that produced the CTB-INS fusion protein than IgG2a antibodies, demonstrating that the immune response was biased toward an insulin-specific Th2 lymphocyte response and that the observed oral tolerance was mediated by active suppression [2, 53].
According to this study, autoimmune diabetes can be successfully prevented by giving NOD mice very small amounts of food that contains insulin made by plants and conjugated with a CTB subunit. The significance of oral antigen dose and the contribution of CTB to the induction of oral tolerance are highlighted by the efficacy of the plant-produced CTB-INS fusion protein at doses at least 100 times lower than those generally reported for unconjugated autoantigens[48, 54].
Due to their ability to control tolerance to food antigens, limit reactivity to the gut microbiota, and mediate tolerance to food antigens, dendritic cells (DCs) are essential for an efficient response to intestinal infections. Intestinal DCs can promote regulatory and effector T cell responses despite their diversity. The actions taken by functionally different DC subsets and how environmental cues influence those actions determine the results in a particular circumstance. DCs continuously sample luminal content to look for infections, but the relevance of the various paths by which this occurs is not entirely known. The distinct traits of intestinal DCs are controlled by local host, nutritional, and microbial cues. The ability to produce all-trans retinoic acid (RA) and activate T lymphocytes with gut tropism are two examples of these traits[55]. Due to their ability to activate TGF beta and generate RA, intestinal DC subsets are potent inducible Treg inducers in the steady state. Responses elicited by steady-state intestinal DCs are not entirely regulatory in character, despite the presence of effector T cells with a preference for commensal bacteria in healthy mucosa and their ability to be locally controlled to stop inflammation[56].
To enhance effector responses in an infection or maintain inflammation in a disease, intestinal DCs probably need to both change the current DC population and attract new populations. The etiology of inflammatory bowel disease can be linked to immune pathways, and in inflamed intestinal tissue, DCs show enhanced microbial recognition machinery expression, activation, and mediator synthesis. Intestinal DCs may be targeted for better vaccination responses or disease treatment[57].
Tryptophan is an essential amino acid, and indoleamine 2, 3-dioxygenase (IDO1) is the first and only catabolic enzyme that can degrade it. IDO breaks down the aromatic indole ring of tryptophan, resulting in the production of a number of tryptophan breakdown metabolites known as "kynurenines." These chemicals have been found to significantly influence immune regulation. Since tryptophan must be received through diet, managing how it is metabolized can significantly affect how the body uses energy and how the immune system responds[58]. By depleting tryptophan, reducing cellular energy generally, and producing released kynurenines, which are known to effectively trigger pro-inflammatory T-cell death, indoleamine 2, 3-dioxygenase was discovered to limit DC maturation(Figure 1)[41].
Th17 cells are characterized by their production of interleukin-17 (IL-17) and other pro-inflammatory cytokines. They are involved in immune responses against pathogens at mucosal surfaces, but their dysregulation can lead to chronic inflammation and tissue damage, promoting autoimmune disorders[59].
According to earlier research, transgenic DCs with high levels of IDO1 expression and tryptophan metabolites (such as l-kynurenine, 3-hydroxykynurenine, and 3-hydroxyanthranilic acid) can permanently decrease allogeneic T-cell proliferation in vitro including Th 17[60-62] and lead to an increased recruitment of CD4+CD25+Foxp3+ T regulatory (Treg) cells within the site of antigen presentation[63-65]. IDO1 was found to be significantly up-regulated in vaccine-inoculated DCs in a recent study using immature human dendritic cells (iDC) co-cultured with CTB-INS. The immunosuppressive enzyme's production was greatly boosted by the CTB-INS fusion protein when compared to DCs that had been exposed to proinsulin, CTB, or an unlinked mixture of the two proteins[41].
According to current research, CTB-INS promotes TNFR signaling and induces IDO1 production in human DCs, which may be the mechanism by which it modulates DC activation. Examining IDO1 production caused by CTB-INS in vaccine-stimulated DCs was made possible by blocking the TNFR pathway[42]. In this recent study, peptides comprising the amino acid sequences of the TRAF2, 3 and TRAF6 binding sites for CD40 were treated with monocyte derived DCs. Following that, CD40 ligand (CD154) and CTB-INS were used to activate the DCs. Blocking peptides for CD40-TRAF2, 3, and CD40-TRAF6 were added, and this prevented IDO1 production from being upregulated in response to either CD154 ligand or CTB-INS. Combining TRAF2, 3 and TRAF6 inhibitors resulted in the highest decrease in IDO1 production.
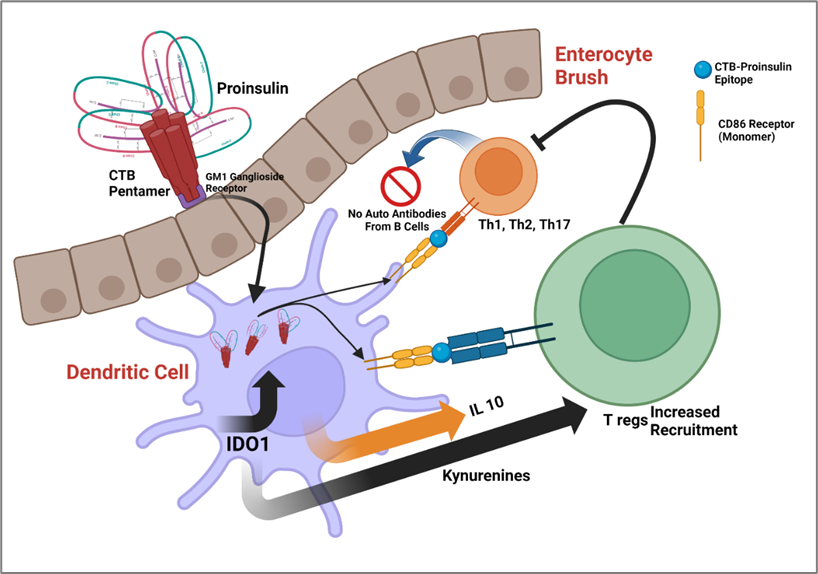
Figure 1. Proposed Mechanism for CTB-INS edible vaccine. Created with BioRender.com. Oral Administration: The CTB-INS vaccine is orally administered, typically through ingestion or ingestion of a plant-based product containing the vaccine. Uptake in the Gut: After oral administration, the vaccine components, including the cholera toxin B subunit (CTB) and the insulin antigen (INS), are taken up by specialized cells in the gut, such as M cells or dendritic cells (DCs). Presentation to Immune Cells: The gut-associated DCs capture the vaccine antigens and migrate to the lymphoid tissues, such as Peyer's patches, mesenteric lymph nodes, or other lymphoid organs. Production of IDO: Indoleamine 2,3-dioxygenase (IDO) is an enzyme involved in the metabolism of tryptophan. The presence of IDO and IL10 induced by the CTB-INS vaccine promotes the migration of Tregs in the gut. IDO plays a key role in immune tolerance by regulating T cell responses and maintaining immune balance through Treg migration, which play a crucial role in immune regulation and tolerance via the inhibition of Th1, Th2 and Th17 and B cell autoantibody production.
The specific immune-unresponsive condition is enhanced in mice after oral administration of the cholera toxin B (CTB) subunit conjugated to the autoantigen insulin. Oral tolerance is what it is known as, and it can suppress autoimmune type 1 diabetes (T1D). The mechanism through which the CTB-insulin (CTB-INS) protein functions as a treatment for T1D in vivo, however, is still unknown. However, recent articles have provided a number of hints. Within organized lymphoid tissue, intestinal cDCs are involved in mediating tolerance to dietary antigens, limiting responsiveness to the gut microbiota, and being necessary for an effective response to intestinal infections. Although intestinal DCs are diverse, they together induce responses from regulatory and effector T cells. In nonobese diabetic (NOD) mice, oral treatment of the CTB-INS protein was recently found to generate exceptional tolerance, postpone the onset of diabetic symptoms, and decrease the start of T1D. Through the non-canonical NF-ĸB pathway, it has been demonstrated to increase levels of the immunosuppressive and tolerogenic enzyme IDO1 in dendritic cells and reduce levels of CD86 costimulatory factors[41, 42]. The by-products of resulting tryptophan catabolism known as kynurenines are known to cause the increase of CD4+CD25+Foxp3+ T regulatory (Treg) cells within the GALT.
-
Conclusion
Edible vaccines are ingested as food and offer advantages in safety and production. They rely on the plant's life cycle and hold potential for controlling diseases. The mechanism of action is still being studied, but evidence suggests that oral administration can induce immune responses and suppress autoimmune diabetes. Intestinal dendritic cells play a role in tolerance and response to infections. While concerns exist about genetically modified plants and cross-contamination, thorough monitoring can address these risks. Edible vaccines have significant benefits and require further research for enhanced disease control. Plant-based vaccine production holds promise for preventing global epidemics.
References
- Arakawa, T.; Chong, D.K.; Langridge, W.H. Efficacy of a food plant-based oral cholera toxin B subunit vaccine. Nat. Biotechnol. 1998, 16, 292–297.
- Arakawa, T.; Yu, J.; Langridge, W.H. Food plant-delivered cholera toxin B subunit for vaccination and immunotolerization. Adv. Exp. Med. Biol. 1999, 464, 161–178.
- Yuki, Y.; Nojima, M.; Hosono, O.; Tanaka, H.; Kimura, Y.; Satoh, T.; Imoto, S.; Uematsu, S.; Kurokawa, S.; Kashima, K.; et al. Oral MucoRice-CTB vaccine for safety and microbiota-dependent immunogenicity in humans: A phase 1 randomised trial. Lancet Microbe 2021, 2, e429–e440.
- Trentham, D.E.; Dynesius-Trentham, R.A.; Orav, E.J.; Combitchi, D.; Lorenzo, C.; Sewell, K.L.; Hafler, D.A.; Weiner, H.L. Effects of oral administration of type II collagen on rheumatoid arthritis. Science 1993, 261, 1727–1730.
- Weiner, H.L.; Mackin, G.A.; Matsui, M.; Orav, E.J.; Khoury, S.J.; Dawson, D.M.; Hafler, D.A. Double-blind pilot trial of oral tolerization with myelin antigens in multiple sclerosis. Science 1993, 259, 1321–1324.
- Weiner, H.L.; Friedman, A.; Miller, A.; Khoury, S.J.; al-Sabbagh, A.; Santos, L.; Sayegh, M.; Nussenblatt, R.B.; Trentham, D.E.; Hafler, D.A. Oral tolerance: Immunologic mechanisms and treatment of animal and human organ-specific autoimmune diseases by oral administration of autoantigens. Annu. Rev. Immunol. 1994, 12, 809–837.
- Zhang, Z.J.; Davidson, L.; Eisenbarth, G.; Weiner, H.L. Suppression of diabetes in nonobese diabetic mice by oral administration of porcine insulin. Proc. Natl. Acad. Sci. USA 1991, 88, 10252–10256.
- Carel, J.C.; Bougnères, P.; Vardi, P. Suppression of diabetes in nonobese diabetic mice by oral administration of porcine insulin. J. Endocrinol. Investig. 1994, 17, 573–580.
- Sun, J.B.; Holmgren, J.; Czerkinsky, C. Cholera toxin B subunit: An efficient transmucosal carrier-delivery system for induction of peripheral immunological tolerance. Proc. Natl. Acad. Sci. USA 1994, 91, 10795–10799.
- Holmgren, J.; Adamsson, J.; Anjuère, F.; Clemens, J.; Czerkinsky, C.; Eriksson, K.; Flach, C.F.; George-Chandy, A.; Harandi, A.M.; Lebens, M.; et al. Mucosal adjuvants and anti-infection and anti-immunopathology vaccines based on cholera toxin, cholera toxin B subunit and CpG DNA. Immunol. Lett. 2005, 97, 181–188.
- Sun, J.B.; Czerkinsky, C.; Holmgren, J. Mucosally induced immunological tolerance, regulatory T cells and the adjuvant effect by cholera toxin B subunit. Scand. J. Immunol. 2010, 71, 1–11.
- Sun, J.B.; Rask, C.; Olsson, T.; Holmgren, J.; Czerkinsky, C. Treatment of experimental autoimmune encephalomyelitis by feeding myelin basic protein conjugated to cholera toxin B subunit. Proc. Natl. Acad. Sci. USA 1996, 93, 7196–7201.
- Lönnroth, I.; Holmgren, J. Subunit structure of cholera toxin. J. Gen. Microbiol. 1973, 76, 417–427.
- Sixma, T.K.; Kalk, K.H.; van Zanten, B.A.; Dauter, Z.; Kingma, J.; Witholt, B.; Hol, W.G. Refined structure of Escherichia coli heat-labile enterotoxin, a close relative of cholera toxin. J. Mol. Biol. 1993, 230, 890–918.
- Chester, M.A. IUPAC-IUB Joint Commission on Biochemical Nomenclature (JCBN). Nomenclature of glycolipids–recommendations 1997. Eur. J. Biochem. 1998, 257, 293–298.
- Schön, A.; Freire, E. Thermodynamics of intersubunit interactions in cholera toxin upon binding to the oligosaccharide portion of its cell surface receptor, ganglioside GM1. Biochemistry 1989, 28, 5019–5024.
- Goins, B.; Freire, E. Thermal stability and intersubunit interactions of cholera toxin in solution and in association with its cell-surface receptor ganglioside GM1. Biochemistry 1988, 27, 2046–2052.
- Kozireski-Chuback, D.; Wu, G.; Ledeen, R.W. Developmental appearance of nuclear GM1 in neurons of the central and peripheral nervous systems. Brain Res. Dev. Brain Res. 1999, 115, 201–208.
- Moreno-Altamirano, M.M.; Aguilar-Carmona, I.; Sánchez-García, F.J. Expression of GM1, a marker of lipid rafts, defines two subsets of human monocytes with differential endocytic capacity and lipopolysaccharide responsiveness. Immunology 2007, 120, 536–543.
- Francis, M.L.; Ryan, J.; Jobling, M.G.; Holmes, R.K.; Moss, J.; Mond, J.J. Cyclic AMP-independent effects of cholera toxin on B cell activation. II. Binding of ganglioside GM1 induces B cell activation. J. Immunol. 1992, 148, 1999–2005.
- Li, S.; Wei, Z.; Chen, J.; Chen, Y.; Lv, Z.; Yu, W.; Meng, Q.; Jin, Y. Oral administration of a fusion protein between the cholera toxin B subunit and the 42-amino acid isoform of amyloid-β peptide produced in silkworm pupae protects against Alzheimer's disease in mice. PLoS ONE 2014, 9, e113585.
- Gong, Z.; Jin, Y.; Zhang, Y. Suppression of diabetes in non-obese diabetic (NOD) mice by oral administration of a cholera toxin B subunit-insulin B chain fusion protein vaccine produced in silkworm. Vaccine 2007, 25, 1444–1451.
- Gong, Z.H.; Jin, H.Q.; Jin, Y.F.; Zhang, Y.Z. Expression of cholera toxin B subunit and assembly as functional oligomers in silkworm. J. Biochem. Mol. Biol. 2005, 38, 717–724.
- Gong, Z.; Jin, Y.; Zhang, Y. Oral administration of a cholera toxin B subunit-insulin fusion protein produced in silkworm protects against autoimmune diabetes. J. Biotechnol. 2005, 119, 93–105.
- Liljeqvist, S.; Ståhl, S.; Andréoni, C.; Binz, H.; Uhlén, M.; Murby, M. Fusions to the cholera toxin B subunit: Influence on pentamerization and GM1 binding. J. Immunol. Methods 1997, 210, 125–135.
- Hajishengallis, G.; Hollingshead, S.K.; Koga, T.; Russell, M.W. Mucosal immunization with a bacterial protein antigen genetically coupled to cholera toxin A2/B subunits. J. Immunol. 1995, 154, 4322–4332.
- Lavelle, E.C.; Ward, R.W. Mucosal vaccines-fortifying the frontiers. Nat. Rev. Immunol. 2022, 22, 236–250.
- Mbongue, J.C.; Nicholas, D.A.; Zhang, K.; Kim, N.S.; Hamilton, B.N.; Larios, M.; Zhang, G.; Umezawa, K.; Firek, A.F.; Langridge, W.H. Induction of indoleamine 2, 3-dioxygenase in human dendritic cells by a cholera toxin B subunit-proinsulin vaccine. PLoS ONE 2015, 10, e0118562.
- Kim, N.S.; Mbongue, J.C.; Nicholas, D.A.; Esebanmen, G.E.; Unternaehrer, J.J.; Firek, A.F.; Langridge, W.H. Chimeric Vaccine Stimulation of Human Dendritic Cell Indoleamine 2, 3-Dioxygenase Occurs via the Non-Canonical NF-κB Pathway. PLoS ONE 2016, 11, e0147509.
- Dénes, B.; Fodor, I.; Langridge, W.H. Autoantigens plus interleukin-10 suppress diabetes autoimmunity. Diabetes Technol. Ther. 2010, 12, 649–661.
- Langridge, W.; Dénes, B.; Fodor, I. Cholera toxin B subunit modulation of mucosal vaccines for infectious and autoimmune diseases. Curr. Opin. Investig. Drugs 2010, 11, 919–928.
- Dénes, B.; Fodor, I.; Langridge, W.H. Persistent suppression of type 1 diabetes by a multicomponent vaccine containing a cholera toxin B subunit-autoantigen fusion protein and complete Freund’s adjuvant. Clin. Dev. Immunol. 2013, 2013, 578786.
- Meng, Q.; Wang, W.; Shi, X.; Jin, Y.; Zhang, Y. Protection against autoimmune diabetes by silkworm-produced GFP-tagged CTB-insulin fusion protein. Clin. Dev. Immunol. 2011, 2011, 831704.
- Kohli, N.; Westerveld, D.R.; Ayache, A.C.; Verma, A.; Shil, P.; Prasad, T.; Zhu, P.; Chan, S.L.; Li, Q.; Daniell, H. Oral delivery of bioencapsulated proteins across blood-brain and blood-retinal barriers. Mol. Ther. 2014, 22, 535–546.
- Arakawa, T.; Chong, D.K.; Merritt, J.L.; Langridge, W.H. Expression of cholera toxin B subunit oligomers in transgenic potato plants. Transgenic Res. 1997, 6, 403–413.
- Mor, T.S.; Gómez-Lim, M.A.; Palmer, K.E. Perspective: Edible vaccines—A concept coming of age. Trends Microbiol. 1998, 6, 449–453.
- Daniell, H.; Lee, S.B.; Panchal, T.; Wiebe, P.O. Expression of the native cholera toxin B subunit gene and assembly as functional oligomers in transgenic tobacco chloroplasts. J. Mol. Biol. 2001, 311, 1001–1009.
- Matoba, N.; Kajiura, H.; Cherni, I.; Doran, J.D.; Bomsel, M.; Fujiyama, K.; Mor, T.S. Biochemical and immunological characterization of the plant-derived candidate human immunodeficiency virus type 1 mucosal vaccine CTB-MPR. Plant Biotechnol. J. 2009, 7, 129–145.
- Matoba, N.; Magérus, A.; Geyer, B.C.; Zhang, Y.; Muralidharan, M.; Alfsen, A.; Arntzen, C.J.; Bomsel, M.; Mor, T.S. A mucosally targeted subunit vaccine candidate eliciting HIV-1 transcytosis-blocking Abs. Proc. Natl. Acad. Sci. USA 2004, 101, 13584–13589.
- Matoba, N.; Griffin, T.A.; Mittman, M.; Doran, J.D.; Alfsen, A.; Montefiori, D.C.; Hanson, C.V.; Bomsel, M.; Mor, T.S. Transcytosis-blocking abs elicited by an oligomeric immunogen based on the membrane proximal region of HIV-1 gp41 target non-neutralizing epitopes. Curr. HIV Res. 2008, 6, 218–229.
- Nochi, T.; Takagi, H.; Yuki, Y.; Yang, L.; Masumura, T.; Mejima, M.; Nakanishi, U.; Matsumura, A.; Uozumi, A.; Hiroi, T.; et al. Rice-based mucosal vaccine as a global strategy for cold-chain- and needle-free vaccination. Proc. Natl. Acad. Sci. USA 2007, 104, 10986–10991.
- Nochi, T.; Yuki, Y.; Katakai, Y.; Shibata, H.; Tokuhara, D.; Mejima, M.; Kurokawa, S.; Takahashi, Y.; Nakanishi, U.; Ono, F.; et al. A rice-based oral cholera vaccine induces macaque-specific systemic neutralizing antibodies but does not influence pre-existing intestinal immunity. J. Immunol. 2009, 183, 6538–6544.
- Yuki, Y.; Tokuhara, D.; Nochi, T.; Yasuda, H.; Mejima, M.; Kurokawa, S.; Takahashi, Y.; Kataoka, N.; Nakanishi, U.; Hagiwara, Y.; et al. Oral MucoRice expressing double-mutant cholera toxin A and B subunits induces toxin-specific neutralising immunity. Vaccine 2009, 27, 5982–5988.
- Kurokawa, S.; Kuroda, M.; Mejima, M.; Nakamura, R.; Takahashi, Y.; Sagara, H.; Takeyama, N.; Satoh, S.; Kiyono, H.; Teshima, R.; et al. RNAi-mediated suppression of endogenous storage proteins leads to a change in localization of overexpressed cholera toxin B-subunit and the allergen protein RAG2 in rice seeds. Plant Cell Rep. 2014, 33, 75–87.
- Lakshmi, P.S.; Verma, D.; Yang, X.; Lloyd, B.; Daniell, H. Low cost tuberculosis vaccine antigens in capsules: Expression in chloroplasts, bio-encapsulation, stability and functional evaluation in vitro. PLoS ONE 2013, 8, e54708.
- Carter, J.E.; Yu, J.; Choi, N.W.; Hough, J.; Henderson, D.; He, D.; Langridge, W.H. Bacterial and plant enterotoxin B subunit-autoantigen fusion proteins suppress diabetes insulitis. Mol. Biotechnol. 2006, 32, 1–15.





