
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Shanthi Krishnasamy | -- | 2452 | 2023-06-15 07:56:28 | | | |
| 2 | Lindsay Dong | Meta information modification | 2452 | 2023-06-16 03:26:09 | | |
Video Upload Options
Patients with psoriasis have a poor nutritional status, and they are at risk of nutrient deficiencies. However, these health aspects are not routinely assessed and may increase the risk of malnutrition among these patients. Therefore, additional assessments, such as body composition and dietary assessment, are needed to determine the nutritional status to provide a suitable intervention.
1. Introduction
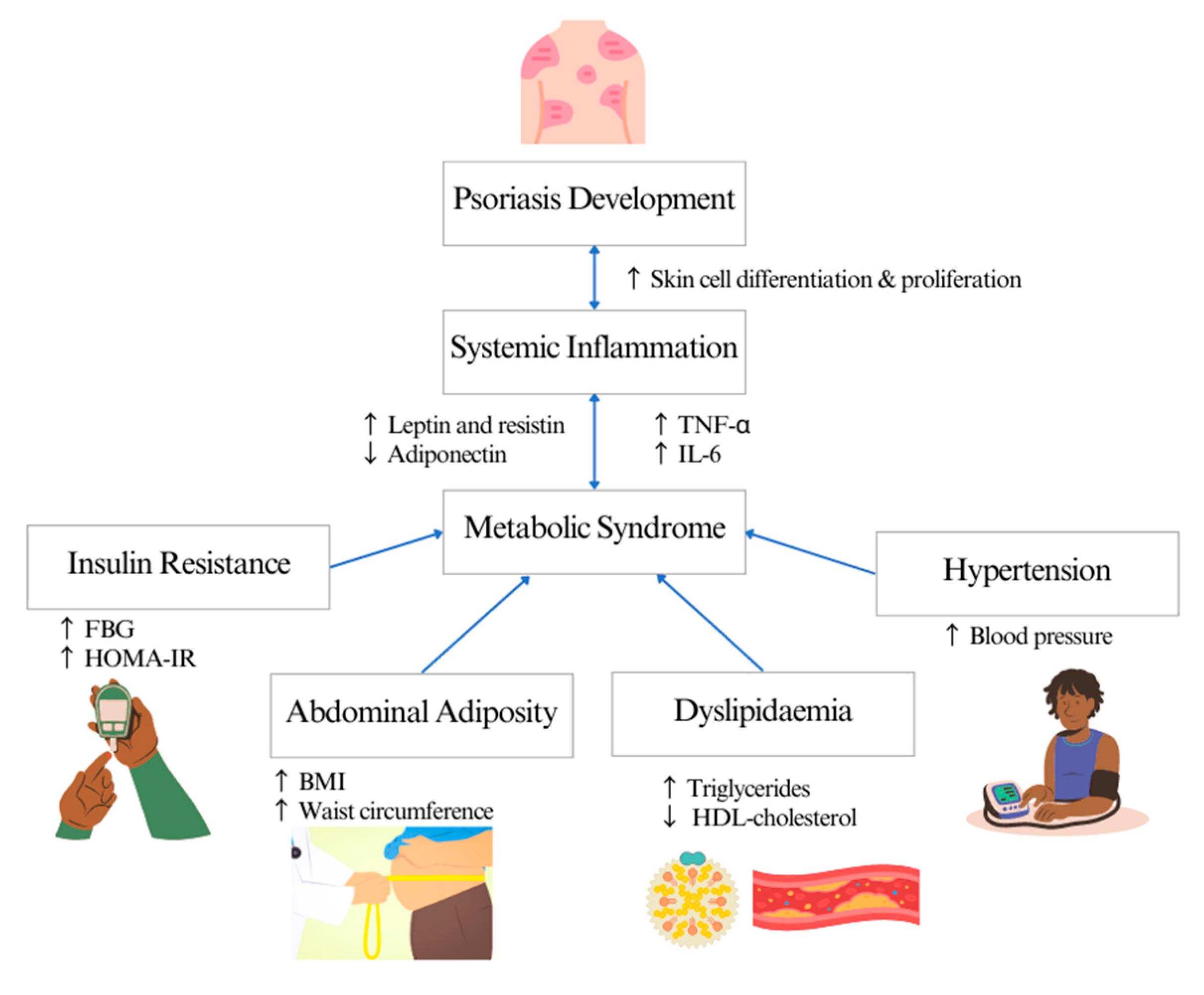
2. Metabolic Syndrome Screening and Nutritional Status of Psoriasis
Patients with psoriasis were mostly overweight or obese. An increased body mass could potentially decrease the drug distribution in the body, reducing the effectiveness of systemic or biological treatment [26][27]. In addition, a higher BMI also increases psoriasis severity [27][28][29] and induces inflammation [24][30], thus increasing the risk of comorbidities in patients.
3. Conclusions
References
- WHO. Global Report on Psoriasis; World Health Organization: Geneva, Switzerland, 2016; ISBN 978-92-4-156518-9.
- Michalek, I.M.; Loring, B.; John, S.M. A Systematic Review of Worldwide Epidemiology of Psoriasis. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 205–212.
- Armstrong, A.W.; Read, C. Pathophysiology, Clinical Presentation, and Treatment of Psoriasis: A Review. JAMA 2020, 323, 1945–1960.
- Fredriksson, T.; Pettersson, U. Severe Psoriasis—Oral Therapy with a New Retinoid. Dermatology 1978, 157, 238–244.
- Gisondi, P.; Bellinato, F.; Girolomoni, G.; Albanesi, C. Pathogenesis of Chronic Plaque Psoriasis and Its Intersection With Cardio-Metabolic Comorbidities. Front. Pharmacol. 2020, 11, 117.
- Kanda, N.; Hoashi, T.; Saeki, H. Nutrition and Psoriasis. Int. J. Mol. Sci. 2020, 21, 5405.
- Kunz, M.; Simon, J.C.; Saalbach, A. Psoriasis: Obesity and Fatty Acids. Front. Immunol. 2019, 10, 1807.
- Bu, J.; Ding, R.; Zhou, L.; Chen, X.; Shen, E. Epidemiology of Psoriasis and Comorbid Diseases: A Narrative Review. Front. Immunol. 2022, 13, 880201.
- Takeshita, J.; Grewal, S.; Langan, S.M.; Mehta, N.N.; Ogdie, A.; Van Voorhees, A.S.; Gelfand, J.M. Psoriasis and Comorbid Diseases. J. Am. Acad. Dermatol. 2017, 76, 393–403.
- Vanderpuye-Orgle, J.; Zhao, Y.; Lu, J.; Shrestha, A.; Sexton, A.; Seabury, S.; Lebwohl, M. Evaluating the Economic Burden of Psoriasis in the United States. J. Am. Acad. Dermatol. 2015, 72, 961–967.e5.
- Gisondi, P.; Fostini, A.C.; Fossà, I.; Girolomoni, G.; Targher, G. Psoriasis and the Metabolic Syndrome. Clin. Dermatol. 2018, 36, 21–28.
- Alberti, K.G.; Zimmet, P.Z. Definition, Diagnosis and Classification of Diabetes Mellitus and Its Complications. Part 1: Diagnosis and Classification of Diabetes Mellitus Provisional Report of a WHO Consultation. Diabet. Med. J. Br. Diabet. Assoc. 1998, 15, 539–553.
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497.
- Balkau, B.; Charles, M.A. Comment on the Provisional Report from the WHO Consultation. European Group for the Study of Insulin Resistance (EGIR). Diabet. Med. J. Br. Diabet. Assoc. 1999, 16, 442–443.
- Einhorn, D.; Reaven, G.M.; Cobin, R.H.; Ford, E.; Ganda, O.P.; Handelsman, Y.; Hellman, R.; Jellinger, P.S.; Kendall, D.; Krauss, R.M.; et al. American College of Endocrinology Position Statement on the Insulin Resistance Syndrome. Endocr. Pract. 2003, 9, 237–252.
- Lemieux, I.; Després, J.-P. Metabolic Syndrome: Past, Present and Future. Nutrients 2020, 12, 3501.
- Gelfand, J.M.; Yeung, H. Metabolic syndrome in patients with psoriatic disease. J. Rheumatol. Suppl. 2012, 89, 24–28.
- Wu, J.J.; Kavanaugh, A.; Lebwohl, M.G.; Gniadecki, R.; Merola, J.F. Psoriasis and metabolic syndrome: Implications for the management and treatment of psoriasis. J. Eur. Acad. Dermatol. Venereol. 2022, 36, 797–806.
- Rodríguez-Cerdeira, C.; Cordeiro-Rodríguez, M.; Carnero-Gregorio, M.; López-Barcenas, A.; Martínez-Herrera, E.; Fabbrocini, G.; Sinani, A.; Arenas-Guzmán, R.; González-Cespón, J.L. Biomarkers of Inflammation in Obesity-Psoriatic Patients. Mediat. Inflamm. 2019, 28, 7353420.
- Blake, T.; Gullick, N.J.; Hutchinson, C.E.; Barber, T.M. Psoriatic Disease and Body Composition: A Systematic Review and Narrative Synthesis. PLoS ONE 2020, 15, e0237598.
- Reber, E.; Gomes, F.; Vasiloglou, M.F.; Schuetz, P.; Stanga, Z. Nutritional Risk Screening and Assessment. J. Clin. Med. 2019, 8, 1065.
- Truijen, S.P.M.; Hayhoe, R.P.G.; Hooper, L.; Schoenmakers, I.; Forbes, A.; Welch, A.A. Predicting Malnutrition Risk with Data from Routinely Measured Clinical Biochemical Diagnostic Tests in Free-Living Older Populations. Nutrients 2021, 13, 1883.
- Solis, M.Y.; de Melo, N.S.; Macedo, M.E.M.; Carneiro, F.P.; Sabbag, C.Y.; Lancha Júnior, A.H.; Frangella, V.S. Nutritional Status and Food Intake of Patients with Systemic Psoriasis and Psoriatic Arthritis Associated. Einstein 2012, 10, 44–52.
- Barrea, L.; Macchia, P.E.; Tarantino, G.; Di Somma, C.; Pane, E.; Balato, N.; Napolitano, M.; Colao, A.; Savastano, S. Nutrition: A Key Environmental Dietary Factor in Clinical Severity and Cardio-Metabolic Risk in Psoriatic Male Patients Evaluated by 7-Day Food-Frequency Questionnaire. J. Transl. Med. 2015, 13, 303.
- Atayoglu, A.T.; Çapar, A.G.; Basmisirlioglu, E.; Yasar, Y.; Aykemat, Y.; Guner Atayoglu, A.; Inanc, N. Investigation of the Relationship between the Disease Severity and Quality of Life of Patients with psoriasis and Their Anthropometric Measurements and Diets. Healthcare 2022, 10, 2323.
- Carrascosa, J.M.; Rocamora, V.; Fernandez-Torres, R.M.; Jimenez-Puya, R.; Moreno, J.C.; Coll-Puigserver, N.; Fonseca, E. Obesity and Psoriasis: Inflammatory Nature of Obesity, Relationship between Psoriasis and Obesity, and Therapeutic Implications. Actas Dermo-Sifiliográficas 2014, 105, 31–44.
- Fleming, P.; Kraft, J.; Gulliver, W.P.; Lynde, C. The Relationship of Obesity With the Severity of Psoriasis: A Systematic Review. J. Cutan. Med. Surg. 2015, 19, 450–456.
- Lin, I.-C.; Heck, J.E.; Chen, L.; Feldman, S.R. Psoriasis Severity and Cardiometabolic Risk Factors in a Representative US National Study. Am. J. Clin. Dermatol. 2021, 22, 719–730.
- Paroutoglou, K.; Papadavid, E.; Christodoulatos, G.S.; Dalamaga, M. Deciphering the Association Between Psoriasis and Obesity: Current Evidence and Treatment Considerations. Curr. Obes. Rep. 2020, 9, 165–178.
- Zorena, K.; Jachimowicz-Duda, O.; Ślęzak, D.; Robakowska, M.; Mrugacz, M. Adipokines and Obesity. Potential Link to Metabolic Disorders and Chronic Complications. Int. J. Mol. Sci. 2020, 21, 3570.
- Barrea, L.; Megna, M.; Cacciapuoti, S.; Frias-Toral, E.; Fabbrocini, G.; Savastano, S.; Colao, A.; Muscogiuri, G. Very Low-Calorie Ketogenic Diet (VLCKD) in Patients with Psoriasis and Obesity: An Update for Dermatologists and Nutritionists. Crit. Rev. Food Sci. Nutr. 2022, 62, 398–414.
- Gisondi, P.; Del Giglio, M.; Girolomoni, G. Considerations for Systemic Treatment of Psoriasis in Obese Patients. Am. J. Clin. Dermatol. 2016, 17, 609–615.
- Jensen, P.; Zachariae, C.; Christensen, R.; Geiker, N.R.W.; Schaadt, B.K.; Stender, S.; Astrup, A.; Hansen, P.R.; Skov, L. Effect of Weight Loss on the Cardiovascular Risk Profile of Obese Patients with Psoriasis. Acta Derm. Venereol. 2014, 94, 691–694.
- Madden, A.M.; Smith, S. Body Composition and Morphological Assessment of Nutritional Status in Adults: A Review of Anthropometric Variables. J. Hum. Nutr. Diet. 2016, 29, 7–25.
- Han, J.H.; Lee, J.H.; Han, K.D.; Kim, H.-N.; Bang, C.H.; Park, Y.M.; Lee, J.Y.; Kim, T.Y. Increased Risk of Psoriasis in Subjects with Abdominal Obesity: A Nationwide Population-Based Study. J. Dermatol. 2019, 46, 695–701.
- Sajja, A.; Abdelrahman, K.M.; Reddy, A.S.; Dey, A.K.; Uceda, D.E.; Lateef, S.S.; Sorokin, A.V.; Teague, H.L.; Chung, J.; Rivers, J.; et al. Chronic Inflammation in Psoriasis Promotes Visceral Adiposity Associated with Noncalcified Coronary Burden over Time. J. Clin. Investig. 2020, 5, e142534.
- Wolk, K.; Sabat, R. Adipokines in Psoriasis: An Important Link between Skin Inflammation and Metabolic Alterations. Rev. Endocr. Metab. Disord. 2016, 17, 305–317.
- Ross, R.; Neeland, I.J.; Yamashita, S.; Shai, I.; Seidell, J.; Magni, P.; Santos, R.D.; Arsenault, B.; Cuevas, A.; Hu, F.B.; et al. Waist Circumference as a Vital Sign in Clinical Practice: A Consensus Statement from the IAS and ICCR Working Group on Visceral Obesity. Nat. Rev. Endocrinol. 2020, 16, 177–189.
- Khalil, S.F.; Mohktar, M.S.; Ibrahim, F. The Theory and Fundamentals of Bioimpedance Analysis in Clinical Status Monitoring and Diagnosis of Diseases. Sensors 2014, 14, 10895–10928.
- von Berens, Å.; Obling, S.R.; Nydahl, M.; Koochek, A.; Lissner, L.; Skoog, I.; Frändin, K.; Skoglund, E.; Rothenberg, E.; Cederholm, T. Sarcopenic Obesity and Associations with Mortality in Older Women and Men–A Prospective Observational Study. BMC Geriatr. 2020, 20, 199.
- Wagenaar, C.A.; Dekker, L.H.; Navis, G.J. Prevalence of Sarcopenic Obesity and Sarcopenic Overweight in the General Population: The Lifelines Cohort Study. Clin. Nutr. 2021, 40, 4422–4429.
- Chen, X.; Xiang, H.; Tan, L.; Zhou, J.; Tang, J.; Hu, X.; Yang, M. Psoriasis Is Associated With Myosteatosis but Not Sarcopenia: A Case-Control Study. Front. Med. 2021, 8, 754932.
- Kim, S.H.; Jeong, J.B.; Kang, J.; Ahn, D.-W.; Kim, J.W.; Kim, B.G.; Lee, K.L.; Oh, S.; Yoon, S.H.; Park, S.J.; et al. Association between Sarcopenia Level and Metabolic Syndrome. PLoS ONE 2021, 16, e0248856.
- Park, S.-J.; Ryu, S.-Y.; Park, J.; Choi, S.-W. Association of Sarcopenia with Metabolic Syndrome in Korean Population Using 2009–2010 Korea National Health and Nutrition Examination Survey. Metab. Syndr. Relat. Disord. 2019, 17, 494–499.
- Zhang, H.; Lin, S.; Gao, T.; Zhong, F.; Cai, J.; Sun, Y.; Ma, A. Association between Sarcopenia and Metabolic Syndrome in Middle-Aged and Older Non-Obese Adults: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 364.
- Xiao, Y.; Jing, D.; Tang, Z.; Peng, C.; Yin, M.; Liu, H.; Chen, X.; Shen, M. Serum Lipids and Risk of Incident Psoriasis: A Prospective Cohort Study from the UK Biobank Study and Mendelian Randomization Analysis. J. Investig. Dermatol. 2022, 142, 3192–3199.e12.
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis Model Assessment: Insulin Resistance and Beta-Cell Function from Fasting Plasma Glucose and Insulin Concentrations in Man. Diabetologia 1985, 28, 412–419.
- Gutch, M.; Kumar, S.; Razi, S.M.; Gupta, K.K.; Gupta, A. Assessment of Insulin Sensitivity/Resistance. Indian J. Endocrinol. Metab. 2015, 19, 160–164.
- Leighton, E.; Sainsbury, C.A.; Jones, G.C. A Practical Review of C-Peptide Testing in Diabetes. Diabetes Ther. 2017, 8, 475–487.
- Gyldenløve, M.; Storgaard, H.; Holst, J.J.; Vilsbøll, T.; Knop, F.K.; Skov, L. Patients with Psoriasis Are Insulin Resistant. J. Am. Acad. Dermatol. 2015, 72, 599–605.
- Karadag, A.S.; Yavuz, B.; Ertugrul, D.T.; Akin, K.O.; Yalcin, A.A.; Deveci, O.S.; Ata, N.; Kucukazman, M.; Dal, K. Is Psoriasis a Pre-Atherosclerotic Disease? Increased Insulin Resistance and Impaired Endothelial Function in Patients with Psoriasis. Int. J. Dermatol. 2010, 49, 642–646.
- Katz, A.; Nambi, S.S.; Mather, K.; Baron, A.D.; Follmann, D.A.; Sullivan, G.; Quon, M.J. Quantitative Insulin Sensitivity Check Index: A Simple, Accurate Method for Assessing Insulin Sensitivity in Humans. J. Clin. Endocrinol. Metab. 2000, 85, 2402–2410.
- Lind, L.; Vessby, B.; Sundström, J. The Apolipoprotein B/AI Ratio and the Metabolic Syndrome Independently Predict Risk for Myocardial Infarction in Middle-Aged Men. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 406–410.
- McQueen, M.J.; Hawken, S.; Wang, X.; Ounpuu, S.; Sniderman, A.; Probstfield, J.; Steyn, K.; Sanderson, J.E.; Hasani, M.; Volkova, E.; et al. Lipids, Lipoproteins, and Apolipoproteins as Risk Markers of Myocardial Infarction in 52 Countries (the INTERHEART Study): A Case-Control Study. Lancet Lond. Engl. 2008, 372, 224–233.
- Nemati, H.; Khodarahmi, R.; Rahmani, A.; Ebrahimi, A.; Amani, M.; Eftekhari, K. Serum Lipid Profile in Psoriatic Patients: Correlation between Vascular Adhesion Protein 1 and Lipoprotein (a). Cell Biochem. Funct. 2013, 31, 36–40.
- Ramezani, M.; Zavattaro, E.; Sadeghi, M. Evaluation of Serum Lipid, Lipoprotein, and Apolipoprotein Levels in Psoriatic Patients: A Systematic Review and Meta-Analysis of Case-Control Studies. Adv. Dermatol. Allergol. Dermatol. Alergol. 2019, 36, 692–702.
- Anyfanti, P.; Margouta, A.; Goulas, K.; Gavriilaki, M.; Lazaridou, E.; Patsatsi, A.; Gkaliagkousi, E. Endothelial Dysfunction in Psoriasis: An Updated Review. Front. Med. 2022, 9, 864185.
- Marongiu, F.; Sorano, G.G.; Bibbò, C.; Pistis, M.P.; Conti, M.; Mulas, P.; Balestrieri, A.; Biggio, P. Abnormalities of Blood Coagulation and Fibrinolysis in Psoriasis. Dermatology 1994, 189, 32–37.
- Baran, A.; Flisiak, I.; Jaroszewicz, J.; Świderska, M. Effect of Psoriasis Activity on Serum Adiponectin and Leptin Levels. Adv. Dermatol. Allergol./Postępy Dermatol. I Alergol. 2015, 32, 101–106.
- Shibata, S.; Tada, Y.; Hau, C.S.; Mitsui, A.; Kamata, M.; Asano, Y.; Sugaya, M.; Kadono, T.; Masamoto, Y.; Kurokawa, M.; et al. Adiponectin Regulates Psoriasiform Skin Inflammation by Suppressing IL-17 Production from Γδ-T Cells. Nat. Commun. 2015, 6, 7687.
- Qu, R.; Chen, X.; Hu, J.; Fu, Y.; Peng, J.; Li, Y.; Chen, J.; Li, P.; Liu, L.; Cao, J.; et al. Ghrelin Protects against Contact Dermatitis and Psoriasiform Skin Inflammation by Antagonizing TNF-α/NF-ΚB Signaling Pathways. Sci. Rep. 2019, 9, 1348.
- Ozdemir, M.; Yüksel, M.; Gökbel, H.; Okudan, N.; Mevlitoğlu, I. Serum Leptin, Adiponectin, Resistin and Ghrelin Levels in Psoriatic Patients Treated with Cyclosporin. J. Dermatol. 2012, 39, 443–448.
- Al-Harbi, N.O.; Nadeem, A.; Ansari, M.A.; Al-Harbi, M.M.; Alotaibi, M.R.; AlSaad, A.M.S.; Ahmad, S.F. Psoriasis-like Inflammation Leads to Renal Dysfunction via Upregulation of NADPH Oxidases and Inducible Nitric Oxide Synthase. Int. Immunopharmacol. 2017, 46, 1–8.
- Conti, A.; Giovannini, L.; Mandel, V.D.; Odorici, G.; Lasagni, C.; Bigi, L.; Pellacani, G.; Cappelli, G. Chronic Kidney Disease in Psoriasis: A Cohort Study. JDDG J. Der Dtsch. Dermatol. Ges. 2020, 18, 438–445.
- Duan, X.; Liu, J.; Mu, Y.; Liu, T.; Chen, Y.; Yu, R.; Xiong, X.; Wu, T. A Systematic Review and Meta-Analysis of the Association between Psoriasis and Hypertension with Adjustment for Covariates. Medicine 2020, 99, e19303.
- Kim, H.-N.; Han, K.; Song, S.-W.; Lee, J.H. Hypertension and Risk of Psoriasis Incidence: An 11-Year Nationwide Population-Based Cohort Study. PLoS ONE 2018, 13, e0202854.
- Bulbul Sen, B.; Atci, N.; Rifaioglu, E.N.; Ekiz, O.; Kartal, I.; Buyukkaya, E.; Kurt, M.; Karakas, M.F.; Buyukkaya, S.; Akcay, A.B.; et al. Increased Epicardial Fat Tissue Is a Marker of Subclinical Atherosclerosis in Patients with Psoriasis. Br. J. Dermatol. 2013, 169, 1081–1086.
- Girisha, B.S.; Shibina, S.; Raghuraja, U.; Subramanyam, K. Carotid Intima-Media Thickness and Epicardial Fat Thickness Predict Precoronary Artery Disease Status in Psoriasis. Indian J. Dermatol. Venereol. Leprol. 2021, 87, 357–363.
- Barrea, L.; Savanelli, M.C.; Di Somma, C.; Napolitano, M.; Megna, M.; Colao, A.; Savastano, S. Vitamin D and Its Role in Psoriasis: An Overview of the Dermatologist and Nutritionist. Rev. Endocr. Metab. Disord. 2017, 18, 195–205.
- Crosta, M.L.; Caldarola, G.; Fraietta, S.; Craba, A.; Benedetti, C.; Coco, V.; Janiri, L.; Rinaldi, L.; De Simone, C. Psychopathology and Eating Disorders in Patients with Psoriasis. G. Ital. Dermatol. E Venereol. 2014, 149, 355–361.
- Musumeci, M.L.; Nasca, M.R.; Boscaglia, S.; Micali, G. The Role of Lifestyle and Nutrition in Psoriasis: Current Status of Knowledge and Interventions. Dermatol. Ther. 2022, 35, e15685.
- Pona, A.; Haidari, W.; Kolli, S.S.; Feldman, S.R. Diet and Psoriasis. Dermatol. Online J. 2019, 25, 13030/qt1p37435s.
- Conway, J.M.; Ingwersen, L.A.; Moshfegh, A.J. Accuracy of Dietary Recall Using the USDA Five-Step Multiple-Pass Method in Men: An Observational Validation Study. J. Am. Diet. Assoc. 2004, 104, 595–603.
- Shim, J.-S.; Oh, K.; Kim, H.C. Dietary Assessment Methods in Epidemiologic Studies. Epidemiol. Health 2014, 36, e2014009.
- Chung, M.; Bartholomew, E.; Yeroushalmi, S.; Hakimi, M.; Bhutani, T.; Liao, W. Dietary Intervention and Supplements in the Management of Psoriasis: Current Perspectives. Psoriasis Targets Ther. 2022, 12, 151–176.
- Garbicz, J.; Całyniuk, B.; Górski, M.; Buczkowska, M.; Piecuch, M.; Kulik, A.; Rozentryt, P. Nutritional Therapy in Persons Suffering from Psoriasis. Nutrients 2021, 14, 119.
- Katsimbri, P.; Korakas, E.; Kountouri, A.; Ikonomidis, I.; Tsougos, E.; Vlachos, D.; Papadavid, E.; Raptis, A.; Lambadiari, V. The Effect of Antioxidant and Anti-Inflammatory Capacity of Diet on Psoriasis and Psoriatic Arthritis Phenotype: Nutrition as Therapeutic Tool? Antioxidants 2021, 10, 157.





