Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Isabel Legaz | -- | 1956 | 2023-06-14 10:25:30 | | | |
| 2 | Camila Xu | Meta information modification | 1956 | 2023-06-14 10:41:10 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Jiménez-Coll, V.; El Kaaoui El Band, J.; Llorente, S.; González-López, R.; Fernández-González, M.; Martínez-Banaclocha, H.; Galián, J.A.; Botella, C.; Moya-Quiles, M.R.; Minguela, A.; et al. cfDNA Analysis. Encyclopedia. Available online: https://encyclopedia.pub/entry/45555 (accessed on 26 March 2026).
Jiménez-Coll V, El Kaaoui El Band J, Llorente S, González-López R, Fernández-González M, Martínez-Banaclocha H, et al. cfDNA Analysis. Encyclopedia. Available at: https://encyclopedia.pub/entry/45555. Accessed March 26, 2026.
Jiménez-Coll, Victor, Jaouad El Kaaoui El Band, Santiago Llorente, Rosana González-López, Marina Fernández-González, Helios Martínez-Banaclocha, José Antonio Galián, Carmen Botella, María Rosa Moya-Quiles, Alfredo Minguela, et al. "cfDNA Analysis" Encyclopedia, https://encyclopedia.pub/entry/45555 (accessed March 26, 2026).
Jiménez-Coll, V., El Kaaoui El Band, J., Llorente, S., González-López, R., Fernández-González, M., Martínez-Banaclocha, H., Galián, J.A., Botella, C., Moya-Quiles, M.R., Minguela, A., Legaz, I., & Muro, M. (2023, June 14). cfDNA Analysis. In Encyclopedia. https://encyclopedia.pub/entry/45555
Jiménez-Coll, Victor, et al. "cfDNA Analysis." Encyclopedia. Web. 14 June, 2023.
Copy Citation
Degraded DNA fragments released into the blood or other fluids are known as cfDNA. Its first detection dates back to 1948 in patients with systemic lupus erythematosus.
donor-derived cell-free DNA (cfDNA)
graft injury
acute rejection
organ transplant
1. Introduction
In kidney transplantation, a biopsy is currently the gold standard for monitoring the transplanted organ. A biopsy involves taking a small tissue sample from the transplanted kidney, which is then examined under a microscope to assess organ health. This is done to assess for any signs of rejection or other problems that may be occurring in the transplanted kidney. However, this is far from an ideal screening method given its invasive nature and the discomfort it can cause the patient. Large-scale studies in renal transplantation show that approximately 1% of biopsies generate major complications, with a risk of macroscopic hematuria greater than 3.5% [1]. Most biopsy-related complications, such as pain and bleeding, are minor and localized, and can be managed conservatively [2]. The most severe complication, however, is the risk of perforation of the collecting system or the kidney itself, which can result in severe hemorrhage, sepsis, and even death. To minimize the risk of complications, careful patient selection, proper imaging guidance, and specialized instruments and techniques are essential [3][4]. Imaging techniques, such as ultrasound and computed tomography (CT), are essential for accurate needle placement. Ultrasound imaging is the most commonly used modality for needle guidance due to its versatility, cost-effectiveness, and relative safety. It allows for real-time visualization of the renal transplant and the surrounding anatomy, making it ideal for guiding percutaneous needle placement. CT imaging can also be used but is typically reserved for more complex cases with insufficient ultrasound imaging.
In addition, with current immunosuppressive therapies, the detection of subclinical rejection is too infrequent to justify this risk, which has meant that many units no longer perform these routine biopsies [5], thus raising the urgent need to find a new non-invasive biomarker that allows the detection of said rejection in order to intervene in time or modify immunosuppression. In response to this need, recent studies have shown that non-invasive biomarkers could be a viable option for detecting subclinical rejection [6][7][8][9]. These non-invasive biomarkers include urinary and serum markers such as urinary albumin-to-creatinine ratio (UACR) and donor-specific antibodies (DSA). Additionally, imaging techniques, such as magnetic resonance imaging (MRI) and ultrasound (US), have been used to detect graft changes that may signal rejection [10][11][12]. Finally, genetic and epigenetic biomarkers, such as microRNAs, have also been used to detect subclinical rejection [13][14][15]. Ultimately, using these non-invasive biomarkers could help identify and intervene in cases of subclinical rejection earlier, thus avoiding more serious complications.
2. Types de Cell-Free DNA
Degraded DNA fragments released into the blood or other fluids are known as cfDNA. Its first detection dates back to 1948 in patients with systemic lupus erythematosus [16]. It would not be until 1970 that this new biomarker would begin to be considered helpful for the clinic, as researchers observed differences in its concentration depending on the health status of the individual studied and began to see its application in cancer patients by allowing the detection of fragments of tumor DNA in the blood [17]. Its interest increased when it was discovered that tumor cells not only released cfDNA into the bloodstream but that these fragments also had the genetic and epigenetic changes of the tumor cells from which they had originated [18]. Shortly after, analysis of fetal cfDNA in maternal plasma began to be used to detect Rh mismatches and chromosomal aneuploidies [19].
Recent literature shows that different cfDNA types can be used as biomarkers of various disease states [20][21]. The following stand out for their relevance: ccf mtDNA (circulating cell-free mitochondrial DNA), ctDNA (circulating tumor DNA), cffDNA (cell-free fetal DNA), and dd-cfDNA (donor-derived cell-free DNA). These types and their applications are listed in Table 1.
Table 1. Different types of cfDNA and their main clinical applications.
| Type of Cell-Free DNA | Abbreviations | Potential Application |
|---|---|---|
| Circulating cell-free mitochondrial DNA | ccf mtDNA | Diagnostic and predictive markers in various disease states, markers of cell death, and non-specific tissue damage |
| Circulating tumor DNA | ctDNA | Marker in oncological diagnostics, monitoring of tumor development |
| Cell-free fetal DNA | cffDNA | Prenatal diagnostics, detection of fetal defects |
| Donor-derived cell-free DNA | dd-cfDNA | Evaluation of post-transplant complications |
Extracted from [22].
3. History of Donor-Derived Cell-Free DNA
Non-patient cfDNA from a transplanted organ is known as circulating dd-cfDNA and can be detected in both blood and urine. Initially, the dd-cfDNA concentration increases to values greater than 5% after transplantation; however, these decrease rapidly and are practically undetectable after a week. Therefore, the presence of dd-cfDNA in the blood of the transplanted patient after a short period may be related to possible complications [1][23]. If the transplanted individual rejects the graft, the concentration of dd-cfDNA increases up to five times more than in healthy controls. In addition, these increased levels can be identified before any other clinical or biochemical symptom or complication, which is why it is of great interest for the early approach to subclinical rejection [24].
In 1998, this interesting biomarker was detected for the first time in the plasma and urine of solid organ recipients, identifying DNA from the Y chromosome in women transplanted with organs from male donors [24][25]. After this discovery, the technology began to evolve, emerging quantitative PCR techniques oriented to typing HLA genes. However, they had reproducibility problems and could not distinguish donor and recipient if they shared typings [5][26].
It would not be until 2011 that a method to detect dd-cfDNA employing digital PCR was devised based on analyzing the differences in SNPs between donor and recipient [27]. This strategy was optimized in 2016 with other authors [28] who, based on the principles of allelic imbalance, were able to measure dd-cfDNA levels by genotyping only the recipientm and between 150,000 and 600,000 SNPs from the donor, therefore, not being necessary to genotype the latter [15] altogether. Later that same year, other authors demonstrated that it was possible to quantify cfDNA levels by analyzing only 266 carefully chosen SNPs to minimize the probability that two unrelated individuals share them [29]. However, although these discoveries have laid the foundations for commercial dd-cfDNA detection kits, they have a series of limitations that have not yet been resolved, such as the impossibility of detecting dd-cfDNA levels in the transplantation of identical twins or differentiating the presence of more than two different genomes, as occurs in the case of retransplanted patients [20][30].
4. Commercial Tests for dd-cfDNA Detection
Three commercial dd-cfDNA detection kits are available for clinical use in kidney transplantation; Allosure from CareDx, Prospera from Natera, and Trac from Viracor Eurofins [22][26][31]. The most commonly used and described in various publications is CareDX, where a panel of 266 SNPs in 22 somatic chromosomes was used to study 102 kidney transplant recipients, 27 with rejection confirmed by biopsy [30]. In the said test, a 1% dd-cfDNA was established as a cut-off to discriminate the presence or absence of active rejection, and it had a specificity of 85%, a sensitivity of 59%, a positive predictive value (PPV) of 61% and an 84% of accuracy negative predictive value (NPV). In this study, TCMR was insufficiently detected, as confirmed by Huang in an independent study with the same test [32]. One reason may be using relatively long amplicons (100–130 bp) in the employed test. According to Clausen et al. [33], the recommended amplicon length is 85.4 bp (66–103 bp).
Prospera de Natera’s dd-cfDNA levels were determined in the following study by detecting 13,392 SNPs on four chromosomes [27][34], and a retrospective study was performed on 178 kidney transplant patients diagnosed with rejection. Acute T-lymphocyte-mediated (TCMR) or antibody-mediated (ABMR). Based on the results obtained, a cut-off for the presence of rejection more significant than 1% of dd-cfDNA was established, as in the Allosure method, with a specificity of 73%, a sensitivity of 89%, a PPV of 52%, and an NPV of 95% [35]. TCMR was well detected, presumably due to shorter amplicon size. Similar results were obtained in the Trifecta study [36].
These two studies would be the most relevant to date at the level of bibliographic background; however, a large number of companies are trying to develop dd-cfDNA kits, and that is where the Eurofins TRAC (Transplant Rejection Allograft Check) study would come in, which uses NGS techniques and recipient genotype data to determine the percentage of dd-cfDNA from the donated organ. In said study, biomarkers were determined in 77 kidney transplant patients with a cut-off of 0.70% to discriminate active rejection and sensitivity values of 58%, specificity of 85%, PPV of 55%, and NPV of 86% (Eurofins study) [35].
To clarify and inform the particular process, a schematic illustration of cfDNA isolation and analysis is shown in Figure 1.
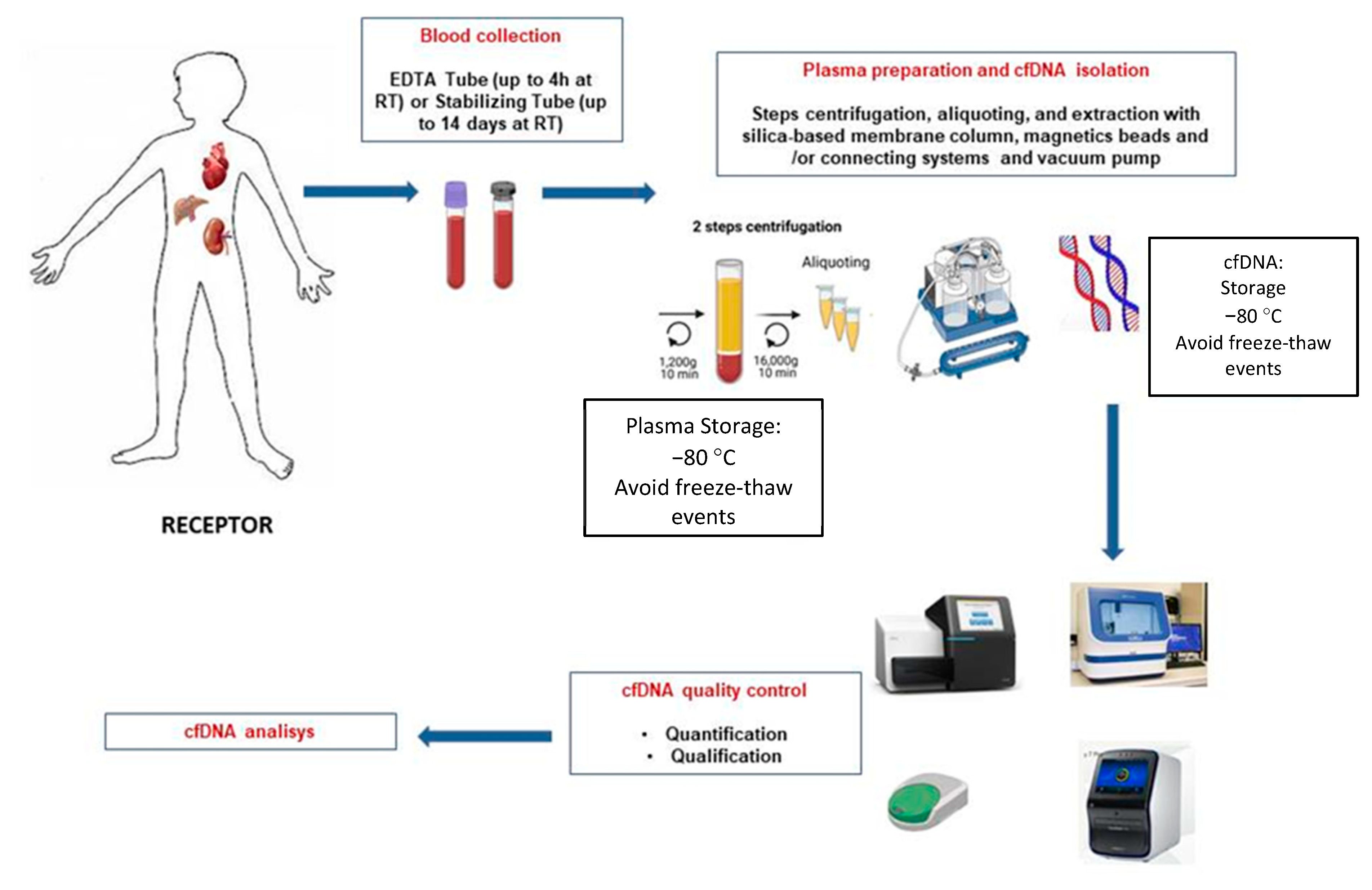
Figure 1. Schematic overview of the different steps for blood sample processing of graft recipients and cfDNA extraction.
These commercial kits for determining dd-cfDNA represent a significant advance in the early detection of subclinical rejection; however, their main drawback is that their prohibitive price means that they are still not valid as a detection technique screening [30][37]. Another group carried out an economic analysis in which commercial dd-cfDNA tests were used as screening for subclinical rejection in kidney transplant patients and concluded that this system was not cost-efficient and that, in order to reach so, the price of each test should be less than US$700, instead of the US$2200–2800 it currently ranges from [35]. That is why new technologies aimed at minimizing this cost are beginning to emerge, such as those based on dd-PCR [38], which reduce the price to less than US$400. However, this technology is relatively recent and has few studies that endorse it [39].
5. Current Status of dd-cfDNA
Leaving economic aspects aside, dd-cfDNA has the problem of having a relatively low PPV (although it should be mentioned that predictive values also depend on the variable prevalence of rejection in the study populations), which makes it difficult to interpret a high level of dd-cfDNA in the absence of additional clinical information, which is why it is often used, together with the analysis of leukocyte expression markers. However, its NPV is much higher, which allows it to play an essential role in avoiding unnecessary biopsies [40]. In addition, it is important to remember that other types of pathologies, independent of rejection, can raise dd-cfDNA levels, so it is essential to evaluate the results within the patient’s clinical context [30][41][42][43][44][45]. The absolute quantification of dd-cfDNA (cp/mL) has the advantage of not being affected by changes in the cfDNA receptor [46][47]. Leukopenia and leukocytosis can alter the dd-cfDNA fraction and produce false positive or negative results [38][48][49]. An example of these facts was the studies carried out by another group in 2020, which retrospectively studied elevations in dd-cfDNA levels in patients with BK viremia; however, these studies did not reach statistical significance [50]. Another group worked along the same lines, observing a positive correlation between BK viremia and increased dd-cfDNA levels in a small cohort of 10 patients [51].
In addition, since the initial validation studies were carried out at the specific moments in which rejection was suspected, there is still not a good understanding of how dd-cfDNA levels naturally evolve post-transplant. In addition, various factors both in the recipient (such as the panel reactive antibody [PRA]) and in the donor (living transplant or after circulatory death [DCD]) can influence dd-cfDNA levels and cause increases in the levels of dd-cfDNA themselves in the absence of suspicion of rejection [30]. Another physiological factor influencing dd-cfDNA that is being studied is obesity, with a retrospective study (in 2020) showing an inverse relationship between morbid obesity and dd-cfDNA levels [52].
The predictive value of dd-cfDNA has been extensively analyzed in acute cellular rejection (ACR) cases and AMR, the two main mechanisms of allograft damage. Among the multiple studies carried out, the most relevant was the so-called DART (Diagnosing Acute Rejection in Kidney Transplant), carried out in the United States with a representative sample of the kidney transplant population (n = 384) [34], which reports a better predictor of AMR than of ACR, since the levels of the biomarker in the first case are much higher, ranging between 1.4% and 2.9%. In comparison, for ACR, they are around 1.2%.
References
- Schwarz, A.; Gwinner, W.; Hiss, M.; Radermacher, J.; Mengel, M.; Haller, H. Safety and Adequacy of Renal Transplant Protocol Biopsies. Am. J. Transplant. 2005, 5, 1992–1996.
- Volpe, A.; Kachura, J.R.; Geddie, W.R.; Evans, A.J.; Gharajeh, A.; Saravanan, A.; Jewett, M.A.S. Techniques, Safety and Accuracy of Sampling of Renal Tumors by Fine Needle Aspiration and Core Biopsy. J. Urol. 2007, 178, 379–386.
- Kim, K.R.; Thomas, S. Complications of image-guided thermal ablation of liver and kidney neoplasms. Semin. Intervent. Radiol. 2014, 31, 138–148.
- Michel, M.S.; Trojan, L.; Rassweiler, J.J. Complications in Percutaneous Nephrolithotomy. Eur. Urol. 2007, 51, 899–906.
- Rush, D.N.; Gibson, I.W. Subclinical inflammation in renal transplantation. Transplantation 2019, 103, E139–E145.
- Mac, Q.D.; Mathews, D.V.; Kahla, J.A.; Stoffers, C.M.; Delmas, O.M.; Holt, B.A.; Adams, A.B.; Kwong, G.A. Non-invasive early detection of acute transplant rejection via nanosensors of granzyme B activity. Nat. Biomed. Eng. 2019, 3, 281–291.
- Hirt-Minkowski, P.; De Serres, S.A.; Ho, J. Developing renal allograft surveillance strategies-urinary biomarkers of cellular rejection. Can. J. Kidney Health Dis. 2015, 2, 28.
- Jamshaid, F.; Froghi, S.; Di Cocco, P.; Dor, F.J.M.F. Novel non-invasive biomarkers diagnostic of acute rejection in renal transplant recipients: A systematic review. Int. J. Clin. Pract. 2018, 72, e13220.
- Baumann, A.K.; Beck, J.; Kirchner, T.; Hartleben, B.; Schütz, E.; Oellerich, M.; Wedemeyer, H.; Jaeckel, E.; Taubert, R. Elevated fractional donor-derived cell-free DNA during subclinical graft injury after liver transplantation. Liver Transplant. 2022, 28, 1911–1919.
- Neimatallah, M.A.; Dong, Q.; Schoenberg, S.O.; Cho, K.J.; Prince, M.R. Magnetic Resonance Imaging in Renal Transplantation. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 1999, 10, 357–368.
- Hollis, E.; Shehata, M.; Khalifa, F.; Abou El-Ghar, M.; El-Diasty, T.; El-Baz, A. Towards non-invasive diagnostic techniques for early detection of acute renal transplant rejection: A review. Egypt. J. Radiol. Nucl. Med. 2017, 48, 257–269.
- Farag, A.; El-Baz, A.; Yuksel, S.E.; El-Ghar, M.A.; Eldiasty, T. A framework for the detection of acute renal rejection with dynamic contrast enhanced magnetic resonance imaging. In Proceedings of the 3rd IEEE International Symposium on Biomedical Imaging: Nano to Macro, Arlington, VA, USA, 6–9 April 2006; 2006, pp. 418–421.
- Bontha, S.V.; Maluf, D.G.; Mueller, T.F.; Mas, V.R. Systems Biology in Kidney Transplantation: The Application of Multi-Omics to a Complex Model. Am. J. Transplant. 2017, 17, 11–21.
- Asvapromtada, S.; Sonoda, H.; Kinouchi, M.; Oshikawa, S.; Takahashi, S.; Hoshino, Y.; Sinlapadeelerdkul, T.; Yokota-Ikeda, N.; Matsuzaki, T.; Ikeda, M. Recent Advances on Biomarkers of Early and Late Kidney Graft Dysfunction. Int. J. Mol. Sci. 2020, 21, 5404.
- Salvadori, M.; Tsalouchos, A. Biomarkers in renal transplantation: An updated review. World J. Transplant. 2017, 7, 161.
- Tug, S.; Helmig, S.; Menke, J.; Zahn, D.; Kubiak, T.; Schwarting, A.; Simon, P. Correlation between cell free DNA levels and medical evaluation of disease progression in systemic lupus erythematosus patients. Cell Immunol. 2014, 292, 32–39.
- Ulrich, B.C.; Guibert, N. Towards a comprehensive framework for cell-free DNA analysis: Lessons from TRACERx. Ann. Transl. Med. 2017, 5, 446–451.
- Arneth, B. Update on the types and usage of liquid biopsies in the clinical setting: A systematic review. BMC Cancer 2018, 18, 527.
- Jeon, S.H.; Lee, H.J.; Bae, K.; Yoon, K.A.; Lee, E.S.; Cho, Y. Efficient Capture and Isolation of Tumor-Related Circulating Cell-Free DNA from Cancer Patients Using Electroactive Conducting Polymer Nanowire Platforms. Theranostics 2016, 6, 828.
- Jimenez-Coll, V.; Llorente, S.; Boix, F.; Alfaro, R.; Galián, J.A.; Martinez-Banaclocha, H.; Botella, C.; Moya-Quiles, M.R.; Muro-Pérez, M.; Minguela, A.; et al. Monitoring of Serological, Cellular and Genomic Biomarkers in Transplantation, Computational Prediction Models and Role of Cell-Free DNA in Transplant Outcome. Int. J. Mol. Sci. 2023, 24, 3908.
- Pascual, J.; Attard, G.; Bidard, F.C.; Curigliano, G.; De Mattos-Arruda, L.; Diehn, M.; Italiano, A.; Lindberg, J.; Merker, J.D.; Montagut, C.; et al. ESMO recommendations on the use of circulating tumour DNA assays for patients with cancer: A report from the ESMO Precision Medicine Working Group. Ann. Oncol. 2022, 33, 750–768.
- Bloom, R.D.; Bromberg, J.S.; Poggio, E.D.; Bunnapradist, S.; Langone, A.J.; Sood, P.; Matas, A.J.; Mehta, S.; Mannon, R.B.; Sharfuddin, A.; et al. Cell-Free DNA and active rejection in kidney allografts. J. Am. Soc. Nephrol. 2017, 28, 2221–2232.
- Starzl, T.E.; Murase, N.; Ildstad, S.; Ricordi, C.; Demetris, A.J.; Trucco, M. Cell migration, chimerism, and graft acceptance. Lancet 1992, 339, 1579.
- Beck, J.; Bierau, S.; Balzer, S.; Andag, R.; Kanzow, P.; Schmitz, J.; Gaedcke, J.; Moerer, O.; Slotta, J.E.; Walson, P.; et al. Digital Droplet PCR for Rapid Quantification of Donor DNA in the Circulation of Transplant Recipients as a Potential Universal Biomarker of Graft Injury. Clin. Chem. 2013, 59, 1732–1741.
- Lo, Y.M.D.; Tein, M.S.C.; Pang, C.C.P.; Yeung, C.K.; Tong, K.L.; Magnus Hjelm, N. Presence of donor-specific DNA in plasma of kidney and liver-transplant recipients. Lancet 1998, 351, 1329–1330.
- Kueht, M.L.; Dongur, L.P.; Cusick, M.; Stevenson, H.L.; Mujtaba, M. The Current State of Donor-Derived Cell-Free DNA Use in Allograft Monitoring in Kidney Transplantation. J. Pers. Med. 2022, 12, 1700.
- Gadi, V.K.; Nelson, J.L.; Boespflug, N.D.; Guthrie, K.A.; Kuhr, C.S. Soluble Donor DNA Concentrations in Recipient Serum Correlate with Pancreas-Kidney Rejection. Clin. Chem. 2006, 52, 379–382.
- Snyder, T.M.; Khush, K.K.; Valantine, H.A.; Quake, S.R. Universal noninvasive detection of solid organ transplant rejection. Proc. Natl. Acad. Sci. USA 2011, 108, 6229–6234.
- Sharon, E.; Shi, H.; Kharbanda, S.; Koh, W.; Martin, L.R.; Khush, K.K.; Valantine, H.; Pritchard, J.K.; De Vlaminck, I. Quantification of transplant-derived circulating cell-free DNA in absence of a donor genotype. PLoS Comput. Biol. 2017, 13, e1005629.
- Grskovic, M.; Hiller, D.J.; Eubank, L.A.; Sninsky, J.J.; Christopherson, C.; Collins, J.P.; Thompson, K.; Song, M.; Wang, Y.S.; Ross, D.; et al. Validation of a Clinical-Grade Assay to Measure Donor-Derived Cell-Free DNA in Solid Organ Transplant Recipients. J. Mol. Diagn. 2016, 18, 890–902.
- Huang, E.; Jordan, S.C. Donor-derived cell-free DNA in kidney transplantation: Evolving concepts and potential limitations. Kidney Int. 2022, 101, 676–677.
- Huang, E.; Sethi, S.; Peng, A.; Najjar, R.; Mirocha, J.; Haas, M.; Vo, A.; Jordan, S.C. Early clinical experience using donor-derived cell-free DNA to detect rejection in kidney transplant recipients. Am. J. Transplant. 2019, 19, 1663–1670.
- Clausen, F.B.; Jørgensen, K.M.C.L.; Wardil, L.W.; Nielsen, L.K.; Krog, G.R. Droplet digital PCR-based testing for donor-derived cell-free DNA in transplanted patients as noninvasive marker of allograft health: Methodological aspects. PLoS ONE 2023, 18, e0282332.
- 30876—Viracor TRAC® Kidney dd-cfDNA | Clinical | Eurofins-Viracor. Available online: https://www.eurofins-viracor.com/clinical/test-menu/30876-viracor-trac-kidney-dd-cfdna/ (accessed on 17 February 2023).
- Sigdel, T.K.; Archila, F.A.; Constantin, T.; Prins, S.A.; Liberto, J.; Damm, I.; Towfighi, P.; Navarro, S.; Kirkizlar, E.; Demko, Z.P.; et al. Optimizing Detection of Kidney Transplant Injury by Assessment of Donor-Derived Cell-Free DNA via Massively Multiplex PCR. J. Clin. Med. 2018, 8, 19.
- Halloran, P.F.; Reeve, J.; Madill-Thomsen, K.S.; Demko, Z.; Prewett, A.; Billings, P. The Trifecta Study: Comparing Plasma Levels of Donor-derived Cell-Free DNA with the Molecular Phenotype of Kidney Transplant Biopsies. J. Am. Soc. Nephrol. 2022, 33, 387–400.
- Muro, M. ¿Es el ADN libre circulante (cfDNA) derivado del donante (dd-cfDNA) un nuevo biomarcador para el rechazo de aloinjertos en trasplante? Inmunología 2020, 39, 23–26.
- Oellerich, M.; Sherwood, K.; Keown, P.; Schütz, E.; Beck, J.; Stegbauer, J.; Rump, L.C.; Walson, P.D. Liquid biopsies: Donor-derived cell-free DNA for the detection of kidney allograft injury. Nat. Rev. Nephrol. 2021, 17, 591–603.
- Puttarajappa, C.M.; Mehta, R.B.; Roberts, M.S.; Smith, K.J.; Hariharan, S. Economic analysis of screening for subclinical rejection in kidney transplantation using protocol biopsies and noninvasive biomarkers. Am. J. Transplant. 2021, 21, 186–197.
- Porrini, E.; Ruggenenti, P.; Luis-Lima, S.; Carrara, F.; Jiménez, A.; de Vries, A.P.J.; Torres, A.; Gaspari, F.; Remuzzi, G. Estimated GFR: Time for a critical appraisal. Nat. Rev. Nephrol. 2018, 15, 177–190.
- Filippone, E.J.; Farber, J.L. The Monitoring of Donor-derived Cell-free DNA in Kidney Transplantation. Transplantation 2021, 105, 509–516.
- Kataria, A.; Kumar, D.; Gupta, G. Donor-derived Cell-free DNA in Solid-organ Transplant Diagnostics: Indications, Limitations, and Future Directions. Transplantation 2021, 105, 1203–1211.
- O’Callaghan, J.M.; Knight, S.R. Noninvasive biomarkers in monitoring kidney allograft health. Curr. Opin. Organ Transplant. 2019, 24, 411–415.
- Rosenheck, J.P.; Keller, B.C.; Fehringer, G.; Demko, Z.P.; Bohrade, S.M.; Ross, D.J. Why Cell-Free DNA Can Be a “Game Changer” for Lung Allograft Monitoring for Rejection and Infection. Curr. Pulmonol. Rep. 2022, 11, 75–85.
- Oellerich, M.; Budde, K.; Osmanodja, B.; Bornemann-Kolatzki, K.; Beck, J.; Schütz, E.; Walson, P.D. Donor-derived cell-free DNA as a diagnostic tool in transplantation. Front. Genet. 2022, 13, 1031894.
- Oellerich, M.; Shipkova, M.; Asendorf, T.; Walson, P.D.; Schauerte, V.; Mettenmeyer, N.; Kabakchiev, M.; Hasche, G.; Gröne, H.J.; Friede, T.; et al. Absolute quantification of donor-derived cell-free DNA as a marker of rejection and graft injury in kidney transplantation: Results from a prospective observational study. Am. J. Transplant. 2019, 19, 3087–3099.
- Whitlam, J.B.; Ling, L.; Skene, A.; Kanellis, J.; Ierino, F.L.; Slater, H.R.; Bruno, D.L.; Power, D.A. Diagnostic application of kidney allograft-derived absolute cell-free DNA levels during transplant dysfunction. Am. J. Transplant. 2019, 19, 1037–1049.
- Oellerich, M.; Wu, A.; Halloran, P.F.; De Vlaminck, I.; Keller, M.; Agbor-Enoh, S. Molecular Approaches to Transplant Monitoring; Is the Horizon Here? Clin. Chem. 2021, 67, 1443–1449.
- Oellerich, M.; Budde, K.; Osmanodja, B.; Bornemann-Kolatzki, K.; Beck, J.; Schütz, E.; Walson, P.D. Donor-Derived Cell-free DNA for Personalized Immunosuppression in Renal Transplantation. Ther. Drug Monit. 2023, 45, 20–25.
- Lum, E.L.; Nieves-Borrero, K.; Homkrailas, P.; Lee, S.; Danovitch, G.; Bunnapradist, S. Single center experience comparing two clinically available donor derived cell free DNA tests and review of literature. Transplant. Rep. 2021, 6, 100079.
- Goussous, N.; Xie, W.; Dawany, N.; Scalea, J.R.; Bartosic, A.; Haririan, A.; Kalil, R.; Drachenberg, C.; Costa, N.; Weir, M.R.; et al. Donor-derived Cell-free DNA in Infections in Kidney Transplant Recipients: Case Series. Transplant. Direct 2020, 6, e568.
- Kant, S.; Bromberg, J.; Haas, M.; Brennan, D. Donor-derived Cell-free DNA and the Prediction of BK Virus-associated Nephropathy. Transplant. Direct 2020, 6, e622.
More
Information
Subjects:
Immunology
Contributors
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.2K
Revisions:
2 times
(View History)
Update Date:
20 Jun 2023
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





