
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Samuel Jacob | -- | 1806 | 2023-06-06 08:17:07 | | | |
| 2 | Conner Chen | Meta information modification | 1806 | 2023-06-07 07:13:25 | | |
Video Upload Options
The exploration of natural substrates for microbial conversion to synthesize industrial platform and fuel chemicals seems to be inevitable within a circular bioeconomy context. Hemicellulose is a natural carbohydrate polymer consisting of a variety of pentose (C5) sugar monomers such as arabinose, mannose, erythrose, and xylose. Among the C5 sugars, L-arabinose (L-Ara) is the second-most-abundant pentose sugar in the lignocellulosic biomass after xylose. L-Ara has been used as an industrial carbon source to produce several value-added chemicals such as putrescine, which is used to synthesize polymers in the textile industry; sugar alcohols that are used as sweeteners in diet foods; and amino acids such as L-lysine, L-glutamate, L-arginine, and L-ornithine, which are used in nutritional supplements, fertilizers, and other products in the food and beverage industries. L-Ara, a natural non-caloric sweetener, is used as a substitute in the food and beverage industry, when the risk of blood sugar and lipid levels could be reduced. Major use of L-Ara is also found in the medical and pharmaceutical sectors to treat several conditions, including mineral absorption disorder, constipation, and diabetes, among others.
1. Introduction
| ValueAdded Products | Yield | Microbe | ‘C’ Source | Reference |
|---|---|---|---|---|
| Organic acids | Lactic Acid: 12.1 g/L Fumaric Acid: 7.4 g/L Acetic Acid: 4.5 g/L |
Lactobacillus sakei WiKim31 | Kimchi cabbage waste | [15] |
| Putrescine | 19 g/L | Clostridium glutamicum PUT21 | Glucose | [16] |
| Amino acids | L-Lysine: 9.9 g/L L-Ornithine: 25.8 g/L L-Arginine: 8.4 g/L |
C. glutamicum ARG1 | Glucose and L-Ara | [6] |
2. Abundance and Significance of L-Ara as a Bioresource
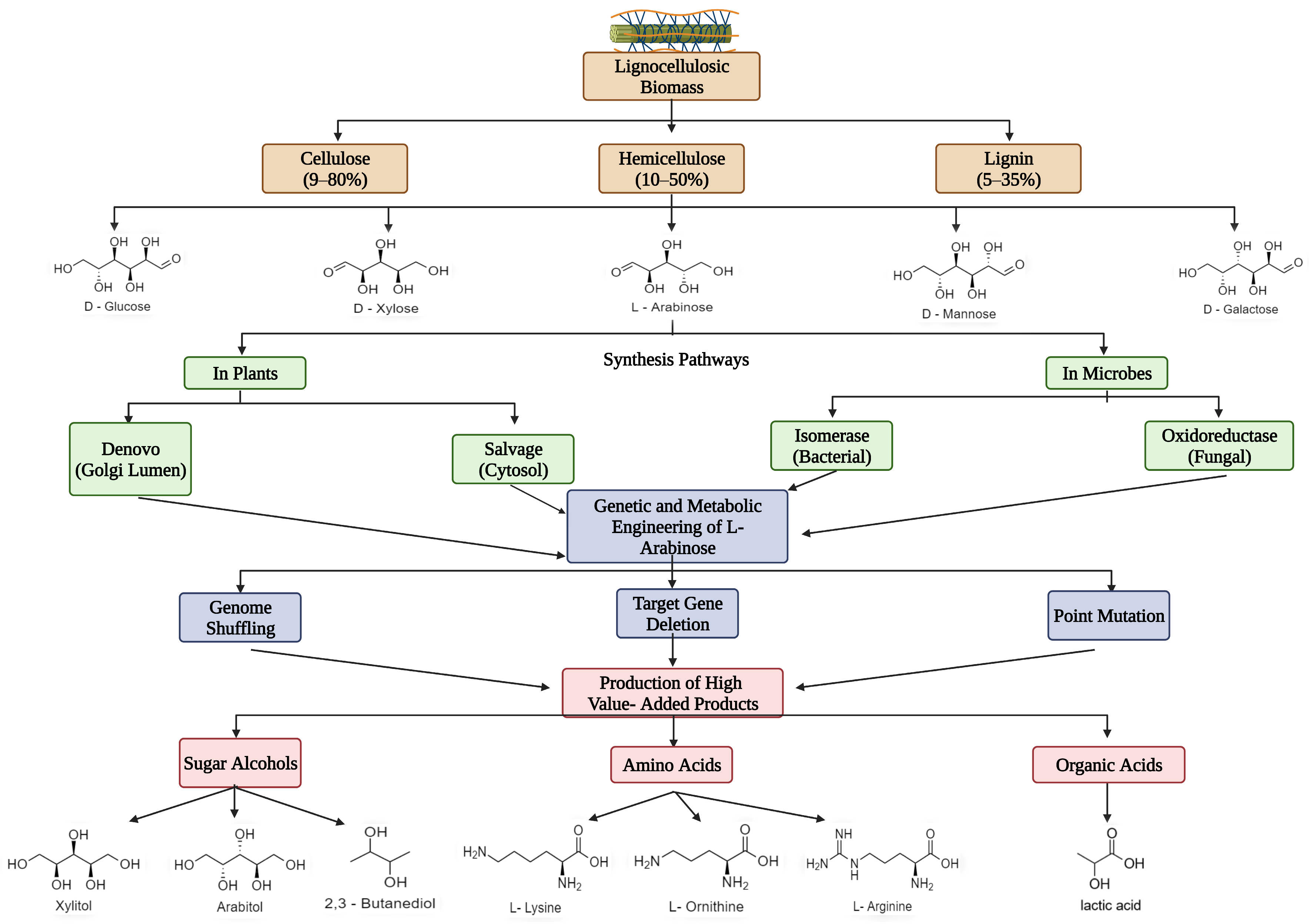
| Feedstock | Lignin (%) | Hemicellulose (%) | Cellulose (%) | Pre-Treatment Method Used to Obtain L-Ara | Applications | References |
|---|---|---|---|---|---|---|
| Sugarcane bagasse | 25–32 | 19–24 | 32–43 | Acid hydrolysis resulted in 2.78 g/L of L-Ara | Food coatings, hydrogels, packaging films, cationic biopolymers, and other biomedical uses | [32][33][34] |
| Corn stover | 19 | 22 | 36 | Acid hydrolysis resulted in 38.2% L-Ara yield in 8 h reaction time | Advanced biofuels and livestock feed | [35][36][37] |
| Rice straw | 15 | 18 | 35 | Combined pre-treatment methods resulted in 2.7–4.5% of L-Arayield | Biofuel and ethanol production | [38][39][40] |
| Water hyacinth | 10 | 35 | 25 | Sulphuric acid treatment resulted in 33.3 g/L yield of L-Ara | Biothanol production using Pichia stipitis | [41][42] |
| Wheat straw | 16–25 | 23–24 | 28–39 | Hot water and NaOH treatment resulted in 2.37 ± 0.09% of L-Ara | Adsorbents, packing materials, bioplastic industry, and several other industries | [43][44][45] |
| Sugar beet molasses | 6 | 30 | 22–24 | Acid alkali pretreatment along with ultrafiltration resulted in 92% recovery of L-Ara | Food industry, as a bakery or confectionery product, apart from being utilized as a ruminant feed | [46][47] |
| Apple pomace | 19 | 10 | 12 | Sulphuric acid treatment resulted in 90% yield of L-Ara | Bioethanol, animal feed, citric acid, and several other applications | [48][49][50] |
| Orange peels | 20 | 9 | 69 | Acid alkali treatment to extract L-Ara | Bioethanol, essential oils, and biogas | [51][52] |
| Carrot pomace | 17 | 7 | 28 | Acid treatment to extract L-Ara | Fertilizer, feed for livestock, dietary fiber, and production of biofuels | [53][54] |
| Tomato pomace | 7 | 31 | 38 | Acid treatment to extract L-Ara | Fertilizer, feed for livestock, dietary fiber, and production of biofuels | [54][55][56] |
2.1. 2,3-Butanediol
2.2. Other Value-Added Products
References
- Francois, J.; Alkim, C.; Morin, N. Engineering microbial pathways for production of bio-based chemicals from lignocellulosi/.c sugars: Current status and perspectives. Biotechnol. Biofuels 2020, 13, 118.
- Paul, S.; Dutta, A. Challenges and opportunities of lignocellulosic biomass for anaerobic digestion. Resour. Conserv. Recycl. 2018, 130, 164–174.
- Holtzapple, M.T. Cellulose, hemicelluloses, and lignin. In Encyclopedia of Food Science, Food Technology, and Nutrition; Macrae, R., Robinson, R.K., Sadler, M.J., Eds.; Academic Press: London, UK, 1993; pp. 2731–2738.
- Sekeri, S.H.; Ibrahim, M.N.M.; Umar, K.; Yaqoob, A.A.; Azmi, M.N.; Hussin, M.H.; Othman, M.B.H.; Malik, M.F.I.A. Preparation and characterization of nanosized lignin from oil palm (Elaeis guineensis) biomass as a novel emulsifying agent. Int. J. Biol. Macromol. 2020, 164, 3114–3124.
- Yaqoob, A.A.; Sekeri, S.H.; Othman, M.B.H.; Ibrahim, M.N.M.; Feizi, Z.H. Thermal degradation and kinetics stability studies of oil palm (Elaeis Guineensis) biomass-derived lignin nanoparticle and its application as an emulsifying agent. Arab. J. Chem. 2021, 14, 103182.
- Schneider, J.; Niermann, K.; Wendisch, V. Production of the amino acids L-glutamate, L-lysine, L-ornithine and L-arginine from L-Ara by recombinant Corynebacterium glutamicum. J. Biotechnol. 2011, 154, 191–198.
- Meiswinkel, T.; Gopinath, V.; Lindner, S.; Nampoothiri, K.; Wendisch, V. Accelerated pentose utilization by Corynebacterium glutamicum for accelerated production of lysine, glutamate, ornithine and putrescine. Microb. Biotechnol. 2012, 6, 131–140.
- Rao, J.; Lv, Z.; Chen, G.; Peng, F. Hemicellulose: Structure, Chemical Modification, and Application. Prog. Polym. Sci. 2023, 140, 101675.
- Pauly, M.; Gille, S.; Liu, L.; Mansoori, N.; de Souza, A.; Schultink, A.; Xiong, G. Hemicellulose biosynthesis. Planta 2013, 238, 627–642.
- Banu, J.R.; Kavitha, P.S.; Tyagi, V.K.; Gunasekaran, M.; Karthikeyan, O.P.; Kumar, G. Lignocellulosic biomass based biorefinery: A successful platform towards circular bioeconomy. Fuel 2021, 302, 121086.
- Market Watch. Food Grade L-Arabinose Market Demand by 2030. Available online: https://www.marketwatch.com/press-release/food-grade-l-arabinose-market-demand-by-2030-2023-04-06 (accessed on 10 April 2023).
- Lane, S.; Xu, H.; Oh, E.J.; Kim, H.; Lesmana, A.; Jeong, D.; Zhang, G.; Tsai, C.S.; Jin, Y.S.; Kim, S.R. Glucose repression can be alleviated by reducing glucose phosphorylation rate in Saccharomyces cerevisiae. Sci. Rep. 2018, 8, 2613.
- Mohamed, E.T.; Mundhada, H.; Landberg, J.; Cann, I.; Mackie, R.I.; Nielsen, A.T.; Herrgard, M.J.; Feist, A.M. Generation of an E. coli platform strain for improved sucrose utilization using adaptive laboratory evolution. Microb. Cell Fact. 2019, 18, 116.
- Ceroni, F.; Carbonell, P.; François, J.M.; Haynes, K.A. Editorial–Synthetic biology: Engineering complexity and refactoring cell capabilities. Front. Bioeng. Biotechnol. 2015, 3, 120.
- Kim, H.M.; Park, J.H.; Choi, I.S.; Wi, S.G.; Ha, S.; Chun, H.H.; Hwang, I.M.; Chang, J.Y.; Choi, H.-J.; Kim, J.-C.; et al. Effective approach to organic acid production from agricultural kimchi cabbage waste and its potential application. PLoS ONE 2018, 13, e0207801.
- Schneider, J.; Eberhardt, D.; Wendisch, V.F. Improving putrescine production by Corynebacterium glutamicum by fine-tuning ornithine transcarbamoylase activity using a plasmid addiction system. Appl. Microbiol. Biotechnol. 2012, 95, 169–178.
- Venkateswar Rao, L.; Goli, J.; Gentela, J.; Koti, S. Bioconversion of lignocellulosic biomass to xylitol: An overview. Bioresour. Technol. 2016, 213, 299–310.
- Safian, M.T.-U.; Sekeri, S.H.; Yaqoob, A.A.; Serra, A.; Jamudin, M.D.; Ibrahim, M.N.M. Utilization of lignocellulosic biomass: A practical journey towards the development of emulsifying agent. Talanta 2022, 239, 123109.
- Gírio, F.; Fonseca, C.; Carvalheiro, F.; Duarte, L.; Marques, S.; Bogel-Łukasik, R. Hemicelluloses for fuel ethanol: A review. Bioresour. Technol. 2010, 101, 4775–4800.
- Fehér, C. Novel approaches for biotechnological production and application of L-arabinose. J. Carbohydr. Chem. 2018, 37, 251–284.
- Kennedy, M.; List, D.; Lu, Y.; Foo, L.Y.; Newman, R.H.; Sims, I.M.; Bain, P.J.S.; Hamilton, B.; Fenton, G. Apple pomace and products derived from apple pomace: Use, composition and analysis. In Modern Methods of Plant Analysis, Analysis of Plant Waste Materials; Linskens, H.F., Jackson, J.F., Eds.; Springer-Verlag: Berlin, Germany, 1999; Volume 20, pp. 75–119.
- Doran, J.B.; Cripe, J.; Sutton, M.; Foster, B. Fermentations of pectin rich biomass with recombinant bacteria to produce fuel ethanol. Appl. Biochem. Biotechnol. 2000, 84, 141–152.
- Mohnen, D. Pectin structure and biosynthesis. Curr. Opin. Plant Biol. 2008, 11, 266–277.
- Zhou, W.; Widmer, W.; Grohmann, K. Developments in ethanol production from citrus peel waste. Proc. Fla. State Hort. Soc. 2008, 121, 307–310.
- Edwards, M.; Doran-Peterson, J. Pectin-rich biomass as feedstock for fuel ethanol production. Appl. Microbiol. Biotechnol. 2012, 95, 565–575.
- Seiboth, B.; Metz, B. Fungal arabinan and L-arabinose metabolism. Appl. Microbiol. Biotechnol. 2011, 89, 1665–1673.
- Hollmann, J.; Lindhauer, M. Pilot-scale isolation of glucuronoarabinoxylans from wheat bran. Carbohydr. Polym. 2005, 59, 225–230.
- Fehér, C. Integrated process of arabinose biopurification and xylitol fermentation based on the diverse action of Candida boidinii. Chem. Biochem. Eng. Q. 2016, 29, 587–597.
- Kühnel, S.; Schols, H.; Gruppen, H. Aiming for the complete utilization of sugar-beet pulp: Examination of the effects of mild acid and hydrothermal pretreatment followed by enzymatic digestion. Biotechnol. Biofuels 2011, 4, 14.
- Treimo, J.; Westereng, B.; Horn, S.J.; Forssell, P.; Robertson, J.A.; Faulds, C.B.; Waldron, K.W.; Buchert, J.; Eijsink, V.G.H. Enzymatic solubilization of brewers’ spent grain by combined action of carbohydrases and peptidases. J. Agric. Food Chem. 2009, 57, 3316–3324.
- Gottschalk, L.; Oliveira, R.; Bon, E. Cellulases, xylanases, β-glucosidase and ferulic acid esterase produced by Trichoderma and Aspergillus act synergistically in the hydrolysis of sugarcane bagasse. Biochem. Eng. J. 2010, 51, 72–78.
- Sakdaronnarong, C.; Jonglertjunya, W. Rice straw and sugarcane bagasse degradation mimicking lignocellulose decay in nature: An alternative approach to biorefinery. ScienceAsia 2012, 38, 364.
- Sabiha-Hanim, S.; Siti-Norsafurah, A.M. Physical properties of hemicellulose films from sugarcane bagasse. Procedia Eng. 2012, 42, 1390–1395.
- Tsigie, Y.; Wang, C.; Truong, C.; Ju, Y. Lipid production from Yarrowia lipolytica Po1g grown in sugarcane bagasse hydrolysate. Bioresour. Technol. 2011, 102, 9216–9222.
- Yang, S.; Zhang, Y.; Yue, W.; Wang, W.; Wang, Y.-Y.; Yuan, T.-Q.; Sun, R.-C. Valorization of lignin and cellulose in acid-steam-exploded corn stover by a moderate alkaline ethanol post-treatment based on an integrated biorefinery concept. Biotechnol. Biofuels. 2016, 9, 238.
- Cellulosic Biofuel Process Can also Improve Ruminant Forage Digestibility. MSU Extension. Available online: https://www.canr.msu.edu/news/cellulosic_biofuel_process_can_also_improve_ruminant_forage_digestibility. (accessed on 28 April 2022).
- Fehér, A. Combined approaches to xylose production from corn stover by dilute acid hydrolysis. Chem. Biochem. Eng. Q. 2017, 31, 77–87.
- Jiang, M.; Zhao, M.; Zhou, Z.; Huang, T.; Chen, X.; Wang, Y. Isolation of cellulose with ionic liquid from steam exploded rice straw. Ind. Crops Prod. 2011, 33, 734–738.
- Pinzi, S.; Dorado, M. Vegetable-based feedstocks for biofuels production. In Handbook of Biofuels Production: Processes and Technologies; Luque, R., Campelo, J., Clark, J., Eds.; Woodhead Publishing Ltd.: Cambridge, UK, 2011; pp. 61–94.
- Roberto, I.; Mussatto, S.; Rodrigues, R. Dilute-acid hydrolysis for optimization of xylose recovery from rice straw in a semi-pilot reactor. Ind. Crops. Prod. 2003, 17, 171–176.
- Nigam, J.N. Bioconversion of water-hyacinth (Eichhornia crassipes) hemicellulose acid hydrolysate to motor fuel ethanol by xylose-fermenting yeast. J. Biotechnol. 2002, 97, 107–116.
- Alfaro, J.R.; Daza, L.T.; Lindado, G.; Peláez, H.C.; Córdoba, Á.P. Acid hydrolysis of water hyacinth to obtaining fermentable sugars. Cienc. Tecnol. Futuro. 2013, 5, 101–112.
- Carvalheiro, F.; Silva-Fernandes, T.; Duarte, L.C.; Gírio, F.M. Wheat straw autohydrolysis: Process optimization and products characterization. Appl. Biochem. Biotechnol. 2009, 153, 84–93.
- Farhat, W.; Venditti, R.; Hubbe, M.; Taha, M.; Becquart, F.; Ayoub, A. A review of water-resistant hemicellulose-based materials: Processing and applications. Chem. Sus. Chem. 2016, 10, 305–323.
- Tozluoğlu, A.; Özyurek, Ö.; Çöpür, Y.; Özdemir, H. Integrated production of biofilm, bioethanol, and papermaking pulp from wheat Straw. Bioresour. 2015, 10, 7834–7854.
- Olmos, J.C.; Hansen, M.Z. Enzymatic depolymerization of sugar beet pulp: Production and characterization of pectin and pectic-oligosaccharides as a potential source for functional carbohydrates. Chem. Eng. J. 2012, 192, 29–36.
- Saric, L.; Filipcev, B.; Simurina, O.; Plavsic, D. Sugar beet molasses: Properties and applications in osmotic dehydration of fruits and vegetables. Food Feed Res. 2016, 43, 135–144.
- Dinand, E.; Chanzy, H.; Vignon, M. Parenchymal cell cellulose from sugar beet pulp: Preparation and properties. Cellulose 1996, 3, 183–188.
- Eveleigh, D.E. Comprehensive biotechnology: The principles, applications and regulations of biotechnology in industry, agriculture and medicine. In The Principles of Biotechnology: Scientific Fundamentals; Moo-Young, M., Bull, A.T., Dalton, H., Eds.; Pergamon Press: Oxford, UK, 1985; Volume 1, p. 688.
- Gama, R.; Dyk, J.V.; Pletschke, B. Optimisation of enzymatic hydrolysis of apple pomace for production of biofuel and biorefinery chemicals using commercial enzymes. 3 Biotech 2015, 5, 1075–1087.
- Ayala, J.R.; Montero, G.; Coronado, M.A.; García, C.; Curiel-Alvarez, M.A.; León, J.A.; Sagaste, C.A.; Montes, D.G. Characterization of orange peel waste and valorization to obtain reducing sugars. Molecules 2021, 26, 1348.
- Torrado, A.M.; Cortés, S.; Salgado, J.M.; Max, B.; Rodríguez, N.; Bibbins, B.P.; Converti, A.; Domínguez, J.M. Citric acid production from orange peel wastes by solid-state fermentation. Braz. J. Microbiol. 2011, 42, 394–409.
- Nawirska, A.; Kwaśniewska, M. Dietary fibre fractions from fruit and vegetable processing waste. Food Chem. 2005, 91, 221–225.
- Szymańska-Chargot, M.; Chylińska, M.; Gdula, K.; Kozioł, A.; Zdunek, A. Isolation and characterization of cellulose from different fruit and vegetable pomaces. Polymers 2017, 9, 495.
- Kheiralla, Z.H.; El-Gendy, N.S.; Ahmed, H.A.; Shaltout Th, H.; Hussein, M.M.D. Upgrading of Tomato (Solanum lycopersicum) Agroindustrial Wastes. J. Microb. Biochem. Technol. 2018, 10, 46–48.
- Del Valle, M.; Cámara, M.; Torija, M. Chemical characterization of tomato pomace. J. Sci. Food Agric. 2006, 86, 1232–1236.
- Song, C.W.; Park, J.M.; Chung, S.C.; Lee, S.Y.; Song, H. Microbial production of 2,3-butanediol for industrial applications. J. Ind. Microbiol. Biotechnol. 2019, 46, 1583–1601.
- Ma, C.; Wang, A.; Qin, J.; Li, L.; Ai, X.; Jiang, T.; Tang, H.; Xu, P. Enhanced 2, 3-butanediol production by Klebsiella pneumoniae SDM. Appl. Microbiol. Biotechnol. 2009, 82, 49–57.
- Yang, Z.; Zhang, Z. Recent advances on production of 2, 3-butanediol using engineered microbes. Biotechnol. Adv. 2019, 37, 569–578.
- Saha, B.C.; Bothast, R.J. Production of 2,3-butanediol by newly isolated Enterobacter cloacae. Appl. Microbiol. Biotechnol. 1999, 52, 321–326.
- Białkowska, A.M. Strategies for efficient and economical 2, 3-butanediol production: New trends in this field. World J. Microbiol. Biotechnol. 2016, 32, 1–14.
- Leuchtenberger, W.; Huthmacher, K.; Drauz, K. Biotechnological production of amino acids and derivatives: Current status and prospects. Appl. Microbiol. Biotechnol. 2005, 69, 1–8.
- Hahn-Hagerdal, B.; Karhumaa, K.; Fonseca, C.; Spencer-Martins, I.; Gorwa-Grauslund, M.F. Towards industrial pentose-fermenting yeast strains. Appl. Microbiol. Biotechnol. 2007, 74, 937–953.
- Gopinath, V.; Meiswinkel, T.M.; Wendisch, V.F.; Nampoothiri, K.M. Amino acid production from rice straw and wheat bran hydrolysates by recombinant pentose-utilizing Corynebacterium glutamicum. Appl. Microbiol. Biotechnol. 2011, 92, 985–996.
- Sasaki, M.; Jojima, T.; Kawaguchi, H.; Inui, M.; Yukawa, H. Engineering of pentose transport in Corynebacterium glutamicum to improve simultaneous utilization of mixed sugars. Appl. Microbiol. Biotechnol. 2009, 85, 105–115.





